Standard Operation Manual

1
Estonian University of Life Sciences
Institute of Veterinary Medicine
Standard Operation Manual
Document nr:
Author: Brian Lassen and Animal Sciences
Infectious Diseases
Kinnitas:
Version nr.: 1.0
Responsible: Brian Lassen Date: 31.08.2011 Page. nr. 1 (5)
Ziehl-Neelsen contrast acid-fast staining for detection of Cryptosporidium spp. in faeces
1. GENERAL
Methodology for drawing up terms of reference are shorter, which compiles the work of carrying out important points. Job description is the numbers are stored according to work instructions and methodologies for the file or dossier annexed to the methodology. May prepare an abbreviated work guidelines for use directly in the work place to guide the work carried out at present.
1.1. PERSONS RESPONSIBLE
Prepare work instructions:
Confirms the work instruction:
Brian Lassen
Arvo Viltrop
Work instructions and methodologies to be responsible for updating the file: Brian Lassen
Place the code ..................... Location Guide confirms .....................
Place to monitor the timeliness of the Code ..................
1.2. STORAGE AND DISTRIBUTION
Approved by the original work instructions, work instructions stored in the file (No. ...).
Job in a laboratory copies of work instructions, which you kept copies of the file-TJ [No ... (Copy)].
................. responsible for keeping up with the work instructions for issuing copies of the laboratory workers.
2. STRUCTURE AND CONTENT OF WORK INSTRUCTIONS
2.1. USAGE / PURPOSE OF THE INVESTIGATION
The metod can give a semi-quantitative estimate of a Cryptosporidium spp. infection from mammal faeces.
2.2. PRINCIPLE OF THE METHOD
Using 2 colours (red for gram positive and acid proof organisms, and green as a general background staining) it is possible to find the small Cryptosporidium oocysts in faeces. These oocysts are very difficult to find during microscopy in a normal clear flotation liquid. s
2.3. EQUIPMENT AND SUPPLIES
Technical equipment:
Microscope slide: 76x26 mm with 20 mm frosted end area
Microscope slide: 76x26 mm
Cover glass: 24x32x0.1 mm
Pencil
Ziehl-Neelsen colouring cradles (2) and bowls (4) and 6000012 ]
Microscope
[Quantum]
[Quantum]
[Quantum]
[Hounisen: 6000010
2
2.3.1. Equipment calibration procedures none
2.4. CULTURE MEDIA, CHEMICALS, REAGENTS
Fixation liquid: methanol (flammable, poisonous) with 3% HCl (corrosive)
Carbolic fuxine solution (dangerous for health) [Carl Roth: A130.1, Akrom]
Sulfuric Acid : 10% sulphuric acid (H
2
SO
4
corrosive)
Malachite Green 5% (dangerous for health)
Pertex: clear glue applied before cover glass to preserve sample (dangerous for health) [Histolab]
2.5. PROCEDURE
2.5.1. Pretreatment of samples (if necessary) - the required amount of sample collection, centrifugation, inactivation, and fixation, the rate of Concentration, purification, extraction, etc.. samples and storage conditions
2.5.2. Control- and reference material
To establish control samples to be compared with test samples previously identified faces collected and identified for Cryptosporidium or Cryptosporidium free are smeared on microscope slides, fixated in methanol/HCl, but not coloured. They can then be stored separately (4 degrees Celsius) and coloured with the test samples.
Including controls ensure: the effect of the colouring is evaluated every time. Colouring liquid is changed once a week or more frequently if needed.
Researchers always have access to positive controls. This helps identifying contaminations of fungus and protozoa that makes diagnosis of Cryptosporidium oocyst difficult.
2.5.3. PROCEDURE -
Warning: Through all stages of the proceedure care should be taken to avoid infections, contaminate the laboratory, and take the zoonotic parasite out of the laboratory. Clean working area thourougly and materials following investigation (instructions below).
Faces sample
Ziehl Neelsen containers: (A) fixation liquid, (B) carbolic fuxine solution, (C) clean water, (D) sulphuric acid 10%, (E) malachite green solution 5%
1.
A small sample of the faces (~0.1 gram) is smeared between a normal microscope slides and a slide with frosted end by rubbing them against each other (flat side against flat side). The preparation must be thin and even. Mark frosted area with farm, animal number, species, and date.
