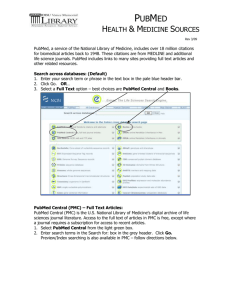
the Nature Immunology Immune Evasion Web Focus
advertisement

Focus on Immune Evasion Volume 3 No 11 November 2002 Welcome to the Nature Immunology Immune Evasion Web Focus <> The immune system has evolved a myriad of mechanisms to detect and eradicate pathogens and tumors from the body. In spite of the excellent array of preventative measures at hand, everyone has suffered microbial infections or knows an individual who has developed cancer. This is because the most successful tumors and pathogens have coevolved with us and developed a panoply of ploys to escape from both the innate and adaptive immune responses. Our understanding of immune evasion strategies is continuing to grow, although much still remains to be discovered. Nature Immunology feel that this is an ideal time to pause and take stock of how far we’ve come and where we are heading. Thus, from 1 November 2002 until 1 February 2003, our Focus on Immune Evasion, which examines recent progress in the field of immune escape, is free to all registrants. We have deliberately avoided focusing on one system, but instead provide a series of comprehensive reviews on microbial and tumor immune evasion. The juxtaposition of the various mechanisms used in these different systems reveals many parallels. It is our hope that this thorough analysis of immune escape will promote cross-fertilization between these areas and help propel the field forward. Similar to our previous web focuses, we include a regularly updated Round-up section that features recent papers on immune evasion, an annotated list of landmark or classic articles on immune escape and links to major articles published by the Nature journals in this area. These features can be reached through the navigational bar at the left. It is obvious that researchers have barely scratched the surface of this rich field. We expect years of exciting discoveries that will aid in our abilities to apply this knowledge. Better vaccines and treatments, through the intelligent boosting of immune responses and specifically foiling of a pathogen’s attacks, may finally enable us to gain the upper hand in our constant struggle with these moving targets. Focus on Immune Evasion Overview Nature Immunology 3, 987 - 989 (2002) doi:10.1038/ni1102-987 © Nature America, Inc. Volume 3 No 11 November 2002 <> Immunology taught by Darwin Rodney E. Phillips The Peter Medawar Building for Pathogen Research, University of Oxford, South Parks Road, Oxford OX1 3SY, UK. rodney.phillips@ndm.ox.ac.uk. This Focus issue brings together the cornucopia of strategies that pathogens and tumors utilize to avoid immune recognition. Here Rodney Phillips discusses some general principles that emerge from this analysis. Nothing in biology makes sense except in the light of evolution. Theodisius Dobzhansky We are still struggling to understand the implications of Charles Darwin's monumental achievements. The most profound biological insight of the 19th century created a framework for understanding variation in organisms that has yet to be fully realized. Before Darwin, biological difference was arrayed before us in endless, unstructured confusion. Darwin made this variation a highly legitimate subject for analysis by recorded observation but also by experimental science. Nearly 150 years after the publication of the Origin of the Species1, we are still laboring to understand evolution and selection in many biological systems. Twentieth century science gave us tools, which, for the first time, allowed us to look directly at the biochemistry that enables genetic variation2. But we are still often unable to explain the mechanisms that underlie clashes between one organism and another. Darwin envisaged selection operating over millennia, with a tempo dictated by brutish struggles in a very competitive world. When complex organisms like humans come under threat from foreign microorganisms and parasites or from within by malignant cellular change, a Darwinian framework is essential if we are to understand these much-accelerated evolutionary encounters. The rapid growth of microorganisms and the huge potential for mutation and recombination in RNA genomes can produce high-speed versions of Darwin's original conception. If we define immunity as the organism's sum capacity to resist attack, then with a tempo that can be as short as days or as long as decades, this defensive resistance constitutes a formidable selection force on malignant cells or pathogenic agents. Immune selection determines the repertoire of bacteria that colonize the skin, the time it takes to die from HIV infection and contributes to a tumor-free youth in humans. When assaulted by a new foreign organism, the immune system produces three basic outcomes: early, complete expulsion of the foreigner; overwhelming infection with failure of control; or persistence with potential long-term carriage or induction of disease. It is this last scenario that allows the immune response to become a sustained selection force, although the dynamics of short clashes can still select for evasive properties in some highly mutable pathogens3. Burnet, inspired by Lewis Thomas, thought the adaptive immune response was involved in continuous "surveillance" for malignant transformation in cells4. As reviewed by Schreiber and colleagues in this issue of Nature Immunology, the concept was developed, refuted and has now had a renaissance. A key prediction of the immune surveillance hypothesis is that breakthrough tumors will have immune-evasion properties. Like microorganisms, tumors require adaptations that allow evasion of the immune response before they can grow in an immunocompetent host. What are the mechanisms of immune escape? The reviews contained in this Focus issue of Nature Immunology describe immune-escape by pathogens and tumors. Some principles emerge from this extensive catalog. Dormancy There is no better way to hide from immunity than to minimize antigen expression. The integrated DNA form of a retrovirus, the minimal protein production of a herpesvirus and the quiescent forms of mycobacteria all have this capacity. But the strategy has limits. Although some cells harboring HIV proviruses may indeed express no viral proteins, others become highly activated and viral proteins are readily processed for presentation to CD8+ killer T lymphocytes5. Successful immune evasion under these selective conditions then depends on other strategies such as antigenic variation 6. Thus microorganisms can have an array of immune-evasion strategies each designed for specific settings. Sequestration Cellular and humoral forms of immunity are pervasive, although the "surveillance" does have blind spots. Some organisms have evolved to occupy special niches where immunity may not penetrate or is frustrated. Malarial parasites have an asexual phase within red blood cells. These complex organisms pay the price of adaptation to life in a biochemically simple environment; the lack of major histocompatibility complex (MHC) class I molecules on the red blood cell surface means that the presence of the plasmodia will not be announced to the immune system in the form of antigenic peptides bound to MHC. Other organisms, such as Mycobacterium tuberculosis, also live within cells and show very low turnover. But how this is achieved is not understood. Genome comparisons between M. tuberculosis and M. leprae provide a clue7. Less than half the genome of M. leprae contains functional genes, but there are many pseudogenes with intact counterparts in M. tuberculosis. Gene deletion and decay have eliminated many important metabolic pathways. The potential evolutionary advantage of this is apparent: M. leprae and its metabolically "crippled" state has a doubling time of approximately 14 days compared to a faster rate in M. tuberculosis. This provides some hint as to why M. leprae has such a remarkably persistent, indolent lifestyle. Gene loss in certain organisms seems to have led to an enhanced capacity for highly specialized sequestration. Salmonella enterica Typhi has 145 fewer functional genes than its relative S. enterica Typhimurium. Several of the lost genes are responsible for bacterial attachment; thus, loss of a specific function may enhance the ability of a microorganism to colonize sites such as the gall bladder, where persistence is favored. Many pathogens appear to enhance virulence by the paradox of gene reduction8. This process of gene loss may produce an irreversible spiral if DNA cannot be reacquired to complement the heavily diminished genome of such organisms ("Muller's ratchet") and so lead to an evolutionary dead end9. This sort of evolutionary decline reflects the "blindness" of adaptation. Small changes in viral genomes, which alter receptor usage, may also enhance the capacity for survival. HIV envelope polymorphisms, which allow binding to dendritic cells, may allow these cells to be used as a "Trojan horse", ferrying the virus away from mucosal surfaces to lymph nodes10. Failure of antigen display Burnet's persuasive arguments in favor of immune surveillance were predicated on the existence of a cellular immune response capable of sensing dangerous antigenic change within cells, wherever they might lie in the body4. The vigor of allograft rejection was viewed by Thomas as a purely artificial and contrived demonstration that histocompatibility was a safety mechanism to prevent transmission of cancer11. How could these difficult concepts be reconciled? And what is the evolutionary role of the adaptive immune system? © Science Photo Library/Photo Researchers, Inc. Engraving of Charles Robert Darwin (1809–1882). Some of this uncertainty was clarified when Zinkernagel and Doherty defined the genetic significance of MHC class I12. These highly polymorphic molecules dictated or "restricted" the response of T cells to foreign antigen. Subsequently, Townsend, in a tour de force of experimental biology, showed unequivocally that MHC class I acted as a molecular clasp, displaying on the cell surface short peptides derived from viruses13, 14. We now know that the vast majority of MHC class I molecules display peptides derived from self, that is, normal cellular proteins. Here was a major clue concerning how the mutant, defective or parasitized cells could alert the policing lymphocytes of the cellular immune response. When a virus harnesses intracellular machinery, a fraction of viral protein is diverted into an active and elaborate process, which loads MHC class I with peptide. Unless this pathway of "processing and presentation" is blocked or subverted, the killer lymphocyte response will inevitably sense intracellular infection and destroy the parasitized cell. In cancer, a more speculative extrapolation was that dangerous mutation would also be signaled to the killer T cells when altered self-peptides arrived at the malignant cell surface bound to MHC class I. Reviews published in this issue of Nature Immunology provide overwhelming evidence that viruses, which have paramount success in establishing chronic infection, have evolved remarkably ingenious ways of subverting almost every part of the biochemistry of the MHC class I pathway. The mechanism may be as simple as a protein sequence rendered indigestible by the peptide-liberating proteasome, as with Epstein-Barr virus proteins, or the acquisition of proteins that interfere with antigen processing at several levels, as with cytomegalovirus (CMV). Indeed, human CMV represents amongst the best circumstantial evidence for the coevolution of pathogen and host 15. Immune-surveillance theories imply that cancer cells might carry phenotypic evidence of immune evasion. In this issue of Nature Immunology, Khong and Restifo review the impressive evidence that tumor cells carry HLA class I deletions, which can arise through many mechanisms. They summarize the mounting, if not overwhelming, evidence that encounters with tumors bearing Fas can prove fatal to anticancer T cells. Evolutionary reasoning would argue that the acquisition of antiimmune cell weapons implies that T lymphocytes are a significant selection force in at least some cancers. The reduction of MHC class I density on cells is detected by a specialized class of lymphocytes with the capacity to lyse these cells directly. These natural killer (NK) cells provide antiviral and antitumor defense. Successful outgrowth of virus or tumor may necessitate ways of subverting this cell population too. Strominger and colleagues review the mechanisms of escape from NK cell surveillance. Antigenic variation Enormous genetic variation is the hallmark of many successful pathogens. Variation is also characteristic of bacteria such as Staphylococcus aureus, which cause asymptomatic carriage as well as disease16. High-throughput sequencing has begun to reveal how variable many microorganisms really are. When subjected to intensive sequencing, some pathogens—such as HIV and meningococcus—are so variable that new analytic tools are required to make some sense of their genetic complexity. One masterly survey of epidemic meningitis identified nine "genoclouds" (a frequent genotype plus its epidemiologically associated descendents) that have been responsible for three pandemic waves of disease since the 1960s17. The authors argue convincingly that loci under positive selection are highly antigenic and represent immune escape from herd immunity17. Many of the variants identified are not fixed over the long term, presumably because of diminished fitness as compared with the parental genotype17. The concept that immunity may shape the detectable diversity in complex genotypes is not new, but in the HIV field this has been highly controversial. One group has argued consistently that the cellular immune response (or indeed any natural selection force) does not positively select for HIV variants18. In this issue of Nature Immunology, Yewdell and Hill review recent evidence for retroviral immune selection and emphasize the definitive studies that have described positive selection for SIV antigenic variants as exerted by cytolytic T lymphocytes (CTLs)19. Another study provides strong genetic evidence that HLA class I is closely linked to HIV polymorphism in a large cohort20. After 12 years of controversy6, the current balance of evidence favors the view that CTLs exert positive selection and so shape the HIV quasispecies in individuals and perhaps also in populations20. Vaccine-induced immunity has been linked with antigenic polymorphism in bacteria. In 1953 mass pertussis vaccination was introduced in the Netherlands, but the disease has remained endemic with occasional outbreaks, including a remarkable increase since 1996. Sequence comparisons between the vaccine and recent isolates showed nonsynonymous mutations in a key antigencoding allele; this finding has been put forward as an explanation for poor vaccine performance in the face of the new prevailing strains21. There is similar evidence for meningococcal vaccine failure22 and for the vaccine-induced CD8+ response in the SIV model23. Antigenic variation represents the most severe barrier to lasting successful preventative vaccination in many infectious diseases, including, par excellence, HIV24. Conclusion Warm-blooded, long-lived vertebrate hosts such as humans have survived because of the extraordinary capacity of the immune response to repel potentially harmful microorganisms. It is clear that pathogens (and tumors) have highly evolved ways of subverting host immunity and that although shaped (or "sculpted") by the immune response, the dangerous outgrowth often prevails. In this century as we reflect upon the achievements of Watson and Crick, Burnet, Medawar, Zinkernagel and Doherty, Townsend and the rest, it must humble modern biologists to think how far we have to go before we master the challenges posed by Charles Darwin. Indeed we are still learning to be his contemporaries. References 1. Darwin, C. On the Origin of Species by Means of Natural Selection, or the Preservation of Favoured Races in the Struggle for Life. (Murray, London, 1859). 2. Watson, J.D. & Crick, F.H.C. Molecular structure of nucleic acids. Nature 171, 4356-4357 (1953). 3. Price, D.A. et al. Positive selection of HIV-1 cytotoxic T lymphocyte escape variants during primary infection. Proc. Natl. Acad. Sci. USA 94, 1890-1895 (1997). | Article | PubMed | ISI | 4. Burnet, F.M. The concept of immunological surveillance. Prog. Exp. Tumour Res. 13, 1-27 (1970) | ISI | 5. Ho, D.D. et al. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature 373, 123-126 (1995). | PubMed | ISI | 6. Phillips, R.E. et al. Human immunodeficiency virus genetic variation that can escape cytotoxic T cell recognition. Nature 354, 453-459 (1991). | PubMed | ISI | 7. Cole, S.T. et al. Massive gene decay in the leprosy bacillus. Nature 49, 1007-1011 (2001). | Article | 8. Ochman, H. & Moran, N.A. Genes lost and genes found: evolution of bacterial pathogenesis and symbiosis. Science 292, 1096-1098 (2001). | Article | PubMed | ISI | 9. Andersson, D.I. & Hughes, D. Muller's ratchet decreases fitness of a DNA-based microbe. Proc. Natl. Acad. Sci. USA. 93, 906-907 (1996). | Article | PubMed | ISI | 10. Baranowski, E., Ruiz-Jarabo, C.M. & Domingo, E. Evolution of cell recognition by viruses. Science 292, 1102-1105 (2001). | Article | PubMed | ISI | 11. Thomas, L. On immunosurveillance in human cancer. Yale J. Biol. Med. 55, 329-333 (1982). | PubMed | ISI | 12. Zinkernagel, R.M. & Doherty, P.C. Restriction of in vitro T cell-mediated cytotoxicity in lymphocytic choriomeningitis within a syngeneic or semiallogeneic system. Nature 248, 701-702 (1974). | PubMed | ISI | 13. Townsend, A.R., Rothbard, J., Gotch, F.M., Bahadur, G. & Wraith, D. The epitopes of influenza nucleoprotein recognized by cytotoxic T lymphocytes can be defined with short synthetic peptides. Cell 44, 959-968 (1986). | PubMed | ISI | 14. Townsend, A. et al. Association of class I major histocompatibility heavy and light chains induced by viral peptides. Nature 340, 443-448 (1989). | PubMed | ISI | 15. Woolhouse, M.E.J., Webster, J.P., Domingo, E., Charlesworth, B. & Levine, B.R. Biological and biomedical implications of the coevolution of pathogens and their hosts. Nature Genet. (in the press, 2002). 