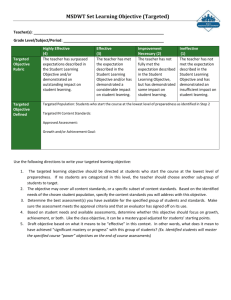
a sample chapter
advertisement

CHAPTER 38 Targeted Therapies to Treat Cancer Katherine L. Byar and M. Linda Workman http://evolve.elsevier.com/KeeHayes/pharmacology/ • Case Studies • Content Updates • Frequently Asked Questions • Student Review Activities • WebLinks • Additional Reference Material • NCLEX Examination Review Questions • • • • • • • Pharmacology Animations IV Therapy Checklists Medication Error Checklists Drug Calculation Problems Electronic Calculators Top 200 Drugs with Pronunciations References from the Textbook OBJECTIVES • Compare the mechanisms of action of targeted therapies for cancer with those of standard chemotherapy drugs. • Distinguish among the different types of targeted therapies for cancer treatment with regard to indications, possible side effects and adverse effects, route of administration, and nursing responsibilities. • Apply the nursing process related to the needs of clients receiving targeted therapies for cancer. • Develop a teaching plan for clients and their families about the use and side effects of targeted therapy for cancer. OUTLINE Objectives Key Terms Pathophysiology Normal Cell Growth Regulation Cancer Cell Loss of Growth Regulation Targeted Therapy Drugs Tyrosine kinases inhibitors Multikinase inhibitors Nursing Process: Tyrosine Kinases Inhibitors and Multikinase Inhibitors Epidermal growth factor receptor inhibitors Vascular endothelial growth factor/receptor inhibitors Nursing Process: Epidermal Growth Factor Receptor Inhibitors and Vascular Endothelial Growth Factor/ Receptor Inhibitors Proteasome inhibitors Angiogenesis inhibitors Monoclonal antibodies Nursing Process: Proteasome Inhibitors, Angiogenesis Inhibitors, and Monoclonal Antibodies Key Websites Critical Thinking Case Study NCLEX Study Questions KEY TERMS apoptosis, p. 000 cyclin-dependent kinase inhibitors, p. 000 cyclins, p. 000 cytotoxic, p. 000 gene expression, p. 000 gene overexpression, p. 000 mitosis, p. 000 monoclonal antibody, p. 000 proteasome, p. 000 signal transduction, p. 000 suppressor gene, p. 000 targeted therapy telomeric DNA, p. 000 transcription factors, p. 000 tyrosine kinase inhibitor, p. 000 tyrosine kinase, p. 000 38-1 38-2 CHAPTER 38 Targeted Therapies to Treat Cancer As described in Chapter 37 and in the Unit 12 opener, cancers develop from normal cells that have sustained gene damage. Cancer cells differ from normal cells in many ways, especially in their unrelenting growth and invasive spread (metastasis). This excessive growth serves no useful function, and causes death when normal tissues are invaded by cancer cells to the extent that vital organs can no longer perform their life-sustaining functions. Traditional chemotherapy is generalized, systemic, chemical, cytotoxic treatment that directly kills or severely damages cells. Mechanisms of action for different categories of chemotherapy drugs vary, but the overall outcome is the inhibition of cell division (mitosis) and cell death. Often, these drugs damage the cell DNA to prevent DNA replication and formation of new cancer cells (see Chapter 37). This form of treatment has improved cancer control and increased longterm survival. Although these drugs have some selectivity for exerting cytotoxic effects on cancer cells, the drugs also have a toxic impact on many normal cells. As a result, acute side effects of traditional combination chemotherapy are uncomfortable and can be life-threatening. Thus the cancer cell– killing effects of traditional chemotherapy is limited by the dosages and scheduling regimens needed to reduce toxic side effects on normal cells. Targeted therapy for cancer treatment differs from traditional cancer chemotherapy by taking advantage of biologic features, such as cellular receptors, enzymes, pathways, or other molecular proteins of cancer cells that either are not present or are present in much smaller quantities in normal cells. Thus, targeted therapies are more specific in their mechanisms and effects than traditional cancer chemotherapy agents. The National Cancer Institute’s (NCI) definition of targeted therapies is drugs or other substances that block the growth and spread of cancer by interfering with specific molecules involved in tumor growth and progression. It is important to remember that, unlike traditional cytotoxic chemotherapy (which exerts its effects by damaging the DNA of nearly any cell), targeted therapies require a specific molecular target as the recipient of their effects. The increased specificity of targeted therapies also means that more tests are required on cancer cells to determine whether or not a target is present in sufficient amounts to make the targeted therapy effective. For example, not all breast cancer cells have the molecular target for trastuzumab (Herceptin), nor do all leukemia cells have the molecular target for imatinib (Gleevec). Cancers that do not have sufficient quantities of the specific molecular target will not respond to targeted therapy. PATHOPHYSIOLOGY Normal Cell Growth Regulation A critical aspect of human health is the tight regulation of cell division, cell function, and cell death. For tissues composed of cells that can undergo mitosis, such as the bone marrow, cells perform their physiologic functions, age, and eventually die. (Mitosis is cell division in which one cell [the parent cell] divides and forms two new daughter cells that are identical to each other and to the original parent cell.) For example, optimum hematopoietic function requires that the majority of blood-producing cells in the marrow at any one time be at their peak functional level. This requires a balance between cell growth (cell division) and cell death within any tissue or organ. Too few functional cells within an organ lead to decreased organ efficiency. Too many functional cells within an organ reduce organ efficiency by using excessive energy and resources. Similarly, too many poorly functional cells within an organ drain resources and reduce overall organ function. Thus, ensuring optimal organ function requires that just the right amount of healthy cells is present in the organ workforce. The main force of this regulation is genetic control over cell division and cell death. Genetic Control over Cell Division As discussed in Chapter 37, the process of cell division (cellular reproduction, mitosis) involves entering the cell cycle. Normal cells capable of cell division enter the cell cycle only when needed to replace dead or poorly functional cells. As shown in Figure XII-1 at the beginning of Unit 12, which depicts the cell cycle, to enter the cell cycle and continue to progress through each phase of the cycle to mitosis, cell cycle checkpoints must be overcome. Initially, general factors determine whether or not the cell enters the cell cycle, including the following: • Whether or not the cell has retained the ability to undergo mitosis • Recognition of the need for more cells in the specific tissue where the cell resides • Adequate nutrition (especially protein, glucose, and oxygen) to support the cells that already exist as well as new cells • Adequate energy supplies or production • Adequate sources of substances needed in the synthesis of more membranes, more DNA, intracellular proteins, and organelles Signal transduction is a method of communication that allows events, conditions, and substances outside of the cell to influence the cell’s decision to divide, not to divide, or to perform its designated function. Some of the known external and internal signaling substances that promote cell division are enzymes, growth factors; adhesion proteins; steroid hormones; and cell-to-cell physical, chemical, and electrical interactions. Figure 38-1 shows a segment of a cell with one interconnecting signal transduction pathway that, when activated, leads to gene activation for those proteins that promote cell division. This pathway can be activated by growth factors binding to their receptors, the interaction of certain drugs with the cell plasma membrane, the presence of adhesion proteins, changes in ion movement (especially sodium and calcium), ligand binding, and other cell-to-cell interactions. When any of these conditions activate the signal transduction pathway, the amount of tyrosine kinases inside the cell is increased. Tyrosine kinases (TKs) are a family of enzymes that activate other substances by adding a phosphate group (PO4) to 38-3 CHAPTER 38 Targeted Therapies to Treat Cancer Growth factor Drug entry site Growth factor receptor Membrane “sticky zone” Tyrosine kinases Activation of transcription factors Ligand-associated receptor Cell adhesion and contact factors Cell plasma membrane Tyrosine kinase Activation of transcription factors Nucleus Increased production of cyclins; Decreased production of suppressor gene products More cell division DNA and genes FIGURE 38-1 Simplified sample signal transduction pathway promoting cell division. them, a process known as phosphorylation. There are many different TKs. Some are unique to the cell type; others may be present only in cancer cells that express a specific gene mutation. Regardless of how a pro-cell division signal transduction pathway is activated, the result is an increase in TK levels that propagate the signal by activating a variety of transcription factors within the pathway. Transcription factors for cell division are substances that enter the nucleus and signal the cell that mitosis is needed. Many different types of substances serve as transcription factors. The overall response is greater expression of oncogene products (cyclins) that promote cell division, and the reduced expression of suppressor gene products that inhibit cell division. When the cell responds to these mitotic signals indicating that cell division is needed and resources are adequate, the cell leaves G0 and enters the G1 phase of the cell cycle. Once the cell has entered G1, whether or not it can progress to the next phase is determined by the presence of specific cyclins (Figure 38-2). Cyclins are part of a family of proteins that, when active, stimulate the cell to move through the cell cycle. They are the products of oncogenes, and most are activated when a phosphorous group is added to the cyclin chemical structure. (Removal of a phosphorous molecule from a cyclin [dephosphorylation] inhibits its activity). Kinases that activate cyclins are cyclin-dependent kinases (CDKs). CDKs combine with cyclins to form complexes that initiate cell mechanisms to complete cell division. In normal cells, the oncogenes that produce cyclins are carefully regulated by suppressor gene products so that cell division only occurs when it is needed and to the degree that it is needed. Cyclin-A/CDK Cyclin-E/CDK Cyclin-B S G1 G2 Cyclin-B/Cdc2 M Cyclin-D/CDK Cyclin-A/Cdc2 Aurora kinase G0 FIGURE 38-2 Cyclin activity promoting progression through the cell cycle. The amount of cyclin and the type of cyclins, as well as which specific CDK is present in the cell during cell division, vary by the phase of the cycle. It is these differences in types of cyclins and CDKs that determines when or if a particular cell moves from one phase of the cycle to the next. Many 38-4 CHAPTER 38 Targeted Therapies to Treat Cancer Growth factor inhibitor Drug entry site Membrane “sticky zone” Ligand-associated receptor Cell adhesion and contact factors Cell membrane contacted on all sides with other cells Growth factor receptor Tyrosine kinases Activation of transcription factors Cell plasma membrane Tyrosine kinases Activation of transcription factors Nucleus Decreased production of cyclins; Increased production of suppressor gene products Less cell division DNA and genes FIGURE 38-3 External signals that inhibit the sample signal transduction pathway, resulting in greatly reduced cell division. different groups of cyclins have been identified, with the D group being most well understood. A common signal for entering and starting the cell cycle at G1 is the combining of a cyclin-D with the appropriate cyclindependent kinase (CDK), forming a cyclin-D/CDK complex (see Figure 38-2). Movement of the cell from G1 into the S, G2, and M phases of the cell cycle is regulated by the continued presence of pro-cell division transcription factors that promote DNA transcription and increased synthesis of specific pro-cell division cyclins and CDKs. Even when cell division is needed, the process is well controlled in normal cells. Proteins synthesized by suppressor genes determine how much oncogene expression is needed to allow cell division to occur but not lead to excessive cell division. Such control is exemplified by normal wound repair. For example, when a person falls and scrapes skin from the knee, the skin cells at the edge of the wound are signaled to divide and fill in the gap. When the wound area is closed, cell division normally stops. The person does not have uncontrolled cell division in this area to the extent that a large skin flap develops over and past the wound site. When cell division is not needed, external signals, such as growth factor inhibitors and the surrounding of a cell plasma membrane with other cells, send signals that are inhibitory to the pro–cell division signal transduction pathway (Figure 38-3). The result of this inhibition leads to low levels of TKs and reduced levels of pro-cell division transcription factors. Instead, suppressor gene activity is increased, resulting in production of more suppressor gene products that inhibit the synthesis of cyclins and CDKs by oncogenes. There are many suppressor genes, and, although all are present in every cell type, specific suppressor genes may be more active in selected types of tissues. For example, the BRCA1 suppressor gene appears most active in suppressing excessive cell division in breast, ovary, and genitourinary tract tissues. One of the most well-characterized suppressor genes is the Tp53 suppressor gene. Its gene product restricts the entry of cells into the cell cycle and restricts progression through the cell cycle for many cell types. Without suppressor gene products, oncogenes would be overexpressed continually, leading to uncontrolled and unneeded cell division. Internal cell conditions, such as poor cell nutrition and reduced energy stores, can trigger the activation of suppressor genes to disrupt the pro–cell division signal transduction pathway even when external conditions indicate a need for cell division (Figure 38-4). Thus, healthy and active suppressor genes guard against cell division when it is not in the body’s best interest. Genetic Control over Cell Death As some cells age, they begin to function less optimally. When a cell is damaged, reduced function occurs at an