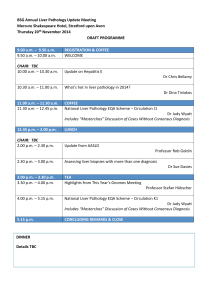
Adverse versus non-adverse – the role of adaptation
advertisement

Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Adverse versus non-adverse – the role of adaptation John R Foster FRCPath, ToxPath Sciences Ltd, Congleton, United Kingdom Hosted by: European Society of Toxicologic Pathology Co-sponsored by: British Society of Toxicological Pathology, French Society of Toxicological Pathology and Society of Toxicologic Pathology Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Adaptive versus adverse? • • • • “Adaptive” and “adverse” changes are not mutually exclusive conditions! An adaptive change permits an organism to respond to an environmental change and is always to the benefit of the organism – not always desirable! An adaptive change can sometimes result in a change in the function of the organ – smoking-induced mucous metaplasia of bronchial mucosa What can be a beneficial, adaptive change acutely, can develop into an adverse change in the longer term. page 2 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 When adaptive becomes adverse • An adaptive change can develop into an adverse one if environmental conditions become so altered as to require the change for the survival of the organism • The adverse effect normally develops secondarily to, or as a progression of, the initial adaptive change • An examples of this is alcohol-induced liver damage: – – – – alcohol-induced increased clearance Hepatic damage Hepatic fibrosis Hepatic cirrhosis – Death. Page 3 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Adaptation is homeostasis in action – a normal response! Correcting mechanism Stress Correcting mechanism Return to optimum Stress Return to optimum Optimum state Correcting mechanism Correcting mechanism The system is one in dynamic equilibrium Page 4 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Normal, adaptation, and pathology Response to the environment Homeostasis Normal variability Stable change Unstable change Adaptation (Non-adverse) = represents amount of energy required to maintain the state Pathology (Adverse) Page 5 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Two examples of “Adaptive” changes • Liver hypertrophy/hyperplasia – – – – Histopathology Biochemical changes Serum biochemistry/functional changes Biomarkers • Nasal toxicity to irritants – Histopathology – Function? Page 6 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Liver as an organ designed for interacting with the environment Front line for chemicals entering body through food. Has developed processes through evolution to deal with xenobiotics Shows adaptive changes to increased metabolic demand – liver growth, hypertrophy/hyperplasia Liver growth is one of the most common response to xenobiotics. It is the normal adaptation to its environment but……….. Can show adverse changes to overtly toxic doses of xenobiotics - necrosis and fibrosis. Page 7 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 What happens at high doses of chemicals that cause liver enlargement at lower doses? • Virtually all chemicals that cause only liver growth/hypertrophy/ hyperplasia at low doses will induce degeneration at higher doses. • Clearly the presence of degeneration is an adverse response • It is accepted that functional replacement may not be total, and fibrotic reactions that occur in this case result in permanent loss of function. • Under these circumstances the changes observed are clearly adverse and a NOAEL is not present at dose levels where these effects occur. Page 17 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 What are the accompanying signs of saturation and toxicity in the liver Histopathology - zonal necrosis, increased apoptosis/single cell necrosis, excess fatty accumulation, inflammatory cells Clinical biochemistry - increased release of liver enzymes into the plasma – transaminases, ALP, GGT etc Decreased plasma albumin and g-globulins Page 19 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Current approaches to defining increased liver weight as adverse • In the presence of histopathology, additional to hepatocellular hypertrophy, the increased liver weight is adverse. • The presence of statistically significant changes in serum parameters indicative of cytotoxicity, ALT ↑, AST ↑, ALP ↑, bilirubin ↑, albumin ↓, g-globulins ↓ • In the absence of the above, the definition of adverse, based upon increased liver weight, seems to be dependent upon the magnitude of response: – <10% increased weight not associated with liver cancer in rodents whereas ≥50% was associated with cancer (Carmichael et