16. Day, N.P.J. et al. Link between virulence and ecological abundance in natural populations of Staphylococcus aureus. Science 292, 114-116 (2001). | Article | PubMed | ISI | 17. Zhu, P. et al. Fit genotypes and escape variants of subgroup III Neisseria meningitides during three pandemics of epidemic meningitis. Proc. Natl. Acad. Sci. USA 98, 5234-5239 (2001). | Article | PubMed | ISI | 18. Sala, M. & Wain-Hobson, S. Are RNA viruses adapting or merely changing? J. Mol. Evol. 51, 12-20 (2000). | PubMed | ISI | 19. Allen, T.M. et al. Nature 407, 386-390 (2000). | Article | PubMed | ISI | 20. Moore, C.B. et al. Evidence of HIV-1 adaptation to HLA-restricted immune responses at a population level. Science 296, 1439-1443 (2002). | Article | PubMed | ISI | 21. Van Loo, I.H.M. & Mooi, F.R. Changes in the Dutch Bordetella pertussis population in the first 20 years after the introduction of whole-cell vaccines. Microbiology 148, 2011-2018 (2002). | PubMed | ISI | 22. Martin, S.L. et al. Effect of sequence variation in meningococcal pora outer membrane protein on the effectiveness of a hexavalent pora outer membrane vesicle vaccine. Vaccine 18, 2476-2481 (2000). | Article | PubMed | ISI | 23. Barouch, D.H. et al. Eventual AIDS vaccine failure in a rhesus monkey by viral escape from cytotoxic T lymphocytes. Nature 415, 335-339 (2002). | Article | PubMed | ISI | 24. Gaschen, B. et al. Diversity considerations in HIV-1 vaccine selection. Science 296, 2354-2360 (2002). | Article | PubMed | ISI | Acknowledgments. I thank C. Bangham, M. Maiden, D. Watkins and P. Klenerman for lively discussions over many years. The Peter Medawar Building for Pathogen Research was built by the Wellcome Trust for the University of Oxford. Supported by the Wellcome Trust. Reviews Cancer immunoediting: from immunosurveillance to tumor escape G P Dunn, A T Bruce, H Ikeda, L J Old & R D Schreiber doi:10.1038/ni1102-991 p 991 | Abstract | Full text | PDF (787K) | Natural selection of tumor variants in the generation of "tumor escape" phenotypes H T Khong & N P Restifo doi:10.1038/ni1102-999 p 999 | Abstract | Full text | PDF (720K) | Viral evasion of natural killer cells p 1006 J S Orange, M S Fassett, L A Koopman, J E Boyson & J L Strominger doi:10.1038/ni1102-1006 | Abstract | Full text | PDF (283K) | To kill or be killed: viral evasion of apoptosis C A Benedict, P S Norris & C F Ware doi:10.1038/ni1102-1013 p 1013 | Abstract | Full text | PDF (860K) | Viral interference with antigen presentation J W Yewdell & A B Hill doi:10.1038/ni1102-1019 p 1019 | Abstract | Full text | PDF (696K) | Chronic bacterial infections: living with unwanted guests D Young, T Hussell & G Dougan doi:10.1038/ni1102-1026 p 1026 | Abstract | Full text | PDF (1022K) | Bacterial strategies for overcoming host innate and adaptive immune responses M W Hornef, M J Wick, M Rhen & S Normark doi:10.1038/ni1102-1033 p 1033 | Abstract | Full text | PDF (669K) | Evasion of innate immunity by parasitic protozoa D Sacks & A Sher doi:10.1038/ni1102-1041 | Abstract | Full text | PDF (1006K) | p 1041 Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 991 - 998 (2002) doi:10.1038/ni1102-991 © Nature America, Inc. <> Cancer immunoediting: from immunosurveillance to tumor escape Gavin P. Dunn1, Allen T. Bruce1, Hiroaki Ikeda1, Lloyd J. Old2 & Robert D. Schreiber1 1 Department of Pathology and Immunology, Center for Immunology, Washington University School of Medicine, 660 South Euclid Avenue, St. Louis, MO 63110, USA. 2 Ludwig Institute for Cancer Research, New York Branch at Memorial Sloan-Kettering Cancer Center, New York, NY 10021, USA. Correspondence should be addressed to R D Schreiber schreiber@immunology.wustl.edu The concept that the immune system can recognize and destroy nascent transformed cells was originally embodied in the cancer immunosurveillance hypothesis of Burnet and Thomas. This hypothesis was abandoned shortly afterwards because of the absence of strong experimental evidence supporting the concept. New data, however, clearly show the existence of cancer immunosurveillance and also indicate that it may function as a component of a more general process of cancer immunoediting. This process is responsible for both eliminating tumors and sculpting the immunogenic phenotypes of tumors that eventually form in immunocompetent hosts. In this review, we will summarize the historical and experimental basis of cancer immunoediting and discuss its dual roles in promoting host protection against cancer and facilitating tumor escape from immune destruction. Since the formalized introduction of the cancer immunosurveillance concept, the idea that the immune system may have a protective role in tumor development has been hotly debated. Recent work, however, has lent new support to the idea's central principle that the immune system can indeed prevent tumor formation. At the same time, this work has shown that the immune system also functions to promote or select tumor variants with reduced immunogenicity, thereby providing developing tumors with a mechanism to escape immunologic detection and elimination. These findings have led to the development of the cancer immunoediting hypothesis, a refinement of cancer immunosurveillance that takes a broader view of immune system–tumor interactions by acknowledging both the host-protecting and tumor-sculpting actions of the immune system on developing tumors. Here we discuss first the history that led to formulation of the original cancer immunosurveillance concept and then review the data that prompted the wide-scale abandonment of the hypothesis. We continue with a summary of the data underlying the development of the cancer immunoediting concept and present a unifying model that proposes the molecular and cellular dynamics of the process. We conclude by highlighting the critical implications that immunoediting may have on the development and treatment of cancer in humans. The cancer immunosurveillance hypothesis Paul Ehrlich was one of the first to conceive the idea that the immune system could repress a potentially "overwhelming frequency" of carcinomas1. The idea of immune control of neoplastic disease was not vigorously pursued, however, until the midpoint of the twentieth century. To a large extent, the revisiting of the Ehrlich proposal had to await the maturation of the developing field of immunology. In the 1950s, the work of Medawar and colleagues clarified the critical role for cellular components of immunity in mediating allograft rejection2. This work cast doubt on data that were used to argue for the existence of tumor antigens. Specifically, although it was generally accepted that the immune response was capable of recognizing and destroying transplanted tumors derived from noninbred strains of mice, it soon became clear that the underlying mechanism was one of allograft rejection rather than tumor-specific rejection. With the availability of inbred strains of mice, the idea that tumors were immunologically distinguishable from normal cells could be critically tested. The demonstration that mice could be immunized against syngeneic transplants of tumors induced by chemical carcinogens, viruses or other means established the existence of "tumorspecific antigens"3, 4 and provided a key cornerstone of the cancer immunosurveillance hypothesis. Clearly, there could be no tumor immunosurveillance if there were no distinctive structures on tumor cells that could be recognized by the immune system. These emerging discoveries were incorporated into the formal hypothesis of "cancer immunosurveillance" proposed by Sir Macfarlane Burnet and Lewis Thomas. In 1957, Burnet stated5: It is by no means inconceivable that small accumulations of tumour cells may develop and because of their possession of new antigenic potentialities provoke an effective immunological reaction with regression of the tumour and no clinical hint of its existence. At about the same time, Thomas suggested that the primary function of cellular immunity was in fact not to promote allograft rejection but rather to protect from neoplastic disease, thereby maintaining tissue homeostasis in complex multicellular organisms6. These speculations formed the evolutionary framework that ultimately resulted in the development of the immunosurveillance concept, which was defined by Burnet as follows7, 8: In large, long-lived animals, like most of the warm-blooded vertebrates, inheritable genetic changes must be common in somatic cells and a proportion of these changes will represent a step toward malignancy. It is an evolutionary necessity that there should be some mechanism for eliminating or inactivating such potentially dangerous mutant cells and it is postulated that this mechanism is of immunological character. Both Burnet and Thomas speculated that lymphocytes acted as sentinels in recognizing and eliminating continuously arising, nascent transformed cells7. Challenging the immunosurveillance hypothesis The introduction of the immunosurveillance hypothesis was rapidly followed by many experiments aimed at testing its logical predictions—namely, that hosts with impaired immune systems would exhibit increased incidences of spontaneous or chemically induced tumors. The earliest approaches explored whether tumor development in mice was influenced by experimental immunosuppression of the host9, 10. Whereas several groups used neonatal thymectomy to induce immunosuppression in mice, they could not reach a consensus as to the effects of this treatment on the incidence of either chemically induced10-13 or spontaneous tumors10, 14, 15. Other groups who compromised the immune system of mice using either heterologous anti-lymphocyte serum or pharmacologic methods obtained similar discordant results10. It also became clear that mice with induced immunodeficiencies showed a high susceptibility to virally induced tumors and a greater tendency to develop spontaneous lymphomas compared with immunocompetent mice. The prevailing view about these observations, however, was that they reflected the greater susceptibility of immunocompromised hosts to infectious agents, such as transforming viruses. The greater frequency of lymphomas could also be ascribed to chronic antigenic stimulation from defective control of bacterial or viral infections, resulting in increased lymphocyte proliferation, somatic mutation and eventually the formation of lymphomas10, 16. As a group, these studies were inconclusive and therefore failed either to prove or disprove the immunosurveillance hypothesis. The identification and characterization of the athymic nude mouse17, 18 enabled researchers to study tumor formation in a host with a genetic immunologic impairment. The work of Osias Stutman represents one of the most extensive uses of nude mice to explore whether immunosurveillance occurs in a physiologic setting. Stutman found that CBA/H strain nude mice did not form more chemically induced tumors compared with their wild-type counterparts, nor did they show a shortened tumor latency period after carcinogen injection. For example, in one experiment, nude mice or immunocompetent mice that were heterozygous for the nude mutation were injected subcutaneously with 0.1 mg of the chemical carcinogen methylcholanthrene (MCA) at birth and were monitored for tumor incidence19. After 120 days, seven of 39 control mice formed tumors at the injection site with a mean time to tumor appearance of 95 days. Of the nude mice tested, five of 27 formed tumors with a mean time to tumor appearance of 90 days. The similarity between immunocompetent and nude mice was maintained in subsequent experiments that employed mice of different ages, different doses of carcinogen and even in experiments where the observation period was extended out to 420 days 20, 21. At least one other group corroborated these results 22. In addition, Stutman showed that there were no statistically significant differences in the incidence of spontaneous nonviral tumor formation between nude and wild-type mice23. These findings were supported by a study of Rygaard and Povlsen that showed no differences in spontaneous tumor formation in 10,800 nude mice over a study period of 3–7 months24, 25. The results using nude mice were thus more conclusive than those based on experimental immunosuppression and failed to uphold the central predictions of the immunosurveillance concept. Based on the limited understanding of the immunologic defects in the nude mouse available at that time, these results were highly convincing and thus led to the abandonment of the immunosurveillance hypothesis. In hindsight, however, there are important caveats to these experiments. First, it is now clear that nude mice are not completely immunocompromised. Although nude mice possess fewer T cells than wild-type mice, they have detectable populations of functional T cell receptor–bearing lymphocytes26-28. Therefore, it is not possible to predict the functional effect of these T cells on tumor formation when compared with wild-type controls. Second, the strain of mice used in the Stutman experiments may have been highly sensitive to MCA-induced tumor formation. MCA requires biotransformation from its pro-form into its carcinogenic form by the aryl hydrocarbon hydroxylase enzyme system. It is now known that various inbred mouse strains produce distinctive patterns of these enzyme isoforms with different specific activities 29. The CBA/H strain mouse used in the Stutman studies expresses a form of the enzyme with a high specific activity30, and thus cellular transformation induced by MCA might occur in these mice at a rate that overwhelms host immunological defense mechanisms. Third, even though the study of Rygaard and Povlsen used large numbers of nude mice, monitoring periods of 3–7 months were probably too short to see spontaneous tumor formation in the face of fully functional intrinsic tumor suppressor systems (such as p53). Last, these studies were carried out before the discovery of other lymphocyte populations such as natural killer (NK) cells, which are not thymus dependent, and may develop extrathymically31. T cells, a subset of which The lack of convincing support for the immunosurveillance concept left room for other theories on the possible functions of immune cells during the development of neoplasia. For example, Prehn proposed that the immune system can actually promote the growth of tumors. In this "immunostimulation theory" 32, the presence of tumor rejection antigens (TRAs) on tumors is explained by the argument that the immune cells that recognize these TRAs provide a positive growth signal to the tumor cells. Although this theory was speculative rather than evidence based, it reflected the mood that the proponents of the immunosurveillance theory were overstating their case. Especially after the Stutman experiments, enthusiasm for the original immunosurveillance concept began to wane. Thomas later noted, "The greatest trouble with the idea of immunosurveillance is that it cannot be shown to exist in experimental animals"33. During the ensuing years, little interest was paid to the possibility that the immune system could prevent the development of nonvirally induced tumors. By all intents and purposes, the cancer immunosurveillance concept was considered dead by 1978, and the field of tumor immunology moved on to study other issues such as the definition and molecular nature of mouse and human tumor antigens, the immune response to known tumor antigens and the development of immunotherapeutic strategies to treat cancer. This view relegated immunosurveillance to the historical dust bin and was clearly echoed in a major review that appeared in 2000, which listed the six critical hurdles that a developing tumor must circumvent to grow and survive 34. No significant mention was made of the natural immune response against tumors. The renaissance of cancer immunosurveillance Between the mid-1970s and 1990s, researchers made several experimental attempts to resurrect the cancer immunosurveillance concept. The discovery of NK cells led to a considerable amount of enthusiasm over the possibility that they functioned as the effectors of immunosurveillance 35. This enthusiasm was dampened, however, when a precise definition and understanding of these cells was difficult to obtain. Subsequently, others repeated the MCA induction experiments of Stutman using nude mice on a BALB/c background36. When they injected nude and control mice with different doses of MCA and monitored them for tumor development, nude mice formed more tumors than controls. These data were limited, however, in their statistical power because of the small experimental group sizes and the magnitude of the differences. Similar suggestive results were obtained when tumor formation induced by MCA was compared between groups of wild-type BALB/c mice and immunodeficient CB-17 severe-combined immunodeficiency (SCID) mice37. The latter lack a functionally active subunit of the DNA-dependant protein kinase (DNA-PK) enzyme (DNA-PKcs) that is required to generate rearranged antigen receptors in lymphocytes38, and thus they are unable to develop antigen-specific immune responses. However, DNA-PKcs (and the intact DNA-PK enzyme) is normally expressed in all cells and is important in repairing double-stranded DNA breaks39, which includes those that can potentially be transforming. Thus, it was not possible to interpret the basis for the increased tumor incidence observed in SCID mice because of an inability to differentiate between tumor suppressor roles for the immune system or for the DNA-PK enzyme itself. Between 1994 and 1998, two key findings incited renewed interest in the process of cancer immunosurveillance. First, endogenously produced interferon (IFN- ) was shown to protect the host against the growth of transplanted tumors and the formation of primary chemically induced and spontaneous tumors. Using tumor transplantation approaches, researchers found that immunogenic fibrosarcomas grow faster and more efficiently in mice treated with neutralizing monoclonal antibodies specific for IFN- 40. Overexpression of a dominant-negative mutant of the IFNreceptor subunit (IFNGR1) in sarcomas such as Meth A (BALB/c derived) or MCA-207 (C57BL/6 derived) completely ablated tumor sensitivity to this cytokine, and the tumors displayed enhanced tumorigenicity and reduced immunogenicity when transplanted into naïve syngeneic hosts 40. Experiments based on models of MCA-induced tumor formation showed that 129/SvEv mice that were lacking either the IFN- receptor or signal transducer and activator of transcription 1 (STAT1, the transcription factor that is important in mediating IFN- receptor signaling) were found to be approximately 10–20 times more sensitive than wild-type mice to the tumor-inducing capacity of MCA, developed more tumors than their wild-type counterparts and showed a shortened tumor latency period41. Subsequent independent experiments using mice on a different genetic background that lacked the gene encoding IFN- confirmed these results42. Similarly, mice lacking the genes encoding the tumor suppressor p53 and the IFNGR1 subunit of the IFN- receptor formed a wider spectrum of tumors as compared with IFN- -sensitive mice lacking only p5341. In addition, IFN- -/- mice on a C57BL/6 background showed an increased incidence of disseminated lymphomas despite the presence of a normal p53 tumor suppressor gene, and IFNBALB/c background showed a low incidence of lung adenocarcinomas43. -/- mice on a The second key finding was the observation that C57BL/6 mice lacking perforin (perforin -/-) were more prone to MCA-induced tumor formation compared with their wild-type counterparts. Perforin is a component of the cytolytic granules of cytotoxic T cells and NK cells that is important in mediating lymphocyte-dependent killing of many different target cells including tumor cells 44. After challenge with MCA, perforin-/- mice developed significantly more tumors compared with perforin-sufficient mice treated in the same manner42, 45, 46. Untreated perforin-/- mice also showed a high incidence of spontaneous disseminated lymphomas, which was greater on a p53+/- background47, and a low incidence of spontaneous lung adenocarcinomas43. Taken together, these observations showed that components of the immune system were involved in controlling primary tumor development. The definitive work supporting the existence of a cancer immunosurveillance process that is dependent on both IFN- and lymphocytes came through the use of gene-targeted mice that lack recombination activating gene 1 (RAG-1) or RAG-2. Like DNA-PK, these enzymes are involved in the repair of double-stranded DNA breaks, but unlike DNA-PK, they are expressed exclusively in the lymphoid compartment. Mice deficient in either of these genes fail to rearrange lymphocyte antigen receptors and thus completely lack natural killer T (NKT), T and B cells 48. In contrast, DNA repair mechanisms in the nonlymphoid compartment of these mice are completely normal. For the first time, inbred strains of mice were available that carried genetically defined mutations affecting specific tissues that led to an absence of lymphocytes bearing antigen receptors. These mice enabled researchers to carry out carcinogenesis experiments that could unequivocally be interpreted. After MCA injection, 129/SvEv RAG-2-/- mice developed sarcomas more rapidly and with greater frequency than genetically matched wild-type controls49. After 160 days, 30 of 52 RAG2-/- mice formed tumors, compared with 11 of 57 wild-type mice. In addition, RAG-2-/- mice aged in a specific pathogen free mouse facility formed far more spontaneous epithelial tumors than did wildtype mice housed in the same room 49 (and unpublished data). Specifically, 26 of 26 RAG-2-/- mice aged 13–24 months developed spontaneous neoplasia, predominantly of the intestine; eight of these mice had premalignant intestinal adenomas, 17 had intestinal adenocarcinomas and one had both an intestinal adenoma and a lung adenocarcinoma. In contrast, only five of 20 wild-type mice aged 13–24 months developed spontaneous neoplasia, which was predominantly benign. Three of the wild-type mice developed adenomas of the Harderian gland, lung and intestine, respectively, whereas the other two developed an adenocarcinoma of the Harderian gland and an endometrial stromal carcinoma. Thus, lymphocytes in a mouse not only protect the host against formation of primary sarcomas that are chemically induced but also prevent the development of spontaneous epithelial tumors. The overlap between the tumor suppressor pathways that depend on IFN- and lymphocytes was defined by comparing tumor formation in 129/SvEv mice lacking IFN- responsiveness (IFNGR1 receptor-/- or STAT1-/- mice), lymphocytes (RAG-2-/- mice) or both RAG-2 and STAT1 (RkSk mice)49. Each of the four lines of gene-targeted mice formed three times more chemically induced tumors than syngeneic wild-type mice when injected with 0.1 mg of MCA. Because no significant differences were detected between any of the groups of deficient mice, the researchers concluded that the two extrinsic tumor suppressor mechanisms heavily overlapped. RkSk mice, however, additionally developed spontaneous breast tumors that were not observed in wild-type or RAG-2-/mice, therefore demonstrating that the overlap between the two tumor suppressor pathways was not complete. Similar findings were made in carcinogenesis experiments with mice that lacked perforin, IFN- or both, in which a small increase was observed in tumor induction in the doubly deficient mice compared with mice lacking only one of the two components 42. Additional studies have used other inbred mouse lines with targeted disruptions in genes encoding critical components of the immune system. They not only support the importance of immune system control of tumor formation but also suggest the involvement of both the innate and adaptive immune compartments in cancer immunosurveillance. Specifically, genetic, immunochemical or functional ablations of NKT, T cells, NK cells, T cells, IFN- or interleukin 12 (IL-12) all lead to increased susceptibility of the host to tumors (Table 1). One of these studies has shed light on the specific subsets of lymphocytes