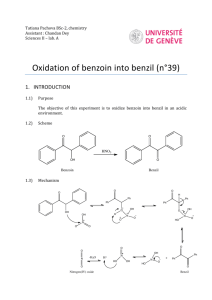
Stereochemistry of the sodium borohydride reduction of benzoin
advertisement

Stereochemistry of the sodium borohydride reduction of benzoin Introduction: The purpose of this two part experiment is to first synthesize 1,2-diphenyl-1,2-ethanediol by the reduction of benzoin using sodium borohydride then use this product to prepare the cyclic acetal of the product where stereochemical information can be derived from H1-NMR spectroscopy. In this experiment, metal hydride reduction is performed, a recognition of symmetry elements in molecules is a key aspect, and the identification of stereocenters is crucial to the identification of stereoisomers. In the above reaction, racemic benzoin is reduced using NaBH4 to obtain glycol hydrobenzoin. It is evident from the above reaction that from the initial reaction two pathways can occur. A chiral diastereomer of the diol can form, or the meso compound can form. The meso isomer will have two non-identical methyl groups that will produce different HNMR signals. It is possible to determine which product is obtained by using H1-NMR analysis. For the H1-NMR to be useful a couple conditions must be satisfied; the chiral carbons of the product must carry one hydrogen atom each and these hydrogens must be non-equivalent, second, their coupling to other atoms in the molecule must be few. To distinguish between the Erythro and Threo these diastereomers must have a preferred conformation under experimental conditions. H1-NMR spectrometry consists of exposing a product to a magnetic field with a specific frequency. In this case, the frequency used is 500 MHz. When nuclei are exposed to a very large magnetic field, the majority of them are aligned with the magnetic field. This is known as the nuclear spin. However, when the magnetic field or frequency is adjusted, some nuclei will change direction opposed to the magnetic field, giving rise to a signal on the spectrum. Analysis of the signal not only can determine fragments but their coupling constants as well. This is a useful tool for the determination of compounds. Results: Mechanism for the reduction of a carbonyl group using sodium borohydride: Table #1: Amount of Reagents Used in Part A Table #2: Amount of Reagents Used in Part B Table #3: Physical Properties Table #4: Final Mass of the Product Discussion: The first week of the lab is solely dedicated to the preparation glycol hydrobenzoin via a reduction of benzoin using sodium borohydride. 2.00g of benzoin is dissolved in 20ml of absolute ethanol in a 125 erlenmeyer flask. Once dissolved a cloud precipitate forms. The reducing agent (sodium borohydride) is then added in small portions ( as to control the amount of foam that builds). Once the addition is complete an additional 5 ml is then added and stirring then continues for 15 minutes. The reaction is then kept cool by being placed in ice and allowed to stir on a magnetic stirrer. The contents of the flask are then diluted with 30 ml of distilled water and then the solution is precipitated using 6M HCL. Again the HCL must be added drop wise as to control the amount of foaming. Once the reaction is complete, an additional 10 ml of distilled water is added and the solution is stirred for an additional 10 minutes. The filtrate is then collected via suction filtration and placed into a vile to be used in the second part of the experiment. During the second week of the lab the diol obtained is used to synthesize the actinide of 1,2-diphenyl-1,2-ethanediol and determine which diastereomer is obtained, In fact for the synthesis in both parts A and B there exists two different isomers as discussed in the introduction. Using the recorded physical properties it is possible to determine which pathway was obtained experimentally. Using 1.00g of diol, it is added and mixed with 0.310g of anhydrous iron chloride in 30 ml of anhydrous acetone in a 100 ml round bottom flask and heated under reflux for 20 minutes. It is important to note that all anhydrous reagents are used because they are “dried” and well prevent unwarranted side reactions. If water were to contaminate the reaction it would lead to the cleavage of the solute and give a separate reaction yielding impurities. The mixture is then cooled for 5 to 10 minutes and then it is diluted with 10ml K2CO3. The mixture is then transferred to a separatory funnel where the organic layers will be clearly identifiable after the addition of dichloromethane and separated. The phases were distinguishable the moment the two 20ml portions of dichloromethane were added to the funnel. The lower organic layer was then drained into a beaker and dried using anhydrous NasSO4 . The solvent must then be evaporated out so the solution is heated until only a few ml of the organic compound remain. The solution is then placed in an ice bath for crystallization. After crystallization, it is then vacuum filtered and dried. The mass, mp, and NHM are then recorded. Conclusion: Solely from the H NMR spectrum, the two distinguishable and identifiable peaks show that they are exactly identical to the NMR sheet obtained from literature. It is evident that since the literature value is that of the meso form, it follows that the product obtained experimentally is of the meso form.