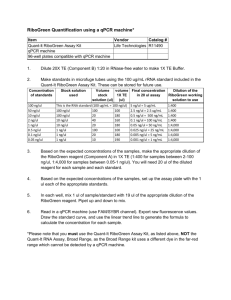
Increased insulin-stimulated myocardial glucose oxidation despite
advertisement

Online Data Supplement Ussher JR, et al. Insulin-stimulated cardiac glucose oxidation is increased in high fat diet-induced obese mice lacking malonyl CoA decarboxylase ONLINE APPENDIX John R. Ussher, B.Sc.1, Timothy R. Koves, Ph.D.2, Jagdip S. Jaswal, Ph.D.1, Liyan Zhang, Ph.D.1, Olga Ilkayeva, Ph.D.2, Jason R. B. Dyck, Ph.D.1, Deborah M. Muoio, Ph.D.2, and Gary D. Lopaschuk, Ph.D.1† 1 2 Cardiovascular Research Group, University of Alberta, Edmonton, Canada Sarah W. Stedman Nutrition and Metabolism Center, Duke University, Durham, North Carolina, U.S.A. †Address for correspondence: Dr. Gary Lopaschuk, email: gary.lopaschuk@ualberta.ca Running Title: Myocardial triglycerides and glucose oxidation 1 Online Data Supplement Ussher JR, et al. RESEARCH DESIGN AND METHODS: Isolated Working Heart Perfusions: In a parallel study, 12-week-old WT or MCD-/- mice were placed on a standard chow, or high fat diet for a 10-week period. At the end of week 10, animals were euthanized via an intraperitoneal injection of sodium pentobarbital (12 mg) and the hearts were subsequently excised and cannulated via the aorta and left atrium, as described previously (1; 2). After equilibration in the Langendorff mode, hearts were switched to the working mode and perfused with modified Krebs-Henseleit buffer containing 118.5 mM NaCl, 25 mM NaHCO3, 4.7 mM KCl, 1.2 mM MgSO4, 1.2 mM KH2PO4, 2.5 mM CaCl2, 0.5 mM EDTA, 5 mM [U-14C]glucose, and 1.2 mM [9,10-3H]palmitate pre-bound to 3% fatty acid free bovine serum albumin (BSA). Hearts were perfused at an 11.5 mmHg left atrial preload and a 60 mmHg aortic afterload. Hearts were perfused for 30 min in the absence of insulin, followed by an additional 30 min in the presence of insulin (100 U/mL). The rates of glucose and palmitate oxidation were measured by quantitative collection of 14CO2 and 3H2O, respectively, as previously described (3; 4). At the end of the 60 min aerobic perfusion protocol, hearts were frozen in liquid N2 and stored at -80oC until used for biochemical analyses. In Vivo Cardiac Function: In vivo cardiac function (cardiac output, tei index) and M-mode ventricular wall measurements (left ventricular wall thickness and diameters) were assessed in isoflurane anaesthetized mice with a Vevo 770 high-resolution echocardiography imaging system equipped with a 30-MHz transducer (RMV-707B; VisualSonics, Toronto, Canada). Intramyocardial Metabolic Profiling: Mass spectrometry-based metabolic profiling was performed as described previously (5; 6) to determine myocardial levels of acylcarnitines and organic acids. Extraction of long chain CoA and short chain CoA esters via high performance liquid chromatography (HPLC) was performed as previously described (1; 7). TG was extracted with a 2:1 chloroform-methanol solution and quantified with a commercially available enzymatic assay kit (Wako Pure Chemical Industries) as previously described (1). Ceramides were extracted as previously described (8). In brief, tissue was extracted with 1 mL of a 1:1:1 chloroform-methanol-1 N HCl in the presence of 0.3 mL saline solution. The resulting organic phase was separated and dried under N2. 0.5 mL of 1 M KOH in 90% (v/v) methanol is added and samples are heated at 90oC for 1 hr to deacylate ceramide into sphingosine. Samples are then extracted with 1 M HCl in methanol, chloroform, and 1 M aqueous NaCl. The resulting organic phase is dried under N2, redissolved in methanol, and derivatized to o-Phthalaldehyde to generate a fluorescent compound that is separated by HPLC and quantified by fluorescence spectrometry. Determination of High Energy Phosphates: Myocardial levels of adenosine triphosphate (ATP) and adenosine monophosphate (AMP) were assessed via extraction with 6% perchloric acid and the homogenate was centrifuged for 5 min at 12,000 x g. The resulting supernatant was brought to a pH range from 5 – 7 with 0.5 M K2CO3 and subsequently centrifuged for 2 min at 10,000 x g. The resulting supernatant was separated via HPLC as previously described (3). PDH Activity: PDH activities were measured using a revised protocol based on the radiometric assay described by Constantin-Teodosiu et al. (9). Briefly, for measurement of active PDC, frozen gastrocnemius muscle tissue was homogenized in buffer containing 200 mM sucrose, 50 mM KCl, 5 mM EGTA, 50 2 Online Data Supplement Ussher JR, et al. mM Tris HCl, 50 mM NaF, 50 mM sodium pyrophosphate (NaPPi), 5 mM dicholoroacetate, and 0.1% Triton X-100 (pH 7.8). For assay of "total" PDC activity (dephosphorylated), frozen tissue was homogenized in buffer containing 1 mM CaCl2 but in the absence of NaF, NaPPi, and EGTA. The total PDC samples were incubated in 0.8 mM MgCl2 at 37°C for 20 min. Both active and total samples were then