Realtime PCR - Personal.psu.edu
advertisement

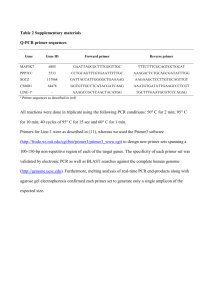
Realtime PCR protocol 1. Reverse Transcription Reverse Transcription is carried out with the M-MLV Reverse Transcriptase (Promega, M1701) and Random Primers (Promega, C1181): 1. Prepare the following RNA/primer mixture in each tube: Total RNA 1-2 g random hexamers (0.5ug/l) 1 l DEPC H2O to 15 l 2. Incubate the samples at 70°C for 5 min and then on ice for at least 1 min. 3. Prepare reaction master mixture. For each reaction: M-MLV 5X Reaction Buffer 5 l 10 mM dNTP 1.25 l M-MLV RT (200units / l) 1 l DEPC H2O 2.75 l 4. Add the reaction mixture to the RNA/primer mixture (final volume is 25 l), mix briefly. 5. Incubate the tubes at 37°C for 60 min. 6. 1:4 dilute 1st strand cDNA product with dd H2O (add 100l dd H2O to 25l RT product), and then store at -20°C until use for real-time PCR. 2. Real-time PCR 1. Normalize the primer concentrations and mix gene-specific forward and reverse primer pair. Each primer (forward or reverse) concentration in the mixture is 5 . 2. Set up the experiment (Don’t forget to add detectors and select all the wells!) and the following PCR program (default protocol) on ABI Prism SDS 7000. Select correct reaction volume (35 or 40l) and then click on the dissociation protocol. 1) 50°C 2 min, 1 cycle 1 2) 95°C 10 min, 1 cycle 3) 95 °C 15 s -> 60 °C 1min, 40 cycles 4) 60°C 20 min, 1 cycle 3. A real-time PCR reaction mixture can be either 35 l or 40 l. Prepare the following mixture in each optical tube. Do two replications for each reaction. 17.5 l SYBR Green PCR Mix* (2x) 20 l SYBR Green PCR Mix (2x) 4 l diluted cDNA or DNA (5-50ng) 4 l diluted cDNA or DNA OR 3 l primer pair mix (5 each 3 l primer pair mix (5 M each primer) primer) 10.5 l H2O 13 l H2O * Strongly recommend preparation of a Master Mix except different template samples or primer pair mix. You may load Master Mix first and then load different template samples or primer pair mix into each well. 4. After PCR is finished, analyze the real-time PCR result with the SDS 7000 software. Check to see if there is any bimodal dissociation curve or abnormal amplification plot. Note: * Preparation of SYBR Green PCR Mix (2x): qPCR Core kit for SYBR® Green I, 500rxns, (Eurogentec, RT-SN73-05) Component Volume 10x Reaction Buffer 2.5ml 50mM MgCl2 solution 1.75ml 5mM dNTP solution 1ml Hot GoldStar enzyme (5U/µl) 125µl 1/2000 dilution (in DMSO)of Sybr Green I stock 750µl Water 6.375ml Total Mix 12.5ml Aliquot and store at -20°C. You can also use pre-made 2x master mix: SYBR Green PCR master mix, 200 reactions (Applied Biosystems) Or qPCR MasterMix Plus for SYBR® green I w/o UNG (Eurogentec, RT-SN2X-03WOU+). 2