earlier cell age. In normal tissues that are capable of cell division, damaged cells and older cells respond to signals for apoptosis, which is programmed cell death, intended to ensure that tissues and organs contain only healthy and optimally functional cells. Apoptosis is under strict genetic control in normal tissues so that healthy functional cells do not self-destruct faster than they can be replaced, and that older or damaged cells unable to perform vital functions do not overpopulate a tissue and CHAPTER 38 Targeted Therapies to Treat Cancer Growth factor Drug entry site Growth factor receptor Membrane “sticky zone” Tyrosine kinases Activation of transcription factors Ligand-associated receptor 38-5 Cell adhesion and contact factors Cell plasma membrane Tyrosine kinases Activation of transcription factors Activation of suppressor gene products Nucleus Decreased production of cyclins; Increased production of suppressor gene products Less cell division DNA and genes FIGURE 38-4 Suppressor gene activity inhibiting the sample signal transduction pathway, resulting in greatly reduced cell division. reduce organ efficiency. Maintenance of optimally functional organs depends on a balance of cell division with apoptosis. The signals for apoptosis may come from the aging cell with the loss of telomeric DNA. Telomeric DNA is special DNA that caps the ends of each chromosome much like plastic tips cap the ends of a shoelace to prevent raveling (Figure 38-5). The function of telomeric DNA is to maintain the integrity of the double DNA strands within each chromosome. With each round of cell division, the telomeric DNA shortens. When the cell has undergone its lifespan’s worth of cell divisions, the telomeric DNA that capped the chromosomes is gone, allowing the DNA to unravel and fragment. These processes then trigger genetic and other intracellular signals for self-destruction through the action of autoenzymes, especially caspase 9. Activated caspase 9 leads to a cascade reaction for rapid activation of many more types of caspase enzymes. These enzymes degrade the cell’s internal structures and cause the plasma membrane to lyse and break the cell into small fragments that are removed from the body by white blood cells. Apoptosis is regulated by different gene products, including those of the Tp53 suppressor gene (Tp53 stands for tumor protein 53). When cells reach a certain age or experience DNA damage, the Tp53 gene is expressed and older or damaged cells either undergo apoptosis or are prohibited from progression through the cell cycle. Growth Regulation and Cancer Loss of Genetic Control of Cell Growth. As stated earlier, cancers develop from cells that were once normal. Normal cells become cancer cells when external or internal conditions Chromosome Telomeres Chromosome end segment 3,000–20,000 bases FIGURE 38-5 Chromosome with telomeric DNA and chromosome end segment with telomeric DNA. lead to gene damage. Although cancer can develop from a normal cell that has sustained damage to its oncogenes, most commonly, one or more suppressor genes are damaged and are no longer able to control oncogene expression. As a result of excessive oncogene expression, cyclins and CDKs are overproduced and cell division occurs when it is not needed. Excessive cell division from gene damage/mutations appears to be self-perpetuating, leading to further gene mutations that do the following: • Allow one or more specific pro-cell division signal transduction pathways to become more active • Allow greater expression of cancer cell membrane receptors that trigger pro-cell division signal transduction pathways • Increase or amplify the production of specific TKs and transcription factors Loss of Apoptosis. Mutations that occur in suppressor genes as a result of DNA damage inactivate the suppressor genes, 38-6 CHAPTER 38 Targeted Therapies to Treat Cancer preventing them from controlling oncogene activity. Some suppressor genes, such as the Tp53 gene, also regulate apoptosis. Inactivation of suppressor genes regulating apoptosis makes cancer cells unresponsive to apoptotic signals. These cancer cells are now resistant to natural cell death, a feature known as cellular immortality. The combined effects of lack of regulation for cell division and the loss of apoptosis result in cancer cells having no balance between cell division and apoptosis. This unbalanced condition favors continuous cancer cell division. TARGETED THERAPY DRUGS Targeted cancer therapies are a relatively new approach in cancer treatment. They work by interfering with cancer cell growth and division in different ways. These drugs are broadly known as signal transduction inhibitors. Targeted therapies generally inhibit cancer cell division by blocking a cancer membrane receptor, blocking tyrosine kinase activity, interfering with signal transduction, stimulating an immune system attack on cancer cells, or inducing the cell to undergo apoptosis. These agents may be used as monotherapy (as a single agent), in combination with traditional chemotherapy, and with radiotherapy. Some targeted therapies are oral and are self-administered at home where it is convenient for clients; others are administered parenterally. TABLE 38-1 The rapid identification of specific cancer cell targets in recent years has lead to increased development of targeted therapies, and the number of drugs approved for cancer targeted therapy has more than tripled in the last 5 years. Some are in relatively common use, and others are used less frequently or for rarer cancer types. Management of client issues related to targeted therapies is an evolving area of study. With many targeted therapies being new to the market, costs can be high and may not be covered by insurance. New targeted therapies are approved frequently. Older targeted therapy drugs may be newly approved for use in a different way or with a different cancer type. Expect to see indications for use of different targeted therapies in additional cancer types in the future. Because newly approved therapies have been less widely used and their adverse effects are not yet fully characterized, this chapter focuses on those therapies that have been in use for a year or more. There are several major classes of targeted cancer therapies, based on their most common mechanism of action (Table 38-1). Some targeted therapies have more than one action. Those discussed in this chapter include the following: tyrosine kinases inhibitors, multikinase inhibitors, epidermal growth factor receptor inhibitors, vascular endothelial growth factor/receptor inhibitors, proteasome inhibitors, angiogenesis inhibitors, and monoclonal antibodies. TARGETED THERAPIES FOR CANCER TREATMENT DRUG TYPE AND NAME ROUTE AND DOSAGE USES AND CONSIDERATIONS TYROSINE KINASE INHIBITORS (TKIs) dasatinib (Sprycel) For leukemia: A: PO 140 mg once daily with or without a meal. imatinib mesylate (Gleevec) For (leukemia; myelodysplastic syndrome): A: PO: recommended starting dose is 400 mg/d for chronic phase and 600 mg/d for accelerated phase or blast crisis. Dose increases to max of 800 mg (given as 400 mg twice daily); may be considered if tolerated for accelerated phase or blast crisis For GIST: A: PO: recommended starting dose is 400 mg/d C: PO: >3 years: 260 mg/m2/day PO given as a single daily dose, or the dose may be divided given once in the morning and once in the evening. Approved for chronic myelogenous leukemia (CML) and acute lymphocytic leukemia (ALL). Do not crush or cut tablets; if tablets are crushed or broken, handle with gloves. Administer antacids 2 hours before and 2 hours after each dose; Avoid H2 blockers and proton pump inhibitors (PPIs) Pregnancy category: D; PB: 95%; t½: 3 to 5 h Approved for Philadelphia chromosome–positive CML, metastatic malignant gastrointestinal stromal tumors (GIST), ALL, chronic eosinophilic leukemia (CEL), and myelodysplastic syndrome (MDS). Avoid pregnancy and breastfeeding. Take with a meal and a large glass of water to minimize gastric irritation. Pregnancy category: D; PB: 95%; t½: 18 to 40 h MULTIKINASE INHIBITORS (MKIs) sorafenib (Nexavar) For hepatocellular cancer and renal cell carcinoma: A: oral 400 mg twice daily Approved for hepatocellular cancer and advanced renal cell carcinoma. Administer orally without food (at least 1 hour before or 2 hours after a meal). Pregnancy Category: D; PB: 99.5%: t½: 25 to 48 h CHAPTER 38 Targeted Therapies to Treat Cancer TABLE 38-1 38-7 TARGETED THERAPIES FOR CANCER TREATMENT—cont’d DRUG TYPE AND NAME ROUTE AND DOSAGE MULTIKINASE INHIBITORS (MKIS)—cont’d sunitinib (Stutent) For gastrointestinal stromal tumors: A: PO 50 mg PO once daily on a schedule of 4 weeks on treatment then 2 weeks off. For renal cell carcinoma: A: PO 50 mg once daily on a schedule of 4 weeks on treatment then 2 weeks off. USES AND CONSIDERATIONS Approved for GIST and advanced renal cell carcinoma (RCC). Sunitinib can only be obtained directly from McKesson Specialty. Administer orally with or without food. Pregnancy Category: D; PB: 90%; t½ of sunitinib is 40 to 60 h, and that of its major metabolite is 80 to 110 h. EPIDERMAL GROWTH FACTOR/RECEPTOR (EGRF) INHIBITORS cetuximab (Erbitux) For colorectal and head and neck cancer: A: IV: initial infusion dose is 400 mg/m2 IV over 2 hours (with a maximum rate of 5 mL/min). Then continue weekly infusions of 250 mg/m2 IV over 60 minutes (with a maximum rate of 5 mL/min). erlotinib (Tarceva) For non–small cell lung cancer: A: PO: 150 mg once daily For pancreatic cancer: A: PO: 100 mg once daily in combination with gemcitabine (1000 mg/m2 IV: Cycle 1—Days 1, 8, 15, 22, 29, 36, and 43 of an 8-week cycle; Cycle 2 and subsequent cycles—Days 1, 8, and 15 of a 4-week cycle) For NSCLC: A: PO: 250 mg once daily Gefitinib (Iressa) panitumumab (Vectibix) For colorectal cancer A: IV 6 mg/kg administered over 60 minutes (administer doses >1000 mg over 90 minutes) given once every 2 weeks. trastuzumab (Herceptin) For breast cancer: A: IV 4 mg/kg infused over 90 minutes on week 1. If the initial infusion is tolerated, starting at week 2 administer 2 mg/kg IV over at least 30 minutes once weekly Approved for EGFR-expressing colorectal and head and neck cancers. Administer only as an IV infusion with an infusion controller, never as a bolus or as an IV push. Administer with low-protein– binding 0.22-micron filter. Do not shake or further dilute vial. Do not mix with other drugs. Premedicate the client with an H1 antagonist (e.g., diphenhydramine 50 mg IV) 30 to 60 minutes before the first dose of cetuximab. Pregnancy Category: C; t½: 41 to 214 h Approved for non–small cell lung cancer (NSCLC) and pancreatic cancer. Give the drug on an empty stomach, 1 hour before or 2 hours after ingestion of food (administering with food increases the risk for side effects). Administer at the same time each day between meals. Pregnancy Category: D; PB: 93%; t½: 36 h Approved for NSCLC. Administer tablet with water without regard for meals. Pregnancy Category: D; PB: 90%; t½: 48 h Approved for EGFR-expressing metastatic colorectal cancer. Administer only as an IV infusion with an IV controller and never as a bolus or an IV push injection. Administer with low-protein– binding 0.2-micron or 0.22-micron in-line filter. Flush line with 0.9% sodium chloride injection before and after administration. Do not mix drug with other drugs or other infusions. Infuse over 60 minutes through a peripheral line or indwelling catheter (doses over 1,000 mg should be infused over 90 minutes. Pregnancy Category: C; t½: 7.5 d Approved for breast cancer that overexpresses EGFR2 (HER2/neu receptor). Do not administer IV push or as a bolus. Monitor clients for infusion-related reactions such as fever or chills, respiratory distress, or severe hypersensitivity reactions. Interrupt the infusion for clients who experience dyspnea or significant hypotension. Pregnancy Category: D; t½: 25 d Continued 38-8 CHAPTER 38 Targeted Therapies to Treat Cancer TABLE 38-1 TARGETED THERAPIES FOR CANCER TREATMENT—cont’d DRUG TYPE AND NAME ROUTE AND DOSAGE USES AND CONSIDERATIONS VASCULAR ENDOTHELIAL GROWTH FACTOR/RECEPTOR (VEGRF) INHIBITORS For metastatic breast cancer: A: IV 10 mg/kg on days 1 and 15 in combination with paclitaxel (90 mg/m2 IV on days 1, 8, and 15) every 28 days; or 7.5 mg/kg to 15 mg/kg on day 1 in combination with docetaxel (100 mg/m2 IV), repeated every 3 weeks. For colorectal cancer: A: IV 5 or 10 mg/kg over 60 or 90 minutes every 14 days in combination with 5-fluorouracil– based chemotherapy. For non–small cell lung cancer: A: IV 15 mg/kg over 60 or 90 minutes every 3 weeks in combination with carboplatin and paclitaxel. For metastatic renal cell carcinoma: A: IV 10 mg/kg IV every 2 weeks in combination with interferon alfa (9 million units subcutaneously 3 times/week up to 52 weeks) PROTEASOME INHIBITORS Approved for metastatic breast cancer, colorectal cancer, NSCLC, and RCC. Administer as an IV infusion and never as an IV push or bolus. Mix infusions with only 0.9% sodium chloride and never with dextrose solutions. Discard any unused portion left in the vial, as the product contains no preservatives. Pregnancy Category: C; t½: 11 to 50 d bortezomib (Velcade) Approved for mantle cell lymphoma (MCL) and multiple myeloma. At least 72 hours should elapse between consecutive doses of bortezomib. Administered as an intravenous injection bolus over 3 to 5 seconds. Reconstitute for injection each vial with 3.5 mL of 0.9% sodium chloride for a final concentration of 1 mg/mL. The final product should be a clear, colorless, solution. Pregnancy Category: D; PB: 83%: t½: 9 to 15 h bevacizumab (Avastin) For mantle cell lymphoma: 1.3 mg/m2 IV bolus on days 1, 4, 8, and 11 followed by a 10-day rest period (days 12 to 21). For extended therapy of more than 8 cycles, administer bortezomib on the standard 3-week cycle or on days 1, 8, 15, and 22 followed by a 13-day rest period (days 23 to 35). For multiple myeloma: A: IV Treatment is administered for nine 6-week cycles. In cycles 1 to 4, 1.3 mg/m2/dose IV bolus is given on days 1, 4, 8, and 11 followed by a 10-day rest period (days 12 to 21) and again on days 22, 25, 29, and 32 followed by a 10-day rest period (days 33 to 42) in combination with melphalan (9 mg/m2/day on days 1 to 4) and prednisone (60 mg/m2/day on days 1 to 4); this 6-week cycle is considered one course. In cycles 5 to 9, bortezomib 1.3 mg/m2/dose IV bolus is given on days 1, 8, 22, and 29 in combination with melphalan (9 mg/m2/day, days 1 to 4) and prednisone (60 mg/m2/day, days 1 to 4); this 6-week cycle is considered one course. CHAPTER 38 Targeted Therapies to Treat Cancer TABLE 38-1 38-9 TARGETED THERAPIES FOR CANCER TREATMENT—cont’d DRUG TYPE AND NAME ROUTE AND