
MasteringPhysics: Print View with Answers
10/19/15, 6:18 AM
Signed in as Andrew Bagley , Instructor
APPY2 (4th Period)
My Courses
Overview
College Physics: A Strategic Approach , 3e
Knight/Jones/Field
Assignments
Equations of State
Sign Out
( MPBAGLEY44076 )
Course Settings
Course Home
Help
Roster
InstructorItem
Resources
Gradebook
Library
eText
Study Area
[ Edit ]
Summary View
Diagnostics View
Print View with Answers
Equations of State
Due: 10:00am on Monday, October 19, 2015
You will receive no credit for items you complete after the assignment is due. Grading Policy
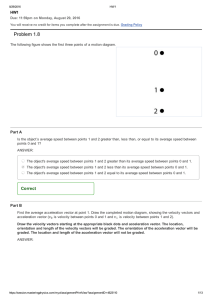
Problem 12.6
Description: An ideal gas is at T. (a) If we times the average kinetic energy of the gas atoms, what is the new
temperature in degree(s) C?
An ideal gas is at 50 ∘ C.
Part A
If we double the average kinetic energy of the gas atoms, what is the new temperature in ∘ C?
ANSWER:
T=
= 373
∘
C
Problem 12.7
Description: An ideal gas is at T. The gas is cooled, reducing the thermal energy by p%. (a) What is the new
temperature in degree(s) C?
An ideal gas is at 40 ∘ C. The gas is cooled, reducing the thermal energy by 30%.
Part A
What is the new temperature in ∘ C?
Express your answer using two significant figures.
ANSWER:
https://session.masteringphysics.com/myct/assignmentPrintView?assignmentID=4015016
Page 1 of 5
MasteringPhysics: Print View with Answers
10/19/15, 6:18 AM
= -54
T=
Also accepted:
∘
C
= -53.9,
= -54
Problem 12.8
Description: An ideal gas at 0 degree(s) C consists of 1.0 * 10^23 atoms. E of thermal energy are added to the gas.
(a) What is the new temperature in degree(s) C?
An ideal gas at 0∘ C consists of 1.0 × 1023 atoms. 12
J of thermal energy are added to the gas.
Part A
What is the new temperature in ∘ C?
Express your answer using two significant figures.
ANSWER:
T=
= 5.8
Also accepted:
∘
C
= 5.80,
= 5.8
Problem 12.9
Description: An ideal gas at 20 degree(s) C consists of 2.2 * 10^22 atoms. E of thermal energy are removed from
the gas. (a) What is the new temperature in degree(s) C?
An ideal gas at 20∘ C consists of 2.2 × 1022 atoms. 6.9
J of thermal energy are removed from the gas.
Part A
What is the new temperature in ∘ C?
Express your answer using two significant figures.
ANSWER:
https://session.masteringphysics.com/myct/assignmentPrintView?assignmentID=4015016
Page 2 of 5
MasteringPhysics: Print View with Answers
T=
10/19/15, 6:18 AM
= 4.8
∘
C
Also accepted:
= 4.85,
= 4.8
Problem 12.17
Description: nu of gas at a temperature of T degree(s) C fills a V container. (a) What is the gas pressure?
1.0
mol of gas at a temperature of -120 ∘ C fills a 2.7 L container.
Part A
What is the gas pressure?
Express your answer using two significant figures.
ANSWER:
P=
Also accepted:
= 4.6
atm
= 4.65,
= 4.6
Problem 12.18
Description: The lowest pressure ever obtained in a laboratory setting is 4.0 * 10^( - 11) Pa. (a) At this pressure,
how many molecules of air would there be in a T experimental chamber with a volume of V?
The lowest pressure ever obtained in a laboratory setting is 4.0 × 10−11
Pa.
Part A
At this pressure, how many molecules of air would there be in a 20 ∘ C experimental chamber with a volume of
0.020 m 3 ?
Express your answer using two significant figures.
ANSWER:
https://session.masteringphysics.com/myct/assignmentPrintView?assignmentID=4015016
Page 3 of 5
MasteringPhysics: Print View with Answers
N=
Also accepted:
10/19/15, 6:18 AM
= 2.0×108
= 1.98×108,
= 2.0×108
A message from your instructor...
This one is tougher. Try it for practice or an extra challenge! Your won't lose points for skipping or getting it wrong.
Problem 12.15
Description: nu of helium are in a V cylinder. The pressure gauge on the cylinder reads P. (a) What is the
temperature of the gas in degree(s)(C)? (b) What is the average kinetic energy of a helium atom?
7.9
mol of helium are in a 18 L cylinder. The pressure gauge on the cylinder reads 67 psi .
Part A
What is the temperature of the gas in ∘ C?
Express your answer using two significant figures.
ANSWER:
T=
= -120
Also accepted:
∘
C
= -119,
= -120
Part B
What is the average kinetic energy of a helium atom?
Express your answer to two significant figures and include the appropriate units.
ANSWER:
https://session.masteringphysics.com/myct/assignmentPrintView?assignmentID=4015016
Page 4 of 5
MasteringPhysics: Print View with Answers
10/19/15, 6:18 AM
= 3.2×10−21
E=
= 3.20×10−21
Also accepted:
Copyright © 2015 Pearson. All rights reserved.
Legal Notice
Privacy Policy
Permissions
https://session.masteringphysics.com/myct/assignmentPrintView?assignmentID=4015016
Support
Page 5 of 5

![Exercise 13.5 Ch 13 Supplemental Questions [ Edit ]](http://s3.studylib.net/store/data/008723409_1-750fc29f92bbd773f55116c1586e6c0c-300x300.png)

![Exercise 5.4 Exercise 5.22 Ch 5 Suppl [ Edit ]](http://s3.studylib.net/store/data/008438115_1-3aed605f845895db933e7996751ae029-300x300.png)

![Problem 3.43 Problem 3.34 Exercise 3.26 Practice Midterm1 [ Edit ]](http://s2.studylib.net/store/data/018396221_1-18b467faac38a28f28568b021e7db428-300x300.png)


![Introduction to Potential Energy Week 7: Chapter 7 [ Edit ]](http://s3.studylib.net/store/data/008792948_1-badd398e755a79911d65229f3f5aeaa5-300x300.png)
![Exercise 14.2 Exercise 14.7 Week 13: Chapter 14 [ Edit ]](http://s3.studylib.net/store/data/008705527_1-add416cf87c7140418a6211e7e753c1a-300x300.png)
![± Gravity on Another Planet Exercise 13.4 Week 12: Chapter 13 [ Edit ]](http://s3.studylib.net/store/data/008723408_1-c9fc9635e4cb7663e66f365b74cb0ff9-300x300.png)