storage and stability
advertisement

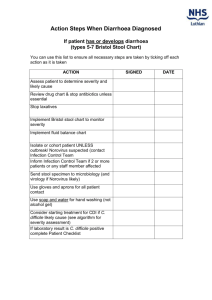
One Step Fecal Occult Blood Test WARNINGS & PRECAUTION Cat. No: 1) In vitro diagnostic use for professional use only. 2) Do not use test kit beyond the expiry date. 3) The test device should not be reused. 4) Patient specimens may contain infectious agents and should be handled as though capable of transmitting disease. Wear disposable gloves throughout the specimen collection and assay procedures. INTENDED USE The One Step Cassette Style Fecal Occult Blood Test is a simple, direct binding immunoassay for the rapid and qualitative detection of human occult blood in feces. The sensitivity is very high with the ability to detect 50 ng/mL of human hemoglobin in feces. In addition, the accuracy of the test is not affected by interfering substances, and dietary restriction is not necessary. For professional laboratory use only. SUMMARY AND EXPLAINATION The test principle is an immunochromatographic sandwich method, which employ two specific monoclonal antibodies to selectively identify hemoglobin in test samples. The result is very specific, and easier to interpret than those of guaiac-based test. The assay is conducted by adding diluted stool specimen to the test device and observing the formation of colored lines. The specimen migrates via capillary action along the membrane to react with the colored conjugate. Positive specimens react with the specific colored antibody conjugates and form a colored line at the test line region of the membrane. Absence of this colored line suggests a negative result. To serve as a procedural control, a colored line will always appear at the control line region if the test has been performed properly. REAGENTS 1) 2) 3) Test reagent One Step Fecal Occult Blood Test Sample Collection Device with buffer Instructions for use C T SPECIMEN COLLECTION Collect stool sample by using the special sample collection device provided. First, unscrew the top of the sample collection device, take out the sample collection stick, and collect the sample by dipping the stick into 3 different places of the stool sample. Then, put the sample collection stick back in the sample collection device and screw together tightly. INTERPRETATION OF RESULTS N Negative: Only one colored band appears on the control (C) region. No apparent band on the test (T) region. This means that FOB level is below 50ng/mL. Positive: In addition to a pink colored control (C) band, a distinct pink colored band will also appear in the test (T) region. This indicates that the FOB concentration is more than 50ng/mL. Please consult your physician to perform a much more detailed examination. Invalid: None of line appears or no line appears in the control (C) region. An invalid result may be due to improper testing procedures or deterioration of the kit components. Repeat the assay sequence using a new device. If the sample cannot be tested on the day of collection, store the stool sample at 4°C. Stir and bring the specimen to room temperature before testing. DIRECTIONS FOR USE 1. 2. 3. 4. Control region: Goat anti-mouse (IgG) polyclonal antibody Test region: mouse monoclonal anti-hemoglobin antibody A 5. Colloidal gold conjugate of monoclonal anti-hemoglobin antibody B squeeze Stool sample MATERIALS PROVIDED Coated Antibodies: Labeled Antibody: break off the tip shake Break point The assay is designed to detect lower levels of colorectal bleeding than other biochemical detection methods. PRINCIPLE OF TEST Dip stool sample 6. Remove the test device from its foil pouch by tearing along the notch and use it as soon as possible. Specimen collection. See also SPECIMEN COLLECTION Shake the sample collection device several times. Holding the sample collection device upright, carefully break off the tip of collection device. Squeeze 3-4 drops (~100ul) of the sample solution on the test sample pad, as in the illustration. Read the test results after 5-10 minutes. It is important that the background is clear before the result is read. Do not read results after more than 20 minutes. C T Negative C T C T Positive C T Invalid STORAGE AND STABILITY The test kit can be stored at temperatures between 2 to 30C in the sealed pouch to the date of expiration. The test kit should be kept away from direct sunlight, moisture and heat. The expiration dating was established under these storage conditions. Revision date: March 2006 QUALITY CONTROL A colored line appearing in the control region (C) is the internal procedural control. It confirms sufficient specimen volume and correct