2.
Air drying: Thin faces smears are dried horizontally (~15 minutes) before placing the sample in the Ziehl-Neelsen cradle.
3.
Fixation: The cradle is lowered into a Ziehl-Neelsen container (A) with fixation liquid.
Fixate for 2 minutes.
4.
Air drying like in pt. 2b. (samples can be stored for 2-3 weeks at 4 degrees Celsius at this point)
5.
Colouring: Move the microscope slides with fixated smears to a new cradle and put it into a
Ziehl-Neelsen container (B) with carbolic fuxine solution for 20 minutes.
6.
Wash: The cradle is placed in a Ziehl-Neelsen container (C) with clean water and washed gently by moving cradle up and down 5 times.
7.
Removing of excess colour: The cradle is moved to a Ziehl-Neelsen container (D) with sulphuric acid for 10 seconds
3
8.
Wash: as in pt. 6.
9.
Repeat point 7-8 once more. Change water.
10.
Contrast colouring: Put the cradle from the water to a Ziehl-Neelsen container with malachite green (E) for 5 minutes.
11.
Wash: dip shortly in a Ziehl-Nielsen container (C) with clean water.
12.
The samples are put on a piece of paper to dry a little.
13.
Optional finish: 1-2 drops of Pertex is put on the sample smear and a covering glass is placed on top. The samples are placed in a fume hood until next day.
14.
Microscopically examination (40x objective). At least 25 visible areas are examined. Look at areas with faecal smears.
15.
Average result (semi-quantitative evaluation) is noted.
Waste
Used solutions of carbolic fuxine solution, malachite green and methanol/HCl and plastics with dangerous chemicals must be stored in bottles marked with “Chemical Waste”. When disposing of full bottles contact Aavo Karus (Chemical department), room 106, Kreutzwaldi
62.
Sulphuric acid can be disposed in the sink with plenty of water.
Pertex must be stored in a place with ventilation (fumes contains Xylen).
Personal danger and preventing infections
Cryptosporidium is infective upon leaving the host, in contrast to other coccidia such as Eim eria spp. and Isospora spp. Host specificity is low and can therefore be transmitted from animals to humans (zoononis), especially C. parvum . Remember this when dealing with materials (wear gloves, clean after yourself and wash hands frequently).
Symptoms of cryptosporidiosis occur about 1 week after infections as stomach cramps, flue-like symptoms, and watery diarrhoea (may vary). The symptoms last 2-3 weeks where after the infection is normally subdued (shedding of oocysts in faeces may still occur longer though). Seek doctor if suspecting an infection, and make sure to keep a good hygiene since the infection is highly infectious (both to humans and animals if C. parvum ).
After dealing with faces and intestinal samples infected with Cryptosporidium , tables and other contaminated surfaces are cleaned with alcohol or iodine solution. Cryptosporidia are killed/inactivated by hot water (>80 degrees).
2.6. RESULTS
2.6.1. Calculation of results: calculation formulas and explanations of the symbols
Oocysts per visual area (average) (examine 3 areas with visual faecal smear and divide number of counted oocysts with 3) at 40x objective.
4
2.6.2. Expression of results and interpretation
Identification of Cryptosporidia
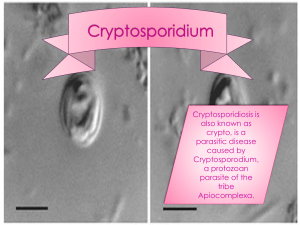
In Ziehl-Neelsen stained smears the oocysts round, often irregular, and red (like a red raisin or red blood cells in structure (red ring with pinkish center)). Yeast may also absorb the red colour but are clear, smoothly coloured, and vary greatly in size. The colouring will most often be stronger in the periphery of the oocyst and homogenously coloured. Most oocysts will contain 1-3 dark granula. In some oocysts inner structures may be seen (sporozoites). There are 14 species of Cryptosporidia .
Most species can be determined morphologically (but PCR is needed for specifics).
