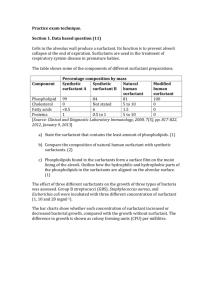
Thermodynamic Analysis of Micelle Formation Using Surfactants Introduction Surfactants, or surface active agents, are amphiphilic molecules that contain a polar head group and a non‐polar tail group, as shown in Figure 1. The hydrophobic group is typically a linear or branched alkyl chain while the head group of the surfactant molecule can be either a non‐ionic, cationic, or anionic group. Because of the distinct differences between these regions of the molecule, surfactants like to reside at interfaces (i.e., solid‐liquid, liquid‐gas, liquid‐liquid interfaces). The inherent ability of these molecules to become surface active yields many unique properties that can be used in many every day products. These applications include soaps, laundry detergents, shampoos, fabric softeners, bactericides, cosmetics, pharmaceuticals, foods and beverages, pesticides, enhanced oil recovery, foams, inkjet printing, and particle synthesis (Salager 2002). Hydrophilic Head (polar)
Hydrophobic Tail (non-polar)
Figure 1: The structure of a surfactant molecule has two distinct parts. The hydrophilic head is polar and likes to come into contact with an aqueous phase. The hydrophobic tail is non‐polar and resists interactions with an aqueous phase. Anionic surfactants are the most widely used, accounting for 50 – 60% of all applications (Salager 2002). Non‐ionic surfactants are the next most important class of surfactants used in approximately 20 – 45% of applications. Cationic and zwitterionic (containing both anion and cation groups) are more expensive and, therefore, have far fewer industrial applications. At low concentrations, surfactant molecules are randomly distributed in the aqueous phase. However, these surfactant molecules self‐assemble into organized structures known as micelles at higher concentrations. In an aqueous solution, the polar head groups will be on the outside of a spherical micelle while the hydrophobic tails aggregate into the core, as shown in Figure 2. The dimensions of the micelles are on the order of twice the length of a surfactant molecule. For example, the size of sodium dodecyl sulfate (SDS) monomer is approximately 1 nm, resulting in micelle diameters of approximately 2 nm (Katz et al. 2008). Because of the distinct hydrophobic core established in a micelle, an aqueous solution containing a high concentration of surfactant can solubilize hydrophobic molecules (e.g., oils) that would normally be insoluble in water. As larger amounts of hydrophobic molecules are solubilized, the micelle swells until a small pool of a distinct phase is established. These swelled states are known as microemulsions, which also have a number of important applications. 1 Thermodynamic Analysis of Micelle Formation Using Surfactants Figure 2: The aggregation of surfactant molecules in an aqueous solution creates a micelle with a hydrophobic region that can solubilize other hydrophobic molecules. As this region becomes larger and creates a distinct phase it becomes known as a microemulsion. Aggregation of surfactants into organized structures The formation of aggregate structures in surfactant solutions is primarily dictated by the concentration of the surfactant. As shown in Figure 3, dilute concentrations of the monomers (surfactant molecules) remain as free molecules in solution. Above a threshold value, known as the critical micelle concentration (CMC), the hydrophobic tails of the monomer begin to aggregate and form spherical micelles. Further increases in surfactant concentration can drive the formation of other complex aggregate structures, such as rod‐like and worm‐like micelles. In some surfactant systems, lamellar phases and vesicles can be formed. Both of these phases have multilayer stacking of surfactant molecules that establish multiple regions of either hydrophobic or hydrophilic molecules. For example, spherical vesicles contain a hydrophilic core, a hydrophobic shell sandwiched between two organized surfactant layers, and the hydrophobic head group on the outside of the vesicle to disperse them in an aqueous solution. Figure 3: The aggregation of surfactant molecules is dependent on the concentration. A threshold concentration, known as the critical micelle concentration (CMC), is needed to observe aggregation. If the concentration is increased further, other types of aggregated structures may be observed, including rod‐like micelles and worm‐like micelles. 2 Thermodynamic Analysis of Micelle Formation Using Surfactants The monomers in the micelle are not held in place permanently. The micelles are continuously forming and disintegrating. Therefore, there is an exchange of the monomers with the bulk solution that establishes an equilibrium between micelles and monomers, as shown in Figure 4. This phenomenon can be important to some applications since the stability of the micelle can be related to the solubility rate of hydrophobic molecules, droplet size, and foamability. Kmic