expressing T cell receptors (TCR) that are important in tumor surveillance. The relative contributions of and T cells in blocking primary tumor formation cell-/- were explored in T (lacking the TCR -chain) or T cell-/- (lacking the TCR -chain) 50 mice . MCA treatment of either type of mouse increased the incidence of fibrosarcomas and spindle cell carcinomas as compared with wild-type controls. These data showed that both T cell subsets are critical for protecting the host in this particular model of tumor development. In an initiation and promotion model of skin tumorigenesis induced by 7,12-dimethylbenzanthracene (DMBA) and 12-O-tetradecanoylphorbol-13-acetate (TPA), however, T cell-/- mice showed a greater susceptibility to tumor formation and a higher incidence of papilloma-to-carcinoma progression than wild-type mice, whereas T cell-/- mice did not. This result suggests that immunosurveillance may be a heterogeneous process requiring the actions of different immune effectors in a manner that is dependent on the tumor's cell type of origin, mechanism of transformation, anatomical localization and mechanism of immunologic recognition. In sum, the large amount of recent data obtained by many independent groups overwhelmingly supports the basic tenets of the cancer immunosurveillance concept as originally envisaged by Burnet and Thomas—namely, that the unmanipulated immune system is capable of recognizing and eliminating primary tumors and that lymphocytes and the cytokines they produce are important in this process. High resolution image and legend (93K) Cancer immunosurveillance in humans If indeed cancer immunosurveillance exists in mice, does it exist in humans? The logical prediction from the immunosurveillance hypothesis is that immunodeficient or immunosuppressed humans should show greater incidences of cancer. Early follow-up studies of transplant patients who were immunosuppressed51 and individuals with primary immunodeficiencies52 showed that they had a significantly higher relative risk for cancer development. Based on long-term studies of patients, it is clear that some of this higher risk was due to the development of tumors of viral origin 53. For example, in the transplant registries from Cincinnatti53, Scandinavia54, and Australia and New Zealand55, particularly high incidence ratios have been observed for non-Hodgkin's lymphoma, Kaposi's sarcoma and carcinomas of the genitourinary and anogenital regions. Many of these cancers have a viral etiology and are linked to infection with Epstein-Barr virus, human herpesvirus 8 and human papilloma virus, respectively56. These malignancies are now occurring with increasing frequency in AIDS patients56. The increased incidence of virally induced tumors in immunocompromised patients represents one immunosurveillance function that is not contested: natural protection against infectious organisms. These data, however, do not invalidate the Table 1. Enhance possibility that the immune system protects humans against development of tumors of nonviral origin. Clearly, the difficulty in assessing cancer immunosurveillance in immunodeficient humans arises from their greater susceptibility to endemic viruses and other pathogens as well as viral reactivation. This can confound the likelihood of finding spontaneous tumors of nonviral origin, which develop slowly because of the shortened life spans and other intercurrent medical problems of these patients. Greater relative risk ratios have, however, been observed for a broad subset of tumors with no apparent viral etiology. Approximately fourfold increases in the incidence of de novo malignant melanoma after organ transplantation have been reported57, 58. A review of data accumulated by the Cincinnati Transplant Tumor Registry from 1968 to 1995 found a twofold greater risk in transplant patients for developing melanoma over that of the general population 59. In addition, whereas only 0.3–0.4% of melanomas occur in the general pediatric population, the occurrence in pediatric transplant patients was 4% 59. The same database showed that transplant patients were three times more likely to develop non-Kaposi's sarcomas60. When neoplasia occurrence was assessed in 608 cardiac transplant patients at the University of Pittsburgh between 1980 and 1993, the prevalence of lung tumors was 25-fold higher than in the general population61. In Australia and New Zealand, the tracking of 925 patients who received cadaveric renal transplants from 1965 to 1998 showed increased risk ratios for development of colon, pancreatic, lung and endocrine tumors as well as malignant melanomas55. In addition, assessment of 5,692 renal transplant patients from 1964 to 1982 in Finland, Denmark, Norway and Sweden showed higher standardized cancer incidence ratios for colon, lung, bladder, kidney, ureter and endocrine tumors as well as malignant melanomas as compared with the general population54. Thus, individuals with severe deficits of immunity indeed have a higher probability of developing a variety of cancers with no known viral etiology. In addition to the supporting epidemiological data described above, there is accumulating evidence showing a positive correlation between the presence of lymphocytes in a tumor and increased patient survival. Some of the most convincing evidence comes from the study of cutaneous melanomas. Three categories were established for tumor lymphocyte infiltration during the vertical growth phase of cutaneous melanoma (brisk, nonbrisk and absent)62. Sorting more than 500 patients with primary melanoma who had 5-, 8- or 10-year follow-ups into these categories and comparing their survival statistics showed that patients in the brisk tumor infiltrating lymphocyte (TIL) response category survived one and one-half to three times longer than patients in the absent TIL response group; patients in the nonbrisk response group had intermediate survival times62, 63. Researchers obtained the same prognostic correlation when the presence of TILs in melanomas that had metastasized to the lymph nodes was used as the criterion64. Similar correlations between the presence of TILs and patient survival have also been made that involved more than 3,400 patients with cancer of the breast65, bladder66, colon67, 68, prostate69, ovary70 or rectum71 and for neuroblastoma72. In some cases, the correlation has been refined to show that CD8+ T cells are the relevant lymphocyte population that affect survival68. In sum, the data obtained from both mouse and human studies provide strong support for the existence and physiologic relevance of cancer immunosurveillance. These findings have rekindled generalized interest in this process and have stimulated much work aimed at defining its immunologic components. Thus, the new millennium has witnessed the resurrection of this old and formerly controversial idea. Immunologic sculpting during tumor development Given the existence of cancer immunosurveillance, why then do cancers occur in immunocompetent individuals? It has long been thought that the immune system functions during tumor formation to select for tumor variants that are better suited to survive in an immunologically intact environment, very much like it does with viruses, bacteria and parasites. Many studies have shown the immunoselective effects of repassage of transplantable tumors through immunocompetent hosts and the generation of tumor variants with reduced immunogenicity. Two particularly informative studies used P815 mastocytomas73 and ultraviolet-induced 1591 fibrosarcomas74. In contrast, few studies have compared the immunogenic characteristics of tumors originally generated in the presence or absence of a functional immune system. This issue was difficult to study in the past because matched sets of tumors derived from isogenic immunodeficient and control immunocompetent hosts were not generally available. A recent study, however, was carried out using many MCA-induced sarcomas obtained from either RAG-2-/- or wild-type mice on a pure 129/SvEv background49. When tumors isolated from wild-type or RAG-2-/- mice were transplanted into RAG-2-/- recipients, they all grew with similar kinetics, which indicates that there were no inherent growth differences between tumors raised in the presence or absence of an intact immune system. In addition, 17 of 17 sarcomas originally isolated from wild-type mice were capable of establishing progressively growing tumors when transplanted into naïve immunocompetent 129/SvEv hosts. In contrast, 8 of 20 tumors originally generated in RAG-2-/- mice were rejected when transplanted into immunocompetent hosts, even when they were injected at a high cell number. Thus, tumors formed in the absence of an intact immune system are, as a group, more immunogenic than tumors that arise in immunocompetent hosts. Other more limited experiments have reached similar conclusions. MCA-induced sarcomas derived from nude75 or SCID mice37 were rejected more frequently than similar tumors derived from wildtype mice when transplanted into wild-type hosts. In addition, two MCA-induced sarcomas derived from TCR J 281-/- mice, which lack a major population of NKT cells, grew more slowly when transplanted into wild-type hosts than did sarcomas originally isolated from wild-type mice46. In contrast, these tumors grew in a comparable manner when transplanted into TCR J 281-/recipients. Finally, lymphomas derived from perforin-/- mice grew avidly when transplanted into perforin-/- mice, but most were rejected when transplanted into wild-type mice43. Taken together, these results show that tumors are imprinted by the immunologic environment in which they form. This imprinting process can often result in the generation of tumors that are better able to withstand the tumor-suppressing actions of the immune system by eliminating tumor cells of intrinsically high immunogenicity but leaving behind tumor variants of reduced immunogenicity (or that have acquired other mechanisms to evade or suppress immune attack) that have a better chance of surviving in the immunocompetent host. The alterations that must occur during the immunologic sculpting of a developing tumor are probably facilitated by the inherent genetic instability of tumors76. Some of the likely targets of the immunologic process that sculpts tumors are genes encoding tumor antigens, components of the major histocompatibility complex (MHC) pathways that process and present antigens or components of the IFN- receptor signaling pathway. Tumors lacking antigen processing and presentation components are commonly found (see Review by Khong and Restifo in this issue), and tumors have been identified that lack functional expression of at least three components of the IFN- receptor signaling pathway41 (unpublished data). It is likely that immunologic sculpting of tumors occurs continuously, but the major effects of this process are most prominent early when the tumor is perhaps histologically—but not clinically—detectable. It follows, then, that the immunogenicity of most tumors that are clinically apparent has already been modified to some degree by their interactions with the immune system. Cancer immunoediting Because the immune system exerts both host-protecting and tumor-sculpting effects on developing tumors, the term cancer immunosurveillance may no longer be appropriate to accurately describe the process because, in its original form, it was thought to function only to protect the host and to act only at the earliest stages of cellular transformation. Rather, we have proposed the use of the broader term "cancer immunoediting" to describe more appropriately the dual host-protecting and tumor-sculpting actions of the immune system that not only prevent but also shape neoplastic disease. We envisage the scope of this process to be very broad such that it can promote complete elimination of some tumors, generate a nonprotective immune state to others or favor the development of immunologic anergy, tolerance or indifference. We envision important roles for components of both the innate and adaptive immune systems in this process. Much work is needed to define the molecular and cellular dynamics of cancer immunoediting. A proposed basic framework for this process is illustrated based on recent data from several groups (Figs. 1 and 2). Although it was not possible to discuss in detail the contributions from each laboratory that were used in the construction of these models because of space limitations, we have incorporated key references in the model descriptions to acknowledge the work of these groups. The models are presented in the hope that they will stimulate additional research aimed at identifying the pathways that lead from cancer immunosurveillance to tumor escape. High resolution image and legend (56K) Figure 1. The three Es of cancer immunoediting. Cancer immunoediting encompasses three process. (a) Elimination corresponds to immunosurveillance. (b) Equilibrium represents the process by which the immune system iteratively selects and/or promotes the generation of tumor cell variants with increasing capacities to survive immune attack. (c) Escape is the process wherein the immunologically sculpted tumor expands in an uncontrolled manner in the immunocompetent host. In a and b, developing tumor cells (blue), tumor cell variants (red) and underlying stroma and nontransformed cells (gray) are shown; in c, additional tumor variants (orange) that have formed as a result of the equilibrium process are shown. Different lymphocyte populations are as marked. The small orange circles represent cytokines and the white flashes represent cytotoxic activity of lymphocytes against tumor cells. High resolution image and legend (128K) Figure 2. A proposed model for the elimination phase of the cancer immunoediting process. (a) The initiation of the response in which lymphocytes that participate in innate immunity (NKT, NK and T cells) recognize transformed cells that have accumulated above a threshold that has yet to be defined and are stimulated to produce IFN- . (b) The initial IFN- starts a cascade of innate immune reactions that involve (i) the induction of chemokines, including the angiostatic chemokines (CXCL10 (1P10), CXCL9 (MIG) AND CXCL11 (I-TAC)) that block neovascularization in the tumor and that also effect the recruitment of NK cells, dendritic cells, macrophages and other immune effector cells to the tumor site; (ii) an antiproliferative action of IFN- on the developing tumor and (iii) the activation of cytocidal activity in macrophages and NK cells entering the tumor. These events result in some tumor cell death by both immunologic and nonimmunologic mechanisms. Dead tumor cells or tumor cell debris (blue squares) are ingested by dendritic cells and are trafficked to the draining lymph node. (c) Tumor growth is kept in check by the cytocidal activities of NK cells and activated macrophages while CD4+ and CD8+ T cells that are specific for tumor antigens develop in the draining lymph node. (d) Tumor-specific CD4+ and CD8+ T cells home to the tumor along a chemokine gradient where they recognize and destroy tumor cells expressing distinctive tumor antigens. Tumor cells (blue); nontransformed cells (gray); dead tumor cells (white to gray gradient circles surrounded by a dashed black line); lymphocytes, dendritic cells (DC) and macrophages (Mac) are marked and colored appropriately. We envisage cancer immunoediting as a result of three processes: elimination, equilibrium and escape. We call these the three Es of cancer immunoediting (Fig. 1). Immunosurveillance occurs during the elimination process, whereas the Darwinian selection of tumor variants occurs during the equilibrium process. This in turn can ultimately lead to escape and the appearance of clinically apparent tumors. The elimination process encompasses the original concept of cancer immunosurveillance (Figs. 1a and 2). As such, when it is successful in deleting the developing tumor, it represents the complete editing process without progression to the subsequent phases. In the first phase of elimination (Fig. 2a), once solid tumors reach a certain size, they begin to grow invasively and require an enhanced blood supply that arises as a consequence of the production of stromagenic and angiogenic proteins77. Invasive growth causes minor disruptions within the surrounding tissue that induce inflammatory signals leading to recruitment of cells of the innate immune system (NKT, NK, T cells, macrophages and dendritic cells) into the site50, 78, 79. Structures on the transformed cells (either expressed as a result of the transformation process itself or induced by the ongoing but limited inflammatory response) are recognized by infiltrating lymphocytes such as NKT, NK or T cells, which are then stimulated to produce IFN- 80-82. In the second phase (Fig. 2b), the IFN- that was initially produced may induce a limited amount of tumor death by means of antiproliferative83 and apopotic84 mechanisms. However, it also induces the production of the chemokines CXCL10 (interferon-inducible protein-10, IP-10), CXCL9 (monokine induced by IFN- , MIG) and CXCL11 (interferon-inducible T cell chemoattractant, I-TAC) from the tumor cells themselves as well as from surrounding normal host tissues85-87. At least some of these chemokines have potent angiostatic capacities and thus block the formation of new blood vessels within the tumor, which leads to even more tumor cell death88-91. Tumor cell debris formed as either a direct or indirect consequence of IFN- production at the tumor is then ingested by local dendritic cells, which home to draining lymph nodes. Chemokines produced during the escalating inflammatory process recruit more NK cells and macrophages to the site. In the third phase (Fig. 2c), the tumor-infiltrating NK cells and macrophages transactivate one another by reciprocal production of IFN- and IL-12, and kill more of the tumor by mechanisms involving tumor necrosis factor–related apoptosis-inducing ligand, perforin and reactive oxygen and nitrogen intermediates 46, 92-95. In the draining lymph node, the newly immigrated dendritic cells induce tumor-specific CD4+ T helper cells expressing IFN(TH1 cells) that in turn facilitate the development of tumor-specific CD8+ T cells96-99. In the fourth phase (Fig. 2d), tumor-specific CD4+ and CD8+ T cells home to the tumor site, where the cytolytic T lymphocytes destroy the remaining antigen-bearing tumor cells whose immunogenicities have been enhanced by exposure to locally produced IFN- 49. In the equilibrium process (Fig. 1b), the host immune system and any tumor cell variant that has survived the elimination process enter into a dynamic equilibrium. In this process, lymphocytes and IFN- exert potent selection pressure on the tumor cells that is enough to contain, but not fully extinguish, a tumor bed containing many genetically unstable and rapidly mutating tumor cells. During this period of Darwinian selection, many of the original escape variants of the tumor cell are destroyed, but new variants arise carrying different mutations that provide them with increased resistance to immune attack. It is likely that equilibrium is the longest of the three processes and may occur over a period of many years. In the escape process (Fig. 1c), surviving tumor variants that have acquired insensitivity to immunologic detection and/or elimination through genetic or epigenetic changes begin to expand in an uncontrolled manner. This results in clinically observable malignant disease that, if left unchecked, results in the death of the host. Conclusions and implications We have summarized here the events that led to the development of the concept of cancer immunoediting and described the evolution of the concept from its birth in immunosurveillance to its present form. The original formulation of the cancer immunosurveillance hypothesis by Burnet and Thomas was indeed as prescient as it was powerful. Although it was based on an emerging understanding of the existence of tumor antigens and the laws of transplantation immunity, it came at a time when not enough was understood about mouse models of immunodeficiency. This led to the unwitting use of imperfect animal models to critically test and prematurely abandon the concept. The concept was resurrected nearly three decades later by the use of genetically defined mouse models of complete immunodeficiency coupled with an enhanced understanding of the molecular nature of tumor antigens. The incorporation of immunosurveillance into the cancer immunoediting concept came about by the finding that the immune system not only protects the host against tumor development but also can sculpt the immunogenic phenotype of a developing tumor. Thus, we now recognize a process that has both positive and negative effects on host–tumor relationships. Clearly, more work is needed to define the process on a molecular and cellular basis. There are four immediate implications of the cancer immunoediting concept that directly apply to human cancer. First, we may need to examine whether immunity plays a role in the development of human cancers associated with known abnormalities in genes encoding such proteins as p53, BRCA1, APC (adenomatous polyposis coli) Ras and others. Second, it will be important to reexamine the carcinogenic potential of many compounds that have never been tested in experimental animals that are immunodeficient and thus may have been incorrectly labeled as noncarcinogenic. Third, the relation between immunologic defects and increased incidences of cancer associated with aging needs to be explored. Last, we need a way to assess the extent to which a tumor has been edited. Perhaps one of the most important contributions of the original immunosurveillance concept was the furious experimentation that followed in its wake. In this respect, we hope for a similar fate for the cancer immunoediting concept—new oeuvres framed by the three Es that test every prediction of this hypothesis and, in eliciting a greater understanding of host–tumor interactions, will ultimately have profound effects on our understanding and treatment of cancer. References 1. Ehrlich, P. Ueber den jetzigen stand der Karzinomforschung. Ned. Tijdschr. Geneeskd. 