Effects of single dose intranasal oxytocin on social cognition in
advertisement

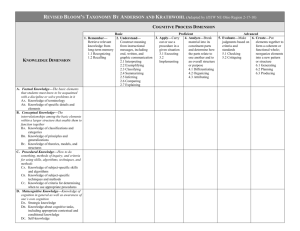
Schizophrenia Research 147 (2013) 393–397 Contents lists available at SciVerse ScienceDirect Schizophrenia Research journal homepage: www.elsevier.com/locate/schres Effects of single dose intranasal oxytocin on social cognition in schizophrenia☆ Michael C. Davis a, b, Junghee Lee a, b, William P. Horan b, Angelika D. Clarke a, b, Mark R. McGee b, Michael F. Green a, b, Stephen R. Marder a, b,⁎ a b UCLA Semel Institute for Neuroscience and Human Behavior, Los Angeles, CA, United States Department of Veterans Affairs VISN 22 MIRECC, Los Angeles, CA, United States a r t i c l e i n f o Article history: Received 7 February 2013 Received in revised form 17 April 2013 Accepted 18 April 2013 Available online 12 May 2013 Keywords: Schizophrenia Social cognition Oxytocin Clinical trial a b s t r a c t Deficits in social cognition are common in schizophrenia and predict poor community functioning. Given the current limitations of psychosocial treatments and the lack of pharmacological treatments for social cognitive deficits, the development of novel therapeutic agents could greatly enhance functional recovery in schizophrenia. This study evaluated whether a single dose of intranasal oxytocin acutely improves social cognitive functioning in schizophrenia. Twenty-three male veterans with schizophrenia completed baseline assessments of social cognition that were divided into lower-level (facial affect perception, social perception, detection of lies) and higher-level (detection of sarcasm and deception, empathy) processes. One week later, patients received the same battery after being randomized to a single dose of 40 IU intranasal oxytocin or placebo. Though the groups did not differ significantly on the social cognition composite score, oxytocin improved performance for the higher-level social cognitive tasks (Cohen's d = 1.0, p = 0.045). Subjects were unable to accurately guess which treatment they had received. The improvements found in higher-level social cognition encourage further studies into the therapeutic potential of oxytocin in schizophrenia. Published by Elsevier B.V. 1. Introduction Schizophrenia is a serious mental illness associated with substantial social and occupational dysfunction (Tandon et al., 2008). While positive psychotic symptoms of schizophrenia often respond to antipsychotic medications, negative symptoms and cognitive impairments are difficult to treat, necessitating novel interventions. In this work, we focus on social cognition, which can be defined as the mental operations that underlie social interactions, including perceiving, interpreting, and generating responses to the intentions, dispositions, and behaviors of others (Mortimer, 2009; Grant et al., 2012). Impairments in social cognition predict multiple aspects of community functioning in schizophrenia (Fett et al., 2011). There are currently two broad approaches to improve social cognition in schizophrenia: psychosocial and pharmacological interventions. While psychosocial interventions (training exercises that target domains of social cognition) have shown some benefit, improvements have typically been limited to a narrow subset of social cognitive processes (most notably, facial affect recognition) (Andreasen, 1982). Pharmacological trials have yielded mixed results, and no medication has consistently improved ☆ Clinical trial registration: Does acute oxytocin administration enhance social cognition in individuals with schizophrenia? http://clinicaltrials.gov/ ClinicalTrials.gov identifier: NCT01312272. ⁎ Corresponding author at: VA VISN 22 MIRECC, Building 210A, 11301 Wilshire Blvd., Los Angeles, CA 90073, United States. Tel.: +1 310 268 3647; fax: +1 310 268 4056. E-mail address: marder@ucla.edu (S.R. Marder). 