M onomers Ð M icelles Figure 4: Surfactant molecules are continuously exchanged with the micelle and an equilibrium is established. Below the CMC, the monomer concentration increases linearly with the surfactant concentration, as shown in Figure 5. At the CMC, micelles begin to form. The addition of surfactant beyond the CMC causes the equilibrium in Figure 4 to be driven further to the right. Therefore, concentrations above the CMC have a constant monomer concentration but an increasing number of micelles. micelles
monomers
Figure 5: Relationship between monomer and micelle concentration in solution. Below the CMC, the number of monomers in solution increases. Above the CMC, the monomers begin to aggregate into micelles. Therefore, the concentration of micelles starts to increase while the concentration of monomers remains constant. 3 Thermodynamic Analysis of Micelle Formation Using Surfactants Determination of CMC Being able to identify the exact concentration where micelles form is important to their effectiveness. Many techniques can be used to determine the CMC, including absorbance, conductivity, and Nuclear Magnetic Resonance (NMR). In all of these methods, the concentration is slowly adjusted to look for a critical phenomenon. When conductivity is used to find the CMC, the conductivity of the solution increases linearly with total surfactant concentration, as shown in Figure 6. However, the slope of the lines has an inflection point that indicates the CMC. Figure 7 shows how absorbance can be used to determine the CMC. In these experiments, a dye is added to the surfactant solution that is only solubilized in the hydrophobic region of a micelle. Therefore, a distinct color change designates the concentration where micelles form. Figure 6: Measurement of the CMC using conductivity. 1
2
3
4
5
6
7
8
9
10
11
12
13
14
15
Figure 7: Measurement of the CMC using UV‐Vis absorbance. A dye can be used that is solubilized into the hydrophobic region of the micelle. Therefore, a color change is observed only at concentrations above the CMC. 4 Thermodynamic Analysis of Micelle Formation Using Surfactants Thermodynamics of micelle formation The monomers aggregate above the CMC because it is energetically favorable. Although it requires energy to establish a new interface in the system (between the hydrophobic region of the surfactant and water), the savings in energy by minimizing the interactions between the hydrophobic tail and water molecules compensates once enough molecules aggregate together (i.e., above the CMC). The thermodynamics of this process include both enthalpic and entropic contributions to the Gibbs free energy. The standard Gibbs free energy change for this process can be related to the equilibrium constant, Kmic , ¢mic Go = ¡RT ln (Kmic ). (1) To relate the equilibrium constant to the concentrations, the degree of micelle ionization, ®, must be known. The degree of micelle ionization relates the total charge of a micelle (m) to the number of surfactant molecules (n) that aggregate to form the micelle. When the micelle contains a large number of surfactant molecules ( n > 50 ), then the standard Gibbs free energy for micellization can be approximated as ¢mic Go ¼ RT (2 ¡ ®) ln (CMC). (2) For the purposes of this experiment, the degree of micelle ionization can be approximated to follow ® = 0:0014T ¡ 0:202 where T is in Kelvin. The standard enthalpy change of the process can be obtained using the Gibbs‐Helmholtz relation ¡¢H
@¢G = T
. =
@T
T2
(3) The standard entropy change of the process can be obtained from the definition of the Gibbs free energy ¢mic Go = ¢mic H o ¡ T ¢mic S o . (4) For additional details on the thermodynamics of surfactant self‐assembly and interface partitioning, see Chapters 16 and 17 of “Intermolecular and Surface Forces” by Jacob N. Israelachvili. Objectives 1. Students will be introduced to key concepts in surfactant chemistry, including micelle formation and solubilization of hydrophobic molecules. 2. Students will be introduced to new laboratory techniques in determining the critical micelle concentration (CMC) for a common surfactant. 3. Students will evaluate the thermodynamic driving forces of molecular self‐assembly in these systems. 5 Thermodynamic Analysis of Micelle Formation Using Surfactants Operating Procedures Conductivity Meter Calibration 1.