5, 273-290 (1909). 2. Silverstein, A.M. A History of Immunology (Academic, San Diego, CA, 1989). 3. Old, L.J. & Boyse, E.A. Immunology of experimental tumors. Annu. Rev. Med. 15, 167-186 (1964). | ISI | 4. Klein, G. Tumor antigens. Annu. Rev. Microbiol. 20, 223-252 (1966). | PubMed | ISI | 5. Burnet, F.M. Cancer--a biological approach. Brit. Med. J. 1, 841-847 (1957). 6. Thomas, L. in Cellular and Humoral Aspects of the Hypersensitive States (ed. Lawrence, H. S.) 529-532 (Hoeber-Harper, New York, 1959). 7. Burnet, F.M. The concept of immunological surveillance. Prog. Exp. Tumor Res. 13, 1-27 (1970). | PubMed | ISI | 8. Burnet, F.M. Immunological factors in the process of carcinogenesis. Br. Med. Bull. 20, 154158 (1964). 9. Kaplan, H.S. Role of immunologic disturbance in human oncogenesis: some facts and fancies. Br. J. Cancer 25, 620-634 (1971). | PubMed | ISI | 10. Stutman, O. Immunodepression and malignancy. Adv. Cancer Res. 22, 261-422 (1975). | PubMed | 11. Grant, G.A. & Miller, J.F. Effect of neonatal thymectomy on the induction of sarcomata in C57BL mice. Nature 205, 1124-1125 (1965). | PubMed | ISI | 12. Nishizuka, Y., Nakakuki, K. & Usui, M. Enhancing effect of thymectomy on hepatotumorigenesis in Swiss mice following neonatal injection of 20-methylcholanthrene. Nature 205, 1236-1238 (1965). | ISI | 13. Trainin, N., Linker-Israeli, M., Small, M. & Boiato-Chen, L. Enhancement of lung adenoma formation by neonatal thymectomy in mice treated with 7,12-dimethylbenz(a)anthracene or urethan. Int. J. Cancer 2, 326-336 (1967). | PubMed | ISI | 14. Burstein, N.A. & Law, L.W. Neonatal thymectomy and non-viral mammary tumours in mice. Nature 231, 450-452 (1971). | PubMed | ISI | 15. Sanford, B.H., Kohn, H.I., Daly, J.J. & Soo, S.F. Long-term spontaneous tumor incidence in neonatally thymectomized mice. J. Immunol. 110, 1437-1439 (1973). | PubMed | ISI | 16. Klein, G. Immunological surveillance against neoplasia. Harvey Lect., 71-102 (1973). | PubMed | 17. Flanagan, S.P. 'Nude', a new hairless gene with pleiotropic effects in the mouse. Genet. Res. 8, 295-309 (1966). | PubMed | ISI | 18. Pantelouris, E.M. Absence of thymus in a mouse mutant. Nature 217, 370-371 (1968). | PubMed | ISI | 19. Stutman, O. Tumor development after 3-methylcholanthrene in immunologically deficient athymic-nude mice. Science 183, 534-536 (1974). | PubMed | ISI | 20. Stutman, O. in Proceedings of the International Workshop on Nude Mice Vol. 1 (eds. Rygaard, J. & Poulsen, C.) 257-264 (Gustav Fischer, Stuttgart 1973). 21. Stutman, O. Chemical carcinogenesis in nude mice: comparison between nude mice from homozygous and heterozygous matings and effect of age and carcinogen dose. J. Natl. Cancer Inst. 2, 353-358 (1979). 22. Outzen, H.C., Custer, R.P., Eaton, G.J. & Prehn, R.T. Spontaneous and induced tumor incidence in germfree "nude" mice. J. Reticuloendothel. Soc. 17, 1-9 (1975). | PubMed | ISI | 23. Stutman, O. in The Nude Mouse in Experimental and Clinical Research (eds. Fogh, J. & Giovanella, B. C.) 411-435 (Academic, New York, 1978). 24. Rygaard, J. & Povlsen, C.O. Is immunological surveillance not a cell-mediated immune function? Transplantation 17, 135-136 (1974). | PubMed | ISI | 25. Rygaard, J. & Povlsen, C.O. The mouse mutant nude does not develop spontaneous tumours. An argument against immunological surveillance. Acta Pathol. Microbiol. Scand. [B] Microbiol. Immunol. 82, 99-106 (1974). | PubMed | ISI | 26. Maleckar, J.R. & Sherman, L.A. The composition of the T cell receptor repertoire in nude mice. J. Immunol. 138, 3873-3876 (1987). | PubMed | ISI | 27. Ikehara, S., Pahwa, R.N., Fernandes, G., Hansen, C.T. & Good, R.A. Functional T cells in athymic nude mice. Proc. Natl. Acad. Sci. USA 81, 886-888 (1984). | PubMed | ISI | 28. Hunig, T. T-cell function and specificity in athymic mice. Immunol. Today 4, 84-87 (1983). | ISI | 29. Heidelberger, C. Chemical carcinogenesis. Annu. Rev. Biochem. 44, 79-121 (1975). | PubMed | ISI | 30. Kouri, R.E. & Nebert, D.W. in Origins of Human Cancer (eds. Hiatt, H. H., Watson, J. D. & Winsten, J. A.) 811-835 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 1977). 31. Hayday, A.C. cells: a right time and a right place for a conserved third way of protection. Annu. Rev. Immunol. 18, 975-1026 (2000). | Article | PubMed | ISI | 32. Prehn, R.T. Perspectives on oncogenesis: does immunity stimulate or inhibit neoplasia? J. Reticuloenothel. Soc. 10, 1-16 (1970). 33. Thomas, L. On immunosurveillance in human cancer. Yale J. Biol. Med. 55, 329-333 (1982). | PubMed | ISI | 34. Hanahan, D. & Weinberg, R.A. The hallmarks of cancer. Cell 100, 57-70 (2000). | PubMed | ISI | 35. Herberman, R.B. & Holden, H.T. Natural cell-mediated immunity. Adv. Cancer Res. 27, 305377 (1978). | PubMed | 36. Engel, A.M. et al. Methylcholanthrene-induced sarcomas in nude mice have short induction times and relatively low levels of surface MHC class I expression. APMIS 104, 629-639 (1996). | PubMed | ISI | 37. Engel, A.M., Svane, I.M., Rygaard, J. & Werdelin, O. MCA sarcomas induced in scid mice are more immunogenic than MCA sarcomas induced in congenic, immunocompetent mice. Scand. J. Immunol. 45, 463-470 (1997). | PubMed | ISI | 38. Schuler, W. et al. Rearrangement of antigen receptor genes is defective in mice with severe combined immune deficiency. Cell 46, 963-972 (1986). | PubMed | ISI | 39. Featherstone, C. & Jackson, S.P. DNA double-strand break repair. Curr. Biol. 9, R759-R761 (1999). | Article | PubMed | ISI | 40. Dighe, A.S., Richards, E., Old, L.J. & Schreiber, R.D. Enhanced in vivo growth and resistance to rejection of tumor cells expressing dominant negative IFN- receptors. Immunity 1, 447-456 (1994). | PubMed | ISI | 41. Kaplan, D.H. et al. Demonstration of an interferon -dependent tumor surveillance system in immunocompetent mice. Proc. Natl. Acad. Sci. USA 95, 7556-7561 (1998). | Article | PubMed | ISI | 42. Street, S.E., Cretney, E. & Smyth, M.J. Perforin and interferon- activities independently control tumor initiation, growth, and metastasis. Blood 97, 192-197 (2001). | Article | PubMed | ISI | 43. Street, S.E., Trapani, J.A., MacGregor, D. & Smyth, M.J. Suppression of lymphoma and epithelial malignancies effected by interferon . J. Exp. Med. 196, 129-134 (2002). | Article | PubMed | ISI | 44. Russell, J.H. & Ley, T.J. Lymphocyte-mediated cytotoxicity. Annu. Rev. Immunol. 20, 323370 (2002). | Article | PubMed | ISI | 45. van den Broek, M.F. et al. Decreased tumor surveillance in perforin-deficient mice. J.Exp.Med. 184, 1781-1790 (1996). | ISI | 46. Smyth, M.J. et al. Differential tumor surveillance by natural killer (NK) and NKT cells. J. Exp. Med. 191, 661-668 (2000). | Article | PubMed | ISI | 47. Smyth, M.J. et al. Perforin-mediated cytotoxicity is critical for surveillance of spontaneous lymphoma. J. Exp. Med. 192, 755-760 (2000). | Article | PubMed | ISI | 48. Shinkai, Y. et al. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell 68, 855-867 (1992). | PubMed | ISI | 49. Shankaran, V. et al. IFN- and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410, 1107-1111 (2001). | Article | PubMed | ISI | 50. Girardi, M. et al. Regulation of cutaneous malignancy by T cells. Science 294, 605-609 (2001). | Article | PubMed | ISI | 51. Penn, I. Malignant Tumors in Organ Transplant Recipients (Springer-Verlag, New York, 1970). 52. Gatti, R.A. & Good, R.A. Occurrence of malignancy in immunodeficiency diseases. A literature review. Cancer 28, 89-98 (1971). | PubMed | ISI | 53. Penn, I. Posttransplant malignancies. Transplant Proc. 31, 1260-1262 (1999). | Article | PubMed | ISI | 54. Birkeland, S.A. et al. Cancer risk after renal transplantation in the Nordic countries, 19641986. Int. J. Cancer 60, 183-189 (1995). | PubMed | ISI | 55. Sheil, A.G.R. in Kidney Transplantation (ed. Morris, P. J.) 558-570 (Saunders, Philadelphia, 2001). 56. Boshoff, C. & Weiss, R. AIDS-related malignancies. Nature Rev. Cancer 2, 373-382 (2002). | Article | PubMed | 57. Hoover, R.N. in Origins of Human Cancer (eds. Hiatt, H. H., Watson, J. D. & Winsten, J. A.) 369-379 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 1977). 58. Sheil, A.G. Cancer after transplantation. World J. Surg. 10, 389-396 (1986). | PubMed | ISI | 59. Penn, I. Malignant melanoma in organ allograft recipients. Transplantation 61, 274-278 (1996). | PubMed | ISI | 60. Penn, I. Sarcomas in organ allograft recipients. Transplantation 60, 1485-1491 (1995). | PubMed | ISI | 61. Pham, S.M. et al. Solid tumors after heart transplantation: lethality of lung cancer. Ann. Thorac. Surg. 60, 1623-1626 (1995). | Article | PubMed | ISI | 62. Clark, W.H. Jr et al. Model predicting survival in stage I melanoma based on tumor progression. J. Natl. Cancer Inst. 81, 1893-1904 (1989). | PubMed | ISI | 63. Clemente, C.G. et al. Prognostic value of tumor infiltrating lymphocytes in the vertical growth phase of primary cutaneous melanoma. Cancer 77, 1303-1310 (1996). | Article | PubMed | ISI | 64. Mihm, M.C. Jr, Clemente, C.G. & Cascinelli, N. Tumor infiltrating lymphocytes in lymph node melanoma metastases: a histopathologic prognostic indicator and an expression of local immune response. Lab. Invest. 74, 43-47 (1996). | PubMed | ISI | 65. Rilke, F. et al. Prognostic significance of HER-2/neu expression in breast cancer and its relationship to other prognostic factors. Int. J. Cancer 49, 44-49 (1991). | PubMed | ISI | 66. Lipponen, P.K., Eskelinen, M.J., Jauhiainen, K., Harju, E. & Terho, R. Tumour infiltrating lymphocytes as an independent prognostic factor in transitional cell bladder cancer. Eur. J. Cancer 29A, 69-75 (1992). | PubMed | 67. Nacopoulou, L., Azaris, P., Papacharalampous, N. & Davaris, P. Prognostic significance of histologic host response in cancer of the large bowel. Cancer 47, 930-936 (1981). | PubMed | ISI | 68. Naito, Y. et al. CD8+ T cells infiltrated within cancer cell nests as a prognostic factor in human colorectal cancer. Cancer Res. 58, 3491-3494 (1998). | PubMed | ISI | 69. Epstein, N.A. & Fatti, L.P. Prostatic carcinoma: some morphological features affecting prognosis. Cancer 37, 2455-2465 (1976). | PubMed | ISI | 70. Deligdisch, L., Jacobs, A.J. & Cohen, C.J. Histologic correlates of virulence in ovarian adenocarcinoma. II. Morphologic correlates of host response. Am. J. Obstet. Gynecol. 144, 885889 (1982). | PubMed | ISI | 71. Jass, J.R. Lymphocytic infiltration and survival in rectal cancer. J. Clin. Pathol. 39, 585-589 (1986). | PubMed | ISI | 72. Palma, L., Di Lorenzo, N. & Guidetti, B. Lymphocytic infiltrates in primary glioblastomas and recidivous gliomas. Incidence, fate, and relevance to prognosis in 228 operated cases. J. Neurosurg. 49, 854-861 (1978). | PubMed | ISI | 73. Uyttenhove, C., Van Snick, J. & Boon, T. Immunogenic variants obtained by mutagenesis of mouse mastocytoma P815. I. Rejection by syngeneic mice. J. Exp. Med. 152, 1175-1183 (1980). | PubMed | ISI | 74. Urban, J.L., Holland, J.M., Kripke, M.L. & Schreiber, H. Immunoselection of tumor cell variants by mice suppressed with ultraviolet radiation. J. Exp. Med. 156, 1025-1041 (1982). | PubMed | ISI | 75. Svane, I.M. et al. Chemically induced sarcomas from nude mice are more immunogenic than similar sarcomas from congenic normal mice. Eur. J. Immunol. 26, 1844-1850 (1996). | PubMed | ISI | 76. Lengauer, C., Kinzler, K.W. & Vogelstein, B. Genetic instabilities in human cancers. Nature 396, 643-649 (1998). | Article | PubMed | ISI | 77. Hanahan, D. & Folkman, J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 86, 353-364 (1996). | PubMed | ISI | 78. Smyth, M.J., Godfrey, D.I. & Trapani, J.A. A fresh look at tumor immunosurveillance and immunotherapy. Nature Immunol. 2, 293-299 (2001). | Article | PubMed | ISI | 79. Matzinger, P. Tolerance, danger, and the extended family. Annu. Rev. Immunol. 12, 9911045 (1994). | Article | PubMed | ISI | 80. Yokoyama, W.M. Now you see it, now you don't! Nature Immunol. 1, 95-97 (2000). | Article | PubMed | ISI | 81. Cerwenka, A. et al. Retinoic acid early inducible genes define a ligand family for the activating NKG2D receptor in mice. Immunity 12, 721-727 (2000). | PubMed | ISI | 82. Diefenbach, A., Jensen, E.R., Jamieson, A.M. & Raulet, D.H. Rae1 and H60 ligands of the NKG2D receptor stimulate tumour immunity. Nature 413, 165-171 (2001). | Article | PubMed | ISI | 83. Bromberg, J.F., Horvath, C.M., Wen, Z., Schreiber, R.D. & Darnell, J.E. Jr. Transcriptionally active Stat1 is required for the antiproliferative effects of both interferon and interferon . Proc. Natl. Acad. Sci. USA 93, 7673-7678 (1996). | Article | PubMed | ISI | 84. Kumar, A., Commane, M., Flickinger, T.W., Horvath, C.M. & Stark, G.R. Defective TNF- induced apoptosis in STAT1-null cells due to low constitutive levels of caspases. Science 278, 1630-1632 (1997). | Article | PubMed | ISI | 85. Luster, A.D. & Ravetch, J.V. Biochemical characterization of a interferon-inducible cytokine (IP-10). J. Exp. Med. 166, 1084-1097 (1987). | PubMed | ISI | 86. Liao, F. et al. Human Mig chemokine: biochemical and functional characterization. J. Exp. Med. 182, 1301-1314 (1995). | PubMed | ISI | 87. Cole, K.E. et al. Interferon-inducible T cell chemoattractant (I-TAC): a novel non-ELR CXC chemokine with potent activity on activated T cells through selective high affinity binding to CXCR3. J. Exp. Med. 187, 2009-2021 (1998). | Article | PubMed | ISI | 88. Luster, A.D. & Leder, P. IP-10, a -C-X-C- chemokine, elicits a potent thymus-dependent antitumor response in vivo. J. Exp. Med. 178, 1057-1065 (1993). | PubMed | ISI | 89. Sgadari, C., Angiolillo, A.L. & Tosato, G. Inhibition of angiogenesis by interleukin-12 is mediated by the interferon-inducible protein 10. Blood 87, 3877-3882 (1996). | PubMed | ISI | 90. Coughlin, C.M. et al. Tumor cell responses to IFN- affect tumorigenicity and response to IL12 therapy and antiangiogenesis. Immunity 9, 25-34 (1998). | PubMed | ISI | 91. Qin, Z. & Blankenstein, T. CD4+ T cell-mediated tumor rejection involves inhibition of angiogenesis that is dependent on IFN- receptor expression by nonhematopoietic cells. Immunity 12, 677-686 (2000). | PubMed | ISI | 92. Bancroft, G.J., Schreiber, R.D. & Unanue, E.R. Natural immunity: a T-cell-independent pathway of macrophage activation, defined in the scid mouse. Immunol. Rev. 124, 5-24 (1991). | PubMed | ISI | 93. Trinchieri, G. Interleukin-12: a proinflammatory cytokine with immunoregulatory functions that bridge innate resistance and antigen-specific adaptive immunity. Annu. Rev. Immunol. 13, 251-276 (1995). | Article | PubMed | ISI | 94. Ikeda, H., Old, L.J. & Schreiber, R.D. The roles of IFN- in protection against tumor development and cancer immunoediting. Cytokine Growth Factor Rev. 13, 95-109 (2002). | Article | PubMed | ISI | 95. Takeda, K. et al. Critical role for tumor necrosis factor-related apoptosis-inducing ligand in immune surveillance against tumor development. J. Exp. Med. 195, 161-169 (2002). | Article | PubMed | ISI | 96. Pardoll, D.M. Spinning molecular immunology into successful immunotherapy. Nature Rev. Immunol. 2, 227-238 (2002). | Article | PubMed | 97. Gerosa, F. et al. Reciprocal activating interaction between natural killer cells and dendritic cells. J. Exp. Med. 195, 327-333 (2002). | Article | PubMed | ISI | 98. Ferlazzo, G. et al. Human dendritic cells activate resting natural killer (NK) cells and are recognized via the NKp30 receptor by activated NK cells. J. Exp. Med. 195, 343-351 (2002). | Article | PubMed | ISI | 99. Piccioli, D., Sbrana, S., Melandri, E. & Valiante, N.M. Contact-dependent stimulation and inhibition of dendritic cells by natural killer cells. J. Exp. Med. 195, 335-341 (2002). | Article | PubMed | ISI | 100. Smyth, M.J., Crowe, N.Y. & Godfrey, D.I. NK cells and NKT cells collaborate in host protection from methylcholanthrene-induced fibrosarcoma. Int. Immunol. 13, 459-463 (2001). | Article | PubMed | ISI | 101. Noguchi, Y., Jungbluth, A., Richards, E. & Old, L.J. Effect of interleukin 12 on tumor induction by 3-methylcholanthrene. Proc. Natl. Acad. Sci. USA 93, 11798-11801 (1996). | Article | PubMed | ISI | Acknowledgments. Supported by grants from the National Cancer Institute (CA43059 and CA76464 to R. D. S.), the Cancer Research Institute (to R. D. S., H.I and A.B.), the Ludwig Institute for Cancer Research (to R. D. S.), and the National Institute of Allergy and Infectious Diseases (to R. D. S. and G. P. D.). We thank V. Shankaran, K. Sheehan, A. Dighe, D. Kaplan, R. Uppaluri, C. Koebel, J. Bui, E. Stockert, E. Richards, M. White, C. Arthur and C. Brendel for their important roles in developing the cancer immunoediting concept and for helpful comments during the preparation of this manuscript. Table 1. Enhanced susceptibility of immunodeficient mice to formation of chemically induced and spontaneous tumors Figure 1. The three Es of cancer immunoediting. Cancer immunoediting encompasses three process. (a) Elimination corresponds to immunosurveillance. (b) Equilibrium represents the process by which the immune system iteratively selects and/or promotes the generation of tumor cell variants with increasing capacities to survive immune attack. (c) Escape is the process wherein the immunologically sculpted tumor expands in an uncontrolled manner in the immunocompetent host. In a and b, developing tumor cells (blue), tumor cell variants (red) and underlying stroma and nontransformed cells (gray) are shown; in c, additional tumor variants (orange) that have formed as a result of the equilibrium process are shown. Different lymphocyte populations are as marked. The small orange circles represent cytokines and the white flashes represent cytotoxic activity of lymphocytes against tumor cells. Figure 2. A proposed model for the elimination phase of the cancer immunoediting process. (a) The initiation of the response in which lymphocytes that participate in innate immunity (NKT, NK and T cells) recognize transformed cells that have accumulated above a threshold that has yet to be defined and are stimulated to produce IFN- . (b) The initial IFN- starts a cascade of innate immune reactions that involve (i) the induction of chemokines, including the angiostatic chemokines (CXCL10 (1P10), CXCL9 (MIG) AND CXCL11 (I-TAC)) that block neovascularization in the tumor and that also effect the recruitment of NK cells, dendritic cells, macrophages and other immune effector cells to the tumor site; (ii) an antiproliferative action of IFN- on the developing tumor and (iii) the activation of cytocidal activity in macrophages and NK cells entering the tumor. These events result in some tumor cell death by both immunologic and nonimmunologic mechanisms. Dead tumor cells or tumor cell debris (blue squares) are ingested by dendritic cells and are trafficked to the draining lymph node. (c) Tumor growth is kept in check by the cytocidal activities of NK cells and activated macrophages while CD4+ and CD8+ T cells that are specific for tumor antigens develop in the draining lymph node. (d) Tumor-specific CD4+ and CD8+ T cells home to the tumor along a chemokine gradient where they recognize and destroy tumor cells expressing distinctive tumor antigens. Tumor cells (blue); nontransformed cells (gray); dead tumor cells (white to gray gradient circles surrounded by a dashed black line); lymphocytes, dendritic cells (DC) and macrophages (Mac) are marked and colored appropriately. Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 999 - 1005 (2002) doi:10.1038/ni1102-999 © Nature America, Inc. <> Natural selection of tumor variants in the generation of "tumor escape" phenotypes Hung T. Khong & Nicholas P. Restifo National Cancer Institute, National Institutes of Health, Bethesda, MD 20892, USA. Correspondence should be addressed to H T Khong khong@nih.gov or N P Restifo restifo@nih.gov The idea that tumors must "escape" from immune recognition contains the implicit assumption that tumors can be destroyed by immune responses either spontaneously or as the result of immunotherapeutic intervention. Simply put, there is no need for tumor escape without immunological pressure. Here, we review evidence supporting the immune escape hypothesis and critically explore the mechanisms that may allow such escape to occur. We discuss the idea that the central engine for generating immunoresistant tumor cell variants is the genomic instability and dysregulation that is characteristic of the transformed genome. "Natural selection" of heterogeneous tumor cells results in the survival and proliferation of variants that happen to possess genetic and epigenetic traits that facilitate their growth and immune evasion. Tumor escape variants are likely to emerge after treatment with increasingly effective immunotherapies. Progress in molecular and cellular immunology during the past two decades has advanced our understanding of tumor-host interactions and opened extraordinary opportunities for the development of antitumor immunotherapies. The identification of tumor antigens has allowed us, for the first time, to tailor therapies to specific molecular targets expressed on tumor cells. In addition, advances in recombinant biotechnology have enabled us to design and develop more effective cancer vaccines for active immunization and cellular therapy for adoptive-transfer treatments. However, consistently effective immunotherapy has not yet been developed for any type of malignancy. Although some maintain that the lack of broad success with current strategies is due to the "escape" of tumor cells from the immunotherapies directed against them, it is also possible—and even likely—that current immune-based treatments for malignancy are simply ineffective. For example, current T cell–based immunotherapies may not be eliciting enough of the right kinds of T cells in the right places at the right time to allow them to be consistently effective. To fully understand the potential role played by tumor escape in the failure of immunotherapeutic treatments in cancer patients, it is worth briefly discussing the basic principles of immunosurveillance and its potential role in shaping tumor phenotypes. Assumptions of the tumor escape hypothesis It is not often explicitly stated that the tumor escape hypothesis assumes that if left unchecked, the immune system would indeed spontaneously attack and destroy tumor. Simply put, immune escape implies immune attack. A Review by Schreiber and colleagues in this issue examines the evidence for immunosurveillance and attempts to resolve the long-standing controversy over its role in the early stages of tumor development. Schreiber's group has demonstrated the existence of an interferon- (IFN- )–dependent extrinsic tumor suppressor system in mice1. However, it is not clear how large a role this plays in the immunosurveillance of tumors because only a minority of human tumor cell lines examined in this study exhibited a permanent and selective IFN- insensitivity1. Two studies have examined the potential roles of natural killer (NK) and T cells bearing the stimulatory lectin-like receptor NKG2D in immunosurveillance. The first study explored antitumor immune responses elicited by tumors cells transduced with the NKG2D ligands, retinoic acid early transcript 1b (Rae-1b) or H-602 and found that NK and/or CD8+ T cells mediated potent antitumor immunity2. The study used NKG2D ligand–transduced cell lines that expressed high amounts of Rae-1b or H-60; NKG2D ligand expression in most spontaneous tumors may be much lower. Nevertheless, these findings demonstrated a possible mechanism of immunosurveillance that may involve NK cells. A second study uncovered a possible role for NKG2D + stages of tumor development3. T cells at the very early A note of caution is useful when interpreting these elegant mouse studies: there may be fundamental differences between the development of tumors injected into a mouse or induced by chemical carcinogens compared to naturally occurring spontaneous tumors in humans. For example, the act of injection itself and the presence of dead tumor cells as well as tissue-culture contaminants, such as fetal bovine serum in the inoculum, may induce local inflammatory reactions. In tumors induced by painting chemical carcinogens onto the skin of highly susceptible mice, Rae1b and H-60 expression may be up-regulated, resulting in their rejection by T cells. In addition, whereas carcinogenesis in humans may be a slow process that occurs over several years, mouse experiments use high doses of carcinogens, which cause transformation in just weeks or months. At these doses, carcinogens may cause massive and rapid mutagenesis, resulting in greatly increased number of neoantigens that may alert the immune system. IFN- , perforin and tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) act as effector molecules in immune surveillance for the prevention of tumor development. Mice that are deficient in lymphocytes and/or in the IFN- signaling pathway have a much higher incidence of carcinogen-induced sarcomas, lymphoma and spontaneous epithelial tumors 4, 5. Other studies have shown that in perforin-deficient mice, there is a higher rate of spontaneous lymphoma and lung adenocarcinoma4, 6. Upon transplantation into syngeneic wild-type mice, these lymphoma cells are promptly rejected by cytotoxic T lymphocyte (CTL)-mediated activity. Inhibition of TRAIL (by blocking antibody) promotes carcinogen-induced tumor development in mice; large numbers of these tumors are TRAIL-sensitive, which is something rarely seen in tumors developed in wild-type mice7. Thus, evidence exists, in some models and under some conditions, that immunosurveillance can play an active role in suppressing the growth of very early tumors. In this paradigm, when tumors do successfully grow, they are thought of as having "escaped" from this immunosurveillance; these early surveillance mechanisms are viewed as "shaping" the tumor's immunological phenotype. Another model that explains why established tumors grow and do not undergo immune-mediated rejection is that tumors function as immunologically normal tissue. In this paradigm, tumor cells appear immunologically as healthy growing cells that do not send out danger signals to activate the immune system because they express neither microbial immune-recognition patterns nor release distress signals to alarm the innate immune cells8. Tumor histology and phenotype may determine whether early tumors ultimately grow as a result of stealth and nonrecognition or as the result of escape and immunological sculpting. Later in the natural history of a tumor, during the progressive growth phase, tumors may become more immune-activating for a variety of reasons (Fig. 1). Tumors can damage or disrupt surrounding tissue or trigger a stress response when they outstrip their oxygen and nutrient supplies. These processes can cause pH imbalance that results from metabolic disturbance, generation of reactive oxygen species (ROS), up-regulation of stress protective factors—such as heat shock proteins—and death by necrosis or apoptosis. Also, as tumors grow progressively, dysregulated genetic and epigenetic events lead to the expression of large numbers of neoantigens. One study estimated that the average malignancy contained more than 10,000 mutations9. All these factors may act as alarm signals to recruit and activate local innate immune cells such as dendritic cells (DCs), macrophages, neutrophils and NK cells, which, in turn, activate T cells to mount an adaptive immune response against tumors. High resolution image and legend (58K) tumor progression. Figure 1. Activation versus suppression during Immune activation during early tumor progression may be triggered by the expression of neoantigens. In addition, the progressive growth of tumors may be associated with the invasion and destruction of surrounding normal tissues. Other factors that may cause immune activation include generation of heat shock proteins (Hsps), which result from cellular stress, and ROS such as OH and H2O2. Conversely, immune activation and function may be hampered by a lack of appropriate costimulation, the presence of immunosuppressive cytokines (for example, VEGF, IL-10 and TGF), and the impact of immunoregulatory cells such as CD4+CD25+ T cells and NKT cells. As tumors grow, these events occur in the presence of massive apoptosis and necrosis of tumor cells, infiltrating immune cells and surrounding stromal cells. The fate of tumors may be dictated by the net effect of immune activation and immune inhibition. Whatever the roles of immunosurveillance, immune attack and immune escape in humans, it is clear that once tumors are clinically detectable, spontaneous regression is exceedingly rare for the vast majority of histologies. Once solid tumors become established, vascularized and clinically detectable, it is simply not clear—given the available evidence—whether they need to "escape" from immune recognition; this is because it is unclear whether most tumors elicit immune responses that can cause tumor destruction8. Indeed, some observations contradict the idea that the immune system spontaneously mounts lethal attacks against tumor cells. If cycles of immune pressure and tumor escape were operative during tumor development, one might expect to observe (either by examination or by imaging modalities) progressive tumor growth that was interspersed with one or more periods of contraction. Ongoing immunological "shaping" or "sculpting" of tumors might be expected to result in the destruction of sensitive cells followed by proliferation of immunoresistant cells that would, in turn, form the bulk of a new tumor emerging from the bed of the old tumor (Fig. 2). However, solid tumors generally do not have growth curves with evidence of significant drops or depressions: tumors simply grow, and then grow larger. High resolution image and legend (38K) Figure 2. Natural selection of tumor variants in the generation of "tumor escape" phenotypes. (a) Genomic instability gives rise to genetic diversity in tumors. Natural selection of tumor variants occurs by differential propagation of tumor subclones in their microenvironment. (b) The same concept also applies to tumor growth after effective immunotherapy. On the other hand, it is also possible that selection is taking place at a cellular level, especially during early tumor development, as discussed above. Ongoing selection of tumor variants on a microscopic or cellular level would not necessarily result in gross or macroscopic changes, such as those easily seen histologically or with commonly used imaging modalities. Similarly, a lack of inflammation at the site of solid tumors may be interpreted by some to cast further doubt on the hypotheses of spontaneous immune attack and immune escape. Although immune cells can be observed in or around tumors, spontaneous local inflammation in an uninfected tumor is generally not seen clinically or histologically. Again, however, it is possible that this inflammation is occurring chronically and on a microscopic level. Thus, there is little direct evidence in mice or humans to support the concept of tumor "shaping" or "sculpting" by the unmanipulated immune system once tumors are vascularized and established. Nevertheless, these hypothesized processes may well turn out to be important upon further investigation. The natural selection of tumor cell variants The setting of partially successful antitumor immunotherapy is one scenario that might be expected to result in tumor escape. One of the most important factors that favors the survival and propagation of evolving organisms is genetic diversity. It is interesting to view the survival of cancer cells in a Darwinian light, where the survival of individual cancer cells is based on heritable variations and differential survival associated with certain genotypes. Natural selection is a passive process by which an organism has a survival advantage simply by possessing certain beneficial genes. Genomic instability, an inherent property shared by all tumors, gives rise to genetic diversity, as manifested by the great degree of heterogeneity seen in human tumors. The law of natural selection is at work when cancer cells that possess genetic and epigenetic traits, which are beneficial to their survival and/or proliferation, have a growth advantage over other cells. The outcome of this passive process is determined by multiple factors in the tumor environment, such as growth factors, nutrient supply and immune pressure. Genomic instability simply creates a vast repertoire of tumor cells that will be selected according to environmental factors. Therefore, terms such as "tumor evasion" or "tumor escape" are misnomers because evasion and escape imply active acquisition of certain characteristics or phenotypes, rather than the differential propagation of tumor subclones (Fig. 2a). The same concept also applies to tumor growth in the face of an effective immunotherapy (Fig. 2b). Loss or down-regulation of HLA class I antigens It is certainly well documented that tumor cells can indeed lose HLA class I molecules through a panoply of mechanisms. Descriptions of HLA loss causing immune escape have a great deal of intuitive appeal, but are nevertheless correlative and indirect. There is little controlled evidence in humans or animals that loss of major histocompatibility complex (MHC) class I molecules actually leads to tumor escape or immunoresistance in the unmanipulated host. Indeed, loss of H-2 expression in transporter associated with antigen processing 1 (TAP-1)– or low-molecular weight protein 2 (LMP-2)–deficient mice does not increase the onset or incidence of a variety of spontaneous tumors10. On the other hand, some recurring tumors lose cell-surface MHC class I both in mice with pre-existing immunity induced by immunization with B7-1–transfected tumor cells11 and in humans after partial responses to T cell–based immunotherapy12. Decreased or absent HLA class I expression is associated with invasive and metastatic lesions13. Total loss of HLA class I expression is not uncommon in many tumors, including melanoma, colorectal carcinoma and prostate adenocarcinoma14. In breast carcinoma, the frequency of total HLA class I loss is >50%15. Thus, it is possible that in antigen-loss, HLA-loss and MHC class I processing– defective variants, the loss or down-regulation of HLA class I antigens occurs as a result of immunological "sculpting" of early tumor lesions (see the Review by Schreiber and colleagues in this issue) or escape from immune attack later in tumor development. Some mechanisms that can result in the loss or down-regulation of HLA class I expression are shown (Fig. 3). The mechanisms that underlie total loss of HLA class I include mutations in one copy of the 2-microglobulin gene in association with loss of heterozygosity (LOH) involving the second allele16. The loss of 2-microglobulin has been observed in patients experiencing objective partial responses after T cell–based immunotherapy12. We recently observed loss of 2microglobulin in tumor cells from two patients: one patient had an apparently complete response to immunotherapy for nearly a decade; the other had experienced an extraordinary response after adoptive transfer of tumor-specific T cells, then had a resurgence of tumor (unpublished observations), although a causal relationship in such clinical scenarios is difficult to demonstrate unequivocally. Other causes of total HLA class I down-regulation include defects in MHC genes and in the antigen processing and transport pathway. Down-regulation of the proteosome multicatalytic complex subunits LMP-2 and LMP-7 and of peptide transporters TAP-1 and TAP-2 have been reported in tumor histologies that include small cell lung carcinoma17, non-small cell lung cancer (NSCLC)18, prostate carcinoma19 and renal cell carcinoma20. In these situations, HLA class I can often be up-regulated by treatment with IFN- . High resolution image and legend (43K) for HLA class I deficiency. Figure 3. Molecular mechanisms responsible Tumor antigens are processed in the proteosome and generate peptides that are transported by TAP to the endoplasmic reticulum (ER); there they bind to certain HLA class I heavy chain in association with 2-microglobulin. The peptide-HLA complexes are then transported through the Golgi to the cell surface. Several different defects are associated with tumor "escape" from immune recognition: (a) defects in components of the antigen-processing machinery (such as LMP-2 and LMP-7) in the proteosome result in HLA class I down-regulation; (b) defects in peptide transporters TAP-1 or TAP-2 cause HLA class I down-regulation; and (c) LOH on chromosome 6 causes HLA class I haplotype loss. Defects in the transcriptional regulation of the HLA class I gene result in HLA class I locus loss. Point mutations or gene deletions involving the HLA class I heavy chain result in HLA class I allelic loss. (d) 2-microglobulin ( 2M) mutation or deletion results in total loss of HLA class I. Tumors can also express selective loss of HLA class I haplotype, locus or allele. HLA haplotype loss can be due to LOH on chromosome 621. Several mechanisms are involved in locus downregulation, which is more frequent with HLA-B than HLA-A antigens. In melanomas, c-Myc oncogene overexpression correlates with selective HLA-B locus down-regulation22. Loss of transcription factor binding to locus-specific regulatory elements can induce HLA-B locus downregulation in colon carcinoma cells23. In melanoma, the gene products of the HLA-C locus are often expressed poorly or not at all24. The defects underlying HLA class I allele–specific loss include mutations in the genes encoding HLA class I heavy chain25. Descriptions of partial or complete losses of HLA class I as mechanisms of immune escape often fail to consider an increased susceptibility to NK cell lysis, which is a direct consequence of such a loss26. Why do HLA class I loss tumor cell variants continue to grow and are not destroyed by NK cells? NK cells express activating receptors, such as NKG2D, which bind to stress-induced ligands (MICA and MICB) that can be up-regulated in a variety of tumors. Activation of NK cells through this signaling pathway can overcome the inhibitory effect of HLA class I–binding receptors (KIRs)27, 28. Thus, although HLA class I–negative tumors should be susceptible to NK killing, the loss or downregulation of MICA or MICB expression by actively growing tumors represents a potential escape strategy, but one that has not yet been demonstrated in human tumors29. Alternative explanations for why tumor cells that have lost HLA class I are not destroyed by NK cells may be derived from the activation-inhibition model (Fig. 1). NK cells are rapidly activated in the presence of stimulatory factors such as interleukin 12 (IL-12), IL-2, IL-15 or type 1 IFNs in response to inflammatory conditions associated with microbial infection. In "sterile" environments, as seen with tumors or transplantation, such stimulatory factors may not be readily available, and the crosstalk between DCs and resting NK cells that induces NK cell activation may not occur30. In addition, a lack of expression of costimulatory molecules—such as B7-1 (also known as CD80), B7-2 (also known as CD86), CD40 and CD70—by tumors may also hinder optimal NK cell activation via CD28 and CD27 costimulation pathways 31-33. It is possible that in some situations, tumors may produce immunomodulatory cytokines, such as transforming growth factor- (TGF- ) or macrophage migration inhibitory factor (MIF)34, which can directly inhibit NK cell activation and function. Loss of tumor antigens and immunodominance Loss of surface antigen expression can occur independently of the dysregulation of HLA class I expression. It is well documented that tumor antigen expression is heterogeneous, even within the same tumor. Decreased expression of melanoma-melanocyte differentiation antigens (MDAs) such as gp100, melanoma antigen recognized by T cells 1 (MART1) and tyrosinase is associated with disease progression35. In one study, cells were MART1+ in 100% of stage I lesions but only 75% of stage IV lesions36. Decreased antigen expression has also been found in residual tumors after peptide vaccination37, 38. With a gp100 peptide (209-2M) vaccine, there was a decrease in gp100 expression in tumors after ztreatment (47% versus 32%), whereas expression of MART1 was unchanged (54% versus 54%)39. In addition, the amount of tumor antigen expressed may also be important for recognition40. Again, these are correlative studies and fully controlled human studies can be difficult or impossible to come by. It seems likely, however, that as T cell–based tumor immunotherapy becomes stronger, escape mechanisms such as antigen loss are likely to become more prominent. The exact mechanisms that control the down-regulation of tumor antigens are not known in most cases; however, the propagation of such antigen loss variants may be facilitated by epitope immunodominance41. The phenomenon of immunodominance may be thought of as the preferential immunodetection of one or a few epitopes among many expressed on a given target. The theory of immunodominance, as it relates to tumor escape, predicts that one of the ways that antigen-loss variants within a tumor are shielded from immune pressure is that the parental tumor cells that carry the immunodominant epitope serve as a red flag for immune attack, thereby diverting attention from the tumor variants. Once the parental cells are eliminated, a new hierarchy is established among the variant subpopulations, and formerly immunorecessive epitopes become dominant 41. A tumor