0920-9964/$ – see front matter. Published by Elsevier B.V. http://dx.doi.org/10.1016/j.schres.2013.04.023 social cognition schizophrenia (Gray and Roth, 2007). Thus, further treatment development is clearly needed in this area. One potential therapeutic target for enhancing social cognition is the oxytocin (OT) system. OT is a nine-amino acid peptide that, in addition to its role in the periphery for regulating lactation and uterine contractions, functions centrally as a neurotransmitter involved in multiple aspects of social behavior (Heinrichs et al., 2009; Meyer-Lindenberg et al., 2011). Given the role of OT in social behavior, and the prominent deficits in social functioning in schizophrenia, a handful of studies have examined the OT system in schizophrenia. Patients with schizophrenia may have altered baseline levels of OT that correlate with symptoms. Specifically, it has been found that lower levels of baseline plasma OT predict negative symptoms (Keri et al., 2009) and also predict the ability of patients with schizophrenia to identify facial expressions (Goldman et al., 2008). OT has also been evaluated as a potential treatment for schizophrenia. For example, regular administration of intranasal OT, added on to antipsychotic therapy, significantly reduced psychotic symptoms of schizophrenia (Feifel et al., 2010; Pedersen et al., 2011; Modabbernia et al., 2013). Beyond symptoms, several groups have also examined the effects of OT on domains of social cognition in schizophrenia, in both acute (single dose) and chronic dosing paradigms. In the two single-dose studies, one found low doses of intranasal OT (10 IU) worsened scores on the Ekman facial affect discrimination task, while a higher 20 IU dose improved scores in patients with polydipsia (Goldman et al., 2011), and the other found a 24 IU dose improved facial affect discrimination (Averbeck et al., 2011). In the study using a 394 M.C. Davis et al. / Schizophrenia Research 147 (2013) 393–397 chronic dosing paradigm, 14 days of intranasal OT administration significantly improved scores on one test of theory of mind, but not other social cognitive measures (Pedersen et al., 2011). Altogether, there is some evidence that OT administration may have therapeutic benefit in the treatment of schizophrenia, but the effects on social cognition are unclear. One important question, arising from the heterogeneity of prior study results, is whether the potential effects of OT on social cognition are specific to particular social cognitive domains. Our current study examined whether a single dose of intranasal OT improved performance across key subdomains of social cognition in schizophrenia. Prior research identified two levels of social cognitive impairment in schizophrenia that are significantly associated with functional capacity and real-world social and work functioning: 1) low-level social cue detection and 2) high-level inferential and regulatory processes (Mancuso et al., 2011). The former factor comprised tasks in which minimal inferential processes are necessary to interpret the presented social information, while the latter factor comprised tasks which required higher-level cognitive processing incorporating knowledge not directly presented in the stimuli. Using a randomized, double-blind, placebo-controlled design, patients completed a social cognitive assessment battery. The primary outcome measure was a composite social cognition score, with secondary analyses examining the low-level and high-level composites separately. In addition to our primary goal of assessing changes in social cognition, our secondary goals included assessing: 1) the subjective experience of intranasal OT treatment; and 2) acute changes in clinical symptoms following intranasal OT treatment. 2. Experimental methods and materials 2.1. Participants Twenty-four male outpatients between the ages of 18 and 56 were recruited from the VA Greater Los Angeles Healthcare System (VAGLAHS). Patients met DSM-IV-TR criteria for schizophrenia, based on clinical interview and medical records. Subjects were clinically stable as indicated by: no psychiatric hospitalizations in the past 6 months; adherent to antipsychotic medication with dosages not varying by >25% over 3 months prior to participation; at least 6 months since any indication of potential danger to self or others; no acute medical problems; and chronic medical conditions consistently treated and stable for >3 months. Exclusion criteria were mental retardation; treatment with electroconvulsive therapy within 6 months prior to participation; history of stroke, traumatic brain injury, or epilepsy; history of substance abuse or dependence within 6 months prior to participation; history of hyponatremia within 6 months prior to participation; or allergic rhinitis or other inflammation of nasal mucosa. Antipsychotic medication type and dose were clinically determined. All participants had the capacity to give informed consent and provided written informed consent in accordance with procedures approved by the Institutional Review Board at VAGLAHS. 