determination of the cation exchange capacity of clays by surface
advertisement

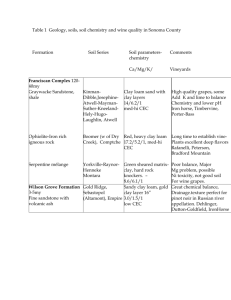
Clay Minerals (1993) 28, 475--481 D E T E R M I N A T I O N OF T H E C A T I O N E X C H A N G E C A P A C I T Y OF C L A Y S BY S U R F A C E T E N S I O N MEASUREMENTS G. B U R R A F A T O AND F. M I A N O * AG1P S.p.A., Drilling Fluids Laboratory, via Maritano 26, 20097 S. Donato Milanese and *ENIRICERCHE S.p.A., via Maritano 26, 20097 S.Donato Milanese, Italy (Received 10 August 1992; revised 11 January 1993) ABSTRACT: The cation exchange capacity (CEC) of clay minerals has been determined by titration of aqueous suspensionof clayswith hexadecylpyridiniumchloride, monitoredusingsurface tension measurements.In order to make the method suitable for an accurate analysisof the CEC of the clays in drilling fluids or in soils, some parameters affectingthe CEC determination (e.g. the presence of carboxymethylcellulose,mixingtime and measurementdevice)have been investigated. The determination of the CEC is an important test in the evaluation of the quality of commercial bentonites. Moreover, CEC is an index of the dispersibility of the formation shales which may affect the properties of drilling fluids and borehole stability (Gray & Darley, 1980). The CEC may be defined as the quantity of exchangeable cations expressed in milliequivalents per 100 g of fully dehydrated mineral (Van Olphen, 1977) or more accurately expressed in mEq per 100 g of ignited weight of clay (Newman, 1987). The principle on which current methods are based is the replacement of the original cations in an aqueous clay suspension. The chemical substance replacing the previous cations in the clay is detectable by some analytical method. Among the known methods, the most relevant are: the ammonium exchange method (Mackenzie, 1951); the colorimetric titration by hexaminocobaltichloride (Bardon et al., 1983); the methylene blue test (API, 1990); and the barium exchange methods (Hendershot & Duquette, 1986). Work by Palumbo & Zucca (pers. comm.) involved comparison of the (Co(NH3)6)C13 and methylene blue methods. The CEC of a series of bentonite clays was consistently greater when tested with the hexaminocobaltichloride technique. The proposed method is based on the strong affinity of hexadecylpyridinium chloride (CPC), a cationic surface-active agent or surfactant, with the clay surface (Greenland & Quirk, 1964). However the tendency of CPC to form a multi-layer on the clay surface is a limiting aspect of the method. Carminati et al., (1990) proposed titration of CPC by monitoring the residual charge potential of the clay with an Electrokinetic Sonic Analyser (ESA) and taking the charge reversal value as the equivalent point of titration. The adsorbing organic cation will replace every available cation on the surface of the clay before an excess of it would remain in solution. Once a mono-layer is formed, the adsorption of a second layer leaves an amount of free surfactant in equilibrium which is able to drop to the lower value of the surface tension of water, which can be detected with high sensitivity and reliability. 9 1993 The MineralogicalSociety 476 G. Burrafato and F. Miano SURFACE TENSION Surface tension is the result of the existence of internal pressure, a force which draws molecules into a liquid and which is perpendicular to the surface. This pressure is caused by the action of the molecular forces and the greater the polarity of a substance, the higher the internal pressure. The unit of measurement of surface tension is mN/m and this force is always tangential to the liquid surface. For a single liquid, the value of surface tension, in the formation of a surface, is established instantaneously (after approx. 