BOS Chemistry Trial Paper 2012 Marking Guidelines
advertisement

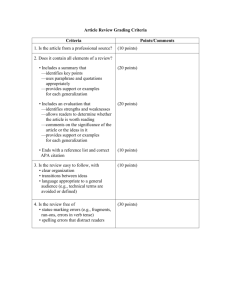
2012 HSC Trial Paper Chemistry Marking Guidelines Section I, Part A Multiple-choice Answer Key Question 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 Answer C D D C B A B B C D A B C D B B C A A B Section I, Part B Question 21(a) Criteria Describes the procedure of the experiment with specific examples Includes at least one relevant chemical equation Describes the procedure of the experiment with specific examples • • • OR • Includes at least one relevant chemical equation Marks 2 1 Question 21(b) Criteria • Identifies an appropriate safety precaution other than using safety goggles AND • Justifies the reason for their choice Marks 1 Question 22(a) • • • Criteria Identifies the importance of the double bond in ethylene Describes the polymerization process of ethylene AND includes a relevant chemical equation Includes at least two examples of products produced from the polymerization of ethylene Identifies the importance of the double bond in ethylene • OR • Briefly describes the polymerization process of ethylene • Includes one example of a product produced from the polymerization of ethylene Marks 2-3 1 Question 22(b) • • Criteria Compares the production of ethanol by fermentation and hydration Identifies ethanol can be produced by fermentation and hydration Marks 2 1 Question 23(a) • Criteria Identifies a purpose of a salt bridge Marks 1 Question 23(b) • • Criteria Accounts for the gradual thinning of the zinc strip Accounts for the development of a reddish-brown solid on the zinc strip Marks 5 AND • Identifies this solid as copper • Explains why the blue copper (II) solution becomes paler over time • Uses relevant chemical equations to explain each of these observations • Accounts for at least three observations • Uses relevant chemical equations to explain these observations • Provides a general description of at least two observations AND • Includes relevant chemical equations • Provides a general description of at least one observation OR • Includes relevant chemical equations 3-4 2 1 Question 24 • • • • • • Criteria States the Bronsted-Lowry definition of an acid and base Uses a relevant chemical equation to indicate the acidic AND basic nature of hydrogen carbonate Identifies hydrogen carbonate as an amphiprotic substance States the Bronsted-Lowry definition of an acid and base Uses a relevant chemical equation to indicate the acidic AND basic nature of hydrogen carbonate Uses a relevant chemical equation to indicate the acidic AND basic nature of hydrogen carbonate Marks 3 2 1 Question 25 Criteria • Outlines the relationship between pH and acidity • Identifies the industrial and/or human sources of pH pollutants such as SO2 and NOx gases AND • Relates this to the difference in the pH of rain in developing and industrialized countries • Explains possible impacts of increased acidity on the marine environment • Identifies the industrial and/or human sources of pH pollutants such as SO2 and NOx gases • Provides possible impacts of increased acidity on the marine environment Question 26(a) Suggested answer Ba(OH)2(aq) + 2HNO3(aq) 2H2O(l) + Ba(NO3)2(aq) (1 mark) Marks 3-4 1-2 Note: must also include the appropriate states. Question 26(b) Suggested answer Moles of nitric acid = volume x concentration = 0.02235L x 0.08M = 0.001788 moles Moles of barium hydroxide required = moles of nitric acid / 2 (refer to the mole ratio in question 26a) = 0.001788 moles / 2 = 0.000894 moles (1 mark) Volume of barium hydroxide required = moles / concentration = moles of barium hydroxide required / concentration of barium hydroxide = 0.000894 moles / 0.14M = 0.006386…L (leave the answer in this form) (1 mark) Question 26(c) Suggested answer Moles of excess (spilt) barium hydroxide = volume x concentration = 0.0135L x 0.14M = 0.00189 moles Consider: Ba(OH)2(aq) Ba2+(aq) + 2OH-(aq) Moles of excess OH= moles of excess barium hydroxide x 2 (refer to the mole ratio in the above equation) = 0.00189 moles x 2 = 0.00378 moles (1 mark) Concentration of excess OH= moles / total volume = moles of excess OH- / (0.02235L + 0.0135L + volume of barium hydroxide required) = 0.00378 moles / (0.02235L + 0.0135L + 0.006386…L) = 0.0895…M (leave the answer in this form) (1 mark) Concentration of excess H+ = (1x10^-14) / concentration of excess OH= (1x10^-14) / 0.0895…M = 1.117…x10^-13M (leave the answer in this form) (1 mark) pH of resulting solution = -log(concentration of excess H+) = -log(1.117…x10^-13M) = 12.95 (2dp) (1 mark) Question 26(d) • • Criteria Identifies the need for a weak, amphiprotic substance to neutralize the spill Suggests an appropriate substance such as sodium hydrogen carbonate Marks 1-2 Question 27(a) (1 mark) Question 27(b) • • • • • • • • • • • • • • • • • Criteria Identifies ALL the reagents used including 1-pentanol, ethanoic acid, concentrated sulphuric acid (catalyst) and boiling chips (optional) Includes the chemical equation for this esterification process (ie. 1pentanol + ethanoic acid water + pentyl ethanoate) Provides a correctly labeled diagram of the process Describes the esterification procedure Explains the purpose of refluxing Explains the purpose of the water bath Explains the need for safety precautions such as gloves and safety goggles, and performing the experiment in a fume cupboard Identifies some of the reagents used Includes the chemical equation for this esterification process Provides a correctly labeled diagram of the process Describes the esterification procedure Explains the purpose of refluxing and/or the water bath Lists the safety precautions to be taken Identifies some of the reagents used Provides a general diagram of the