DOSAGE USES AND CONSIDERATIONS ANGIOGENESIS INHIBITORS temsirolimus (Torisel) For renal cell carcinoma: A: IV 25 mg IV given over 30 to 60 minutes once a week Approved for advanced RCC. Premedicate client with diphenhydramine 25 to 50 mg IV, or a similar antihistamine, approximately 30 minutes before the start of each temsirolimus dose. Two dilutions are required before IV infusion. Use only the supplied diluent for the initial dilution. Use an in-line polyethersulfone filter with a pore size of not greater than 5 microns and an infusion pump. Administer only through polyethylene-lined administration sets. Administer dose over 30 to 60 minutes. Pregnancy Category: D; t½: 17 to 20 d MONOCLONAL ANTIBODIES alemtuzumab (Campath) gemtuzumab ozogamicin (Mylotarg) For chronic lymphocytic leukemia: A: IV Start the drug at 3 mg IV over 2 hours once daily. When the 3-mg dose is tolerated (infusion reactions are grade 2 or less), escalate the daily dose to 10 mg IV once daily and continue until infusion reactions are grade 2 or less, then start the maintenance dose of 30 mg IV given 3 times weekly on alternate days (e.g., Monday, Wednesday, Friday); the total therapy duration including dose escalation is 12 weeks. For acute myelogenous leukemia: A: IV 9 mg/m2 as a 2-hour infusion on days 1 and 15. C: IV 4 to 9 mg/m2/course for up to 3 courses. Approved for chronic lymphocytic leukemia (CLL). Administer as an IV infusion over 2 hours and never as an IV push or bolus dose. Monitor client closely for serious, sometimes fatal, infusion-related reactions. Premedicate the client 30 minutes before the first dose, with any dose escalation, and as needed with diphenhydramine (50 mg) and acetaminophen (500 to 1,000 mg). Withhold alemtuzumab administration if a grade 3 or 4 infusion reaction occurs Pregnancy Category: C; t½: 12 d Approved for CD33-positive acute myelogenous leukemia (AML). Do not administer to clients with known murine (mouse) hypersensitivity. Premedicate the client 30 minutes before administration with oral diphenhydramine (50 mg) and acetaminophen 650 to 1,000 mg (adults). Do not mix gemtuzumab ozogamicin with other drugs, electrolyte solutions, or 5% dextrose. Protect the drug against direct and indirect sunlight and unshielded fluorescent light during preparation and administration of the infusion. Administer as an IV infusion over 2 hours and never as an IV push or bolus dose. Monitor vital signs at least every half hour during the infusion and 4 hours following the infusion. Pregnancy Category: D; t½: 60 h Continued 38-10 CHAPTER 38 Targeted Therapies to Treat Cancer TABLE 38-1 TARGETED THERAPIES FOR CANCER TREATMENT—cont’d DRUG TYPE AND NAME ROUTE AND DOSAGE USES AND CONSIDERATIONS MONOCLONAL ANTIBODIES—cont’d ibritumomab tiuxetin (Zevalin) rituximab (Rituxan) I131 tositumomab (Bexxar) For non-Hodgkin’s lymphoma: A: IV Step 1 Rituximab 250 mg/m2 IV is administered first (see Rituximab monograph). Within 4 hours of completing the rituximab infusion, give 5 mCi (1.6 mg total antibody dose) of In-111 ibritumomab tiuxetan IV over 10 minutes. Step 2 Seven to nine days following step 1, give a second dose of Rituximab 250 mg/m2 IV. Within 4 hours of completing the rituximab infusion, give Y-90 ibritumomab tiuxetan 0.4 mCi/kg (14.8 Mbq/kg) or 0.3 mCi/kg (11.1 MBq/kg) IV over 10 minutes. For non-Hodgkin’s lymphoma (NHL): A: IV 375 mg/m2 once weekly for 4 doses; may be retreated for an additional 4 doses. In combination with CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone), 375 mg/m2 IV on day 1 of each cycle of chemotherapy for up to 8 cycles. For non-Hodgkin’s lymphoma: A: IV Step 1: dosimetric step On day 0, 450 mg unlabeled tositumomab IV over 1 hour followed by iodine I-131 tositumomab 5 mCi (35 mg tositumomab) IV over 20 minutes. Total body gamma counts using a gamma camera are obtained on day 0; day 2, 3, or 4; and on day 6 or 7. Using these counts, perform calculations based on standard internal radiation dosimetry methods to determine the patient-specific activity (in millicuries) of radiolabeled tositumomab required to deliver a maximum tolerated dose of 75 cGy total-body dose. Step 2: therapeutic step 450 mg unlabeled tositumomab IV over 1 hour followed by the patient-specific activity (in millicuries) iodine I-131 labeled to 35 mg of tositumomab IV between day 7 and day 14. Approved for B-cell NHL. Use appropriate precautions for handling, preparing, and administering radiopharmaceuticals. Do not administer In-111 ibritumomab tiuxetan and Y-90 ibritumomab tiuxetan unless the rituximab predose has been administered. Do not give Y-90 ibritumomab tiuxetan to clients with a platelet count <100,000/mm3). Pregnancy Category: D; t½: 30 h Approved for CD20-positive B-cell NHL. Premedicate the client 30 minutes before administration with diphenhydramine (50 mg), acetaminophen 650 to 1,000 mg (adults), and possibly corticosteroids. Do not administer as an IV push or bolus. Administer first IV infusion at an initial rate of 50 mg/h. If no hypersensitivity or infusion-related events occur, increase infusion rate in 50 mg/h increments every 30 minutes, to a maximum of 400 mg/h. Monitor clients closely during infusion for profound hypotension, dyspnea, and any cardiovascular infusion–related reactions. Do not mix rituximab solution with other drugs. Pregnancy Category: C; t½: 31.5 to 52.6 h Approved for relapsed or refractory CD20positive, follicular NHL. Administer thyroid protection agents at least 24 hours before step 1 and step 2. Do not administer to clients with known murine (mouse) hypersensitivity. Premedicate the client 30 minutes before administration with oral diphenhydramine (50 mg) and acetaminophen 650 to 1,000 mg (adults). Use appropriate precautions for handling, preparing, and administering radiopharmaceuticals. Administer through an IV tubing set with a 0.22-micron in-line filter. The same IV tubing set and filter must be used throughout the entire dosimetric or therapeutic step. Administer iodine I-131 tositumomab infusion over 60 minutes. After infusion of the iodine I-131 tositumomab is complete, close the stopcock to the syringe. Flush the extension set and the secondary IV infusion set with 0.9% sodium chloride for injection. Pregnancy Category: ×; t½: 8.5 d CHAPTER 38 Targeted Therapies to Treat Cancer Tyrosine Kinase Inhibitors Several types of targeted therapies have as an outcome the inhibition of tyrosine kinases using a variety of mechanisms. Specific drugs that cause this action as their main mechanisms are referred to as tyrosine kinase inhibitors (TKIs). The most common TKIs currently prescribed are imatinib mesylate (Gleevec) and dasatinib (Sprycel). Table 38-1 lists the TKIs and their dosages, routes, uses, and considerations. Prototype Drug Chart 38-1 lists specific drug information about the TKI imatinib (Gleevec). Newly approved TKIs include pazopanib (Votrient), which is used in advanced renal cell carcinoma, and lapatinib (Tykerb), which is used with other drugs in the treatment of HER2 positive breast cancer. TKIs are chemicals that exert their effects by directly inhibiting only specific types of tyrosine kinases. The types they inhibit are the SRC kinases, which are present in many cells (normal and cancerous) and a very specific type, the BCR-ABL tyrosine kinase. As shown in Figure 38-1, receptors on the cell membrane can activate tyrosine kinases, which then turn on signal transduction pathways promoting cell division. The TKIs are nonreceptor kinase inhibitors because they do not bind to the receptors on the plasma membrane. Instead they work directly on the tyrosine kinase molecule. The SRC kinases are involved in the activation of signal transduction pathways promoting cell division in many cell types. The BCR-ABL tyrosine kinase is present only in cancer cells that have a specific gene mutation resulting from a chromosome structural rearrangement that forms a “Philadelphia chromosome.” This mutation is highly present in chronic myelogenous leukemia (CML) cells and has recently been found in some other cancer cell types. When activated and expressed, BCR-ABL tyrosine kinase turns on a strong pro-cell division signal transduction pathway that leads to proliferation of cancer cells. The TKIs prevent activation of tyrosine kinases, which then inhibits further activation of the signal transduction pathway and stops the proliferation of cancer cells. This action can control the disease but cannot alone eradicate it. Dasatinib Dasatinib (Sprycel) is approved for Philadelphia chromosome– positive chronic myeloid leukemia (CML) and acute lymphocytic leukemia (ALL). Pharmacokinetics. Dasatinib is an oral drug that is readily absorbed from the GI tract and can be taken with or without food. Antacids slow the absorption. The drug is extensively metabolized in the liver by the cytochrome P450 (CYP) 3A4 isoenzyme. Pharmacodynamics. Dasatinib most specifically targets BCR-ABL tyrosine kinase, found in CML cells. The drug blocks the ATP (adenosine triphosphase) binding site on the tyrosine kinase so that it does not become activated. This lack of BCR-ABL tyrosine kinase activation inhibits further activation of the signal transduction pathway and stops the proliferation of cancer cells. The drug has the greatest effects on cancer cells expressing the Philadelphia chromosome genetic mutation. 38-11 Side Effects and Adverse Reactions. Dasatinib often causes electrolyte imbalances, especially low serum levels of phosphorus and calcium. These imbalances may require phosphorus and calcium replacement. ECG abnormalities, especially prolonged QT interval, have been seen in clients taking dasatinib. This drug should be used cautiously in anyone at risk for long QT, such as those who have hypokalemia or hypomagnesemia and those with a family history of long QT syndrome. Fluid retention has also been seen in up to 50% of clients. Hematologic side effects of myelosuppression with anemia, thrombocytopenia, and neutropenia are relatively common. Drug Interactions. Dasatinib activity is affected by CYP3A4 enzyme inhibitors, which decrease dasatinib metabolism, resulting in an increased serum concentration and increased risk for toxicity. Other drugs that strongly increase the serum concentration of dasatinib include atazanavir, clarithromycin, indinavir, itraconazole, ketoconazole, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, triazolobenzodiazepines, and voriconazole. HERBAL ALERT 38-1 Grapefruit juice increases the serum concentration and toxicity of TKIs, EGFRIs, MKIs, proteasome inhibitors, and angiogenesis inhibitors. St. John’s wort reduces the serum concentration and the effectiveness of drugs from these classes. Drugs that enhance the activity of the CYP3A4 enzyme decrease dasatinib serum levels and reduce its effectiveness. Such drugs include aminoglutethimide, barbiturates, carbamazepine, dexamethasone, grisefulvin, modafinil, nafcillin, phenytoin, primidone, rifabutin, and rifampin. Other drugs that appear to reduce the effectiveness of dasatinib include antacids, H2 histamine blockers, and proton pump inhibitors. Dasatinib may interfere with the metabolism of other drugs that use the CYP3A4 enzyme system, such as alfentanil, astemizole, terfenadine, cisapride, cyclosporine, fentanyl, pimozide, quinidine, sirolimus, tacrolimus, and ergot alkaloids. Multikinase Inhibitors The multikinase inhibitors (MKIs) are chemicals that directly inhibit the activity of specific kinases in cancer cells and in cancer cell vasculature. (Recall that kinases are enzymes that activate other proteins, including those that activate signal transduction pathways promoting cancer cell division.) Table 38-1 lists the MKIs and their dosages, routes, uses, and considerations. Sorafenib Sorafenib (Nexavar) is a multikinase inhibitor that specifically targets serine/threonine and receptor tyrosine kinases, which are activated as a result of gene mutations and are most commonly found in pancreatic cancer, colon cancer, and non–small cell lung cancer. In addition, the drug may be used [AQ2] 38-12 CHAPTER 38 Targeted Therapies to Treat Cancer PROTOTYPE DRUG CHART 38-1 Imatinib Mesylate Drug Class Tyrosine kinase inhibitor (TKI) Trade name: Gleevec Pregnancy Category: D Dosage Leukemias; myelodysplastic syndrome: A: PO: Recommended starting dose is 400 mg/d for chronic phase and 600 mg/d for accelerated phase or blast crisis. Dose increases to max of 800 mg (given as 400 mg twice daily); may be considered if tolerated for accelerated phase or blast crisis. C: PO: Children >3 years: 260 mg/m2/day PO given as a single daily dose, or the dose may be divided given once in the morning and once in the evening. For GIST: A: PO: Recommended starting dose is 400 mg/d. Contraindications Clients who may become pregnant, are pregnant, or are breastfeeding; have severe heart failure or severe kidney disease; have moderate to severe liver disease and are also taking another hepatotoxic chemotherapy drug(s); or have severe neutropenia, anemia, or thrombocytopenia Drug-Lab-Food Interactions Drug: Any agent that inhibits cytochrome P450 (CYP) 3A4 may decrease the metabolism of imatinib and increase imatinib concentrations leading to an increased incidence of adverse reactions (examples include ketoconazole, amiodarone, antiretroviral protease inhibitors, dalfopristin quinupristin, mifepristone). Any drug that induces cytochrome P450 (CYP) 3A4 may increase the metabolism of imatinib and decrease imatinib concentrations and clinical effects (examples include barbiturates, bosentan, carbamazepine, dexamethasone, phenobarbital, rifabutin, and rifapentine). Any drug that is metabolized by cytochrome P450 (CYP) 2D6 may have higher than normal blood levels of the drug (examples include amoxapine, atomoxetine, carvedilol, metoprolol, propranolol, timolol, clozapine, codeine, cyclobenzaprine, darifenacin, fenfluramine, dexfenfluramine, dextromethorphan, flecainide, haloperidol, hydrocodone, maprotiline, methadone, methamphetamine, mexiletine, morphine, oxycodone, paroxetine, perphenazine, propafenone, risperidone, thioridazine, tramadol, trazodone, tricyclic antidepressants, and venlafaxine). St. John’s wort decreases imatinib blood levels and may decrease its effectiveness. Taking imatinib with acetaminophen increases the risk for acetaminophen poisoning. Taking imatinib with NSAIDs increases hypertension and disruption of platelet aggregation. Taking imatinib with warfarin increases the risk for severe bleeding. Lab: May alter liver enzyme levels and bilirubin levels Food: Grapefruit juice increases imatinib plasma concentrations Pharmacokinetics Absorption: Bioavailability is 98%. Steady-state levels achieved in 1 to 2 days. Distribution: 95% PB with extensive