2.2. Pharmacological treatment OT nasal spray (50 IU/ml) was compounded by Inland Compounding Pharmacy (Loma Linda, CA). A placebo nasal spray was prepared that was otherwise identical to the active treatment. Nasal sprays were prepared in 3 ml single use vials, calibrated to dispense 0.1 ml per puff. Subjects were instructed to spray 4 puffs into each nostril, for a total dose of 40 IU OT (or equivalent volume of placebo spray). 2.3. Study design At an initial screening visit, informed consent was obtained and demographic data and medical histories were obtained. Brief physical examinations were performed. If subjects were eligible, they were scheduled for a second visit for baseline assessment. At baseline, subjects were assessed with the Positive and Negative Syndrome Scale (PANSS), Clinical Global Impression (CGI), and the social cognition assessment battery, with no pharmacological intervention. The order of administration for social cognitive tests was counter-balanced across subjects. On the third visit, one week later, subjects were randomized (double-blind) to receive either intranasal OT or placebo. Thirty minutes after treatment, each subject completed the same assessments in the same order as in their baseline visit. At the conclusion of the visit, participants were interviewed regarding their subjective experiences of the treatment. 2.4. Social cognition assessments Social cognition was assessed by 4 tests that represented 4 social cognitive domains. The tests are briefly described here and more complete descriptions about the tests and their use in schizophrenia are available elsewhere. 2.4.1. Theory of mind Theory of mind was assessed using Part III of The Awareness of Social Inference Test (TASIT Part III: Social Inference—Enriched) (McDonald et al., 2003), as described previously (Mancuso et al., 2011). This test provides a total score (maximum 64) and subscale scores for lies and sarcasm scenes (maximum 32 each). 2.4.2. Empathy Empathy was assessed using the Emotional Perspective Taking Task (EPTT) (Derntl et al., 2009). In this task, subjects are presented with 60 digital images depicting two individuals in a social interaction, with one individual's face masked. Subjects are asked to infer the emotional expression of the masked face, selecting between two choices. Scenes portray 5 basic emotions as well as neutrality and each image is displayed for 4 s each. The score is the total number correct (maximum = 60). 2.4.3. Social perception Social perception was assessed using the Half Profile of Nonverbal Sensitivity (Half-PONS) (Rosenthal et al., 1979; Ambady et al., 1995), as described previously (Mancuso et al., 2011). The score is the total number correct (maximum = 110). 2.4.4. Facial affect recognition Facial affect recognition was assessed by asking participants to identify facial expressions of emotion in still images from the standardized stimulus set developed by Ekman (2004), as described previously (Mancuso et al., 2011). The score is the total number correct (maximum = 56). 2.5. Clinical assessments Positive and Negative Syndrome Scale (PANSS) for Schizophrenia (Kay et al., 1987): This instrument assesses 30 different symptoms on a scale from 1 to 7 based on clinical interview. For the current study, total scores as well as positive, negative, and general psychopathology subscores were examined. Clinical Global Impression (CGI-S and CGI-I) (Guy, 1976): For the CGI-S (severity scale) the clinician rates the severity of the subject's mental illness, relative to their past experience with patients with the same diagnosis, from 1 (=normal, not at all ill) to 7 (=extremely ill). The CGI-I (improvement scale) requires the clinician to rate on a scale from 1 to 7 how much the mental illness has improved or worsened, relative to a baseline. MIRECC Global Assessment of Functioning (MIRECC GAF): This is a version (Niv et al., 2007) of the Global Assessment of Functioning M.C. Davis et al. / Schizophrenia Research 147 (2013) 393–397 scale in which occupational functioning, social functioning, and symptom severity are scored on a scale of 1–100, with lower scores indicating more impairment. 