Atomoxetine in Children & Adolescents SCP
advertisement

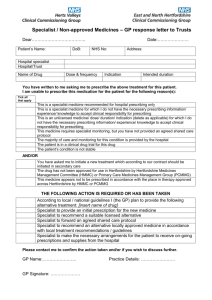
SCP 3 SHARED CARE PROTOCOL Atomoxetine Clinical Indication: Attention Deficit Hyperactivity Disorder (ADHD) in children and adolescents (aged 6 to 18) Version 1: May 2014 due for review: April 2016 Introduction Attention-Deficit Hyperactivity Disorder (ADHD) is diagnosed if the three clinical features -inattention, over-activity and impulsiveness - have been present from an early age, persist in more than one situation (e.g. at home and in school) and impair function. The diagnosis must be made following a comprehensive assessment by an appropriate child psychiatrist and/or a paediatrician with special interest and training in this field. The assessment and management of this condition has been reviewed by SIGN Guideline No. 112, October 2009 and NICE Quality Standard 39, July 2013. Drug therapy with atomoxetine is only one part of the package of care for children with ADHD, which includes behavioural and educational interventions. Shared Care A shared care protocol is used to facilitate the sharing of care and transfer of prescribing. This would usually take place once the patient’s condition is stable; the patient is demonstrably benefiting from the treatment and is free from any significant side effects. GPs should only take on the prescribing when they are confident in the use of the drug, in the context of the protocol. Contingency plans must be in place to enable the patient to receive the recommended treatment, should the GP decline to prescribe. Indication for Therapy Atomoxetine (Strattera ®) is licensed for children of six years or older as part of a comprehensive treatment programme for attention-deficit hyperactivity disorder (ADHD). Treatment must be initiated under the supervision of a child psychiatrist and / or a paediatrician with special interest and training in this field. Preparations Available Atomoxetine (Strattera ®) is available as 10mg, 18mg, 25mg, 40mg, 60mg, 80mg and 100mg capsules. Recommended Dosage and Administration Child aged 6 years and over and adolescent with body weight up to 70kg, initially 500 mcg/kg daily for 7 days increased according to response to usual maintenance dose of 1.2 mg/kg daily. Doses up to 1.8 mg/kg can be used under specialist advice. (Maximum dose 120 mg daily – unlicensed) Child and adolescent with body weight over 70kg, initially 40mg daily for 7 days then increased according to response to usual dose of 80mg – 100mg daily (maximum dose 120mg daily – unlicensed) Total daily dose may be given either as a single dose in the morning or in two divided doses with the last dose given no later than early evening, with or without food. It should be swallowed whole. Dosage must be adjusted in hepatic impairment in accordance with the manufacturers Summary of Product Characteristics. Shared Care Responsibilities Aspects of Care for which the Specialist is responsible Assessment and diagnosis of children with ADHD Liaising with the GP practice to ensure they are willing to prescribe. Making suitable arrangements with the patient in the event the practice are not willing to prescribe. To review the patient at 12 months to identify if treatment needs to be continued. Liaison with GP to agree to share the patient’s care -1Created by amcvean 15/02/2016 533562786 Patient monitoring - initially 3 monthly, then 6 monthly in the longer term. This includes height, weight and blood pressure and pulse. Discontinuation - advising GP when Atomoxetine should be discontinued for patients receiving the drug long term. The specialist will provide necessary supervision and support during the drug discontinuation phase If Atomoxetine is continued beyond the age of 17 then care should be transferred from CAMHS to Adult Psychiatric Services as appropriate Communication to ensure GP is informed of any changes in treatment e.g. patient monitoring, dose alteration etc Aspects of Care for which the General Practitioner is responsible If unwilling to prescribe the drug, to ask if any colleagues are willing to prescribe. If the practice is unwilling to prescribe to relay this decision back to the Specialist in a timely manner for alternative arrangements to be established. To initiate and prescribe the drug if willing to do so under the instruction from the Specialist To establish suitable monitoring as per the above schedule once they take over prescribing Liaison with the Paediatrician/Psychiatrists regarding any complications of treatment Reporting of adverse reactions to Yellow Card Scotland is the responsibility of both the consultant and GP https://yellowcard.mhra.gov.uk/ Adverse Effects The following adverse effects have been reported: Insomnia, somnolence, dizziness Decreased appetite, anorexia, constipation, dyspepsia Occasional abdominal pain, nausea and vomiting Headaches, rash, fatigue Emotional lability, mood swings Temporary growth retardation may occur during prolonged therapy - monitor height and weight Changes in blood pressure and heart rate (usually increased) Drug Interactions Monoamine oxidase inhibitors (MAOIs). Fluoxetine, paroxetine, quinidine, terbinafine – may all increase atomoxetine levels. Salbutamol – action of salbutamol on the cardiovascular system may be potentiated. Atomoxetine may decrease the effectiveness of anti-hypertensive drugs. Drugs that affect noradrenaline – use with caution (venlafaxine, mirtazepine). Drugs that lower seizure threshold – use with caution (e.g. TCA’s, SSRIs, tramadol, neuroleptics). QT prolonging drugs (e.g. erythromycin, methadone, mefloquine, TCA’s, lithium, cisapride). Contra-Indications Do not use in combination with MAOIs or within 2 weeks of stopping MAOIs Do not use with severe cardiovascular or cerebrovascular disorders whose conditions would be expected to deteriorate if they experienced and increase in blood pressure or heart rate. Do not use in patients with narrow angle glaucoma. Do not use in patients with pheochromocytoma. Hypersensitivity to atomoxetine or to any excipients. Precautions Suicide related behaviour has been reported. Patients should be monitored for the appearance or worsening of suicide related behaviour, hostility, irritability, agitation, depression, psychosis or mania. Use with caution in patients with hypertension, tachycardia, cereberovascular or cardiovascular disease. Hepatic effects: Atomoxetine should be discontinued in patients with jaundice or laboratory evidence of liver injury and should not be restarted. Patients and carers should be advised of the risks of hepatic disorders , be told how to recognise symptoms and seek prompt medical -2Created by amcvean 15/02/2016 533562786 attention in the case of abdominal pain, unexplained nausea, malaise, darkening of urine or jaundice. Seizures are a potential risk therefore Atomoxetine should be introduced with caution in patients with a history of seizure. Consider discontinuation in any patient developing seizures or if there is an increase in seizure frequency. Reports of QT interval prolongation have been received with patients with atomoxetine. Atomoxetine should be used with caution in those with congenital or acquired long QT or a family history of QT prolongation. This risk may be enhanced if atomoxetine is used concurrently with other drugs that produce QT prolongation, drugs that can cause electrolyte disturbances and those that inhibit cytochrome P450 2D6 (e.g. fluoxetine, paroxetine, quinidine, terbinafine). Pregnancy. Please refer to the Current Summary of Product Characteristics (SPC) for full details at www.medicines.org.uk Contact Points Borders CAMHS Primary Care Prescribing Team BGH Pharmacy Dr Diana Leaver, Community Paediatrician West Lothian ADHD Service Tel: 01750 23715 Tel: 01896 827708 Tel: 01896 826783 Tel: 01896 826686 Tel: 01506 419666 ext 2734 This information was prepared by the Consultants in the Department of Child and Adolescent Mental Health Service and the Pharmacists at the Royal Edinburgh Hospital through liaison with the General Practice Prescribing Committee, the Paediatric & Neonatal Drug and Therapeutics Committee, Lothian University Hospitals Division, and the Hospital and Specialist Services Medicines Committee, NHS Lothian. Used and adapted with permission. -3Created by amcvean 15/02/2016 533562786