distribution Metabolism: Metabolized mainly in the liver by the cytochrome P450 (CYP) 3A4 and CYP2D6 isoenzymes; t½ of drug is 18 hours, of its major metabolite 40 hours. Excretion: Mainly in the feces Pharmacodynamics Oral absorption is fast and not affected by meal timing. Therapeutic Effects/Uses Approved for chronic myelogenous leukemia (CML), metastatic malignant gastrointestinal stromal tumors (GIST), acute lymphocytic leukemia (ALL), chronic eosinophilic leukemia (CEL), gastrointestinal stromal tumors (GIST), and myelodysplastic syndrome (MDS) Mode of Action: Imatinib competitively inhibits the ATP binding site on tyrosine kinases specific for Abl, PDGF, SCF, and c-Kit, preventing the activation of those specific tyrosine kinases. This action inhibits platelet-derived growth factor (PDGF) and stem cell factor (SCF) mediated cellular events. This leads to reduced cancer cell division and induction of apoptosis. This action results in blockage of downstream EGFR-TK mediated signal transduction pathways, cell cycle arrest, and inhibition of angiogenesis. Side Effects Decreased appetite, diarrhea, difficulty sleeping, headache, heartburn, joint pain, muscle cramps or pain, nausea, and upset stomach. Adverse Reactions Allergic reactions, severe neutropenia increasing the risk for infection, severe thrombocytopenia increasing the risk for excessive bleeding/hemorrhage, liver impairment, kidney impairment. A, adult; PO, by mouth; PB, protein-binding; >, greater than; t½, half-life CHAPTER 38 Targeted Therapies to Treat Cancer in those hepatocellular carcinomas and renal cell carcinomas that overexpress the target. Pharmacokinetics. Sorafenib (Nexavar) is an oral drug whose absorption and bioavailability are inhibited when taken with a high-fat meal. The drug is metabolized in liver, mainly by the CYP3A4 enzyme system. It is 99.5% proteinbound and reaches peak plasma level in about 3 hours. The drug is eliminated in the feces and urine, with a half-life of 25 to 48 hours. Pharmacodynamics. Sofafenib inhibits Raf kinase, vascular endothelial growth factor (VEGF) receptors VEGFR-2 and VEGFR-3, platelet-derived growth factor receptor (PDGFR), Kit receptor tyrosine kinase (KIT), fms-like tyrosine kinase 3 (FLT-3), and RET. When these tyrosine kinase receptors are activated by cytokines or growth factors, a protein-kinase–mediated cascade starts, leading to uncontrolled cellular proliferation. The inhibition of these signaling pathways by sorafenib results in decreased cancer cellular proliferation. In addition, sorafenib specifically inhibits two VEGF receptors (VEGFR-2 and VEGFR-3), which are key receptor tyrosine kinases involved in angiogenesis. This action results in reduced blood vessel formation in cancer cells. This anticancer activity is present only when the drug is present, and cancer growth returns when therapy is stopped. Side Effects and Adverse Reactions. Sorafenib has many common side effects. Hypertension is very common and can occur within the first 6 weeks of therapy. Skin side effects include alopecia, pruritus, dry skin, exfoliative dermatitis, acne, flushing, and palmar-plantar erythrodysesthesia (handfoot syndrome), which also manifest within the first 6 weeks. Other side effects include weight loss, nausea/vomiting, diarrhea, anorexia, constipation, abdominal pain, mucositis, dyspepsia, dysphagia, and mild neutropenia and thrombocytopenia. More severe effects are possible, including pancreatitis, erectile dysfunction, and myocardial ischemia. Drug Interactions. Sorafenib levels are not increased by the presence of other drugs, even those that inhibit the enzyme that metabolizes sorefenib. However, drugs that induce metabolizing enzyme activity can reduce the blood levels and effectiveness of sorafenib. These include rifampin, phenytoin, phenobarbital, carbamazepine, dexamethasone, rifabutin, and rifapentint. Sorafenib can increase the blood levels of rapaglinide, amiodarone, ibuprofen, loperamide, irinotecan, propfol, and warfarin. Sunitinib Sunitinib (Sutent) inhibits more than 80 tyrosine kinases. This inhibition results in regression of tumor growth, especially in clear cell renal cell carcinoma (RCC) and gastrointestinal stromal tumors. Pharmacokinetics. Sunitinib is an oral drug that is well absorbed with or without meals. It is 90% protein-bound and reaches peak plasma levels in 6 to 12 hours. The drug is metabolized in liver, mainly by the CYP3A4 enzyme system, and is eliminated in the feces. The half-life of sunitinib is 40 38-13 to 60 hours, and its major active metabolite has a half-life of 80 to 110 hours. Pharmacodynamics. The action of sunitinib is the inhibition of many receptor tyrosine kinases (RTKs), including those of platelet-derived growth factor receptors (PDGFR), vascular endothelial growth factor receptors (VEGFR1, VEGFR2, VEGFR3), and a variety of others. This inhibition results in decreased cancer cell proliferation and in reduced blood vessel formation in cancer cells. This drug also increases adverse effects on normal tissues, especially hair and skin. Side Effects and Adverse Reactions. Sunitinib side effects and adverse reactions are more widespread as a result of the number of different types of kinases this drug inhibits. Cardiovascular effects include hypertension, peripheral edema, left ventricular dysfunction, prolonged QT interval, and venous thromboembolism. GI effects include nausea/vomiting, diarrhea, stomatitis, dyspepsia, anorexia, constipation, abdominal pain, glossodynia, and flatulence. Neuromuscular effects include fatigue, asthenia, headache, dizziness, peripheral neuropathy, mild arthralgia, limb pain, myalgia, and back pain. Liver impairment may occur with elevated liver enzyme levels and jaundice of the skin and sclerae. Common integumentary changes include lightening of the hair, rash, dry skin, and palmar-plantar erythrodysesthesia (hand-foot syndrome). Endocrine changes may include hypothyroidism and adrenal insufficiency. Hematologic changes may include mild neutropenia and thrombocytopenia. Respiratory-associated effects may include mild dyspnea and cough. Drug Interactions. Sunitinib blood levels and activity are increased by drugs that inhibit the CYP3A4 enzyme levels, including atazanavir, clarithromycin, ketoconazole, itraconazole, indinavir, nefazondone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole, diltiazem, and verapamil. When these drugs are used during sunitinib therapy, side effects of sunitinib are more common and more severe. Drugs that increase sunitinib elimination and reduce its effectiveness include rifampin, phenytoin, phenobarbital, carbamazepine, dexamethasone, rifabutin, and rifapentin. Epidermal Growth Factor/Receptor Inhibitors The epidermal growth factor/receptor inhibitors (EGFRIs) include erlotinib, gefitinib, panitumumab, cetuximab, and trastuzumab. Most EGFRIs ultimately also inhibit tyrosine kinase, but they do it more indirectly than the TKIs. As shown in Figure 38-1, the growth factor receptors on the cell membrane can activate tyrosine kinases, which then turn on signal transduction pathways promoting cell division. The EGFRIs bind to different areas of the epidermal growth factor receptor, blocking its activity so that it cannot activate tyrosine kinase. As a result, the downstream signal transduction pathway for promotion of cell division is inhibited and cell proliferation is severely limited. Table 38-1 lists the EGFRIs and their dosages, routes, uses, and considerations. Prototype Drug Chart 38-2 lists the specific drug information for erlotinib (Tarceva). 38-14 CHAPTER 38 Targeted Therapies to Treat Cancer NURSING PROCESS Tyrosine Kinase Inhibitors and Multikinase Inhibitors for Cancer Treatment Assessment baseline physical condition of the client before initiating targeted therapy regimen and during the treatment period. Assess laboratory studies, including CBC with differential, hepatic and renal studies, electrolytes, and urinalysis at the beginning of therapy and at specified time intervals (ranging from weekly to monthly) during therapy. Conduct a detailed medication history, including a list of all concurrent medications, including prescriptions, over-the-counter medicines, antacids, dietary supplements, vitamins, and herbal supplements to avoid drugdrug interactions. Assess client and family knowledge related to therapeutic regimen. Assess Nursing Diagnoses Knowledge, deficient, related to targeted therapy regimen Skin integrity, impaired, risk for, related to dermatologic effects and toxicities of therapy Infection, risk for, related to bone marrow suppression Fluid volume, deficient, risk for, related to GI effects of therapy Electrolyte imbalance, risk for, related to actions of targeted therapy Planning Client and family will verbalize understanding of targeted therapy as part of an anticancer treatment regimen. and family will demonstrate understanding of the importance of reporting targeted therapy–related side effects and adverse reactions. Client and family will verbalize strategies to minimize risks related to targeted therapy–related side effects. Client’s side effects will be managed to a level that the client can tolerate and are not life-threatening. Client will remain infection-free. Client will have fluid balance and electrolytes within expected normal ranges. Client Nursing Interventions Follow institution guidelines for safe handling, preparing, administering, and dispensing of targeted therapy agents. prescribed premedications according to established protocols for specific targeted therapies. Monitor complete blood cell count with differential and platelet count at baseline, once weekly for the first month, every other week for the second month, and at least every 2 to 3 months thereafter. Monitor electrolytes regularly during treatment and for 8 weeks after treatment has stopped, especially phosphorus Administer and calcium levels, because low levels may require replacement. Monitor liver function and renal function tests at baseline and at least once monthly during therapy. Monitor blood pressure at baseline and at least weekly during therapy. Monitor ejection fraction at baseline and periodically during therapy. Monitor thyroid function if client experiences symptoms of hypothyroidism (cold intolerance, hair loss, decreased memory or mental alertness, constipation, bradycardia). Discontinue therapy prior to elective surgery. For clients receiving sunitinib, monitor adrenal function in clients exposed to stress (e.g., surgery, shock, trauma, or infections). Monitor hands and feet for signs of palmar-plantar erythrodysesthesia (redness, pain, swelling or blisters). Sorafenib potentiates the activity of warfarin. Check the client’s INR weekly. Client Teaching alcohol and nonessential drugs that are cleared by the liver or have liver-toxic effects (e.g., acetaminophen). Report symptoms of adverse effects or severe side effects promptly, especially fever, chills, persistent sore throat, swelling, weight gain, or increasing shortness of breath). Report symptoms of bleeding immediately, including black stools, vomit that looks like coffee grounds, or easy bleeding/bruising. Report symptoms of liver impairment immediately, including stomach/abdominal pain, yellowing eyes or skin, dark urine, or unusual fatigue. Remind women with childbearing potential to avoid pregnancy throughout treatment and for up to 12 months after treatment is completed. Advise breastfeeding women to stop breastfeeding during and for 60 days after therapy. Teach clients that drinking grapefruit juice can increase the blood levels of most targeted therapies and make side effects or adverse effects worse. Instruct them to avoid this food while receiving treatment with targeted therapies. Teach clients to avoid using the herbal St. John Wort while receiving treatment with targeted therapies because this agent decreases the effectiveness of most of these drugs. Teach clients to take imatinib with food or with at least 240 mL of water to reduce gastric irritation. Teach clients to weigh themselves daily and report a weight gain of more than 2 pounds in 1 day or 4 pounds in 1 week to the health care provider. Teach clients to check hands and feet daily for signs of palmar-plantar erythrodysesthesia (redness, pain, swelling, or blisters) and to report these symptoms when they appear. Remind clients receiving sorafenib who also take warfarin to keep all appointments for INR testing. [AQ1] Avoid CHAPTER 38 Targeted Therapies to Treat Cancer 38-15 PROTOTYPE DRUG CHART 38-2 Erlotinib Drug Class Dosage Epidermal growth factor/receptor inhibitor (EGFRI) For non–small cell lung cancer: Trade name: Tarceva A: PO: 150 mg once daily Pregnancy Category: D For pancreatic cancer: A: PO: 100 mg once daily in combination with gemcitabine (1,000 mg/m2 IV Cycle 1—Days 1, 8, 15, 22, 29, 36, and 43 of an 8-week cycle; Cycle 2 and subsequent cycles—Days 1, 8, and 15 of a 4-week cycle) Contraindications Clients who may become pregnant, are pregnant, or are breastfeeding; have preexisting respiratory problems (because pulmonary fibrosis may occur); or those who are dehydrated or have liver impairment (because risk for renal failure is high). Use cautiously in clients who have a history of peptic ulcer disease or diverticulitis (increases the risk for GI perforation). Drug-Lab-Food Interactions Drug: Drugs that increase the blood levels of erlotinib and may lead to increased adverse reactions or toxicities include ketoconazole, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, troleandomycin (TAO), voriconazole. Drugs that reduce the effectiveness of erlotinib include rifampicin, rifabutin, rifapentine, phenytoin, carbamazepine, phenobarbital, H2 histamine blockers, proton pump inhibitors. Lab: Erlotinib may increase INR and lead to increased risk for bleeding. Food: Grapefruit juice increases erlotinib drug levels and increases the risk for adverse effects. St. John’s wort decreases erlotinib blood levels and reduces its effectiveness Cigarette smoking also decreases erlotinib blood levels. Pharmacokinetics Pharmacodynamics Oral absorption is moderately fast and is enhanced with food intake. High Absorption: Bioavailability is 60%. gastric pH levels inhibit oral absorption. Distribution: 93% PB. Steady-state levels are achieved in 7 to 8 days. Metabolism: Liver (CYP3A4 and CYP1A2 enzymes) Excretion: Mainly in the feces Therapeutic Effects/Uses Approved for treatment of non–small cell lung cancer (NSCLC) and pancreatic cancer. Mode of Action: Erlotinib selectively inhibits the activation of the epidermal growth factor receptor-tyrosine kinase (EGFR-TK). Side Effects Diarrhea and skin reactions Adverse Reactions Ocular changes (inflammation, corneal perforation), GI