Species
Size (µm)
Hosts
Cryptosporidium parvum 5.0 x 4.5 Mammals, primarily cattle and humans
Cryptosporidium hominis
Cryptosporidium andersoni
Cryptosporidium canis
Cryptosporidium felis
5.0 x 4.5
7.4 x 5.5
5.0 x 4.7
4.6 x 4.0
Humans, monkeys, also other animals
Cattle and other ruminants
Dogs, also other animals
Cats, also other animals
Cryptosporidium suis
Cryptosporidium baileyi
4.6 x 4.2 Pigs, also found in humans
6.3 x 5.0 Birds
Cryptosporidium meleagridis 4.5 x 4.0 Birds
Cryptosporidium serpentis 6.2 x 5.3 Reptiles
Evaluation of positive samples
1-5 oocysts
6-25 oocysts
>25 oocysts
+ Low 10
4
-10
5
oocysts per gram
++ Moderate 10
5
-10
6
oocysts per gram
+++ Massive >10
6
oocysts per gram
2.6.3. Reliability:
Sensitivity/specificity of Method (Quiles et al. 1996) :
Sensitivity: Pigs: 67.7%, Cattle: 79.3%
Specificity: Pigs: 85.5%, Cattle: 92.9%
Ziehl-Neelsen versus other analytical methods (Geurden et al. 2006).
This method seems to be most sensitive when used to anlyze samples from young calves compared to IFAT and PCR diagnostics. Positives should be confirmed by either IFAT or PCR if of clinical relevance.
2.6.4. Workability characteristics
Staining may vary depending on faeces smear thickness and the age of the solutions used. Thin smears should be aimed at. Staining solutions should be changed weekly if large amounts of samples are processed.
2.7. Differences to this methodology
Adapted from DFVF (Denmark) manual No. V04-03-003
2.8. ToR documents or document and Literature
Quilez J, Sanchez-Acedo C, Clavel A, del Cacho E, Lopez-Bernad F: Comparison of an acid-fast stain and a monoclonal antibody-based immunofluorescence reagent for the detection of Cryptosporidium oocysts in faecal specimens from cattle and pigs, Vet
Parasitol. 1996 Dec 2;67(1-2):75-81
5
. A Bayesian approach for the evaluation of six diagnostic assays and the estimation of Cryptosporidium prevalence in dairy calves. Vet. Res. 37 (2006) 671–682
2.9. SOLUTIONS FOR PREPARATION INSTRUCTIONS
ALL REAGENTS MUST BE HANDLED IN THE FUME HOOD WITH GLOVES AND EYE
PROTECTION!
Fixation liquid: 100 ml concentrated 30% H
2
SO
4
(corrosive) [Carl Roth, Akrom] added TO 900 ml ionized water (ALWAYS add acid TO solution – otherwise an explosion may occur). Solution marked 10% sulphuric acid.
Malachite Green 5% (wt/volume). Made by mixing 5 grams of Brilliant Green crystals [Carl Roth:
C.I. 42040, Akrom] solution with 100 ml ionized water and mix until dissolved.
Carbolic fuxine solution: ready to use [Carl Roth: A130.1, Akrom]
Sulfuric Acid 10% : Add 100 ml concentrated sulphuric acid (H
2
SO
4
corrosive) TO 900 ml of destilled water (ALWAYS add acid TO solution – otherwise an explosion may occur). Warning: the solution gets very hot and needs to cool down in the fume hood.
Pertex: ready to use (dangerous for health) [Histolab]
Safety sheets:
Carbolic fuxine: http://www.carlroth.com/media/_en-de/sdpdf/A130e.PDF
Malachite Green: http://www.carlroth.com/media/_en-de/sdpdf/0324e.PDF
Hydrochloric acid (HCl): http://www.carlroth.com/media/_en-de/sdpdf/9277e.PDF
Sulphuric acid (H
2
SO
4
): http://www.carlroth.com/media/_en-de/sdpdf/X876e.PDF
Pertex: http://62.173.87.186/docs/BDBinDoc.asp?ID
={8B819F53-F4B0-45C9-9175-
3AEB2D3889F2}&Version=1
Contacts for materials and solutions:
Quantum: tellimus@quantum.ee
Hounisen: liselotte@hounisen.dk
Akrom: akro@akrom.ee
Histolab: kristaps.herings@mednet.lv