0.001 s). The surface tension at the liquid-gas or liquid-vapour interface is determined by the methods of the capillary rise, by the weighing or counting of drops, by the maximum bubble pressure, by the break-off of a ring from the surface and by several other methods. Those substances which accumulate in an interface layer are called surface-active agents or surfactants. There is a positive adsorption at the interface and the surfactant must possess a surface tension less than that of the solvent. In this way the accumulation of the substance at the interface results in an advantageous change of surface tension. Surfactants are generally made up of two parts: a polar group (ionic or non-ionic) and a non-polar hydrocarbon chain. The interaction between a cationic surfactant and the clay particles, and the ability of the surfactant to modify the surface tension of a water solution, may lead to a new method for CEC determination. MATERIALS AND METHODS Bentonites (commercial samples) and shales (from well cores or quarry samples) have been used, as were hexadecylpyridinium chloride (CPC, Merck) and commercial sodium carboxymethylcellulose (CMC). The amounts of montmorillonite, kaolinite and illite in the samples were determined by X-ray diffractometry using a Philips PW 1730 instrument. Suspensions of the clay minerals were titrated with an aqueous solution of CPC (0-025 N) and the surface tension measured on the whole suspension. For comparison, the surface tension was also measured on the supernatant after centrifugation at 8000 rpm for 30 min, with a Kruss K10 tensiometer, equipped with a platinum plate or with a platinum ring, at a temperature of 20~ The experimental sequence was as follows: (1) (2) (3) (4) Grind the clay or shale sample. Sieve the sample through a 200 mesh screen. Dry the sample in an oven at 90~ for 2 h. Suspend the clay in deionized water at a concentration of 0-5% w/v (250 mg in 50 cm 3 of water). (5) Titrate 50 cm 3 of the suspension with CPC solution at a concentration of 25 mEq/dm 3. (6) Stir, after every addition, for 20 min. (7) Measure the surface tension. As an alternative, different additions of CPC solution can be made to several different aliquots of suspension and every sample can be centrifuged before carrying out the surface tension measurement on the supernatant. 477 C E C by surface tension measurements RESULTS AND DISCUSSION The change in the surface tension of water whilst adding CPC and, in the absence and presence of 0.5% w/v bentonite A, respectively, is shown in Fig. 1. As shown by Carminati et al. (1990), the high repartition rate of CPC on the clay surface results in a high and constant surface tension value as long as the surfactant does not reach the saturation of the clay surface. Above the CEC, further adsorption of surfactant may occur. In this region the adsorbed surfactant forms a second layer around the clay, in equilibrium with a concentration of free surfactant which tends to its critical micellar concentration. Consequently, a drop in surface tension can be detected easily at the CEC value, the adsorption of CPC exceeding twice the CEC notwithstanding (Greenland & Quirk, 1964). The addition of CMC and other polymers to commercial bentonites (e.g. for drilling fluids) is a very common practice used to obtain better rheological properties in aqueous dispersion and, usually, quantities of up to 1-2 % are added to obtain this result. The effect of such addition had been evaluated and Fig. 2 shows the curves related to the untreated bentonite and to the sample with addition of I and 10% of CMC, respectively (based on the bentonite content). The evaluation of the CMC addition has been performed on bentonite B because, in bentonite A, some CMC is often added by the producer. As can be seen on Fig. 2, the usual quantity of CMC added does not influence the determination, while large amounts of CMC may. The problem could be solved by treating the bentonite with H202 before performing the determination. The influence of the delay time (the time after the addition of the surfactant to the dispersion