perforation, skin desquamation, renal failure, hepatic failure. A, adult; d, day; PB, protein-binding; PO, by mouth. Some of the epidermal growth factor/receptor inhibitors carry a Black Box Warning for adverse effects. A Black Box Warning indicates that the FDA has provided notice that a drug may produce serious or even life-threatening effects in some people in addition to its beneficial effects. This warning is printed on the package insert sheet and is bordered in black. Drugs with this warning can still be used, but prescribers are instructed to make certain that such drugs are prescribed only for clients who meet strict criteria and who understand the serious nature of the possible adverse effects. Gefitinib Gefitinib (Iressa) is a synthetic anilinoquinazoline that selectively inhibits the epidermal growth factor receptor-tyrosine kinase (EGFR-TK). It is most commonly used in the management of advanced non–small cell lung cancer. Pharmacokinetics. Gefitinib is absorbed slowly in the gastrointestinal tract, with 60% reaching systemic circulation. It is metabolized mainly in the liver by the CP3A4 enzyme and is primarily excreted in the feces. The half-life is 48 hours. Pharmacodynamics. Gefitinib selectively inhibits EGFRTK. This action results in blockage of downstream EGFRTK–mediated signal transduction pathways, cell cycle arrest, and inhibition of angiogenesis. Side Effects and Adverse Reactions. Gefitinib can cause conjunctivitis and abnormal eyelash growth. Rash occurs in about 43% of patients. Some patients experience a moderate to low potential for nausea/vomiting, diarrhea, and mouth ulcers. Hypersensitivity with a vesiculobullous rash, toxic epidermal necrolysis, erythema multiforme, angioedema, and urticaria is rare. 38-16 CHAPTER 38 Targeted Therapies to Treat Cancer Drug Interactions. Gefitinib activity is affected by CYP3A4 enzyme inhibitors, which decrease dasatinib metabolism, resulting in an increased serum concentration and increased risk for toxicity. These drugs include atazanavir, clarithromycin, indinavir, itraconazole, ketoconazole, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, and voriconazole. Drugs that lower gefitinib serum levels and reduce its activity include carbamazepine, phenytoin, phenobarbital, rifabutin, and rifampin. Because gefitinib works best in an acid environment, its activity is reduced with the use of H2 histamine blockers and proton pump inhibitors. When this drug is received by clients on warfarin therapy, the effectiveness of warfarin (Coumadin) is greatly increased and bleeding risks are increased. The dosage of warfarin should be adjusted based on INR results. liation, rash, skin fissures, acne, dry skin, and paronychia and other nail disorders. Electrolyte imbalances (specifically hypomagnesemia and hypocalcemia) may occur and require replacement. Severe diarrhea can occur when panitumumab is given along with irinotecan. Severe infusion reactions, including angioedema and anaphylaxis, have occurred with hypotension and bronchospasm. Although this reaction is rare, clients should be monitored closely throughout the infusion. Drug Interactions. This drug has not been shown to be associated with general drug interactions. However, when administered with other EGRFIs, the side effects and toxicities are additive and more severe. Panitumumab is not recommended for use in combination with other antineoplastic drug regimens. Panitumumab Cetuximab (Erbitux) is a partially humanized monoclonal antibody (see Table 38-2). (See the Monoclonal Antibodies section for a more complete description of antibody types.) It is most commonly used for the management of colorectal and head and neck cancers. Pharmacokinetics. Cetuximab is administered as an intravenous infusion. Steady-state concentration occurs by the third weekly infusion. The half-life is long, ranging from 41 to 213 hours, with a mean of 97 hours. Pharmacodynamics. Cetuximab still contains about 30% mouse proteins. It binds specifically to EGFR on both normal and tumor cells and prevents formation of a ligand that usually attaches epidermal growth factor to the receptor. This action prevents the receptor from binding to agonists that activate it. As a result, EGFR-TKs are not activated and the signals are not conducted downstream. Other effects of this drug also include inhibition of cell growth, induction of apoptosis, decreased proinflammatory cytokine and vascular growth factor production, and internalization of the EGFR. Cetuximab may make cancer cells more vulnerable to other cancer chemotherapy agents and radiation therapy. The drug is most effective in causing regression in tumors that have the wild-type K-RAS oncogene and overexpress EGFR. Side Effects and Adverse Reactions. Cetuximab carries a Black Box Warning for infusion reactions, usually with the initial dosing, although severe reactions also have been reported during later infusions. Manifestations include rapid onset of airway obstruction, including bronchospasm, stridor, hoarseness, urticaria, and hypotension. Be aware that patients who live in certain geographic areas, such as North Carolina and Tennessee, have an increase risk of hypersensitivity to cetuximab than patients who live in other geographic locations. Cetuximab is associated with dermatologic changes that typically involve the face, upper chest, and back. An acneform rash occurs in 90% of clients, usually within the first 2 weeks of therapy. Most clients continue to have the rash for at least 28 days after therapy is stopped. Drug Interactions. This drug has not been shown to be associated with general drug interactions. However, when administered with other EGRFIs, the side effects and toxicities are additive and more severe. Panitumumab (Vectibex) is a fully humanized monoclonal antibody (Table 38-2). (See the Monoclonal Antibodies section for a more complete description of antibody types.) It is most commonly used in the management of advanced metastatic colorectal carcinomas that express or overexpress EGFR. Pharmacokinetics. Panitumumab is administered as an intravenous infusion. It is a small immunoglobulin that appears to be eliminated in the feces. The half-life is 7.5 days. Pharmacodynamics. Panitumumab binds strongly to the EGFR when it is overexpressed on malignant cells. It binds specifically to EGFR on both normal and tumor cells, and prevents formation of a ligand (arm) that usually attaches epidermal growth factor to the receptor. As a result, EGFRTKs are not activated and the signals are not conducted downstream. Other effects of this drug also include inhibition of cell growth, induction of apoptosis, and decreased proinflammatory cytokine and vascular growth factor production. In addition, binding of panitumumab to the EGFR causes the receptor to be taken into the cell, preventing it from binding to agonists that activate the receptor. Tumors must express EGFR for clients to be candidates for panitumumab. Side Effects and Adverse Reactions. Panitumumab carries a Black Box Warning for dermatologic problems and toxicity. Manifestations usually occur within the first 2 weeks of therapy and include erythema, acneform rash, pruritus, exfoTABLE 38-2 SUFFIXES OF TARGETED CANCER THERAPIES SUFFIX MEANING mab momab imab A monoclonal antibody Composed of only murine (mouse) proteins Composed of more human proteins (>60%) than murine proteins (~30%) Composed of mostly human proteins (95% or more) and only a few murine proteins (<5%) Composed of only human proteins and no murine proteins A tyrosine kinase inhibitor A proteasome inhibitor zumab umab inib zomib Cetuximab CHAPTER 38 Targeted Therapies to Treat Cancer Trastuzumab Trastuzumab (Herceptin) is a monoclonal antibody that binds to the HER2 protein on the surface of cancer cells that overexpress this receptor. The HER2 receptor is structurally related to the EGFR. The HER2 receptor is overexpressed in some breast, ovarian, and colon cancers. When this receptor is overexpressed, there is increased cancer cell proliferation and increased angiogenesis. Trastuzumab is most commonly used in combination with chemotherapy agents to manage breast, ovarian, and colorectal cancers that have demonstrated overexpression of the HER2 receptor. Pharmacokinetics. Trastuzumab is administered as a weekly intravenous infusion. Its half-life averages 25 days, but it may be present in the blood for as long as 18 weeks after therapy is stopped. Just like most monoclonal antibodies, trastuzumab appears to be eliminated slowly through the feces. Pharmacodynamics. Trastuzumab is a mostly humanized monoclonal antibody that binds to the HER2 protein on the surface of breast cancer, ovarian cancer, and colon cells that overexpress this receptor. This drug specifically inhibits the proliferation of cancer cells that overexpress HER2 receptors. In addition, binding of trastuzumab to the cancer cell receptor increases killing of these cells through attack by immune system cells, especially natural killer cells and monocytes. Side Effects and Adverse Reactions. Trastuzumab carries a Black Box Warning for cardiomyopathy manifesting as congestive heart failure when the drug is used as monotherapy. This risk is increased when the drug is given in combination with other drugs that cause cardiotoxicities, such as the anthracyclines and cyclophosphamide. Hypersensitivity reactions, including anaphylaxis, may occur but are not common. Trastuzumab is associated with pain, asthenia, fever, chills, and nausea during the initial infusion. After the initial infusion, these symptoms usually do not reoccur. Other common side effects include loss of appetite, headache, and muscle aches. Drug Interactions. Trastuzumab can increase the incidence and severity of cardiac dysfunction in clients who receive trastuzumab in combination with anthracyclines and cyclophosphamide. This drug may increase myelosuppressive effects of other antineoplastic agents. Vascular Endothelial Growth Factor/Receptor Inhibitors Bevacizumab The currently approved drug in this class is bevacizumab (Avastin), a humanized monoclonal antibody. Table 38-1 lists the dosages, routes, uses, and considerations for bevacizumab. It binds to vascular endothelial growth factor (VEGF) and prevents the binding of VEGF with its receptors, VEGFR-1 (Flt-1) and VEGFR-2 (Flk-1/KDR), which are found on the surface of endothelial cells. The role of VEGF is critical in angiogenesis, the formation of new blood vessels. In human cancers, the increased expression of VEGF is associated with increased microvascular density, tumor growth, metastasis, and a poor prognosis. The result of bevacizumab therapy is the reduction of microvascular growth and inhibition of metastatic disease progression. Bevacizumab is 38-17 approved for many types of malignancies, all in combination with chemotherapy. Pharmacokinetics. Bevacizumab is administered as an intravenous infusion, usually every 2 to 3 weeks. Half-life is about 20 days (range of 11 to 50 days) and steady-state levels are achieved in about 100 days. Clearance varies with weight, gender, and tumor burden, with men clearing the drug faster than women. Pharmacodynamics. Bevacizumab binds to the vascular endothelial growth factor receptor (VEGFR) on tumors that overexpress this receptor. The result of this binding is the competitive inhibition of vascular endothelial growth factor with the VEGFR. This action leads to reduced tumor vascularity, the development of tumor necrosis, and reduced metastatic potential. Side Effects and Adverse Reactions. Bevacizumab carries a Black Box Warning for GI perforations, wound dehiscence, impaired wound healing, hemorrhage, and fistula formation after surgery. Thus, the drug should not be given within 28 days of after major surgery. Hypertension is a common side effects of bevacizumab. Other side effects are associated with hematopoietic suppression, including neutropenia and thrombocytopenia. Proteinuria can be seen in about 36% of clients, and, in rare situations, nephrotic syndrome has developed. The risk for thromboembolism and deep vein thrombosis is increased among clients taking the drug for colon cancer and non–small cell lung cancer. Drug Interactions. No specific drug interactions have been reported with bevacizumab; however, coadministration of drugs with similar pharmacologic effects, especially traditional chemotherapy agents, may cause additive pharmacologic effects, including toxicity. Proteasome Inhibitors Bortezomib Bortezomib (Velcade) is the major drug in this class. Table 38-1 lists the dosages, routes, uses, and considerations for bortezomib. It works by inhibiting the 26S proteasome, which is part of the most common proteasome pathway. A proteasome is a large complex of proteins in cell fluid (cytoplasm) and cell nucleus that regulates protein expression and the degradation of damaged or old proteins within the cell. Its activity is critical to activation or suppression of cellular functions. This system regulates the expression of substances that mediate cell cycle progression (especially oncogene products and cyclins), suppressor genes (especially the Tp53 gene), and proteins that signal apoptosis. When proteasomes are inhibited, cells are much more likely to undergo apoptosis. Although proteasomes are present in normal and cancer cells, the cancer cells are much more sensitive to the effects of proteasome inhibition than normal cells. Thus, proteasome inhibition results in suppression of cancer cell division and enhancement of cancer cell apoptosis. Bortezomib is most commonly used to manage mantle-cell lymphoma and multiple myeloma. Pharmacokinetics. Bortezomib is administered as an intravenous injection bolus. More than 80% of the drug is protein-bound and distributed to most body tissues, 38-18 CHAPTER 38 Targeted Therapies to Treat Cancer NURSING PROCESS Epidermal Growth Factor Receptor Inhibitors and Vascular Endothelial Growth Factor Receptor Inhibitors for Cancer Treatment Assessment baseline physical condition of the client before initiating targeted therapy regimen and during the treatment period. Assess laboratory studies, including CBC with differential, hepatic and renal studies, electrolytes, and urinalysis at the beginning of therapy and at specified time intervals (ranging from weekly to monthly) during therapy. Conduct a detailed medication history, including a list of all concurrent medications, including prescriptions, over-the-counter medicines, antacids, dietary supplements, vitamins, and herbal supplements to avoid drugdrug interactions. Assess client and family knowledge related to therapeutic regimen. Assess Nursing Diagnoses Knowledge, deficient, related to targeted therapy regimen Skin integrity, impaired, risk