procedural technique. A clear background is an internal negative background control. If the test is working properly, the background in the result area should be white to light pink and not interfere with the ability to read the test result. External controls may also be used to assure that the reagents are working properly and that the assay procedure is followed correctly. It is recommended that a control be tested at regular intervals as good laboratory testing process. Users should follow the appropriate federal, state, and local guidelines concerning the running of external quality controls. LIMITATIONS 1. As with all diagnostic tests, all results must be considered with other clinical information available to the physician. A definite clinical diagnosis should only be made by the physician after all clinical and laboratory findings have been evaluated. This test cannot intended to replace G.I. fibroscope, endoscopy, colonoscopy, or x-ray analysis. 2. This test is limited to the detection of Fecal Occult Blood in human stool sample only. 3. Although the test is highly accurate in detecting human hemoglobin, a low incidence of false positive results may occur. In addition, because many bowl lesions, indulging some polyps and colorectal cancers, may bleed intermittently or not at all, occult blood may not be uniformly distributed throughout a fecal sample. Thus rest results may be negative even when disease is present. PERFORMANCE CHARACTERISTICS Accuracy This test is sensitive for 50 ng/mL human hemoglobin in stool sample. 100 spiked stool samples with concentrations at 0 ng, 25 ng, 50 ng, 75 ng, 100 ng / ml were tested. The results in Table 1 showed that the cutoff was the expected 50 ng/mL. Moreover, two types of abnormal blood (Thalassemia and Sickle Cell) were tested and no false negative issues were raised as results were as expected. One Step FOB Test kit for relative sensitivity and specificity in 648 stool samples. Only 2 samples were discordant, the agreement is 99.67%. (Table 1) Table 1: Cutoff Concentration for IND FOB Test Hemoglobin (ng/mL) # of Samples Negative Postive 0 25 50 75 100 20 20 0 20 20 0 20 2 18 20 0 20 20 0 20 Interfering Substances A performance study was completed to investigate the cross reactivity of other species of hemoglobin and tissue extracts on the FOB Test. The hemoglobin species of bovine, equine, pig, rabbit, sheep, fish, chicken, and goat origin was added to the test device and the results as expected. Hemoglobin of the species was added to normal stool extracts at both 0 and 50 ng/ml human hemoglobin and the results were negative and positive respectively. In addition, the study was repeated with tissue extracts of beef, pig, rabbit, sheep, fish, chicken and goat and no cross reactivity was evident. Lastly, toilet water with the presence of various cleaners (enzymatic to chlorax based) was added to test device and results were as expected following the same protocol as above. A performance study was completed to investigate the interference of dietary substances on the FOB test. Aqueous extracts of raw broccoli, cauliflower, cantaloupe, horseradish, red radish, parsnip, and turnip were added to test device. Also included were dietary iron, vitamin C and horseradish peroxidase. The dietary substance extracts were added to normal stool extracts at both 0 and 50 ng/ml human hemoglobin and the results were as expected. All of the 0 ng/ml Hb samples spiked with interfering substances were negative, while all of the 50ng/ml Hb spiked samples were positive. Table 1: Comparison between Syntron Bioresearch FOB and IND FOB Test IND kits Syntron kits Subtotal + 28 2 30 + - 0 618 618 Subtot al 28 620 648 References: Gorzynski TJ, Krco CJ, David CS., An idiotope expressed on a monoclonal antibody specific for human haemoglobin beta chain and naturally occurring immunoglobulin(s). J Immunogenet. 1985 Dec;12 (6):281-91. Gorzynski TJ, Krco CJ, David CS., The Hb-2d cross-reactive idiotype. A common idiotype expressed by monoclonal and polyclonal antibodies to human haemoglobin. J Immunogenet. 1985 Dec;12(6):267-79. GRAPHICAL SYMBOLS USED Storage temperature Lot number In vitro diagnostic device Expiry date Read instruction before use Manufacturer D Do not reuse Method Comparison To establish the sensitivity and specificity of IND Diagnostic One Step FOB Test kit relative to other rates of qualitative FOB tests, 648 clinic samples were studied. Another commercially available qualitative test kit (Syntron Bioresearch FOB) was used to compare with IND Diagnostic Revision date: March 2006