Indicator Titration
Introduction
One method a chemist can use to investigate acid-base reactions is a titration. An
indicator titration is performed by adding a standard base to an acid of unknown
concentration. The color of the acid is watch carefully. The endpoint of the titration
occurs when the indicator causes the solution to change color (from clear to fuchsia in
this case). This point occurs when the acid and base in solution are stoichiometrically
equivalent. An endpoint can be very useful in determining the concentration of an acid or
base.
Purpose
To titrate a dilute HCI solution with a dilute NaOH solution and to observe the endpoint
and find the concentration of an unknown acid solution.
Objectives
After completing this experiment the student will be able to:
Determine the concentration of a strong acid
Pinpoint sources of error
Explain what effect various sources of error will have on the calculated
conecentration of the acid
Equipment/Materials
HCI (unknown conc.)
0.10 M NaOH
250-mL volumetric flask
50-mL buret
250-mL beaker
100-mL graduated cylinder magnetic stirrer and stirring bar
phenolphthalein indicator
buret clamp
Kimwipes
Safety
• Always wear apron and goggles in the lab.
• Avoid contact of solutions with skin and clothing. If there is contact, wash with water.
• Locate the acid/base safety stations in the laboratory.
• Sodium hydroxide can cause instantaneous, permanent eye damage.
Procedure
1. Obtain a beaker for wash water.
2. Measure 25.00 mL of unknown HCI into a 250-mL beaker and dilute it with 75 mL of
water.
3. Place the beaker on the magnetic stirrer and add a stirring bar.
4. Clean a 50-mL buret and rinse three times with about 5 mL NaOH solution.
5. Fill the buret past 0.00-mL mark with NaOH solution and deliver the excess into a
waste beaker.
10. Add a couple drops of indicator solution TO THE ACID.
11. Begin titrating, add the NaOH quickly at first but as the pink starts to linger add more
slowly.
12. Continue Adding slowly until a final drop cause the solution to turn a very slight pink
and remain so for 10 seconds. Record the volume of NaOH added. This is the endpoint.
13. Repeat three times adding only 50 ml, then 25 ml, and finally 0 ml of water in step 2.
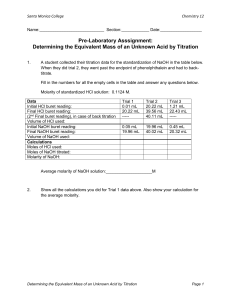
14. Determine the average molarity of the acid solution.
Calculations
a.
b.
d.
e.
f.
Find the moles of base. (vol base (L) x conc base (M/L) = moles)
Write and balance the equation for the reaction of HCI with NaOH
Calculate the moles of acid required to react with the calculated moles of NaOH.
Determine the concentration of acid in moles/L.
Repeat for all trials and average.
Questions
How would the following affect your results? NOTE: “Your results will be off,” is not
an acceptable answer. You are to tell me, “The calculated concentration of the acid will”:
a) increase; b) decrease; c) be unaffected. You will then support your answer with logical
statements (preferably including equations and mathematics—but baby steps).
a. The buret is dirty and drops of NaOH cling to the sidewalls of the buret as it is
drained.
b. The buret is not rinsed with NaOH prior to filling.
c. The buret tip is not filled at the start of the titration.
d. 30 mL of unknown acid are used instead of 25 mL.
e. The buret is not read at eye level. Explain.
f The base is added too rapidly in the equivalence region of the titration and the
solution turns dark pink.
g. 100 mL of distilled water is used instead of 75 mL in step 2.
h. The original volume of base is not at 0.00 mL and a student assumes it is.
 0
0