before reading the value) on the measured surface tension can be evaluated from the data presented in Fig. 3. The three curves illustrate surface tension measurements as a function of delay time for a 0.5% suspension of bentonite A. The first one is related to an 75.0 Surface tension (mN/m) 70.01 \ 65.0 \\\ 60.0 \\ '\\ 55.0 50.0 \\\\\ \\ 45.0 30.0 " 0.0 [] CPC a d d e d to d i s t i l l e d water 0 CPC a d d e d to 0.5 % b e n t o n i t e A J] I I I I J 1.0 2.0 3.0 4.0 5.0 CPC added 6.0 (cma) FI6.1. Surfacetension vs. CPC (25 mEq/dm3) added to distilled water and to a 0-5% w/v suspension of bentonite A. G. Burrafato and F. Miano 478 Surface tension (mN/m) 75.0 _ [] 70.0 C 65.0 j 60.0 55.0 50.0 45.0 4O.0 35.0 i [] Bentonite B O B e n t o n i t e B * 1.0 % C M C A 30.0 0.0 B e n t o n i t e B + 10.0 % C M C I r I I I r i I r 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 CPC added (cma) Fl6.2. Influence of CMC addition on surface tension measurements. Bentonite B, 0-5% wtv. Surface tension (mN/m) 70.0 [ U 0 6 5 . 0 ~- 60.0 55.0 50.0 A 45.0 ~- [] cPc added - 4.0 cm3 (90 % of the calculated CEC) 0 CPC added 9 4.5 cm a(100 % of the calculated CEC) CPC added 9 6.0 cm 3 (130 % of the calculated CEC) 40.0 ~ 0 A 5 L L L L L 10 15 20 25 30 35 40 Delay time (min) FIG. 3, Surface tension vs. delay time on a 0.5% w/v suspension of bentonite A with different amounts of CPC added. CEC by surface tension measurements 479 addition of CPC of - 9 0 % of the calculated CEC, the second one is related to an addition of - 1 0 0 % and the last one to an addition of - 1 3 0 % . In every case, a period of 20 min is sufficient to reach the equilibrium exchange of the organic cation, even at the very critical point at which the total surface is almost completely covered by a monolayer of CPC. To avoid the effect of suspended particles on the surface tension measurements, the suspension was centrifuged at -8000 r.p.m, for 30 min. In this case, different volumes of CPC were added to different aliquots of suspension, and, after centrifugation, the surface tension measurements were carried out on the supernatant. In Fig. 4, two curves illustrate data, obtained while using the platinum plate, on a bentonite A suspension both with and without centrifugation as described above. The CEC values were calculated from the intersection between the straight lines obtained from the best fit analyses. The calculated values, 42-1 and 45.1 mEq/100 g, are very close and the difference is < 7 % . Following the reported procedure, the relationship between the CPC added (cm 3) and the CEC value (mEq/100 g) is: CEC (mEq/100 g) = CPCadde d (cm 3) • 10. The use of different measuring devices can be evaluated from analysis of Fig. 5. The two curves presented are related to different sets of measurements performed with the same tensiometer equipped with a platinum plate or ring. Although the curves are shifted slightly, the calculated CEC values are 45-1 and 45.6 mEq/100 g respectively, showing a difference of --1%, negligible considering the presumed difference in water contents. A comparison between the CEC values obtained from the "methylene blue test" (MBT) and the "surface tension method" for five different bentonites, two shales and a kaolinite sample is reported in Table 1. The latter determinations were performed on the whole Surface tension (mN/m) 80.0 \ 70.0 [] 60.0 50,0 40.0 30,0 20,0 0.0 [] plate, no c e n t r i f u g a t i o n 9 plate, centrifugation r 1.0 __, 2.0 ~ 3.0 _ _ 4.0 5.0 6.0 CPC added (cm 3) FIG.4. CEC determination from surface tension measurements.Bentonite A, 0-5% w/v. 7.0 G. Burrafato and F. Miano 480 suspension, using the platinum plate and without centrifugation. All of the values were o b t a i n e d as a v e r a g e s o f a t l e a s t t h r e e d e t e r m i n a t i o n s a n d in b o t h cases t h e s t a n d a r d d e v i a t i o n is - - 4 % . T h e r e is r e l a t i v e l y g o o d a