process Briefly describes the esterification procedure and identifies the need for refluxing and/or the water bath Lists the safety precautions to be taken Marks 6-7 4-5 3 • • • Identifies some of the reagents used Briefly describes the esterification procedure and includes a diagram Lists some of the safety precautions to be taken 1-2 Question 28(a) • Criteria Identifies two branches of chemistry, such as physical chemistry, analytical chemistry, organic chemistry, inorganic chemistry Marks 1 Question 28(b) Criteria • Identifies a specific chemist and their branch of chemistry • Describes the role/work of this chemist in their occupation AND • Relates this to a chemical principle they use • Identifies a chemist working in a branch of chemistry AND • Describes their role Marks 3 1-2 Question 29(a) • • • • Criteria Accounts for the importance of monitoring heavy metals in waterways and their impacts on the marine environment Compares the use of AAS to monitor heavy metal pollution, in comparison to traditional methods Identifies the importance of monitoring heavy metals in waterways Lists the advantages and disadvantages of using AAS Marks 3 1-2 Question 29(b) Criteria • Includes a balanced chemical equation of the Haber process (ie. N2(g) + 3H2(g) 2NH3(g)) • Accounts for the needs to monitor different aspects of the reaction vessel including temperature, pressure, gas ratio, impurities and contaminants AND • Identifies the consequences if monitoring is neglected • Accounts for the needs to monitor some of the different aspects of the reaction vessel OR • Identifies the consequences if monitoring is neglected Marks 3 1-2 Question 30(a) Criteria Marks • Identifies the detrimental effects of CFCs in the stratosphere to the ozone layer AND • Relates this to the effect on humans and/or plants • Describes how CFCs destroy ozone using a series of relevant chemical equations • Identifies the detrimental effects of CFCs in the stratosphere to the ozone layer • Describes how CFCs destroy ozone 3 1-2 Question 31(b) • • • • Criteria Identifies the sources of CFCs Provides a list of the different steps that have been taken to prevent further release of CFCs, such as the Montreal Protocol, banning of CFCs and HCFCs, HCF replacements) Identifies that even after such steps of prevention, it takes a very long period of time for the ozone to return to its normal state Briefly describes some steps that have been taken to prevent the release of CFCs Marks 2-3 1 Question 35(a) • Criteria Identifies that the molecule is an amino acid Marks 1 Question 35(b) Criteria Correctly identifies a distinguishing test for each carbon compound • Correctly identifies the positive result for ALL chemical tests • Includes at least TWO chemical equations including states • Identifies at least TWO distinguishing tests • Correctly identifies the positive test for at least ONEidentified distinguishing tests • Includes at least ONE correct chemical equation with incorrect or without states OR • Identifies at least THREE distinguishing tests • Incorrectly identifies ONE or more positive test • Includes at least ONE correct chemical equation with incorrect or without states Completes at least TWO of the following: • Identifies at least ONE distinguishing test OR • Identifies the positive test of ONE distinguishing test • Marks 4 2-3 1 OR • Includes ONE chemical equation Question 35(c) Criteria Identifies that both glycosidic bond and a peptide bond is produced by condensation polymerisation • Identifies that a glycosidic bond is formed by two carbohydrates while a peptide bond is formed by two amino acids • Describes the difference between a glycosidic bond and a peptide bond • Correctly does any TWO of the above. • Correctly does any ONE of the above. • Marks 3 2 1 Question 35(d)(i) • Criteria Identifies that the technique used is electrophoresis Marks 1 Question 35(d)(ii) Criteria • Identifies that chromatography requires a solute dissolved in the solvent • Explains how paper chromatography works with reference to polarity • Suggests a possible use of paper chromatography in forensic chemistry • Any TWO of the above. • Any ONE of the above. Marks 3 2 1 Question 35(e) Criteria • Identifies that the DNA structure is a double helix • Identifies that there are two backbones and refers to deoxyribose sugar groups, phosphate groups AND nitrogen bases OR • Identifies that the DNA structure is a double helix • Identifies that there are two backbones and refers to nitrogen bases • Identifies that the four nitrogen bases form hydrogen bonds • Identifies any TWO of the above • Identifies any ONE of the above Marks 3 2 1 Question 35(f) • Criteria Identifies at least THREE instrumental techniques. Marks 6-7 • Discusses the value of each instrumental techniques, including at least TWO advantages and TWO disadvantages of each instrumental technique. Each discussion must refer to cost and time. • Suggests a use of each technique in forensic chemistry. • Includes an evaluative statement in reference to the value of instrumental techniques in forensic chemistry. • Coherent response and logical progression of ideas. • Identifies at least THREE instrumental techniques. • Discusses the value of each instrumental techniques with at least ONE advantage and disadvantage of each technique including reference to either cost and/or time. • Includes an evaluative statement. • Identifies at least ONE instrumental technique. • Discusses the value of each at least ONE instrumental techniques with at least ONE advantage and disadvantage. • Attempts to answer the question. • Identifies ONE instrumental technique. OR • Gives ONE advantage and/or disadvantage of an instrumental technique. OR • Includes an evaluative statement. 4-5 2-3 1