2.6. Subjective experience assessment Subjects completed a brief qualitative interview at the end of their participation, asking whether they thought they had received OT or placebo; whether they experienced any treatment-associated side effects; and whether the social cognition assessment battery was difficult to tolerate. 2.7. Statistical analysis 395 Table 1 Demographic and baseline clinical characteristics. Numbers represent mean values, with standard deviations in parentheses. For sex, ethnicity, and marital status data, numbers represent number of subjects. Chlorpromazine equivalent doses of antipsychotic were calculated using published equivalencies for conventional and atypical antipsychotics (Lehman et al., 2004; Woods, 2003). There were no statistically significant differences in demographic or baseline clinical characteristics between the treatment groups. PANSS = Positive and Negative Syndrome Scale; CGI-S = Clinical Global Impression— Severity; MIRECC GAF = Mental Illness Research, Education, and Clinical Center Global Assessment of Functioning. Age Sex (male) Ethnicity White Latino Asian Black Marital status Single Married Divorced Widowed Years of education Years of psychotic symptoms Antipsychotic (in mg CPZ equivalents) PANSS positive PANSS negative PANSS general PANSS total CGI-S MIRECC GAF: occupational MIRECC GAF: social MIRECC GAF: symptomatic Placebo (n = 12) Oxytocin (n = 11) 48.6 (9.1) 12 48.6 (6.6) 11 3 2 3 4 1 1 0 9 9 0 2 1 12.7 (1.5) 30.2 (10.6) 612.5 (457.5) 18.2 (6.2) 19.8 (5.4) 33.8 (8.3) 71.8 (16.5) 4.4 (1.5) 37.9 (17.4) 55.8 (14.0) 47.1 (16.0) 9 0 1 1 12.7 (1.6) 24.5 (12.0) 742.4 (753.5) 18.1 (6.8) 21.2 (5.9) 36.4 (6.8) 75.6 (13.7) 4.2 (1.1) 35.9 (12.6) 57.3 (15.7) 58.6 (13.2) All analyses were conducted using SAS version 9.3 (Cary, NC). Baseline patient group characteristics were compared using t-tests or chi-square tests, as appropriate. For social cognition assessments, baseline and follow-up raw scores for each measure were standardized to z-scores using the baseline means and standard deviations from the total sample. A social cognition composite score, which was the primary endpoint of this study, was calculated as the mean of z-scores on TASIT Part III, EPTT, Half-PONS, and Facial Affect Recognition tasks. Based on prior findings (Mancuso et al., 2011), a “low level social cognition” score was calculated as the mean of z-scores on TASIT Part III detection of lies, Half-PONS, and Facial Affect Recognition, and a “high level social cognition” score was calculated as the mean of z-scores on TASIT Part III detection of sarcasm and the EPTT. Each endpoint was evaluated using an analysis of covariance (ANCOVA) model with the change from baseline to treatment as the dependent variable. Each model included treatment group as a factor and the baseline value of the corresponding endpoint as a covariate. Estimates of effect size (Cohen's d) were computed using the difference between the OT and placebo mean changes from baseline to treatment and the pooled (OT + placebo) standard deviation of the change from baseline to treatment. Clinical symptoms (PANSS, CGI-S) were evaluated statistically as per social cognition assessments, though without z-score standardization. CGI-I scores were compared between treatment groups using t-tests. Finally, patients' judgments about which treatment they received were examined with chi-square tests. All results at α = 0.05 (two sides) were considered significant. There were no significant differences in clinical symptom improvement between subjects treated with OT vs. placebo, as assessed by PANSS, CGI-S, and CGI-I ratings (data not shown). Patients were unable to accurately predict which treatment they had received (7 of the patients on OT and 7 of those on placebo thought they were taking OT). Side effects of nasal spray treatment were reported in 2/12 subjects receiving placebo (rhinorrhea and mild nasal irritation) and in 2/11 subjects receiving OT (mild tingling on nasal inhalation and mild drowsiness). 