for, related to dermatologic effects and toxicities of therapy Infection, risk for, related to bone marrow suppression Fluid volume, decicient, risk for, related to GI effects of therapy Electrolyte imbalance, risk for, related to actions of targeted therapy Planning Client and family will verbalize understanding of targeted therapy as part of an anticancer treatment regimen. and family will demonstrate understanding of the importance of reporting targeted therapy–related side effects and adverse reactions. Client and family will verbalize strategies to minimize risks related to targeted therapy–related side effects. Client’s side effects will be managed to a level that the client can tolerate and are not life-threatening. Client will remain infection-free. Client will have fluid balance and electrolytes within expected normal ranges. Client Nursing Interventions prescribed premedications according to established protocols for specific targeted therapies. Monitor complete blood cell count with differential and platelet count at baseline, and as often as recommended for specific agents. Monitor renal function tests at baseline and at least once monthly during therapy. Follow institution guidelines for safe handling, preparing, administering, and dispensing targeted therapy agents. Administer Examine client’s skin closely at each visit for the presence of erythema, rash, peeling, or blister formation, and rate the severity of dermatologic reactions. Also determine whether infection is present in any nonintact skin. When administering monoclonal antibodies intravenously, have resuscitation equipment nearby, and stay with the client during the first 15 minutes of the infusion. Thereafter, monitor vital signs every 15 to 30 minutes during the infusion and for 1 hour after infusion is complete. Monitor electrolytes regularly during treatment and for 8 weeks after treatment has stopped, especially phosphorus and calcium levels, because low levels may require replacement. Gefitinib potentiates the activity of warfarin. Check the client’s INR weekly. Before administering trastuzumab, ask whether the client has a known allergy to hamsters or hamster products. With trastuzumab therapy, monitor electrocardiogram and ejection fraction at baseline and periodically during treatment. Use with caution in older adults (older than 65 years of age) because the incidence of serious adverse events is higher among older clients. Monitor blood pressure at least every 2 to 3 weeks during therapy. Monitor more frequently if hypertension develops. Monitor for proteinuria with a urine dipstick before each dose. Clients with a 2+ or greater reading should undergo further assessment, such as 24-hour urine collection. Initiate bevacizumab therapy at least 28 days after major surgery or when the surgical incision is completely healed, whichever occurs latest. Consider discontinuing bevacizumab before elective surgery or delaying the surgery until 2 months after therapy has stopped. Assess lower extremities for deep vein thrombosis at every visit. Client Teaching symptoms of adverse effects or severe side effects promptly, especially fever, chills, persistent sore throat, swelling, weight gain, or increasing shortness of breath). Report symptoms of bleeding immediately, including black stools, vomit that looks like coffee grounds, easy bleeding/bruising. Remind women with childbearing potential to avoid pregnancy throughout treatment and for up to 12 months after treatment is completed. Advise breastfeeding women to stop breastfeeding during and for 60 days after therapy. Teach clients to avoid direct sunlight and tanning beds to prevent worsening of skin side effects. Teach clients to avoid cigarette smoking with erlotinib. Instruct clients taking erlotinib to immediately report worsening of skin rash; severe or persistent diarrhea, nausea, anorexia, or vomiting; onset or worsening of unexplained shortness of breath or cough; or eye irritation. Report CHAPTER 38 Targeted Therapies to Treat Cancer NURSING PROCESS—cont’d Teach clients to avoid taking NSAIDs, such as aspirin (except for low-dose aspirin), celecoxib, ibuprofen, and naproxen, to prevent excessive bleeding. Remind clients receiving gefitinib who also take warfarin to keep all appointments for INR testing. Teach clients to weigh themselves daily and report a weight gain of more than 2 pounds in 1 day or 4 pounds in 1 week to the health care provider. Instruct clients to notify the health care provider if foaming of urine occurs (an indication of protein in the urine). including myocardium. The half-life ranges from 9 to 15 hours. Bortezomib is metabolized by the CYP3A4, CYP2C19, and CYP1A2 enzymes in the liver. At present, elimination pathways are not known. Pharmacodynamics. Bortezomib inhibits the activity of the 26S proteasome. This inhibition causes an increase in cell cycle checkpoint activity, a decrease in cell proliferation, and a greater responsiveness to cellular signals for apoptosis. The overall results are inhibition of cancer cell growth and an increase in cancer cell apoptosis. Side Effects and Adverse Reactions. The most common side effects of bortezomib are nausea, vomiting, anorexia, abdominal pain, bowel changes (constipation or diarrhea), and decreased taste sensation. Most clients also experience either new-onset peripheral neuropathy or worsening of existing peripheral neuropathy, including both loss of sensation and orthostatic hypotension. Other general side effects include headache, insomnia, rash, pruritus, back pain, arthralgia, bone pain, and muscle cramps. Respiratory effects include dyspnea, cough, and pneumonia. Weakness (asthenia) and low-grade fever are relatively common. Moderate to severe thrombocytopenia, anemia, and neutropenia can occur during bortezomib therapy. Rare but serious cardiopulmonary side effects have been reported. These include new onset congestive heart failure or worsening of pre-existing heart failure, decreased left ventricular function, and pulmonary pneumonitis. Additional neurologic side effects may include anxiety, fatigue, headaches, lethargy, dizziness, insomnia, and blurred or double vision. An uncommon adverse event is the development of reversible posterior leukoencephalopathy syndrome (RPLS) in clients receiving bortezomib. RPLS is a reversible neurologic condition that can present as seizures, hypertension, headache, lethargy, confusion, visual impairment (including blindness), or other neurologic disturbances. Because bortezomib causes rapid killing of cancer cells, the development of tumor lysis syndrome (TLS), including hyperkalemia and hyperphosphatemia, is possible. This complication is more likely in clients with a high tumor burden at the start of bortezomib therapy. Drug Interactions. Drugs that inhibit the CYP3A4 enzyme can lead to increased serum levels of bortezomib, which increases the risk for severe side effects. These drugs include ketoconazole, atazanavir, clarithromycin, indinavir, 38-19 Instruct clients to seek medical help immediately if chest pain, symptoms of stroke (e.g., change in mental awareness; inability to talk or move one side of the body; sudden numbness or weakness of the face, arm, or leg; or seizures), severe abdominal pain, or swelling associated with redness or pain in one leg occurs. Teach clients receiving bevacizumab ways to promote venous return and avoid deep vein thrombosis (DVT), such as avoiding becoming dehydrated, not wearing clothing that restricts circulation, and avoiding smoking cigarettes. itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole, amprenavir, aprepitant, diltiazem, fluconazole, verapamil, and cimetidine. Drugs that induce CYP3A4 enzyme activity may lower bortezomib blood serum levels and reduce its effectiveness. These drugs include rifampin, carbamazepine, and phenytoin. Angiogenesis Inhibitors Temsirolimus/Sirolimus Temsirolimus (Torisel) and its active metabolite, sirolimus, is the major drug in this class. The target of this drug is a protein kinase known as the mammalian target of rapamycin (mTOR). When temsirolimus or sirolimus bind to an intracellular protein called FKBP-12, a protein-drug complex forms that directly inhibits the activity of mTOR. With inhibition of mTOR, the concentration of vascular endothelial growth factor (VEGF) is greatly reduced. In addition, by its inhibition of mTOR, a variety of downstream pro-cell–division signal transduction pathways are disrupted, especially in renal cell carcinoma cells. This drug is most commonly used to manage advanced renal cell carcinoma. Pharmacokinetics. Temsirolimus is administered by intravenous infusion. It is extensively metabolized in the liver by CYP3A4 into five active metabolites, including sirolimus. The half-life or temsirolimus is 17 hours, and the half-life of sirolimus is 54 hours. The drug and its metabolites are primarily excreted in the feces. Pharmacodynamics. Temsirolimus and its metabolites bind to an intracellular protein forming a drug-protein complex that inhibits the protein kinase, mTOR, in the tumor cells of renal cell carcinoma (RCC). As a result of this inhibition RCC tumors lose vascularity. Inhibition of mTOR also reduces production of cyclin D so that progression through the cell cycle does not occur. RCC cell proliferation is inhibited, even under hypoxic kidney conditions. Side Effects and Adverse Reactions. Hypersensitivity reactions to temsirolimus are common, and pretreatment is recommended (see the Nursing Process section). Hyperglycemia is very common (in about 89%) in clients receiving temsirolimus, and it may require treatment with oral antidiabetic agents or with insulin. Hematologic side effects of anemia, neutropenia, and thrombocytopenia may be moderate to severe. Other common side effects include headache, insomnia, nausea and vomiting, back pain, arthralgia, myalgia, mucositis, and diarrhea. Integumentary problems 38-20 CHAPTER 38 Targeted Therapies to Treat Cancer such as rash, pruritus, nail disorder, dry skin, acne, and abnormal wound healing may occur. Respiratory adverse effects of interstitial pneumonitis or other interstitial lung disease are possible. Liver impairment and renal impairment have been reported. Cardiovascular effects of edema formation, chest pain, hypertension, venous thromboembolism, and thrombophlebitis are possible. Drug Interactions. Drugs that inhibit the CYP3A4 enzyme can lead to increased serum levels of temsirolimus, which increases the risk for severe side effects. These drugs include ketoconazole, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, ritonavir, saquinavir, telithromycin, voriconazole, amprenavir, aprepitant, diltiazem, fluconazole, verapamil, and cimetidine. Drugs that induce the CYP3A4 enzyme activity may lower temsirolimus blood serum levels and reduce its effectiveness. These drugs include rifampin, carbamazepine, and phenytoin. Clients taking antihypertensive drugs such as angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor antagonists during temsirolimus therapy are at increased risk for angioedema of the face and upper airways. The combination of temsirolimus and sunitinib may result in dose-limiting integumentary toxicities of erythematous maculopapular rash and gout/cellulitis that require hospitalization. The use of live vaccines such as intranasal influenza, measles, mumps, rubella, oral polio, BCG, yellow fever, varicella, and typhoid vaccines should be avoided during treatment with temsirolimus. Monoclonal Antibodies Monoclonal antibodies largely exert their effects on specific cell membrane surface proteins. All of the FDA-approved monoclonal antibodies currently used in cancer treatment are given intravenously because their protein structure would be altered and inactivated in the GI tract. Antigens are normal cell surface proteins or ligands that serve as targets for binding engineered monoclonal antibodies for the treatment of cancer. Ideal target antigens for cancer treatment should be specific to tumor cells; be located on the surface of the tumor cell and not shed into the bloodstream; occur in high numbers; and play a role in tumor cell survival. Monoclonal antibody therapy is aimed specifically at tumor cells expressing the target antigen. The side effects of monoclonal antibodies are related to activation of the immune system, location of the target antigen, and type of monoclonal antibody. Some monoclonal antibodies sensitize tumor cells to chemotherapy and overcome chemotherapy resistance. The function of an antibody or immunoglobulin is to recognize an antigen and then interact with other serum proteins to eliminate the antigen or the cell associated with that antigen. The Y shape of the antibody has two active sites (Fab and Fc) and the following two activities: 1. The Fab portion of the antibody contains the antigenbinding sites that recognize and bind to a specific antigen. 