Chemistry 100: Nuclear Reactions Worksheet – Lead into Gold

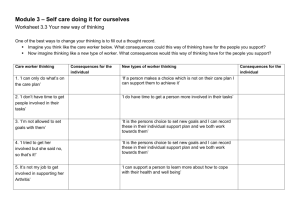
Chemistry 100: Nuclear Reactions Worksheet – Lead into Gold
Team Name: _______________________
Leader:
Recorder:
____________________
____________________
Spokesperson: ____________________
Worker:
Worker:
Worker:
____________________
____________________
____________________
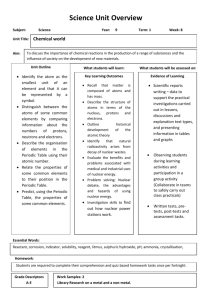
Practical B describes how Rutherford made nitrogen into oxygen and later, other scientists did turn platinum into gold. How do you make this work? Smash two nuclei together:
14
7
N
2
4
He
17
8
O
1
1 p
Why couldn’t the alchemists get this to work? Getting the nitrogen and helium nuclei together is hard because the nuclei are +7 and +2 charged and strongly repel each other.
Rutherford used a particle accelerator (at huge cost – Brookhaven National Laboratory’s big accelerator costs about $80,000 per hour just for electricity).
1.
Read Practical B and then complete the following nuclear equations:
11
5
B
15
7
N
196
78
Pt
1
2
H
197
78
Pt
197
Pt
78
2.
The last paragraph of Practical B is a huge understatement: Only hundreds to tens of thousands of gold atoms can be created at a time. If we are optimistic and get 10000 gold atoms, what is the mass? a.
How many moles of gold is 10000 gold atoms? b.
What is the mass of this gold in grams? c.
If Gold is worth $12.33 per gram (online quote 8/6/03, 10:20 pm). How much is the gold from part (b) worth?