3. Results 4. Discussion Twenty-four subjects were enrolled in the study. One subject randomized to receive OT did not present for his final study visit. Hence, demographic and clinical variables are presented in Table 1 for the 23 remaining subjects. Based on this information, the sample can be characterized as a middle-aged, relatively chronically ill, medicated group of veteran patients who were experiencing moderate to high levels of current psychiatric symptoms and generally low functional outcome. All subjects in the analysis except one had complete social cognitive data at the final visit. There were no statistically significant differences in baseline demographic or clinical variables. Performance on social cognition assessment battery composite scores, as well as individual measures, is presented in Table 2. OT did not significantly improve performance relative to placebo on the total social cognition composite measure, which was our primary endpoint (Fig. 1A). However, when measures were combined into high-level and low-level composite scores (Fig. 1B, C), OT significantly improved performance in high-level social cognition (p = 0.045) with a large effect size (d = 1.00). OT treatment did not improve performance on any individual social cognition measure when compared to placebo, though it reached a trend level of significance in improving the TASIT III sarcasm score. In general, patients later randomized to receive OT scored lower on baseline social cognition measures, particularly the EPTT and Ekman facial affect recognition tasks. In this study, we evaluated the acute effects of an OT treatment across multiple domains of social cognition. The treatment and testing battery were well-tolerated. A single dose of 40 IU intranasal OT did not impact overall social cognitive functioning. It did have a significant effect on high-level (inferential and regulatory processes) social cognition, but not on low level (social cue detection) social cognition. These preliminary findings support further efforts to evaluate the specificity of therapeutic benefits of OT for social cognition in schizophrenia. Our findings were consistent with those of Pedersen et al. (2011), who reported significant improvement on a theory of mind test (i.e., Brüne 2nd order false belief identification), which reflects high-level social cognition. In contrast, Averbeck et al. (2011) reported improved emotion recognition following a single dose of OT in men, while we saw no significant effect on facial affect recognition. Differences in study sample and dosing could potentially explain this, as our sample was older, more symptomatic, and we used a higher (40 IU) dose of intranasal OT (Feifel et al., 2010). Indeed, differential dose–response of intranasal OT is possible, as Goldman et al. (2011) found different effects on emotion detection between 10 IU and 20 IU OT doses. One methodological question concerns the duration of effects following intranasal OT administration. We performed the 1-hour social 396 M.C. Davis et al. / Schizophrenia Research 147 (2013) 393–397 Table 2 Social cognition assessments. Numbers represent z-scores or changes in z-scores, with standard deviations in parentheses. PL = placebo; OT = oxytocin; total social cog composite = mean of scores on TASIT III total, EPTT, Half-PONS, and Ekman faces; high level composite = mean of scores on TASIT III sarcasm and EPTT tasks; low level composite = mean of scores on TASIT III lies, Half-PONS, and Ekman faces tasks; TASIT III = the awareness of social inference test, with total, lies, or sarcasm subscores as indicated; EPTT = emotional perspective taking task; Half-PONS = half-profile of nonverbal sensitivity; Ekman faces = facial affect recognition task. PL baseline z-score OT baseline z-score Total social cog composite High level composite Low level composite 0.19 (0.80) 0.17 (0.61) 0.15 (0.89) −0.19 (0.76) −0.16 (1.1) −0.13 (0.61) Measures TASIT III total Lies Sarcasm EPTT Half-PONS Ekman faces 0.12 0.07 0.08 0.26 0.07 0.29 −0.12 −0.07 −0.08 −0.28 −0.07 −0.27 (0.88) (0.90) (0.84) (0.87) (1.2) (1.0) (1.1) (1.1) (1.2) (1.1) (0.03) (0.93) PL change in z-score 0.10 (0.35) 0.01 (0.39) 0.15 (0.49) −0.01 (0.47) 0.26 (0.51) −0.22 (0.51) 0.24 (0.63) 0.15 (0.77) 0.05 (0.67) OT change in z-score 0.23 (0.36) 