2. The Fc or constant region is the end of the antibody that signals the immune system to destroy the cell the antibody has bound. The binding of antibody to antigen is highly selective, rather like a lock-and-key fit. When an antibody selectively binds to a specific antigen, the antibody/antigen complex acts as a flag or target, drawing other immune cells to the bound cell to destroy it. A monoclonal antibody recognizes only a single unique antigen and is produced by cloning a single cell. Hybridoma technology, developed in 1975, allowed for mass production of monoclonal antibodies. In this technique, a mouse is inoculated with a purified antigen and antibody-producing cells are isolated from the mouse spleen. Then antibody producing cells are mixed with myeloma cells (which are immortal cells unable to produce antibodies). When antibody-producing cells fuse with myeloma cells, the resulting cells can make large quantities of the specific monoclonal antibody. Binding of a monoclonal antibody to its specific target antigen on the cancer cell inactivates or destroys the cancer cell by one or more of the following mechanisms: • Causing neutralization of tumor cell growth by direct interference with normal biologic activities of the antigen, such as signal transduction of cell growth messages. This cytostatic process can slow growth of the tumor cells. • Promoting antibody-dependent cell-mediated cytotoxicity (ADCC) in which the Fc portion of the bound monoclonal antibody recruits effector cells such as phagocytes, T-cells, and natural killer cells of the immune system to release cytokines that destroy the target cell. • Initiating complement-dependent cytotoxicity (CDC), which activates the complement system (a cascade of naturally circulating blood proteins), thus enhancing immune system destruction of antibody-bound cells. • Directly inducing apoptosis, or programmed cell death. • In fully human antibodies, the entire antibody has been engineered to contain only human antibody protein sequences. Human antibodies are usually named with the suffix “umab” (see Table 38-2). Murine monoclonal antibodies are derived from mice. Mouse antibodies have a very short half-life in the human body, are not as effective as human antibodies in eliciting a response from the CDC and ADCC systems, and can cause the development of human anti-mouse antibodies (HAMA) that neutralize and render the mouse antibodies ineffective against the tumor. Mouse antibodies are usually named with the suffix “momab.” Chimeric monoclonal antibodies end with the suffix “ximab” and contain both human and mouse protein sequences (typically 70% human and 30% mouse). Humanized monoclonal antibodies ending in zumab contain human and gene mouse sequences; however they are not considered chimeric because they contain more human sequences than do chimeric monoclonal antibodies, usually 90% to 95% human and 5% to 10% mouse. When monoclonal antibodies are named, not only is the suffix a clue to the make-up, but also you can tell what they target by their names. If a generic monoclonal antibody has a u in the name, then the target is on a tumor cell. Monoclonal CHAPTER 38 Targeted Therapies to Treat Cancer antibodies can be naked, or unconjugated, with nothing attached to them. They also may be conjugated, in which other anticancer agents such as chemotherapy agents, toxins, or radioisotopes are attached with the antibody. Although agents attached to conjugated antibodies are targeted specifically against tumor cells, nearby healthy cells can still be negatively affected, especially if radioisotopes are used. If the specific target antigen is found on normal cells, these antibodies also cause damage to these cells. The five major monoclonal antibodies currently in use for cancer therapy are alemtuzumab, gemtuzumab ozogamicin, ibritumomab tiuxetin, rituximab, and tositumomab. Table 38-1 lists the monoclonal antibodies and their dosages, routes, uses, and considerations. Prototype Drug Chart 38-3 lists specific drug information for rituximab. Ofatumumab (Arzerra) is a newly approved monoclonal antibody for management of chronic lymphocytic leukemia (CLL). 38-21 Alemtuzumab Alemtuzumab (Campath) is an unconjugated, humanized monoclonal antibody against the cell surface antigen CD52. CD52 is found on most B- and T-lymphocytes, the majority of monocytes, macrophages and natural killer or NK cells, some granulocytes, stem cells, and mature spermatozoa. Although normal cells may express this antigen, malignant cells are much more sensitive to the destructive activity of this antibody. This drug is most commonly used for management of chronic lymphocytic leukemia. Pharmacokinetics. Alemtuzumab is administered as an intravenous infusion. Its average half-life is 12 days, and steady-state levels are reached by about 6 weeks. Because all monoclonal antibodies bind to target cell surfaces and are destroyed along with the target cell, they are cleared as debris from the blood by the liver and eliminated in the feces. PROTOTYPE DRUG CHART 38-3 Rituximab Drug Class Dosage Monoclonal antibody Trade name: Rituxan Pregnancy Category: C A: IV: 375 mg/m2 IV once weekly for 4 doses; may be re-treated for an additional 4 doses. In combination with CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone), 375 mg/m2 IV on Day 1 of each cycle of chemotherapy for up to 8 cycles. Contraindications Clients who may become pregnant, are pregnant, or are breastfeeding; have previously had hepatitis B; have active bacterial or viral infection; have moderate to severe renal, liver, and/or cardiac disease; or have preexisting pulmonary fibrosis Drug-Lab-Food Interactions Drug: Potentiation of hypotension when co-administered with antihypertensive drugs Potentiation of bone marrow suppression when co-administered with other drugs that also cause bone marrow suppression, increasing the risk for infection and bleeding Reduces effectiveness of vaccinations Lab: None known Food: None known Pharmacokinetics Pharmacodynamics Antibody binding specifically to the CD20 cell surface antigen on B-lymphocytes Absorption: 100% bioavailability after intravenous infusion Distribution: Throughout extracellular fluid, bone marrow, and secondary lymphoid tissues (primarily the spleen) Metabolism: Degraded by circulating and liverbased phagocytic cells Excretion: As cellular debris in feces Therapeutic Effects/Uses CD20-positive non-Hodgkin’s lymphoma (NHL) Rheumatoid arthritis (lower dosages) Mode of Action: Binds to CD20-positive B-lymphocytes and lymphoma cells, leading to complement-dependent cytotoxicity, antibody-dependent cellular cytotoxicity, and apoptosis. Side Effects Bone marrow suppression with pancytopenia, Tumor lysis syndrome, hypotension, night sweats, joint and muscle aches, headaches, soreness at injection site A, adult; IV, intravenous. Adverse Reactions Infusion reactions Reactivation of dormant viruses, pulmonary fibrosis, cardiac dysrhythmias, heart failure 38-22 CHAPTER 38 Targeted Therapies to Treat Cancer Pharmacodynamics. Alemtuzumab binds to leukemic cells expressing the CD52 cell surface antigen and induces antibody-dependent lysis. T-cell prolymphocytic leukemia cells are most sensitive to this drug, but monocytes and monocytic leukemias are resistant to it, despite expressing similar amounts of antigen. As an unconjugated monoclonal antibody, this drug relies on inducing apoptotic signals and the activation of mechanisms such as complement or T-cells to attack and kill the targeted cells. Alemtuzumab is also associated with the release of tumor necrosis factor (TNF), interleukin-6, and interferon gamma. Side Effects and Adverse Reactions. Alemtuzumab therapy induces fatigue as the most common side effect that is not related to infusion reactions or pancytopenia. This drug carries a Black Box Warning because of profound bone marrow suppression that may require dose interruptions or reduction, based on severity. This suppression is more profound in clients who are also receiving standard cytotoxic chemotherapy. In addition, infusion reactions with fever, nausea, chills, blood pressure changes, hyperglycemia, and hypoxia are common and often require premedication with antihistamines and acetaminophen. Some clients have reduced reactions with subsequent infusions. Drug Interactions. Specific drug interactions have not been reported with this drug; however, concomitant administration of drugs with similar pharmacologic effects may cause additive side effects, including toxicity. Gemtuzumab Gemtuzumab ozogamicin (Mylotarg) is a conjugated humanized monoclonal antibody directed against the CD33 surface antigen that is expressed by leukemic cells and immature myelomonocytic cells. It is conjugated with an antitumor antibiotic called calicheamicin. This drug is most commonly used to manage acute myeloid leukemia (AML) that is positive for the CD33 cell surface protein. Pharmacokinetics. Gemtuzumab ozogamicin is administered as an intravenous infusion. Its half-life is 45 hours for the first dose and 60 hours for the second dose. Because all monoclonal antibodies bind to target cell surfaces and are destroyed along with the target cell, they are cleared as debris from the blood by the liver and eliminated in the feces. Pharmacodynamics. Gemtuzumab ozogamicin is conjugated with calicheamicin, an antitumor antibiotic. The binding of the of the antibody portion of this combination to CD33 antigen on leukemia cells results in the formation of a complex that is internalized by the cell. Upon internalization, calicheamicin is released from the antibody and binds to the minor groove of the cell’s DNA. This action breaks the DNA double strands and causes cell death. Side Effects and Adverse Reactions. Gemtuzumab ozagamicin carries a Black Box Warning for severe hypersensitivity reactions, pulmonary edema, and acute respiratory distress syndrome. This drug can induce hepatotoxicity, most commonly in clients who have received a hematopoietic stem cell transplant and have experienced any degree of hepatic veno-occlusive disease. Just as with all monoclonal antibodies, this drug causes profound bone marrow suppression that may require dose interruptions or reduction, based on severity. The suppression is more profound in clients who are also receiving standard cytotoxic chemotherapy. Infusion reactions with fever, nausea, chills, blood pressure changes, hyperglycemia, and hypoxia are common and often require premedication with antihistamines and acetaminophen. Some clients have reduced reactions with the second and subsequent infusions. Drug Interactions. Specific drug interactions have not been reported with this drug; however, concomitant administration of drugs with similar pharmacologic effects may cause additive side effects, including toxicity. Ibritumomab Tiuxetan Ibritumomab tiuxetan (Zevalin) is a conjugated murine monoclonal antibody. Ibritumomab is the antibody, and tiuxetan is a linker-chelater to which either Indium-111 or Yttrium-90 can be bound. Like rituximab, this drug combination binds specifically to the human B-lymphocytes that express the CD20 cell surface antigens. This drug is most commonly used for management of B-cell types of non-Hodgkin’s lymphoma that express the CD20 cell surface protein. Pharmacokinetics. Ibritumomab tiuxetin is administered as an intravenous infusion. Its physical half-life is 64 hours, and its biologic half-life (determined by radioactivity detection) is 30 hours. The drug is excreted to a slight degree by the kidneys but is mostly eliminated in the feces. Pharmacodynamics. Ibritumomab tiutuxetin binds to the CD20 antigen on B-lymphocytes and lymphoma cells. Following binding with the CD20 antigen, beta wave radioactive emissions from the attached radionuclide, Y-90, induce cellular damage by the formation of free radicals in the target and neighboring cells. This cellular damage prevents cells from dividing and also causes cell death. Side Effects and Adverse Reactions. Ibritumomab tiuxetan carries a Black Box Warning for infusion reactions with fever, nausea, chills, and blood pressure changes. Severe infusion reactions warrant discontinuing the drug. Less severe infusion reactions may be managed using premedication with antihistamines and acetaminophen. Some clients experience reduced reactions with the second and subsequent infusions. Additional Black Box Warnings for this drug include severe cytopenia and severe cutaneous and mucocutaneous reactions, fetal damage if given during pregnancy, and possible second malignancies. Just as with all monoclonal antibodies, this drug causes profound bone marrow suppression that may require dose interruptions or reduction (based on severity) and are more profound in clients who are also receiving standard cytotoxic chemotherapy. Ibritumomab tiuxetan side effects and adverse effects not associated with bone marrow suppression or infusion reaction include nausea, vomiting, diarrhea, abdominal pain, cough, rash, pruritus, and urticaria. Drug Interactions. Ibritumomab tiuxetin increases the risk for bleeding or hemorrhage when used along with anticoagulant or antiplatelet therapy. Other specific drug interactions CHAPTER 38 Targeted Therapies to Treat Cancer are not associated with this drug; however, concomitant administration of drugs with similar pharmacologic effects may cause additive side effects, including toxicity. Tositumomab Tositumomab (Bexxar) is a murine monoclonal antibody conjugated with the radioactive isotope iodine-131 (131I). Like rituximab and ibritumomab tiuxetin, the antibody portion of this drug binds specifically to the human B-lymphocytes that express the CD20 cell surface antigens. This drug is most commonly used to manage B-cell non-Hodgkin’s lymphoma that does not respond to rituximab. Pharmacokinetics. Tositumumab is administered as an intravenous infusion. It has a median blood clearance of 68.2 mg/hour (with 485-mg dosage). The mean total-body effective half-life is 67 hours. The radioisotope (131I) decays over time (half-life is 8 days) and is eliminated in the urine. Pharmacodynamics. Tositumomab induces cytotoxicity by combining the immunologic effects of antibody binding with the preferential targeting of radiation therapy against CD20-positive lymphocytes and lymphoma cells. Actions of the antibody include the induction of complement-mediated cytolysis, antibody-dependent cellular cytotoxicity, and apoptosis. Radiation activity is cytotoxic not only to the cells bound by the radiolabeled antibody, but also to adjacent cells that may not have been bound by the antibody or do not express the target antigen (a process called the cross fire effect). Together, these actions result in sustained depletion of circulating CD20-positive lymphocytes and lymphoma cells. NURSING PROCESS Proteosome Inhibitors, Angiogenesis Inhibitors, and Monoclonal Antibodies for Cancer Treatment Assessment baseline physical condition of the client before initiating targeted therapy regimen and during the treatment period. Assess laboratory studies, including CBC with differential, hepatic and renal studies, electrolytes, and urinalysis at the beginning of therapy and at specified time intervals (ranging from weekly to monthly) during therapy. Conduct a detailed medication history, including a list of all concurrent medications, including prescriptions, over-the counter medicines, antacids, dietary supplements, vitamins, and herbal supplements to avoid drug-drug interactions. Assess client and family knowledge related to therapeutic regimen. Assess Nursing Diagnoses Knowledge, deficient, related to targeted therapy regimen Skin integrity, impaired, risk for, related to dermatologic effects and toxicities of therapy Infection, risk for, related to bone marrow suppression 38-23 Side Effects and Adverse Reactions. Tositumomab carries a Black Box Warning for severe hypersensitivity reactions, severe bone marrow suppression, and fetal damage (when used during pregnancy). Specific tositumomab side effects and adverse effects include asthenia, headache, hypotension, nausea, vomiting, abdominal pain, diarrhea, hypothyroidism, cough, dyspnea, pleural effusion, and pneumonia. Toxicities associated with radioimmunotherapy can be acute, delayed, or long-term. The most common acute toxicities are fever, rigors, fatigue, headache, and nausea, whereas hypotension and allergic reactions are less common. Delayed toxicities include shortness of breath, fever, signs of infection, inflammation, pain with urination, rash, sore joints, and bone marrow suppression. Long-term toxicities