2.
3.
4.
5.
Turn on meter with power key In Conductivity Mode, select calibration (F1‐key) Rinse probe with DI‐Water and pat dry with Kimwipe Place probe into calibration standard (Provided by TA) – Press Start (F3‐key) Wait for reading to stabilize a. If reading matches standard, select “accept” (F2‐key) b. If reading does not match standard, select “edit” (F3‐key) i. Adjust reading to match standard with number keys ii. Once complete select “done” (F2‐key) iii. Selected “Accept” (F3‐key) Conductivity Meter Sample Reading 1. Rinse probe with DI‐Water and pat dry with Kimwipe 2. Place probe in solution and press “measure” 3. Wait for reading to stabilize (display will stop flashing) Repeat process for all samples being collected 4. When complete, store probe in fresh DI Water UV‐Vis Wavelength Scan 1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
Select CarryWin‐UV icon on computer desktop Select “Scan” Icon Load Empty Cuvette into Instrument and hit the “Zero” Button Use “Set Up” Tab to create method Select Wavelength Range for your Scan (600 – 350 nm) Select Scan Speed (Hint – Slower Speeds result in more reliable data) In Baseline Tab, select “Baseline Correction” In Report Tab, select “Include x‐y pairs” Hit “OK” Load Pentane solution into cuvette and collect baseline by selecting “baseline” Key Load Dye Sample into Cuvette and collect Scan with “Start” Key What should you do if the detector reaches maximum with this sample? 12. Export Data by selecting “File” and “Save Data As” 13. Export as ASCii file (Spreadsheet) 6 Thermodynamic Analysis of Micelle Formation Using Surfactants UV‐Vis Single Wavelength Reading 1. Select CarryWin‐UV icon on computer desktop 2. Select “Simple Reading” Icon 3. In Set‐Up Tab select wavelengths to collect Multiple wavelengths can be collected by leaving a space. (i.e., 400 450) 4. Hit OK 5. Load DI Water Filled Cuvette into instrument and hit “Zero” Key 6. Load Sample filled Cuvette into instrument and collect Absorbance values with “Start” Key 7. Load Next Sample into instrument – Hit “Start” Key 8. Repeat for all samples 9. Export Data by selecting “File” and “Save Data As” 10. Export as Rich Text Format file (Word) Experimental Procedures A stock solution of the surfactant, sodium dodecyl sulfate (SDS), will be ready for your use at the start of the experiment. This standard solution will be prepared at an SDS concentration of 35 mM. You will need to prepare multiple concentrations of the surfactant solution to find the CMC at different temperatures using either UV‐Vis spectroscopy or conductivity. At least one CMC must be measured by each technique. The CMC value of SDS at room temperature is around 8 mM. You will need to select enough data points on both sides of the CMC to accurately determine the transition. The final volume of all solutions should be 15 mL to ease measurements with the conductivity meter. All calculations MUST BE DONE BEFORE COMING TO THE LAB! The solutions must be equilibrated for a minimum of 30 min at each temperature so be sure to plan your experiment wisely to collect the required data. After the CMC has been located at each temperature measured, determine the thermodynamic properties associated with micelle formation. Hazards The surfactant, PAN dye (1‐(2‐Pyridylazo)‐2‐naphthol) and pentane are considered irritants. Students should wear eye protection and gloves at all times and avoid skin contact. Pentane is highly flammable and should not be used near open flames. All solutions should be properly disposed of in an aqueous solution container. References Israelachvili, J. N. Intermolecular and Surface Forces; Academic Press: San Diego, CA, 1991. Katz, C.A., Calzola, Z.J., Mbindyo, K.N. Structure and Solvent Properties of Microemulsions; J. Chem. Educ. 85, 263, 2008. Salager, J.‐L. SURFACTANTS: Types and Uses; FIRP BOOKLET # E300‐A, Universidad de los Andes, 2002. 7
 0
0