0.47 (0.53) 0.03 (0.38) 0.18 −0.06 0.26 0.61 0.00 0.22 (0.83) (0.92) (0.74) (0.76) (0.73) (0.45) ANCOVA p-value Effect size (Cohen's d) .869 .045⁎ .088 0.35 1.00 0.27 .623 .226 .082 .439 .405 .807 0.30 0.43 0.78 0.53 0.19 0.29 ⁎ Indicates statistical significance of p b 0.05. cognition assessment battery 30 min after the nasal spray administration. To our knowledge, cerebrospinal fluid (CSF) levels of OT following intranasal administration in humans have not yet been measured. A Total Social Cognition Composite z-score 0.6 0.4 0.2 0.0 -0.2 -0.4 Baseline Placebo B Treatment Oxytocin Low-Level Social Cog. Composite z-score 0.6 0.4 0.2 0 -0.2 -0.4 Baseline Placebo z-score C Treatment Oxytocin High-Level Social Cog. Composite 0.6 0.4 0.2 0 -0.2 -0.4 -0.6 Baseline Placebo Treatment Oxytocin Fig. 1. Changes in social cognition composite scores. Total social cognition composite (1A) comprises the mean of z-scores for TASIT III, EPTT, Half-PONS, and Ekman faces tests. Low-level social cog. composite (1B) comprises the mean of z-scores for TASIT III detection of lies, Half-PONS, and Ekman faces tests. High-level social cog. composite (1C) comprises the mean of z-scores for TASIT III detection of sarcasm and EPTT tests. Treatments that patients received are indicated by line color and symbol. Of note, patients only received a nasal spray treatment at the “Treatment” visit, 30 min before beginning testing, and differences in baseline scores are attributable to randomization with limited sample size. Error bars represent standard errors of the mean. Changes in scores between baseline and treatment visits were not significantly different between OT and placebo in total social cognition composite scores or low-level social cognition composite scores. Participants receiving OT significantly improved high-level social cognition composite scores as compared to placebo (p = 0.045). Vasopressin is a similar in terms of structure and molecular weight to OT and has been shown to be significantly elevated in CSF 80 min after intranasal administration (Born et al., 2002). Furthermore, elevated salivary levels of OT have been reported more than 7 h after intranasal administration in humans (van Ijzendoorn et al., 2012). Hence, we believe that our testing occurred while OT was elevated. We were unable to detect any improvement on positive, negative, or general schizophrenia symptoms following a single dose of OT, but previous reports of improvements in clinical symptoms (Feifel et al., 2010; Pedersen et al., 2011; Modabbernia et al., 2013) were during 2–8 weeks of treatment. We were also interested in the subjective effects of acute OT nasal spray administration, as these could serve to break the blind. Our results indicate that the side effects of 40 IU intranasal OT were minimal and that patients could not tell which treatment they had received. In fact, no reliable side effects were noted in a review of 38 randomized controlled trials (MacDonald et al., 2011). This preliminary study has several limitations. First, the small sample size limited our power to detect potential benefits of OT. Second, a limitation related to the sample size is that subjects randomized to OT tended to have lower baseline social cognition scores, which we tried to address by covarying for baseline score. Third, we used an all-male sample that was generally middle age and chronically mentally ill. Thus we could not evaluate potential differential effects on men versus women (Ditzen et al., in press). It will be important for future studies to test whether OT has different effects on social cognition based on age and/or illness duration, and whether the effects are similar between the sexes. This study raises interesting questions about the mechanism for the improvement in high-level social cognition, and whether the effect extends also low-level social cognition. Indeed, prior studies showing improved trust, mental-state attribution, and empathic accuracy following OT administration (Kosfeld et al., 2005; Bartz et al., 2010; Guastella et al., 2010) have led to the speculation that OT's effects may be specific to higher-order social cognition (Churchland and Winkielman, 2012). Functional neuroimaging approaches conducted during social cognitive assessment following intranasal