variant that has lost the restricting HLA class I allele while retaining the immunodominant antigen could cross-present this antigen to CD8+ CTLs by DCs and maintain an immunodominant response to a "phantom" target at the expense of more appropriate and effective responses to other antigens41. Defective death receptor signaling Two death receptor ligands that play a role in immune surveillance against tumor development are Fas ligand (FasL) and TRAIL7, 42-44. Defective death receptor signaling is a mechanism that may contribute to the survival and proliferation of tumor cells. Death receptors have cytoplasmic sequences called "death domains" that are essential for the transmission of apoptotic signals via the caspase cascade. For example, upon engagement of the death receptor Fas by its ligand, Fasassociated death domain (FADD) engages caspase-8, which, in turn, autoactivates itself and cleaves downstream substrates such as caspase-3, caspase-6 and caspase-745. The caspase-8 inhibitor cellular FLICE-inhibitory protein (cFLIP) is expressed in various tumors. In these cases, cFLIP may render tumor cells resistant to death receptor–mediated apoptosis46. Increased expression of cFLIP by tumor cells may also contribute to immunoresistance to T cells in vivo47. Down-regulation or loss of Fas expression in tumors may also contribute to their resistance to apoptosis. Missense mutations and loss of the gene encoding Fas have been identified in multiple myeloma48, non-Hodgkin's lymphoma (NHL)49 and melanoma50. These mutations disrupt Fas signaling, resulting in a loss-of-function death receptor. In addition, mutations of the genes in the proximal pathways downstream of Fas signaling have been also found in NSCLC and lymphoma 51, 52. These include inactivating mutations of FADD and caspase-10. Most were detected in metastatic lesions of NSCLC. Besides defective death receptor signaling, tumors can also block CTL-mediated cytotoxicity via the perforin pathway by overexpressing PI-9 (also known as SPI-6), a serine protease inhibitor that inactivates granzyme B53. In the case of TRAIL-mediated apoptosis, loss of expression of all TRAIL receptors by one of the following causes: chromosomal loss; loss of caspase-8 by chromosomal loss or mutation; no signaling from death-inducing signaling complex (DISC) due to FADD mutation, X-linked inhibitor of apoptosis protein (XIAP) inhibition of caspase-3 and low second mitochondria–derived activator of caspase or direct IAP-binding protein with low pI SMAC (also known as Diablo) release; and low expression of death receptors by post-transcriptional regulation are associated with tumor resistance to TRAIL-mediated apoptosis54. Thus in tumor cells, there may be defects at multiple sites in the death receptor pathways that can favor tumor escape. Lack of costimulation As discussed above, most tumors seem to grow in a noninflammatory microenvironment that is not conducive to immune activation. Histologically, tumors generally coexist innocuously with normal tissues, apparently without giving or inducing immune-activating signals, especially during the early stage of growth. Recognition of tumor antigens by DCs under these conditions will not lead to DC activation and maturation. In addition, lack of expression of costimulatory molecules by tumor cells may lead to T cell anergy55 and suboptimal activation of NK cells. In an experimental setting, insertion of genes encoding B7-1, B7-2 or both into tumors generally increases the immunogenicity of those tumors but does not necessarily lead to regression56. Immunosuppressive cytokines Activation or inhibition of T cells also depends on the presence or absence of cytokines in their immediate microenvironment. Tumor cells produce a variety of cytokines and chemokines that can negatively effect maturation and function of immune cells. Vascular endothelial growth factor (VEGF) is a cytokine that is secreted by most tumors 57. In vitro studies show VEGF inhibits DC differentiation and maturation through suppression of the transcription factor NF- B in hematopoietic stem cells58. Immunohistochemical staining of gastric carcinoma tissues revealed an inverse correlation between the density of DCs and VEGF expression. This finding was also associated with poor prognosis59. In patients with lung, head and neck, and breast cancers, there was a decrease in the function and number of mature DCs, which was associated with increased plasma concentrations of VEGF60. Besides VEGF, increased concentrations of another cytokine, IL-10, are frequently detected in the serum of patients with cancer. IL-10 can exert an inhibitory effect on DC differentiation from stem cell precursors61. In addition, maturation and the functional status of DCs are also compromised by IL-10. This cytokine also inhibits antigen presentation, IL-12 production and induction of T helper type 1 responses in vivo62, 63. IL-10 also enhances spontaneous DC apoptosis64 as well as susceptibility to autologous NK cell lysis 65. IL-10 may protect tumor cells from CTLs by down-regulation of HLA classes I and II and ICAM-1 (intercellular adhesion molecule 1)66. The loss of HLA class I expression could also be due to IL-10–mediated down-regulation of TAP1 and TAP2 proteins in tumor cells67, 68. The proinflammatory factor prostaglandin E2 (PGE2) is another cytokine that is expressed by tumors as a result of enhanced expression of the enzyme cyclooxygenase 2, which is the rate-limiting enzyme for PGE2 synthesis, in multiple human tumors69-71. PGE2 increases the production of IL-10 by lymphocytes and macrophages and inhibits IL-12 production by macrophages72. High concentrations of TGF- are also frequently found in cancer patients and are associated with disease progression73 and poor responses to immunotherapy74. In addition to potentially being produced by some tumor cell lines, TGF- may also be released by cells dying apoptotically75. TGF- inhibits the activation, proliferation and activity of lymphocytes in vivo76. One point to consider is that tumors may not necessarily produce these cytokines as escape mechanisms: the hypothesized immunosuppressive functions may be mere side-effects of the angiogenic and growth factor functions of these cytokines. Apoptosis of activated T cells One of the more controversial mechanisms of tumor escape is the expression of death receptor ligands by tumor cells. A variety of cancer cells express functional FasL, which induces apoptosis of Fas+-susceptible target cells. These include lung carcinoma77, melanoma78, colon carcinoma79 and hepatocellular carcinoma80. The most recent study on this controversial topic asserts that FasL is coexpressed with melanosomal and lysosomal markers in multivesicular bodies in human melanoma cells81. Some of the data concerning FasL expression on tumor cells have been questioned due to a lack of appropriate negative controls and technical concerns, such as the use of nonspecific antibodies, non-intron–spanning polymerase chain reaction primers without proper controls and functional assays that use the Jurkat cell line, which can themselves be induced to express FasL82, 83. In our own work exploring FasL expression on human melanoma cells, we found no evidence of significant expression either at the mRNA or protein level. In addition, there was no killing of Fas+-susceptible targets by melanoma cells in controlled functional assays84. Another level of complexity in the role of FasL may be that it is actually pro-inflammatory in some circumstances. In fact, all well controlled in vivo experiments with membrane-bound FasL–transfected tumor cells show accelerated rejection accompanied by neutrophil infiltration 85-87. However, the activity of FasL may be modulated if it is processed to a secreted soluble form or if it is elaborated in the presence or absence of certain cytokines within the tumor microenvironment. For example, TGF- can regulate the proinflammatory effects of FasL88. The most crucial role of Fas-FasL interactions in the tumor setting may be the induction of activation-induced cell death (AICD) of antitumor T cells. Upon activation by tumor antigen recognition, T cells express high amounts of FasL, which induces apoptosis of these T cells ("suicide") and between T cells ("fratricide")84, 89 (Fig. 4). High resolution image and legend (55K) Figure 4. Alternative models for the induction of FasL-mediated T cell death after encounter with tumor cells. (a) AICD of T cells after recognition of tumor cells. Tumor recognition leads to activation of T cells and up-regulation of Fas and FasL on T cell surface, which results in the T cell killing of itself ("suicide") and of other T cells ("fratricide"). (b) Proposed tumor "counterattack" model. Tumor cells express functional FasL and kill infiltrating Fas-expressing T cells via Fas-FasL binding, which leads to tumor escape. However, ligation of Fas expressed on innate immune cells such as neutrophils, macrophages and immature DCs by FasL expressed on tumor cells may also lead to release of multiple proinflammatory cytokines and chemokines, setting the stage for tumor rejection. Other TNF family ligands, such as TRAIL, are expressed by tumors, but there is no convincing evidence that they play a major role in the induction of tumor-specific T cell apoptosis in human cancers90. Tumor-associated B7-H1 ligand also induces T cell apoptosis91. However, this study relied heavily on artificial models of B7-H1–transfected tumor cells that expressed high amounts of B7-H1. It remains unclear whether the relatively small increases in specific T cell apoptosis over background caused by endogenously expressed B7-H1 will be enough to enable human tumors to evade immune recognition. The role of suppressor T cells Immunoregulatory CD4+ CD25+ T cells (also known as Treg, TH3 and TR1 cells) control key aspects of immunological tolerance to self-antigens92, 93. Removal of these cells, which constitute 5–10% of CD4+ T cells in humans and rodents, induces autoimmune disease in the ovaries, thyroid gland, salivary glands and the mucosal linings of the stomach and small intestine and accelerate diabetes in nonobsese diabetic mice. Depletion of CD4+CD25+ cells plus injection of an antibody capable of blocking CTLA-4 (cytotoxic T lymphocyte antigen 4), enhanced reactivity to a known tumorassociated antigen (TAA)94. However, based on the available evidence from experimental mouse tumor models, it seems that simply blocking or even eliminating T regulatory cell function will not be enough to treat established tumors95. Identification of the glucocorticoid-induced TNF receptor family–related gene (GITR, also known as TNFRSF18) expressed on T regulatory cells might afford new therapeutic opportunities, as stimulation of GITR with an activating antibody reverses CD4+CD25+ T cell–mediated suppression96, 97. Another possible therapeutic intervention could be blockade of signaling through the molecular pair of TNF-related activation-induced cytokine (TRANCE) and receptor-activator of NF- B (RANK), as these signals are involved in the generation and activation of CD4+CD25+ T regulatory cells98. In addition to CD4+CD25+ T cells, NKT cells can exert immunoinhibitory effects on tumor immunity. CD4+ NKT cells inhibit effective CTL-mediated tumor rejection by IL-13 via the IL-4R–STAT6 (interleukin 4 receptor–signal transducers and activators of transcription 6) pathway99. Another study has identified CD4+DX5+ NKT suppressor cells that regulate the growth of ultraviolet-induced skin cancers and mediate antigen-specific immune suppression100; thus, targeting NKT cells could be another therapeutic option. The challenge for immunotherapists now is to discover how to use an understanding of T regulatory function to enhance other immunotherapeutic interventions. Conclusion Despite recent progress in tumor immunobiology and technical advances in the field of tumor immunotherapy, current immunotherapeutic strategies used in the treatment of patients with cancer have not been successful in most cases. It is not clear whether tumor escape accounts for these failures or whether lack of observed tumor regression is due to inadequacies of the immunotherapies themselves. The balance of immune activation and immune inhibition must favor the former if tumors are to come under immune pressure. However, even in some instances where effective antitumor responses can be achieved with immunotherapy, tumors often recur. It seems likely that as the tumor immunologist's armamentarium becomes more powerful and immunotherapies become more effective, the selection of tumor variants with immunoresistant phenotypes will be observed with greater frequency. Tumor escape may ultimately thwart some dramatic responses to immunotherapy. Indeed, some tumors that recur after successful immunotherapy often possess immunoresistant phenotypes. The challenge for the tumor immunologists now is to understand the mechanisms by which tumors become refractory to immune modulation. The optimal immunotherapy strategy may be to achieve three things concurrently: provision of appropriate immune activating signals, elimination of inhibitory factors and avoidance of the emergence of immunoresistant phenotypes. The latter might be achieved with a combination of modalities sustained for a long enough period of time to complete tumor destruction. References 1. Kaplan, D.H. et al. Demonstration of an interferon -dependent tumor surveillance system in immunocompetent mice. Proc. Natl. Acad. Sci. USA 95, 7556-7561 (1998). | Article | PubMed | ISI | 2. Diefenbach, A., Jensen, E.R., Jamieson, A.M. & Raulet, D.H. Rae1 and H60 ligands of the NKG2D receptor stimulate tumour immunity. Nature 413, 165-171 (2001). | Article | PubMed | ISI | 3. Girardi, M. et al. Regulation of cutaneous malignancy by (2001). | Article | PubMed | ISI | T cells. Science 294, 605-609 4. Street, S.E., Trapani, J.A., MacGregor, D. & Smyth, M.J. Suppression of lymphoma and epithelial malignancies effected by interferon . J. Exp. Med. 196, 129-134 (2002). | Article | PubMed | ISI | 5. Shankaran, V. et al. IFN and lymphocytes prevent primary tumour development and shape tumour immunogenicity. Nature 410, 1107-1111 (2001). | Article | PubMed | ISI | 6. Smyth, M.J. et al. Perforin-mediated cytotoxicity is critical for surveillance of spontaneous lymphoma. J. Exp. Med. 192, 755-760 (2000). | Article | PubMed | ISI | 7. Takeda, K. et al. Critical role for tumor necrosis factor-related apoptosis-inducing ligand in immune surveillance against tumor development. J. Exp. Med. 195, 161-169 (2002). | Article | PubMed | ISI | 8. Restifo, N.P. et al. Assumptions of the tumor 'escape' hypothesis. Semin. Cancer Biol. 12, 81-86 (2002). | Article | PubMed | ISI | 9. Stoler, D.L. et al. The onset and extent of genomic instability in sporadic colorectal tumor progression. Proc. Natl. Acad. Sci. USA 96, 15121-15126 (1999). | Article | PubMed | ISI | 10. Johnsen, A.K. et al. Systemic deficits in transporter for antigen presentation (TAP)-1 or proteasome subunit LMP2 have little or no effect on tumor incidence. Int. J. Cancer 91, 366-372 (2001). | Article | PubMed | ISI | 11. Zheng, P., Sarma, S., Guo, Y. & Liu, Y. Two mechanisms for tumor evasion of preexisting cytotoxic T-cell responses: lessons from recurrent tumors. Cancer Res. 59, 3461-3467 (1999). | PubMed | ISI | 12. Restifo, N.P. et al. Loss of functional 2-microglobulin in metastatic melanomas from five patients receiving immunotherapy. J. Natl. Cancer Inst. 88, 100-108 (1996). | PubMed | ISI | 13. Garrido, F. et al. Implications for immunosurveillance of altered HLA class I phenotypes in human tumours. Immunol. Today 18, 89-95 (1997). | Article | PubMed | ISI | 14. Algarra, I., Collado, A. & Garrido, F. Altered MHC class I antigens in tumors. Int. J. Clin. Lab. Res. 27, 95-102 (1997). | PubMed | ISI | 15. Cabrera, T. et al. High frequency of altered HLA class I phenotypes in invasive breast carcinomas. Hum. Immunol. 50, 127-134 (1996). | Article | PubMed | ISI | 16. Hicklin, D.J. et al. 2-microglobulin mutations, HLA class I antigen loss, and tumor progression in melanoma. J. Clin. Invest. 101, 2720-2729 (1998). | PubMed | ISI | 17. Restifo, N.P. et al. Identification of human cancers deficient in antigen processing. J. Exp. Med. 177, 265-272 (1993). | PubMed | ISI | 18. Korkolopoulou, P., Kaklamanis, L., Pezzella, F., Harris, A.L. & Gatter, K.C. Loss of antigenpresenting molecules (MHC class I and TAP-1) in lung cancer. Br. J. Cancer 73, 148-153 (1996). | PubMed | ISI | 19. Sanda, M.G. et al. Molecular characterization of defective antigen processing in human prostate cancer. J. Natl. Cancer Inst. 87, 280-285 (1995). | PubMed | ISI | 20. Seliger, B. et al. Expression and function of the peptide transporters in escape variants of human renal cell carcinomas. Exp. Hematol. 25, 608-614 (1997). | PubMed | ISI | 21. Ramal, L.M. et al. Molecular strategies to define HLA haplotype loss in microdissected tumor cells. Hum. Immunol. 61, 1001-1012 (2000). | Article | PubMed | ISI | 22. Versteeg, R. et al. Suppression of class I human histocompatibility leukocyte antigen by cmyc is locus specific. J. Exp. Med. 170, 621-635 (1989). | PubMed | ISI | 23. Soong, T.W. & Hui, K.M. Locus-specific transcriptional control of HLA genes. J. Immunol. 149, 2008-2020 (1992). | PubMed | ISI | 24. Marincola, F.M. et al. Locus-specific analysis of human leukocyte antigen class I expression in melanoma cell lines. J. Immunother. Emphasis. Tumor Immunol. 16, 13-23 (1994). | PubMed | ISI | 25. Koopman, L.A., van Der, S., Giphart, M.J. & Fleuren, G.J. Human leukocyte antigen class I gene mutations in cervical cancer. J. Natl. Cancer Inst. 91, 1669-1677 (1999). | Article | PubMed | ISI | 26. Porgador, A., Mandelboim, O., Restifo, N.P. & Strominger, J.L. Natural killer cell lines kill autologous 2-microglobulin-deficient melanoma cells: implications for cancer immunotherapy. Proc. Natl. Acad. Sci. USA 94, 13140-13145 (1997). | Article | PubMed | ISI | 27. Bauer, S. et al. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 285, 727-729 (1999). | Article | PubMed | ISI | 28. Groh, V. et al. Broad tumor-associated expression and recognition by tumor-derived T cells of MICA and MICB. Proc. Natl. Acad. Sci. USA 96, 6879-6884 (1999). | Article | PubMed | ISI | 29. Garrido, F. & Algarra, I. MHC antigens and tumor escape from immune surveillance. Adv. Cancer Res. 83, 117-158 (2001). | PubMed | ISI | 30. Gerosa, F. et al. Reciprocal activating interaction between natural killer cells and dendritic cells. J. Exp. Med. 195, 327-333 (2002). | Article | PubMed | ISI | 31. Galea-Lauri, J. et al. Expression of a variant of CD28 on a subpopulation of human NK cells: implications for B7-mediated stimulation of NK cells. J. Immunol. 163, 62-70 (1999). | PubMed | ISI | 32. Carbone, E. et al. A new mechanism of NK cell cytotoxicity activation: the CD40-CD40 ligand interaction. J. Exp. Med. 185, 2053-2060 (1997). | Article | PubMed | ISI | 33. Takeda, K. et al. CD27-mediated activation of murine NK cells. J. Immunol. 164, 1741-1745 (2000) | PubMed | ISI | 34. Apte, R.S., Mayhew, E. & Niederkorn, J.Y. Local inhibition of natural killer cell activity promotes the progressive growth of intraocular tumors. Invest. Ophthalmol. Vis. Sci. 38, 1277-1282 (1997). | PubMed | ISI | 35. de Vries, T.J. et al. Heterogeneous expression of immunotherapy candidate proteins gp100, MART-1, and tyrosinase in human melanoma cell lines and in human melanocytic lesions. Cancer Res. 57, 3223-3229 (1997). | PubMed | ISI | 36. Hofbauer, G.F., Kamarashev, J., Geertsen, R., Boni, R. & Dummer, R. Melan A/MART-1 immunoreactivity in formalin-fixed paraffin-embedded primary and metastatic melanoma: frequency and distribution. Melanoma Res. 8, 337-343 (1998). | PubMed | ISI | 37. Jager, E. et al. Inverse relationship of melanocyte differentiation antigen expression in melanoma tissues and CD8+ cytotoxic-T-cell responses: evidence for immunoselection of antigenloss variants in vivo. Int. J. Cancer 66, 470-476 (1996). | Article | PubMed | ISI | 38. Lee, K.H. et al. Functional dissociation between local and systemic immune response during anti-melanoma peptide vaccination. J. Immunol. 161, 4183-4194 (1998). | PubMed | ISI | 39. Riker, A. et al. Immune selection after antigen-specific immunotherapy of melanoma. Surgery 126, 112-120 (1999). | Article | PubMed | ISI | 40. Cormier, J.N. et al. Natural variation of the expression of HLA and endogenous antigen modulates CTL recognition in an in vitro melanoma model. Int. J. Cancer 80, 781-790 (1999). | Article | PubMed | ISI | 41. Schreiber, H., Wu, T.H., Nachman, J. & Kast, W.M. Immunodominance and tumor escape. Semin. Cancer Biol. 12, 25-31 (2002). | Article | PubMed | ISI | 42. Straus, S.E. et al. The development of lymphomas in families with autoimmune lymphoproliferative syndrome with germline Fas mutations and defective lymphocyte apoptosis. Blood 98, 194-200 (2001). | Article | PubMed | ISI | 43. Davidson, W.F., Giese, T. & Fredrickson, T.N. Spontaneous development of plasmacytoid tumors in mice with defective Fas-Fas ligand interactions. J. Exp. Med. 187, 1825-1838 (1998). | Article | PubMed | ISI | 44. Takeda, K. et al. Involvement of tumor necrosis factor-related apoptosis-inducing ligand in surveillance of tumor metastasis by liver natural killer cells. Nature Med. 7, 94-100 (2001) | Article | PubMed | ISI | 45. Salvesen, G.S. & Dixit, V.M. Caspase activation: the induced-proximity model. Proc. Natl. Acad. Sci. USA 96, 10964-10967 (1999). | Article | PubMed | ISI | 46. Irmler, M. et al. Inhibition of death receptor signals by cellular FLIP. Nature 388, 190-195 (1997). | Article | PubMed | ISI | 47. Medema, J.P., de Jong, J., van Hall, T., Melief, C.J. & Offringa, R. Immune escape of tumors in vivo by expression of cellular FLICE-inhibitory protein. J. Exp. Med. 190, 1033-1038 (1999). | Article | PubMed | ISI | 48. Landowski, T.H., Qu, N., Buyuksal, I., Painter, J.S. & Dalton, W.S. Mutations in the Fas antigen in patients with multiple myeloma. Blood 90, 4266-4270 (1997). | PubMed | ISI | 49. Gronbaek, K. et al. Somatic Fas mutations in non-Hodgkin's lymphoma: association with extranodal disease and autoimmunity. Blood 92, 3018-3024 (1998). | PubMed | ISI | 50. Shin, M.S. et al. Alterations of Fas (Apo-1/CD95) gene in cutaneous malignant melanoma. Am. J. Pathol. 