al 1997). – NTP database showed that ≥50% increase in liver weight was associated with cancer in rodents (Haseman et al 1997) – Liver enlargement ≥ 40% was associated with hepatic neoplasia in C57Bl/10j mice (Greaves 1996) – Pesticide Safety Directorate/ACP guidance document suggested that increases in liver weight in rodents of <10% were considered to be non-adverse, while increases of >10% were interpreted as being potentially adverse (Andrew 2005) http://www.pesticides.gov.uk/Resources/CRD/MigratedResources/Documents/A/ACP_Paper_on_the_interpretation_of_Liver_Enlargement.pdf Page 10 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Conclusions: Liver enlargement alone • Regulatory agencies in EU regard ≥20% increase in liver weight as adverse, irrespective of duration of study showing the effect and of the species showing it. • Why? • This is a dogmatic stance that lacks sound, scientific, support. • In the absence of additional evidence of cytotoxicity (serum enzymes etc.) hypertrophy alone should be regarded as non-adverse and should be used to set a NOAEL. – At dose levels that induce additional cytotoxicity, chronic induction of regenerative cell replication would occur – a clear risk factor for chronic toxicity/carcinogenicity. Page 11 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Industrial chemical – furfuryl alcohol • Large production volume, chemical intermediate • Eye irritation is limiting in human studies – limited evidence for respiratory effects in man! • Male and female F344/N rats and B6C3F mice exposed by inhalation for 6hr/day for 5 days/week for 2 years (NTP TR482 – 1999). • Non-genotoxic Page 12 14 week study in F344 rats Male Female Exposure (ppm) 0 2 4 8 32 0 2 4 8 32 No examined 10 10 10 10 10 10 10 10 10 10 Squamous metaplasia – respiratory epithelium 0 0 0 5 10 0 0 0 6 10 Mucous hyperplasia – respiratory epithelium 0 0 0 7 10 0 0 0 2 9 Squamous metaplasia – transitional epithelium 0 9 10 10 10 0 9 10 10 10 Degeneration/vacuolation – olfactory 0 1 4 9 10 0 3 7 10 10 Hyperplasia – olfactory 0 0 1 2 9 0 0 0 2 10 Metaplasia – olfactory 0 0 0 0 2 0 0 0 0 5 Surface exudate – olfactory 0 0 0 0 7 0 0 0 2 10 Inflammation 0 0 0 1 10 0 2 0 3 6 • • • No NOEL but changes ≤8 ppm were all minimal grade and focal (level 2 only) No effects on food consumption/body weight – no functional effects at or below 8 ppm! Could 8 ppm be NOAEL? Page 13 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Two year studies - results Male Female 0 2 8 32 0 2 8 32 Suppurative inflammation 3/50 6/50 17/50 44/50 4/49 1/50 5/48 23/49 Hyperplasia – lateral wall 1/50 49/50 50/50 50/50 0/49 39/50 48/48 49/49 Metaplasia – squamous 1/50 1/50 8/50 33/50 0/49 1/50 0/48 24/49 Squamous cell neoplasia 0/50 1/50 1/50 4/50 0/50 0/50 0/50 0/50 Suppurative inflammation 7/50 11/49 27/49 28/50 5/50 12/48 25/49 42/50 Metaplasia – squamous 0/50 6/49 35/49 47/50 1/50 1/48 34/49 46/50 F344 rat B6C3F1 mouse • • • • • • NOEL in male rat at 8ppm for neoplasia – combined nasal tumours No neoplasia in female rat, male or female mouse No NOEL for non-neoplastic findings but…… Squamous metaplasia High incidence of low severity (minimal to mild), and focal to level 2 of the nasal passages at 8 ppm and below! No effects on survival/food consumption/body weight – no obvious functional deficit! NOAEL at 8 ppm? Page 14 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Nasal tissue lesions with furfuryl alcohol • In female rat and mice lesions occur early and do not progress to cancer – can they be considered non-adverse? • Lesion severities at 8 ppm and below were minimal to mild grading and focal to level 2 of nasal passages • Lesions are expected to recover – no recovery study available. • Can they be considered an adaptive response as a consequence of the expected function of the nose in scrubbing out irritant chemicals? • Can a NOAEL be set at 8 ppm? Page 15 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 Summary • Adaptation is an evolutionary strategy for survival • Adaptive changes are not necessarily non-adverse • Decision of “non-adverse” for a change is based upon the impact of the change on the function of the organ. • An adaptive change that results in, or leads to, the long term degeneration of the organ is clearly adverse – hepatic fibrosis; neoplasia. • Adaptive changes are not necessarily reversible. Page 16 Webinar on: “Considerations and Guidance for Adversity Decisions on Pathological Findings in Toxicology and Pathology Reports” based on presentations given at the 4th International ESTP Expert Workshop in Paris, France, June 08+09, 2015 THANK YOU FOR YOUR ATTENTION Page 17