OT administration could help clarify the neural mechanism of OT's effects. Clinically, an exciting future direction will be to test whether intranasal OT could augment the efficacy of social cognitive skills training, which has shown promise for improving some aspects of social cognition in schizophrenia (Horan et al., 2011). Funding source Funding for this project came from a Department of Veterans Affairs VISN 22 MIRECC pilot grant to Dr. Davis. Dr. Davis was supported by the Office of Academic Affiliations, Advanced Fellowship Program in Mental Illness Research and Treatment, Department of Veterans Affairs. M.C. Davis et al. / Schizophrenia Research 147 (2013) 393–397 Contributors Drs. Davis, Green, Horan, Lee, and Marder designed the study. Dr. Davis wrote the protocol, coordinated the study administratively, recruited patients, performed clinical interviews, and wrote the first draft of the manuscript. Ms. Clarke and Mr. McGee coordinated the study administratively. Drs. Davis, Horan, and Lee performed data analysis. All authors contributed to and have approved the final manuscript. Financial disclosures Authors Dr. Davis, Dr. Lee, Dr. Horan, Ms. Clarke, and Mr. McGee have no financial relationships to disclose. Dr. Green reports consultation fees from Abbott Laboratories, Amgen, Biogen, Mnemosyne, and Roche. Dr. Marder reports consultation fees from Amgen, Astellas, Abbott Laboratories, Roche, Targacept, Otsuka, Pfizer, Shire, and Lundbeck; research support from Glaxo Smith Kline, Novartis, Sunovion, and Psychogenics; and is a stockholder of Med Avante. Acknowledgments The authors thank Crystal Gibson, Cory Tripp, Katherine Weiner, Michelle Dolinsky, Amber Tidwell, and Amanda Bender for their assistance in data collection and study administration, and Charles S. Davis for statistical consultation. References Ambady, N., Hallahan, M., Rosenthal, R., 1995. On judging and being judged accurately in zero-acquaintance situations. J. Pers. Soc. Psychol. 69 (3), 518. Andreasen, N.C., 1982. Negative symptoms in schizophrenia. Definition and reliability. Arch. Gen. Psychiatry 39 (7), 784–788. Averbeck, B.B., Bobin, T., Evans, S., Shergill, S.S., 2011. Emotion recognition and oxytocin in patients with schizophrenia. Psychol. Med. 1–8. Bartz, J.A., Zaki, J., Bolger, N., Hollander, E., Ludwig, N.N., Kolevzon, A., Ochsner, K.N., 2010. Oxytocin selectively improves empathic accuracy. Psychol. Sci. 21 (10), 1426–1428. Born, J., Lange, T., Kern, W., McGregor, G.P., Bickel, U., Fehm, H.L., 2002. Sniffing neuropeptides: a transnasal approach to the human brain. Nat. Neurosci. 5 (6), 514–516. Churchland, P.S., Winkielman, P., 2012. Modulating social behavior with oxytocin: how does it work? What does it mean? Horm. Behav. 61 (3), 392–399. Derntl, B., Finkelmeyer, A., Toygar, T.K., Hulsmann, A., Schneider, F., Falkenberg, D.I., Habel, U., 2009. Generalized deficit in all core components of empathy in schizophrenia. Schizophr. Res. 108 (1–3), 197–206. Ditzen, B., Nater, U.M., Schaer, M., La Marca, R., Bodenmann, G., Ehlert, U., Heinrichs, M., 2012. Sex-specific effects of intranasal oxytocin on autonomic nervous system and emotional responses to couple conflict. Soc. Cogn. Affect. Neurosci. http://scan.oxfordjournals.org/ cgi/pmidlookup?view=long&pmid=22842905 (Electronic publication ahead of print). Ekman, P., 2004. Subtle Expression Training Tool (SETT) & Micro Expression Training Tool (METT). Paul Ekman. Feifel, D., Macdonald, K., Nguyen, A., Cobb, P., Warlan, H., Galangue, B., Minassian, A., Becker, O., Cooper, J., Perry, W., Lefebvre, M., Gonzales, J., Hadley, A., 2010. Adjunctive intranasal oxytocin reduces symptoms in schizophrenia patients. Biol. Psychiatry 68 (7), 678–680. Fett, A.K., Viechtbauer, W., Dominguez, M.D., Penn, D.L., van Os, J., Krabbendam, L., 2011. The relationship between neurocognition and social cognition with functional outcomes in schizophrenia: a meta-analysis. Neurosci. Biobehav. Rev. 35 (3), 573–588. Goldman, M., Marlow-O'Connor, M., Torres, I., Carter, C.S., 2008. Diminished plasma oxytocin in schizophrenic patients with neuroendocrine dysfunction and emotional deficits. Schizophr. Res. 98 (1–3), 247–255. Goldman, M.B., Gomes, A.M., Carter, C.S., Lee, R., 2011. Divergent effects of two different doses of intranasal oxytocin on facial affect discrimination in schizophrenic patients with and without polydipsia. Psychopharmacology 216 (1), 101–110. 