154, 1785-1791 (1999). | PubMed | ISI | 51. Shin, M.S. et al. Alterations of Fas-pathway genes associated with nodal metastasis in nonsmall cell lung cancer. Oncogene 21, 4129-4136 (2002). | Article | PubMed | ISI | 52. Shin, M.S. et al. Inactivating mutations of CASP10 gene in non-Hodgkin lymphomas. Blood 99, 4094-4099 (2002). | Article | PubMed | ISI | 53. Medema, J.P. et al. Blockade of the granzyme B/perforin pathway through overexpression of the serine protease inhibitor PI-9/SPI-6 constitutes a mechanism for immune escape by tumors. Proc. Natl. Acad. Sci. USA 98, 11515-11520 (2001). | Article | PubMed | ISI | 54. Hersey, P. & Zhang, X.D. How melanoma cells evade trail-induced apoptosis. Nature Rev. Cancer 1, 142-150 (2001). | Article | PubMed | 55. Schwartz, R.H. A cell culture model for T lymphocyte clonal anergy. Science 248, 1349-1356 (1990). | PubMed | ISI | 56. Chen, L. et al. Tumor immunogenicity determines the effect of B7 costimulation on T cellmediated tumor immunity. J. Exp. Med. 179, 523-532 (1994). | PubMed | ISI | 57. Toi, M. et al. Clinical significance of the determination of angiogenic factors. Eur. J. Cancer 32, 2513-2519 (1996). | Article | ISI | 58. Oyama, T. et al. Vascular endothelial growth factor affects dendritic cell maturation through the inhibition of nuclear factor- B activation in hemopoietic progenitor cells. J. Immunol. 160, 12241232 (1998). | PubMed | ISI | 59. Saito, H., Tsujitani, S., Ikeguchi, M., Maeta, M. & Kaibara, N. Relationship between the expression of vascular endothelial growth factor and the density of dendritic cells in gastric adenocarcinoma tissue. Br. J. Cancer 78, 1573-1577 (1998). | PubMed | ISI | 60. Almand, B. et al. Clinical significance of defective dendritic cell differentiation in cancer. Clin. Cancer Res. 6, 1755-1766 (2000). | PubMed | ISI | 61. Girolomoni, G. & Ricciardi-Castagnoli, P. Dendritic cells hold promise for immunotherapy. Immunol. Today 18, 102-104 (1997). | Article | PubMed | ISI | 62. De Smedt, T. et al. Effect of interleukin-10 on dendritic cell maturation and function. Eur. J. Immunol. 27, 1229-1235 (1997). | PubMed | ISI | 63. Sharma, S. et al. T cell-derived IL-10 promotes lung cancer growth by suppressing both T cell and APC function. J. Immunol. 163, 5020-5028 (1999). | PubMed | ISI | 64. Ludewig, B. et al. Spontaneous apoptosis of dendritic cells is efficiently inhibited by TRAP (CD40-ligand) and TNF- , but strongly enhanced by interleukin-10. Eur. J. Immunol. 25, 19431950 (1995). | PubMed | ISI | 65. Carbone, E. et al. Recognition of autologous dendritic cells by human NK cells. Eur. J. Immunol. 29, 4022-4029 (1999). | Article | PubMed | ISI | 66. Yue, F.Y. et al. Interleukin-10 is a growth factor for human melanoma cells and downregulates HLA class-I, HLA class-II and ICAM-1 molecules. Int. J. Cancer 71, 630-637 (1997). | Article | PubMed | ISI | 67. Salazar-Onfray, F. et al. Down-regulation of the expression and function of the transporter associated with antigen processing in murine tumor cell lines expressing IL-10. J. Immunol. 159, 3195-3202 (1997). | PubMed | ISI | 68. Zeidler, R. et al. Downregulation of TAP1 in B lymphocytes by cellular and Epstein-Barr virusencoded interleukin-10. Blood 90, 2390-2397 (1997). | PubMed | ISI | 69. Ristimaki, A., Honkanen, N., Jankala, H., Sipponen, P. & Harkonen, M. Expression of cyclooxygenase-2 in human gastric carcinoma. Cancer Res. 57, 1276-1280 (1997). | PubMed | ISI | 70. Sano, H. et al. Expression of cyclooxygenase-1 and -2 in human colorectal cancer. Cancer Res. 55, 3785-3789 (1995). | PubMed | ISI | 71. Wolff, H. et al. Expression of cyclooxygenase-2 in human lung carcinoma. Cancer Res. 58, 4997-5001 (1998). | PubMed | ISI | 72. Huang, M. et al. Non-small cell lung cancer cyclooxygenase-2-dependent regulation of cytokine balance in lymphocytes and macrophages: up-regulation of interleukin 10 and downregulation of interleukin 12 production. Cancer Res. 58, 1208-1216 (1998). | PubMed | ISI | 73. Gorsch, S.M., Memoli, V.A., Stukel, T.A., Gold, L.I. & Arrick, B.A. Immunohistochemical staining for transforming growth factor 1 associates with disease progression in human breast cancer. Cancer Res. 52, 6949-6952 (1992). | PubMed | ISI | 74. Doran, T., Stuhlmiller, H., Kim, J.A., Martin, E.W.J. & Triozzi, P.L. Oncogene and cytokine expression of human colorectal tumors responding to immunotherapy. J. Immunother. 20, 372-376 (1997). | PubMed | ISI | 75. Chen, W., Frank, M.E., Jin, W. & Wahl, S.M. TGF- released by apoptotic T cells contributes to an immunosuppressive milieu. Immunity 14, 715-725 (2001) | Article | PubMed | ISI | 76. Fontana, A. et al. Transforming growth factor- inhibits the generation of cytotoxic T cells in virus-infected mice. J. Immunol. 143, 3230-3234 (1989). | PubMed | ISI | 77. Niehans, G.A. et al. Human lung carcinomas express Fas ligand. Cancer Res. 57, 1007-1012 (1997). | PubMed | ISI | 78. Hahne, M. et al. Melanoma cell expression of Fas(Apo-1/CD95) ligand: implications for tumor immune escape. Science 274, 1363-1366 (1996). | Article | PubMed | ISI | 79. O'Connell, J., O'Sullivan, G.C., Collins, J.K. & Shanahan, F. The Fas counterattack: Fasmediated T cell killing by colon cancer cells expressing Fas ligand. J. Exp. Med. 184, 1075-1082 (1996). | PubMed | ISI | 80. Strand, S. et al. Lymphocyte apoptosis induced by CD95 (APO-1/Fas) ligand-expressing tumor cells-a mechanism of immune evasion? Nature Med. 2, 1361-1366 (1996). | PubMed | ISI | 81. Andreola, G. et al. Induction of lymphocyte apoptosis by tumor cell secretion of FasL-bearing microvesicles. J. Exp. Med. 195, 1303-1316 (2002). | Article | PubMed | ISI | 82. Restifo, N.P. Not so Fas: Re-evaluating the mechanisms of immune privilege and tumor escape. Nature Med. 6, 493-495 (2000). | Article | PubMed | ISI | 83. Restifo, N.P. Countering the 'counterattack' hypothesis. Nature Med. 7, 259 (2001). | Article | PubMed | ISI | 84. Chappell, D.B., Zaks, T.Z., Rosenberg, S.A. & Restifo, N.P. Human melanoma cells do not express Fas (Apo-1/CD95) ligand. Cancer Res. 59, 59-62 (1999). | PubMed | ISI | 85. Arai, H., Gordon, D., Nabel, E.G. & Nabel, G.J. Gene transfer of Fas ligand induces tumor regression in vivo. Proc. Natl. Acad. Sci. USA 94, 13862-13867 (1997). | Article | PubMed | ISI | 86. Kang, S.M., Lin, Z., Ascher, N.L. & Stock, P.G. Fas ligand expression on islets as well as multiple cell lines results in accelerated neutrophilic rejection. Transplant. Proc. 30, 538 (1998). | PubMed | 87. Drozdzik, M., Qian, C., Lasarte, J.J., Bilbao, R. & Prieto, J. Antitumor effect of allogenic fibroblasts engineered to express Fas ligand (FasL). Gene Ther. 5, 1622-1630 (1998). | Article | PubMed | ISI | 88. Chen, J.J., Sun, Y. & Nabel, G.J. Regulation of the proinflammatory effects of Fas ligand (CD95L). Science 282, 1714-1717 (1998). | Article | PubMed | ISI | 89. Zaks, T.Z., Chappell, D.B., Rosenberg, S.A. & Restifo, N.P. Fas-mediated suicide of tumorreactive T cells following activation by specific tumor: selective rescue by caspase inhibition. J. Immunol. 162, 3273-3279 (1999). | PubMed | ISI | 90. Cappello, P., Novelli, F., Forni, G. & Giovarelli, M. Death receptor ligands in tumors. J. Immunother. 25, 1-15 (2002). | Article | PubMed | ISI | 91. Dong, H. et al. Tumor-associated B7-H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nature Med. 8, 793-800 (2002). | Article | PubMed | 92. McHugh, R.S. & Shevach, E.M. Cutting edge: depletion of CD4+CD25+ regulatory T cells is necessary, but not sufficient, for induction of organ-specific autoimmune disease. J. Immunol. 168, 5979-5983 (2002). | PubMed | ISI | 93. Sakaguchi, S. et al. Immunologic tolerance maintained by CD25+ CD4+ regulatory T cells: their common role in controlling autoimmunity, tumor immunity, and transplantation tolerance. Immunol. Rev. 182, 18-32 (2001). | Article | PubMed | ISI | 94. Sutmuller, R.P. et al. Synergism of cytotoxic T lymphocyte-associated antigen 4 blockade and depletion of CD25+ regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J. Exp. Med. 194, 823-832 (2001). | Article | PubMed | ISI | 95. Antony, P.A. & Restifo, N.P. Do CD4+ CD25+ immunoregulatory T cells hinder tumor immunotherapy? J. Immunother. 25, 202-206 (2002). | Article | PubMed | ISI | 96. Shimizu, J., Yamazaki, S., Takahashi, T., Ishida, Y. & Sakaguchi, S. Stimulation of CD25+CD4+ regulatory T cells through GITR breaks immunological self-tolerance. Nature Immunol. 3, 135-142 (2002). | Article | PubMed | ISI | 97. McHugh, R.S. et al. CD4+CD25+ immunoregulatory T cells: gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity. 16, 311-323 (2002). | PubMed | ISI | 98. Green, E.A., Choi, Y. & Flavell, R.A. Pancreatic lymph node-derived CD4+CD25+ Treg cells: highly potent regulators of diabetes that require TRANCE-RANK signals. Immunity 16, 183-191 (2002). | PubMed | ISI | 99. Terabe, M. et al. NKT cell-mediated repression of tumor immunosurveillance by IL-13 and the IL-4R-STAT6 pathway. Nature Immunol. 1, 515-520 (2000). | Article | PubMed | ISI | 100. Moodycliffe, A.M., Nghiem, D., Clydesdale, G. & Ullrich, S.E. Immune suppression and skin cancer development: regulation by NKT cells. Nature Immunol. 1, 521-525 (2000). | Article | PubMed | ISI | Figure 1. Activation versus suppression during tumor progression. Immune activation during early tumor progression may be triggered by the expression of neoantigens. In addition, the progressive growth of tumors may be associated with the invasion and destruction of surrounding normal tissues. Other factors that may cause immune activation include generation of heat shock proteins (Hsps), which result from cellular stress, and ROS such as OH- and H2O2. Conversely, immune activation and function may be hampered by a lack of appropriate costimulation, the presence of immunosuppressive cytokines (for example, VEGF, IL-10 and TGF- ), and the impact of immunoregulatory cells such as CD4+CD25+ T cells and NKT cells. As tumors grow, these events occur in the presence of massive apoptosis and necrosis of tumor cells, infiltrating immune cells and surrounding stromal cells. The fate of tumors may be dictated by the net effect of immune activation and immune inhibition. Figure 2. Natural selection of tumor variants in the generation of "tumor escape" phenotypes. (a) Genomic instability gives rise to genetic diversity in tumors. Natural selection of tumor variants occurs by differential propagation of tumor subclones in their microenvironment. (b) The same concept also applies to tumor growth after effective immunotherapy. Figure 3. Molecular mechanisms responsible for HLA class I deficiency. Tumor antigens are processed in the proteosome and generate peptides that are transported by TAP to the endoplasmic reticulum (ER); there they bind to certain HLA class I heavy chain in association with 2-microglobulin. The peptide-HLA complexes are then transported through the Golgi to the cell surface. Several different defects are associated with tumor "escape" from immune recognition: (a) defects in components of the antigen-processing machinery (such as LMP-2 and LMP-7) in the proteosome result in HLA class I down-regulation; (b) defects in peptide transporters TAP-1 or TAP-2 cause HLA class I down-regulation; and (c) LOH on chromosome 6 causes HLA class I haplotype loss. Defects in the transcriptional regulation of the HLA class I gene result in HLA class I locus loss. Point mutations or gene deletions involving the HLA class I heavy chain result in HLA class I allelic loss. (d) 2-microglobulin ( 2M) mutation or deletion results in total loss of HLA class I. Figure 4. Alternative models for the induction of FasL-mediated T cell death after encounter with tumor cells. (a) AICD of T cells after recognition of tumor cells. Tumor recognition leads to activation of T cells and up-regulation of Fas and FasL on T cell surface, which results in the T cell killing of itself ("suicide") and of other T cells ("fratricide"). (b) Proposed tumor "counterattack" model. Tumor cells express functional FasL and kill infiltrating Fas-expressing T cells via Fas-FasL binding, which leads to tumor escape. However, ligation of Fas expressed on innate immune cells such as neutrophils, macrophages and immature DCs by FasL expressed on tumor cells may also lead to release of multiple proinflammatory cytokines and chemokines, setting the stage for tumor rejection. Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 1006 - 1012 (2002) doi:10.1038/ni1102-1006 © Nature America, Inc. <> Viral evasion of natural killer cells Jordan S. Orange, Marlys S. Fassett, Louise A. Koopman, Jonathan E. Boyson & Jack L. Strominger Department of Molecular and Cellular Biology, Harvard University, Cambridge MA, USA. Correspondence should be addressed to J L Strominger jlstrom@fas.harvard.edu Viruses have evolved mechanisms to avoid the host immune system, including means of escaping detection by both the innate and adaptive immune responses. Natural killer (NK) cells are a central component of the innate immune system and are crucial in defense against certain viruses. To attain a state of chronic infection, some successful viruses have developed specific mechanisms to evade detection by and activation of NK cells. These NK cell–specific evasion mechanisms fall into distinct mechanistic categories used in numerous virus families. NK cells are lymphocytes that do not undergo genetic recombination events to increase their affinity for particular ligands, and are thus considered part of the innate immune system. They are capable of mediating cytotoxic activity and of producing cytokines after ligation of a variety of germlineencoded receptors. NK cells mediate direct lysis of target cells by releasing cytotoxic granules containing perforin and granzymes, or by binding to apoptosis-inducing receptors on the target cell. They also secrete cytokines such as interferon- (IFN- ) and tumor necrosis factor- (TNF- ) during infection and inflammation. Several receptors that can activate NK cells have been identified, including the human natural cytotoxicity receptors NKp30, NKp44, NKp461 and Ly49D and Ly49H in the mouse2. Although the specificities of many NK cell–activating receptors are still unknown, some recognize viral products; these include influenza hemagglutinin, recognized by NKp463, and murine cytomegalovirus (MCMV) m157, recognized by Ly49H 4, 5. Other well known molecules can also function as activation receptors in NK cells, including leukocyte function–associated antigen 1 (LFA1)6 and the CD2 family7. NK cell responses are also coordinated and modulated by cytokines, including IFN- , IFN- , interleukin 2 (IL-2), IL-12, IL-15 and IL-188. Because of the possible consequences of NK cell activation, normal host cells must be able to readily and effectively inhibit NK cells. Various inhibitory receptors are consistently expressed by subsets of NK cells, including killer-cell immunoglobulin-like receptors (KIR), immunoglobulin-like inhibitory receptors (ILT) and the lectin-like heterodimer CD94-NKG2A1. These receptors bind to host MHC class I molecules and transmit inhibitory signals to the NK cell through intracellular tyrosine-based inhibitory motifs (ITIMs) contained in their cytoplasmic domains. Signaling via coreceptors with inhibitory potential, such as CD81, may also silence NK cells. Thus, NK cells can mediate powerful effector functions, but are effectively regulated by healthy host cells under normal circumstances. NK cells are activated during a wide variety of viral infections by virus-induced type I IFNs8. Studies in animal models, however, have demonstrated that NK cells are required for clearance of only certain viruses, including herpesviruses. The importance of NK cell defense against these viruses is highlighted by the susceptibility of mice depleted of NK cells to experimental infection and by the invasive or disseminated viral disease that is associated with naturally occurring NK cell deficiencies in humans8-11. Many of these pathogens have effective means of avoiding the adaptive immune response. In eluding T cells, however, these viruses might have increased their susceptibility to NK cell–mediated defenses. Members of the herpesvirus, papillomavirus, retrovirus, poxvirus and flavivirus families have developed mechanisms to evade the NK cell response. These fall into five categories (Fig. 1): expression of virally encoded MHC class I homologs; selective modulation of MHC class I protein expression by viral proteins; virus-mediated inhibition of activating receptor function; production of virally encoded cytokine-binding proteins or cytokine-receptor antagonists; and direct viral effects on NK cells. The putative purpose of these mechanisms is to block NK cell activity. Examples of each strategy (summarized in Table 1) are discussed below. Many other viruses probably use these strategies, and additional viral products may affect NK cell functions. This discussion is limited to viral infections and viral gene products known to affect the NK cell response. High resolution image and legend (69K) Figure 1. Viral mechanisms for evading NK cells. The strategies by which viruses evade NK cells fall into five categories and are depicted in the interaction between a virus infected target cell (left) and an NK cell (right). (a) NK cells can be inhibited by a viral MHC class I homolog with structural similarity to endogenous host class I that binds to inhibitory class I receptors on NK cells. (b) Viruses can inhibit expression of HLA-A and HLA-B, resulting in a relative increase in HLA-C and HLA-E on the surface of the target cell; these inhibit NK cells through the class I inhibitory receptors CD94-NKG2A and KIR, respectively. Alternatively, viral gene expression can result in selectively increased expression of HLA-E, which inhibits NK cells through CD94-NKG2A. (c) Virus-encoded proteins can function as cytokine binding proteins that block the action of NK cell activating cytokines. In addition, viruses can produce homologs, or increase host production of cytokines that inhibit NK cells. (d) NK cell activities can also be avoided by decreased expression of NK cell–activating ligands in virus-infected target cells, which prevent signal transduction via NK cell–activating receptors. To achieve the same end, viruses can encode antagonists of the activating receptor–ligand interaction. (e) Viruses can also directly inhibit NK cells by infecting them or using envelope proteins to ligate NK cell inhibitory receptors. Proteins outlined in red are virally encoded. Each mechanism corresponds to the similarly numbered section of the text where additional details and examples are provided. High resolution image and legend (136K) Table 1. Viral mechanisms of evading NK cell responses Inhibition of NK cells by viral homologs of MHC class I Virus-encoded homologs of cellular MHC class I genes represent the earliest recognized viral mechanism for evading NK cells. Many viruses evade T cell recognition by down-regulating class I molecules on the surface of the host cell (discussed below). In theory this leaves infected cells susceptible to NK lysis owing to the reduced opportunity for class I molecules—human leukocyte antigen C (HLA-C) and HLA-E—to engage NK cell inhibitory receptors. The discovery of a class I homolog, UL18, in the genome of human cytomegalovirus (HCMV)12 has led to speculation that these viral proteins might serve as NK cell decoys and ligate inhibitory receptors to block NK cell cytotoxicity in the absence of host class I molecules. Early studies in which HCMV UL18 was expressed in the HLA-A, HLA-B and HLA-C–deficient B cell line 721.221 demonstrated a CD94-dependent inhibition of killing by various NK cell lines and clones13. Subsequently, a principal ligand of the CD94-NKG2A inhibitory receptor was found to be HLA-E, a class I protein that is expressed only after forming a complex with a peptide nonamer derived mainly from the signal peptides of some of the class I proteins14. Further efforts to identify a nonamer in the UL18 sequence that would form a complex with HLA-E have been unsuccessful15. Thus, the mechanism underlying the reported inhibition of lysis of the human B cell line transfected with UL18 remains unexplained. It was not due to