g r e e m e n t b e t w e e n t h e d a t a o b t a i n e d f r o m t h e t w o d i f f e r e n t m e t h o d s . T a b l e 1 also i l l u s t r a t e s t h a t t h e b e h a v i o u r o f t h e b e n t o n i t e s a n d s h a l e s is n o t l i n e a r l y r e l a t e d t o t h e i r m o n t m o r i l l o n i t e c o n t e n t . T h i s is p r o b a b l y d u e t o d i f f e r e n c e s b e t w e e n c o m m e r c i a l s a m p l e s s u c h as d i f f e r e n t t r e a t m e n t s (e.g. i n o r g a n i c a n d / o r organic activation ) or different mineralogical components that can influence the CEC Surface tension (mN/m) 80.0 0 70.0 60.0' 50.0 \ \ 40.0 30.0 20.0 0.0 I: \ plate, centrifugation ring, centrifugation -' 1.0 I L L I 2.0 3.0 4.0 5.0 _ [ 6.0 CPC added (crn 3) F~6.5. CEC determination from surface tension measurements. Bentonite A, 0.5% w/v. TABLEl, Montmorillonite content and CEC (mEq/100 g) values measured by the surface tension method and the methylene blue test (MBT) for various bentonites and shales. Sample Wyoming (bent.) BDH 1 (bent.) Bentonite A Bentonite B Bentonite C Bentonite D Shale A 2 Shale B Shale C Warren (kaol.) MBT Surface tension -46 _+ 2 65 _+ 3 63 _+ 3 47 _+ 2 35 +_ 1 8 -+ 0-3 -- 98 _+ 4 67 _+ 3 45 _+ 2 56 _+ 2 58 _+ 2 49 _+ 2 34 +_ 1 7 _+ 0.3 8 -+ 0-3 5 _+ 0.2 % montmorillonite 99 92 86 75 91 -35 -9 97% kaolinite BDH Bentonite powder (technical) Prod. 26022. BDH Ltd, England. 2 Illite + kaolinite + chlorite = 28%. 7.0 C E C by surface tension measurements 481 determination. Nevertheless, the analytical setup has been developed principally for evaluation of commercial samples of clays. CONCLUSIONS A new method for the determination of C E C of clays and shales has been proposed. The method is based on the "titration" of a clay suspension with an appropriate surfactant, the first surfactant excess being determined by surface tension measurements. The method can easily be performed in the laboratory and requires only 250 mg of sample for each determination. The results are comparable with those obtained from the methylene blue test (MBT) and the method can also be used for the C E C determination of shales with a very low montmorillonite content. The method did not appear to be influenced by the presence, in the clay samples, of the common additives used commercially to obtain better viscosity yields, and the results show a relatively good level of reproducibility. ACKNOWLEDGMENTS The experimentalwork has been carriedout under the project "Additivesfor DrillingFluidsOptimisation"financed by AGIP SpA and the ENI Group. Dr. Borgarello (Eniricerche)is thanked for helpful discussion. REFERENCES API (1990)RecommendedPractice StandardProcedure for LaboratoryTestingDrillingFluids.API Recommended Practice 13 I. BARDONC. et al. (1983) Recommandationspour la d6terminationexp6rimentalede la capacit6d'6changede cations des milieuxargileux, Revue de l'lnstitute Franqais du Petrole, Vol. 38, no. 5, Sept-Oct, 621~i26. CARM1NATIS., CARNIANIC. • MIANOF. (1990)Surface modificationof montmorillonitein aqueousdispersionswith hexadecylpyridiniumchloride. Coll. Surf., 48, 209-217. GRAYG.R. & DARLEYH.C.H. (1980) Composition and Properties of Oil Well Drilling Fluids. Gulf Publishing Company, Fourth Edition, 21. GREENLANDD.J. & QUIRKJ.P. (1964) Determination of the total specificsurface areas of soils by adsorption of cetylpyridiniumbromide. J. Soil Sci. 15, 178-191. HENDERSnOTW.H. & DUQUE'L'rEM. (1986) A simple barium chloride method for determiningcation exchange capacity and exchangeablecations. Soil Sci. Soc. Am. J. 50, 605~608. MACKENZIER.C. (1951) A micro-methodfor determination of cation exchange capacity of clay. J. Coll. Sci. 6, 219-222. NEWMANA.D.C. (1987) Chemistry of Clays and Clay Minerals, pp. 237-271. MineralogicalSocietyMonograph no. 6, Longman, Essex. VANOLPHENH. (1977) An Introduction to Clay Colloid Chemistry, John Wiley& Sons, (2nd Ed.) New York.