incubated in assay buffer (150 mM Tris · HCl, 0.75 mM EDTA, 0.75 mM nicotinamide adenine dinucleotide, 1.5 mM thiamine pyrophosphate, 5 mM EGTA, 5 mM dichloroacetate, and 0.75 mM CoA), and the reaction was initiated by the addition of pyruvate. The reaction was terminated by perchloric acid. Samples were neutralized and centrifuged, and the resulting supernatant was used for determination of acetyl-CoA content. Acetyl CoA was converted to [14C]citrate and separated from unreacted radioactivity using Dowex resin (50WX8, 100–200 mesh). The amount of acetyl CoA was determined by comparison of acetyl CoA standard curves run in parallel in each experiment. Immunoblot Analysis: Frozen ventricular tissue (25-30 mg) was homogenized in buffer containing 50 mM Tris HCl (pH 8 at 4oC), 1 mM EDTA, 10% glycerol (wt/vol), 0.02% Brij-35 (wt/vol), 1 mM dithiothreitol, protease and phosphatase inhibitors (Sigma). After homogenization for 30 s, the homogenate was left on ice for 10 min before centrifugation at 10,000 x g for 20 min. The resulting supernatant was processed for immunoblotting. Protein concentration of homogenates was determined via Bradford protein assay kit (Bio-Rad). Samples (20 g protein each) were resolved via 8% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto a 0.45 m nitrocellulose membrane. Membranes were blocked with 10% fat free milk for 2 hours and probed with either antiAMPK (Cell Signaling Technologies, 1/1000 dilution in 5% BSA), anti-phosphoThreonine-172 AMPK (Cell Signaling Technologies, 1/1000 dilution), anti-Akt (Cell Signaling Technologies, 1/1000 dilution), anti-phosphoSerine-473 Akt (Cell Signaling Technologies, 1/500 dilution), anti-PGC1 (Santa Cruz Biotechnology, 1/200 dilution), or anti-UCP2 (Alpha Diagnostic International Inc., 1/500 dilution) antibodies in 5% fatty acid free BSA. Membranes were then washed with 1x phosphate buffered saline and subsequently probed with goat anti-rabbit (Santa Cruz Buitechnology, 1/2000 dilution) secondary antibody in 1% fat-free milk. Immunoblots were visualized with the enhanced chemiluminescence Western blot detection kit (Perkin Elmer) and quantified with Quantity One (4.4.0) Software (Biorad Laboratories). Statistical Analysis: All values are presented as mean SE (n observations). The significance of differences was determined by the use of an unpaired, two-tailed Student’s t-test, two-way analysis of variance (ANOVA), followed by Bonferroni's Multiple Comparison Test, and a repeated measures ANOVA where appropriate. Differences were considered significant when P < 0.05. Supplemental Reference List: 1. Atkinson LL, Kozak R, Kelly SE, Onay Besikci A, Russell JC, Lopaschuk GD: Potential mechanisms and consequences of cardiac triacylglycerol accumulation in insulin-resistant rats. Am J Physiol Endocrinol Metab 284:E923-930, 2003 2. Dyck JR, Hopkins TA, Bonnet S, Michelakis ED, Young ME, Watanabe M, Kawase Y, Jishage K, Lopaschuk GD: Absence of malonyl coenzyme A decarboxylase in mice increases cardiac glucose oxidation and protects the heart from ischemic injury. Circulation 114:1721-1728, 2006 3. Liu B, el Alaoui-Talibi Z, Clanachan AS, Schulz R, Lopaschuk GD: Uncoupling of contractile function from mitochondrial TCA cycle activity and MVO2 during reperfusion of ischemic hearts. Am J Physiol 270:H72-80, 1996 3 Online Data Supplement Ussher JR, et al. 4. Liu Q, Docherty JC, Rendell JC, Clanachan AS, Lopaschuk GD: High levels of fatty acids delay the recovery of intracellular pH and cardiac efficiency in post-ischemic hearts by inhibiting glucose oxidation. J Am Coll Cardiol 39:718-725, 2002 5. An J, Muoio DM, Shiota M, Fujimoto Y, Cline GW, Shulman GI, Koves TR, Stevens R, Millington D, Newgard CB: Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and wholeanimal insulin resistance. Nat Med 10:268-274, 2004 6. Haqq AM, Lien LF, Boan J, Arlotto M, Slentz CA, Muehlbauer MJ, Rochon J, Gallup D, McMahon RL, Bain JR, Stevens R, Millington D, Butler MD, Newgard CB, Svetkey LP: The Study of the Effects of Diet on Metabolism and Nutrition (STEDMAN) weight loss project: Rationale and design. Contemp Clin Trials 26:616-625, 2005 7. Gao S, Kinzig KP, Aja S, Scott KA, Keung W, Kelly S, Strynadka K, Chohnan S, Smith WW, Tamashiro KL, Ladenheim EE, Ronnett GV, Tu Y, Birnbaum MJ, Lopaschuk GD, Moran TH: Leptin activates hypothalamic acetyl-CoA carboxylase to inhibit food intake. Proc Natl Acad Sci U S A 104:17358-17363, 2007 8. Bose R, Kolesnick R: Measurement of ceramide levels by the diacylglycerol kinase reaction and by high-performance liquid chromatography-fluorescence spectrometry. Methods Enzymol 322:373-378, 2000 9. Constantin-Teodosiu D, Cederblad G, Hultman E: A sensitive radioisotopic assay of pyruvate dehydrogenase complex in human muscle tissue. Anal Biochem 198:347-351, 1991 4 Online Data Supplement Ussher JR, et al. Supplementary Figure A. B. 5