397 Grant, K.M., LeVan, T.D., Wells, S.M., Li, M., Stoltenberg, S.F., Gendelman, H.E., Carlo, G., Bevins, R.A., 2012. Methamphetamine-associated psychosis. J. Neuroimmune Pharmacol. 7 (1), 113–139. Gray, J.A., Roth, B.L., 2007. Molecular targets for treating cognitive dysfunction in schizophrenia. Schizophr. Bull. 33 (5), 1100–1119. Guastella, A.J., Einfeld, S.L., Gray, K.M., Rinehart, N.J., Tonge, B.J., Lambert, T.J., Hickie, I.B., 2010. Intranasal oxytocin improves emotion recognition for youth with autism spectrum disorders. Biol. Psychiatry 67 (7), 692–694. Guy, W., 1976. Clinical Global Impression Scale. ECDEU Assistant Manual for Psychopharmacology. Heinrichs, M., von Dawans, B., Domes, G., 2009. Oxytocin, vasopressin, and human social behavior. Front. Neuroendocrinol. 30 (4), 548–557. Horan, W.P., Kern, R.S., Tripp, C., Hellemann, G., Wynn, J.K., Bell, M., Marder, S.R., Green, M.F., 2011. Efficacy and specificity of social cognitive skills training for outpatients with psychotic disorders. J. Psychiatr. Res. 45 (8), 1113–1122. Kay, S.R., Fiszbein, A., Opler, L.A., 1987. The Positive and Negative Syndrome Scale (PANSS) for schizophrenia. Schizophr. Bull. 13 (2), 261–276. Keri, S., Kiss, I., Kelemen, O., 2009. Sharing secrets: oxytocin and trust in schizophrenia. Soc. Neurosci. 4 (4), 287–293. Kosfeld, M., Heinrichs, M., Zak, P.J., Fischbacher, U., Fehr, E., 2005. Oxytocin increases trust in humans. Nature 435 (7042), 673–676. Lehman, A.F., Kreyenbuhl, J., Buchanan, R.W., Dickerson, F.B., Dixon, L.B., Goldberg, R., Green-Paden, L.D., Tenhula, W.N., Boerescu, D., Tek, C., Sandson, N., Steinwachs, D.M., 2004. The Schizophrenia Patient Outcomes Research Team (PORT): updated treatment recommendations 2003. Schizophr. Bull. 30 (2), 193–217. MacDonald, E., Dadds, M.R., Brennan, J.L., Williams, K., Levy, F., Cauchi, A.J., 2011. A review of safety, side-effects and subjective reactions to intranasal oxytocin in human research. Psychoneuroendocrinology 36 (8), 1114–1126. Mancuso, F., Horan, W.P., Kern, R.S., Green, M.F., 2011. Social cognition in psychosis: multidimensional structure, clinical correlates, and relationship with functional outcome. Schizophr. Res. 125 (2–3), 143–151. McDonald, S., Flanagan, S., Rollins, J., Kinch, J., 2003. TASIT: a new clinical tool for assessing social perception after traumatic brain injury. J. Head Trauma Rehabil. 18 (3), 219–238. Meyer-Lindenberg, A., Domes, G., Kirsch, P., Heinrichs, M., 2011. Oxytocin and vasopressin in the human brain: social neuropeptides for translational medicine. Nat. Rev. Neurosci. 12 (9), 524–538. Modabbernia, A., Rezaei, F., Salehi, B., Jafarinia, M., Ashrafi, M., Tabrizi, M., Hosseini, S.M., Tajdini, M., Ghaleiha, A., Akhondzadeh, S., 2013. Intranasal oxytocin as an adjunct to risperidone in patients with schizophrenia: an 8-week, randomized, double-blind, placebo-controlled study. CNS Drugs 27 (1), 57–65. Mortimer, A.M., 2009. Update on the management of symptoms in schizophrenia: focus on amisulpride. Neuropsychiatr. Dis. Treat. 5, 267–277. Niv, N., Cohen, A.N., Sullivan, G., Young, A.S., 2007. The MIRECC version of the Global Assessment of Functioning scale: reliability and validity. Psychiatr. Serv. 58 (4), 529–535. Pedersen, C.A., Gibson, C.M., Rau, S.W., Salimi, K., Smedley, K.L., Casey, R.L., Leserman, J., Jarskog, L.F., Penn, D.L., 2011. Intranasal oxytocin reduces psychotic symptoms and improves theory of mind and social perception in schizophrenia. Schizophr. Res. 132 (1), 50–53. Rosenthal, R., Hall, J.A., DiMatteo, M.R., Rogers, P.L., Archer, D., 1979. Sensitivity to Nonverbal Communication: The PONS Test. Johns Hopkins University Press Baltimore. Tandon, R., Keshavan, M.S., Nasrallah, H.A., 2008. Schizophrenia, “Just the Facts”: what we know in 2008 part 1: overview. Schizophr. Res. 100 (1–3), 4–19. van Ijzendoorn, M.H., Bhandari, R., van der Veen, R., Grewen, K.M., BakermansKranenburg, M.J., 2012. Elevated salivary levels of oxytocin persist more than 7 h after intranasal administration. Front. Neurosci. 6, 174. Woods, S.W., 2003. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J. Clin. Psychiatry 64 (6), 663–667.