are myelodysplasia or acute leukemia, secondary malignancies, and hypothyroidism. Just as with other monoclonal antibodies, this drug causes profound bone marrow suppression that may require dose interruptions or reduction, based on severity, and are more profound in clients who are also receiving standard cytotoxic chemotherapy. In addition, infusion reactions with fever, nausea, chills, blood pressure changes, hyperglycemia, and hypoxia are common and often require premedication with antihistamines and acetaminophen. Some clients have reduced reactions with subsequent infusions. Drug Interactions. Tositumomab increases the risk for bleeding or hemorrhage when used along with anticoagulant or antiplatelet therapy. Other specific drug interactions have not been reported with this drug; however, concomitant administration of drugs with similar pharmacologic effects may cause additive side effects, including toxicity. Fluid volume, deficient, risk for, related to GI effects of therapy Electrolyte imbalance, risk for, related to actions of targeted therapy Planning Client and family will verbalize understanding of targeted therapy as part of an anticancer treatment regimen. and family will demonstrate understanding of the importance of reporting targeted therapy–related side effects and adverse reactions. Client and family will verbalize strategies to minimize risks related to targeted therapy–related side effects. Client’s side effects will be managed to a level that the client can tolerate and are not life-threatening. Client will remain infection-free. Client will have fluid balance and electrolytes within expected normal ranges. Client Nursing Interventions prescribed premedications according to established protocols for specific targeted therapies. Monitor complete blood cell count with differential and platelet count at baseline, and as often as recommended for specific agents. Administer Continued 38-24 CHAPTER 38 Targeted Therapies to Treat Cancer NURSING PROCESS—cont’d Monitor liver function tests and renal function tests at baseline and at least once monthly during therapy. Follow institution guidelines for safe handling, preparing, administering, and dispensing of targeted therapy agents. Bortezomib Check the client’s peripheral sensation and blood pressure at each visit throughout the duration of treatment. Ensure that client is adequately hydrated before, during, and after therapy to reduce the risk for tumor lysis syndrome. Discuss with the health care provider the possible need for prophylactic allopurinol therapy. Monitor output and serum potassium levels closely. Check for manifestations of tumor lysis syndrome (decreased urine output, hyperkalemia, and hypertension). Temsirolimus/Sirolimus Ask the client about any known hypersensitivity to temsirolimus or sirolimus. If prescribed, premedicate the client with diphenhydramine 25 to 50 mg IV, or a similar antihistamine, approximately 30 minutes before the start of each dose. Ensure that resuscitation equipment is nearby, and stay with the client during the first 15 minutes of the infusion. Thereafter, monitor vital signs every 15 to 30 minutes during the infusion and for 1 hour after infusion is complete. Check the client’s blood glucose level before, during, and after drug administration. Monitor fasting blood glucose and serum electrolytes at baseline and at least every 2 weeks during therapy. Monitor chest radiograph at baseline and periodically during therapy for onset of pulmonary toxicity. Monitor renal and hepatic function at baseline and then periodically during therapy. Monoclonal Antibodies Infusion reactions are common with intravenous infusion of monoclonal antibodies. Ensure that resuscitation equipment is nearby, and stay with the client during the first 15 minutes of the infusion. Thereafter, monitor vital signs every 15 to 30 minutes during the infusion and for 1 hour after infusion is complete. Monoclonal antibodies against leukocytes can profoundly suppress bone marrow activity and increase the risk for infection. Monitor for signs and symptoms of infection. Clients receiving alemtuzumab are usually prescribed to start antimicrobial prophylaxis beginning on the first day of therapy to reduce the risk for serious infection. Prophylaxis is continued for 2 months after the last dose. Gemtuzumab ozogamicin can cause liver toxicity. Closely monitor liver function tests. clients for hepatitis B infection before the initiation of rituximab therapy. Ensure that clients are adequately hydrated before, during, and after rituximab therapy to reduce the risk for tumor lysis syndrome. Monitor output and serum potassium levels closely. Check for manifestations of tumor lysis syndrome (decreased urine output, hyperkalemia). Screen Client Teaching alcohol and nonessential drugs that are cleared by the liver or have liver-toxic effects (e.g., acetaminophen). Report symptoms of adverse effects or severe side effects promptly, especially fever, chills, persistent sore throat, swelling, weight gain, or increasing shortness of breath). Report symptoms of bleeding immediately, including black stools, vomit that looks like coffee grounds, easy bleeding/bruising. Report symptoms of liver impairment immediately, including stomach/abdominal pain, yellowing eyes or skin, dark urine, or unusual fatigue. Remind women with childbearing potential to avoid pregnancy throughout treatment and for up to 12 months after treatment is completed. Advise breastfeeding women to stop breastfeeding during and for 60 days after therapy. Avoid Bortezomib Teach clients to avoid driving or operating dangerous machinery during periods of blurred or double vision or when fatigue is extreme. Instruct clients (or family members) to immediately report the development of convulsions, persistent headache, reduced eyesight, blood pressure increases, or blurred vision. Teach clients to drink at least 4 liters of fluid daily on the day before therapy, on the days of therapy, and for 2 days following therapy. Stress the importance of keeping fluid intake consistent throughout the 24-hour day, and help clients draw up a schedule of fluid intake. Instruct clients to contact the health care provider or cancer clinic immediately if nausea and vomiting prevent adequate fluid intake so they can be started on parenteral fluids. Temsirolimus/Sirolimus Instruct nondiabetic clients to report symptoms of hyper- glycemia (excessive thirst or any increase in the volume or frequency of urination). Instruct diabetic clients to test blood glucose levels more frequently. If prescribed dosages of oral antidiabetic drugs are not sufficient to maintain target blood glucose levels, instruct the client to go to his or her primary care health care provider for diabetic therapy adjustments. CHAPTER 38 Targeted Therapies to Treat Cancer Instruct clients who develop swelling of the face, lips, or tongue to immediately go to the nearest emergency department. Instruct the client and live-in family members not to receive any vaccinations without consulting the oncologist. Monoclonal Antibodies Teach clients the signs and symptoms of bone marrow depression and infection. Instruct the client receiving alemtuzumab therapy and livein family members not to receive any live-virus vaccinations during therapy or for 2 months after therapy is completed. Teach clients that the monoclonal antibodies conjugated to radioisotopes (ibritumomab tiuxetin and tositumomab) make the client somewhat radioactive for a time after the infusion and pose a radiation hazard to others 38-25 for 4 to 7 days. Teach these clients to use the following precautions for one week after each drug administration: Sleep in a separate bed. Maintain a distance of six feet or more from children and pregnant women. Limit time spent in public places. Use a separate bathroom and sit while urinating. Wash hands frequently, especially after using the toilet or handling genitals. Drink plenty of liquids (at least 3 L daily) Keep eating utensils separate from others. Wash laundry separately from those of others. Avoid using disposable products. Avoid sexual contact and avoid becoming pregnant. Clean spilled urine and dispose of body fluid–contaminated material so that others will not inadvertently handle it. KEY WEBSITES American Cancer Society (ACS): www.cancer.org/docroot/ETO/content/ETO_1_2x_Targeted_ Therapy.asp National Cancer Institute (NCI): www.cancer.gov/cancertopics/ understandingcancer/targetedtherapies/htmlcourse National Comprehensive Cancer Network (NCCN): www.nccn.org/professionals/meetings www.nccn.com/breast_cancer_IV.aspx www.nccn.com/metastatic_colorectal_cancer.aspx CRITICAL THINKING CASE STUDY The client is a 64-year-old man diagnosed with stage III B-cell non-Hodgkin’s lymphoma (NHL). He is scheduled to receive rituximab (Rituxan) and a traditional chemotherapy regimen of cyclophosphamide (Cytoxan), doxorubicin (Adriamycin), vincristine (Oncovin), and prednisolone, a combination known as “CHOP.” The dosage and schedule of rituximab for this client is 600 mg IV on day 1 and 6 before the first cycle of CHOP chemotherapy (which will be administered on day 8). The next 2 doses will be administered 2 days before the third and fifth cycles, and the remaining 2 cycles of rituximab will be administered after the sixth cycle of CHOP on days 134 and 141. He has a friend who is taking imatinib (Gleevec) daily as an oral drug for chronic myelogenous leukemia. The client asks why rituximab must be taken intravenously. He also asks that because he has an implanted port, if his spouse, who is an LPN, could administer the rituximab at home so that he will not have to travel to the cancer center in addition to his standard chemotherapy appointments. 1. What is your best response about why rituximab must be administered intravenously? 2. How are rituximab and imatinib different? 3. Why can his spouse not administer rituximab at home? 4. What side effects are specific for rituximab? 5. What are the most common adverse effects of rituximab? 6. What should you teach this client specifically related to the rituximab therapy? NCLEX STUDY QUESTIONS 1. The nurse works in the oncology clinic and knows that targeted therapies for cancer differ from traditional chemotherapy due to which factor? a. They are more likely to kill cancer cells rather than just slow their growth. b. They are so specific for cancer cells that they do not have effects on normal cells. c. They attack and inactivate specific cellular chemicals or structures that are more commonly found in cancer cells than in normal cells. d. They make the plasma membranes of malignant cells more permeable so that intracellular proteins leak out to the extent that cancer cells are unable to perform their specific functions. 2. The client on chemotherapy for breast cancer asks why she is not receiving trastuzumab (Herceptin) like her sister with breast cancer did. What is the nurse’s best response? a. “Your breast cancer cells are estrogen receptor–positive, and targeted therapy is not needed.” b. “ You are much older than your sister and would not tolerate that treatment well.” c. “The drug is expensive, and your insurance does not cover it.” d. “Your cancer cells do not have the target for trastuzumab.” CHAPTER 38 Targeted Therapies to Treat Cancer 3. Which instruction is important for the nurse to include when teaching the client about imatinib (Gleevec) therapy? a. Do not drink grapefruit juice while taking this drug. b. Go immediately to the emergency department if you develop a headache while taking this drug. c. This drug will only work for about 2 months before your cancer develops resistance to it. d. Be sure to take this drug on an empty stomach, either 1 hour before eating or at least 3 hours after eating. 4. Which of the following is the priority nursing diagnosis for clients receiving epidermal growth factor receptor inhibitors (EGFRIs)? a. Risk for infection related to bone marrow suppression and neutropenia b. Risk for impaired skin integrity related to skin side effects c. Risk for injury related to reduced platelet activity d. Disturbed body image related to alopecia 5. When administering which class of targeted therapies should the nurse be most alert to a possible infusion reaction? a. Tyrosine kinase inhibitors b. Multikinase inhibitors c. Monoclonal antibodies d. Proteasome inhibitors 6. The client taking sunitinib (Sutent) reports that the skin on her hands and feet is red, painful, and has some blisters. What is the nurse’s best action? a. Document the report as the only action because this is a mild side effect of the drug. b. Instruct the client to wear gloves and mittens when going outdoors in cold weather. c. Instruct the client to avoid getting her hands wet and to avoid touching food. d. Notify the oncologist to determine whether or not a dosage reduction is needed. 7. Which action is most important for the nurse to teach a client taking Tositumumab (Bexxar)? a. Avoid drinking alcohol for 1 week after receiving this drug. b. Avoid smoking cigarettes for the entire period you are being treated with this drug. c. Use a separate bathroom and sit while urinating for 1 week after receiving this drug. d. Be sure to take this drug on an empty stomach, either 1 hour before or 2 hours after eating. 8. Which activity should the nurse instruct the client taking erlotinib (Tarceva) to avoid? a. Drinking alcoholic beverages b. Taking aspirin or aspirin-containing drugs c. Exposing himself or herself to crowds or persons who are ill d. Exposing himself or herself to direct sunlight or tanning beds 9. Why should clients taking or receiving targeted therapies for cancer avoid using St. John’s wort? a. This herbal drug increases the blood levels of most targeted therapies and increases the risk for severe side effects or adverse reactions. b. This herbal drug decreases the blood levels of most targeted therapies and reduces their effectiveness. c. Targeted therapies increase the blood levels of St. John’s wort, increasing the risk of an overdose of this herbal agent. d. Targeted therapies bind with St. John’s wort in the intestinal tract, preventing the absorption of both the drug and the herbal agent. 10. The client taking imatinib (Gleevec) has gained 5 pounds in the past week. Is this cause for concern? a. No, weight gain is an expected side effect of this drug because it increases the appetite. b. Yes, weight gain is an indication of slow metabolism and possible hypothyroidism. c. Yes, weight gain is an indication of water retention and possible renal impairment. d. Yes, weight gain is an indication of a drug interaction between imatinib and loop diuretics. Answers: 1, c; 2, d; 3, a; 4, b; 5, c; 6, d; 7, c; 8, d; 9, b; 10, c. 38-26