inadvertent selection of HLA-E–expressing 721.221 cells because immunoprecipitation of class I molecules from the 721.221-UL18 transfectants yielded a heavily glycosylated -chain of the size of UL18 ( 66 kD) and not HLA-E (44 kD)13. In a contrasting report, however, UL18-transfected fibroblasts showed increased susceptibility to lysis by NK cell lines, and fibroblasts infected with wild-type HCMV were lysed more efficiently than those infected with a UL18-deficient HCMV16. Alternatively, the inhibitory activity mediated by UL18 may be due to its binding of ILT-2 (LIR-1), an inhibitory receptor expressed by B cells, monocytes and a subset of NK cells17. Thus, the role of UL18 in evasion of NK cells is not completely understood, but it may be an important mechanism by which certain subpopulations of NK cells are inhibited. Compelling data support the notion that the MCMV MHC class I homolog m144 is involved in the inhibition of NK cells. An m144 deletion strain of MCMV is less virulent than the wild-type virus, and this effect is reversed after in vivo depletion of NK cells18. Transfection of m144 into the Raji cell line results in partial inhibition of antibody-dependent cellular cytotoxicity19. In addition, m144transfected RMA-S lymphoma cells injected into mice are tumorigenic, whereas untransfected RMA-S tumors are rejected by NK cells20. Further investigation of m144 will benefit from identification of the cognate inhibitory receptor. The structures of m144 and UL18 are considerably different (for example, the latter binds peptides, the former does not), so that their receptors, mechanisms and functions may be distinct. A newly described viral class I homolog, MCMV m157, has both activating and inhibitory effects upon NK cells, depending on the mouse strain from which the cells were derived. MCMV m157 is a ligand for the NK-activating receptor Ly49H in MCMV-resistant C57BL/6 mice and is predicted to share sequence and structural similarity with other nonclassical MHC class I genes4, 5. Because a virus-encoded ligand that activates NK cells seems teleologically unsound from the standpoint of the virus, it is notable that m157 binds the putative inhibitory receptor Ly49I on a subset of NK cells in CMV-susceptible 129/J mice4. The significance of m157-induced activating and inhibitory functions is unknown, particularly in noninbred mice, but their existence suggests constant evolution in the host as well as the virus. The dual specificity of MCMV m157 for NK-activating and NK-inhibitory receptors could provide new insight about HCMV UL18. Like m157, UL18 might exert different effects upon NK cells by interacting with distinct receptors in different in vitro or in vivo systems. In addition, the example of m157 is a useful reminder that viral homologs of MHC class I function in a complex, dynamic environment of viral and immune elements during in vivo infection. Additional MHC class I homologs await characterization. Rat cytomegalovirus (RCMV) r144 has not been assayed for direct effects upon NK cell cytotoxicity, but lower virus titers occur in the spleens of rats infected with an r144-deleted strain of RCMV than in those infected with wild-type RCMV21. This suggests that like other class I homologs, r144 may be a ligand for an NK cell inhibitory receptor. Molluscum contagiosum virus (MCV) also encodes a class I homolog, MC080R, which is retained in the endoplasmic reticulum and Golgi of transfected cells 22. Its effects on NK cell activity are presently unknown. Most promising is the recent identification of ten class I homologs in the MCMV genome, in addition to m144 and m1575. These genes were not detected earlier because they have structural rather than sequence similarity to known class I molecules. Their discovery is suggestive of the possibility that additional, undetected class I homologs might exist in other viral genomes as well. Selective modulation of MHC class I allele expression A common feature of many viral infections is the virus-induced modulation of class I expression. Viruses down-modulate class I molecules that are efficient at presenting viral peptides to CD8 + cytotoxic T cells (CTLs), such as HLA-A and HLA-B, to evade CTL-mediated destruction. In contrast, either HLA-C and HLA-E, the dominant ligands for NK cell–inhibitory receptors, are spared from virus-induced clearance from the cell surface or their expression is specifically enhanced. The selectivity of viral proteins for certain class I molecules appears to be indicative of a compromise ensuring that class I expression is diminished only to an extent that will still allow efficient inhibition of NK cells. Such a strategy appears to allow viruses to walk a fine line between the adaptive and innate arms of the immune system. A large number of viral proteins participate in class I downregulation23. Here, the discussion will be limited to viral down-modulation of class I molecules as it relates to NK cell responses. Many viral proteins cause class I molecules to deviate from their normal progression from the endoplasmic reticulum to the cell surface. At least four HCMV proteins, US2, US3, US6 and US11, function in this manner. US2 and US11 show a certain degree of selectivity in their targeting of class I molecules24, 25. In particular, two dominant inhibitory receptor ligands, HLA-C and HLA-E, are resistant to either US2- or US11-mediated degradation, suggesting that virus-infected cells evade NK cell activity by sparing the class I molecules least effective at presenting viral peptides to CTL but most effective at inhibiting NK cells26, 27. In contrast, class I molecules are nonselectively downmodulated from the cell surface by US3 and US6, and these proteins can partially inhibit the cell surface expression of HLA-C and HLA-G28. It is not clear whether the extent of down-modulation mediated by US3 and US6 is sufficient to make infected cells susceptible to NK cell lysis. Notably, none of these US proteins that down-regulate HLA expression inhibit the expression of the viral encoded class I homolog UL18, further demonstrating a selective targeting for NK cell evasion 29. MCMV possesses three genes, m04, m06 and m152, that selectively modulate class I expression; these genes are not homologous to the HCMV US genes and use slightly different modulation mechanisms. This indicates that HCMV and MCMV independently evolved systems to regulate class I expression. Notably, although the m04–class I protein complex inhibits CTLs, it is expressed on the cell surface where it could potentially inhibit NK cells 30. A second mechanism whereby certain viruses modulate class I expression is by accelerating the endocytosis of class I molecules from the cell surface. The nef gene, present in primate lentiviruses such as HIV-1, HIV-2 and SIV, encodes a 27-kD protein that is expressed early after infection and selectively down-modulates the expression of HLA-A and HLA-B, but not HLA-C or HLA-E31, 32. Thus, HIV-infected target cells remain resistant to lysis by NK cells, and their resistance depends on the failure of Nef to down-modulate HLA-C and HLA-E from the cell surface. Nef acts by inducing the clathrin adaptor protein complex to recognize a tyrosine-based sorting motif in the cytoplasmic tail of HLA-A and HLA-B32. The resistance of HLA-C to Nef-induced down-regulation is due to locusspecific tyrosine-to-cysteine and aspartic-acid-to-asparagine substitutions in the cytoplasmic tail31, 32. The SIV Nef protein also down-modulates monkey class I proteins via endocytosis but uses a COOH-terminal region of SIV Nef that is not present in HIV Nef, suggesting once again that viruses have repeatedly evolved multiple mechanisms to modulate class I expression 33. Acceleration of class I molecule endocytosis is also a feature of Kaposi's sarcoma–associated herpesvirus (KSHV). Two KSHV proteins, K3 and K5, induce the rapid endocytosis of class I proteins from the cell surface34, 35. Whereas K3 down-modulates HLA-A, HLA-B, HLA-C and HLA-E, K5 is more selective and down-modulates only HLA-A and HLA-B36. Because K5 has other functions, it is difficult to evaluate the importance of HLA-C and HLA-E resistance to K5 endocytosis with regard to NK cells36. In addition to their specific down-modulation of class I alleles, viruses also up-regulate certain class I alleles to evade NK cells. Previous work suggests that HCMV actively enhances the expression of HLA-E, the ligand for the inhibitory CD94–NKG2A receptor complex15, 37. Cell surface expression of HLA-E requires binding of a nonamer peptide derived from the signal sequence of most HLA molecules38. The HCMV UL40 protein possesses a nonamer peptide homologous to HLA signal sequences and thus can enhance cell surface expression of HLA-E15, 37. HLA-E binds the UL40 peptide in a TAP-independent manner, presumably bypassing the inhibitory effects of the HCMV US6 protein. In HCMV-infected cells, UL40 is necessary to mediate resistance to NK cell lysis in a CD94- and MHC class I–dependent manner39. Additional investigation is required to test the effects of virus-induced MHC modulation on NK cell resistance. One complicating feature is that the retention of HLA-C on the cell surface may actually result in lysis of the virus-infected cells by NK cells possessing the cognate activating KIR but no inhibitory KIR40, 41. Similarly, because HLA-E also binds to the CD94-NKG2C–activating receptor14, UL40-induced up-regulation of HLA-E could potentially activate NK cells. Thus, it is likely that selective HLA regulation contributes to viral evasion of NK cells, but the relative importance of this mechanism is uncertain. Virus-mediated inhibition of activating receptor function In addition to receptors with potent inhibitory capabilities, NK cells have receptors whose ligation can induce cytotoxicity, proliferation and cytokine production 1. Several such activating receptors have been characterized, and some recognize putative ligands that include specific viral products 3-5. An effective viral evasion strategy, therefore, would be to interfere with the process of activating receptor ligation. Several means to this end have been identified. The most commonly documented mechanism of interference with activating receptor function is virus-mediated down-regulation of activating receptor ligands in infected cells. For example, certain strains of HCMV increase the resistance of their infected host cells to NK cell cytotoxicity by downregulating LFA-342. This regulation, which is independent of the virus effects on class I molecules discussed previously, presumably interferes with the binding of LFA-3 to the NK cell–activating receptor, CD2. In addition, MCMV m152 encodes gp40, a protein that presumably inhibits surface expression of the murine ligand for the NKG2D-activating receptor, H-6043. Cells infected with m152-deleted MCMV have greater expression of H-60 and are more readily lysed by NK cells. An additional example of host-cell ligand regulation as an evasion strategy is found in KSHV. Transfection of target cells with the K5 gene of KSHV results in decreased expression of ICAM-1 and B7-2, both of which can serve as ligands for NK cell–activating receptors36, 44. The K5-mediated reduction in the surface expression of these ligands results in target-cell escape from NK cell cytotoxicity in certain in vitro NK cell systems36, but not in others44. K5 is an E2 ubiquitin ligase and thus probably directs the ubiquitination of the NK cell–activating receptor ligands it targets, ultimately resulting in their degradation34. As a group, the host-cell molecules targeted by viruses have functions other than as NK cell–activating receptor ligands and thus these examples, although relevant, are not entirely specific to NK cells. The discovery of ligands for activating receptors specific to NK cells will probably lead to fruitful studies of viral regulation of their expression. A second way that viruses may interfere with activating receptor function is by virus-induced modification of the ligand on target-cell surfaces. Infection of certain target cells by HIV, human T cell lymphotrophic virus I (HTLV-I) or HTLV-II can result in resistance to NK cell lysis associated with sialylation of surface molecules45. Although target-cell binding is not impaired by infection, chemical removal of sialic acid restores susceptibility to NK cell cytotoxicity. These receptor modifications have not been specifically seen on known NK cell ligands, but it can be reasoned that activating receptor ligands would be the most likely targets of virus-induced sialylation. It is to be hoped that follow-up studies will reveal the viral proteins responsible for, and specific cellular targets of, this evasion strategy. A third mechanism for avoiding NK cell activation is antagonism of the activating receptor and ligand interaction. An example is the interaction of an HCMV encoded protein with the NKG2D receptor. NKG2D is an activating receptor expressed on NK cells and subsets of T cells that binds to the UL16-binding protein (ULBP) family of glycosylphosphatidylinositol-linked receptors and MHC class I–related molecules (MIC) in humans, as well as the retinoic acid–inducible early gene 1 (Rae1) protein family and H-60 minor histocompatibility antigen in mice46. Ligation of NKG2D results in signal transduction via DAP10, leading to NK cell cytotoxicity46. HCMV encodes UL16, which in a soluble form is capable of binding ULBP, thus blocking the interaction between the NKG2D activating receptor complex and its cognate activating ligand47, 48. These data suggest a mechanism by which UL16 might inhibit NK cell function in vivo. A final means by which viruses interfere with activating receptor function is through the inhibition of signaling induced by activating-receptor ligation. Although these mechanisms can potentially inhibit many cells, at least one viral protein appears to have some specificity for NK cell activation. HIV-1 Tat can block the NK cell activation and function induced by ligation of LFA-1 on the NK cell surface49, 50. Tat does not affect NK cell adhesion to the target cell, but specifically binds to an L- type calcium channel on NK cells49. Its binding blocks calcium influx and subsequent induction of the calcium-calmodulin kinase II in NK cells, which is required for cytotoxicity50. The search for viral products that specifically block signaling for NK cell cytotoxicity will certainly become more productive as the appreciation of these pathways in NK cells grows. Evasion by modulation of cytokines or chemokines Viruses may subvert NK cell responses through virus-encoded proteins that counteract or modulate the interactions between cytokine or chemokine molecules and their cognate receptors. Numerous poxviruses and herpesviruses encode homologs to known cytokines and chemokines with agonistic or antagonistic function, or secreted proteins or receptors that bind with high affinity to cytokines and chemokines51. Although several examples of this sort of molecular mimicry have been described for different viruses, direct evidence for the involvement of NK cells in this strategy of immune escape in vivo is scarce. Interference with anti-viral NK cell function could involve inhibition or antagonism of cytokines such as IL-12, IL-18, TNF- , IL-1 , IL-1 and IL-15, which participate in inducing NK cell IFN- production and cytotoxicity8. Alternatively, viruses could facilitate overproduction or encode homologs of other cytokines (such as IL-1052) that have an inhibitory effect upon NK cells. Among the chemokines, targets for viral modulation could involve those that directly affect NK cell chemotaxis, including MIP-1 (CCL3), MIP-1 (CCL4), MCP-1 (CCL2), MCP2 (CCL8), MCP-3 (CCL7) and RANTES (CCL5)8, or other chemokines and chemokine receptors involved in recruitment of leukocyte subsets that influence NK cell function. Several virus-produced chemokine homologs have been described that might interfere with NK cell–mediated defense. The putative CC-chemokine homolog m131-m129 (or MCK-2) of MCMV is directly linked to NK cell evasion53, 54. Infection of mice with an m131-m129–deleted mutant virus results in decreased viral burden in the spleen and liver. The increased clearance of this mutant virus is negated by NK cell depletion, suggesting that m131-m129 inhibits NK cell–mediated viral clearance. The exact mechanism underlying this function of m131-m129 is unclear. Another chemokine homolog with possible relevance to NK cells is the broad-spectrum CC, CXC and CX3C chemokine antagonist vMIP-II of KHSV, which could block chemotactic responses of monocytes to RANTES, MIP-1 and MIP-1 55. In addition, the CCR8 antagonist vMIP-I binds to chemokine receptors on NK cells56. Similarly, MCV also encodes a homolog, MC148, that is a narrow-spectrum antagonist of MCP-157. Each of these chemokine antagonists could potentially interfere with NK cell responses. In contrast, virus-encoded homologs of the cytokine IL-10, which function as agonists, have been cloned from HCMV and Epstein-Barr virus (EBV)58, 59. These viral proteins may impair NK cell activation by inhibiting production of type 1 cytokines as well as by directly acting upon NK cells. Specific interference with NK cell activation can also be mediated by cytokine-binding protein homologs. A principal target is IL-18, which is central to NK cell production of IFN- . Ectromelia poxvirus (EV) encodes a cytokine-binding protein with NK cell–specific effects60. The EV p13 protein is homologous to mammalian IL-18BP, binds murine IL-18 and inhibits IL-18 receptor binding and activity. Mice infected with a p13-deleted mutant virus have markedly greater NK cell cytotoxicity than those infected with wild-type EV, as a result of increased NK cell activation. Compared to mice infected with p13-deleted virus, mice infected with wild-type EV have lower IFNproduction and higher IL-10 production, further highlighting the importance of this viral protein in NK cell evasion60. MCV also encodes several gene products that likely modulate host immunity61, 62, including a functional IL-18BP homolog, MC54L63. The evolution of these homologs points to the importance of the NK cell IFN- pathway in the elimination of virus-infected cells, and shows the importance of IL-18 function. Binding and sequestration of IL-18 from its cognate receptor has also been suggested to be involved in human papillomavirus (HPV)–related pathogenesis64, 65. The oncoproteins HPV16 E6 and E7 inhibit IL-18–induced IFN- production in PHA-stimulated PBMC and in an IL-12–stimulated immortalized NK cell line by specific and competitive binding to IL-1865 or IL-18R 64. Other proteins without homology to known cytokine or chemokine receptors or binding proteins that can act as cytokine or chemokine antagonists include murine -herpesvirus 68 M3, which encodes a secreted 44-kD broad-spectrum chemokine binding protein (hvCKBP), and a 35-kD soluble CCchemokine–specific binding protein (vCKBP) encoded by vaccinia virus. Both proteins block the activity of several chemokines, including the NK cell chemoattractants MIP-1 66, 67, MCP-1 and RANTES67, and thus function as inhibitors with potential NK cell specificity. Direct viral effects on NK cells Viruses can exert direct effects on NK cells. In particular, viruses can infect and inhibit or destroy NK cells, or cause inhibition through presumably direct contact with NK cells. These mechanisms should be particularly advantageous to viruses that attain chronically high systemic or organspecific titers. Both HIV and HSV infect NK cells in vitro. HIV can infect cultured NK cells, and is found ex vivo in NK cells from HIV-seropositive individuals68, 69. Although in vitro infection of NK cells with HIV does not affect the overall lytic activity of NK cell cultures, it does greatly reduce viability68. Thus, HIV may evade NK cell responses to some degree by direct infection and induction of cytopathic effects. In contrast, HSV can spread from infected fibroblasts to cultured NK cells, and thus inhibits NK cell cytotoxicity69. This direct infection of NK cells is unusual and suggests that HSV-mediated inhibition of these cells may fall into the category of direct viral effects, although the mechanism is unknown. It will be useful to ascertain whether HSV gene products can specifically interfere with intracellular NK cell signals leading to activation in infected NK cells. Interactions between virus particles and NK cells that do not result in infection may exert other direct effects upon NK cells. One viral envelope protein has recently been implicated in inhibition of NK cells. E2, the major envelope protein of hepatitis C virus (HCV), binds to CD81 (TAPA-1), which induces a costimulatory signal in T cells70. In NK cells, however, ligation of CD81 by immobilized E2 or anti-CD81 inhibits NK cell cytotoxicity, IL-2–induced proliferation and IL-2–, IL-12– or IL-15– induced IFN- production71, 72. In addition, CD81 ligation specifically inhibits ERK and MAPK phosphorylation induced by CD16-mediated activation of NK cells71. These effects of E2 binding to CD81 are specific to NK cells, as opposite activities are induced in T cells. Thus, E2 can mediate specific inhibition of NK cells via an inhibitory receptor, and therefore direct viral binding to NK cells may quell NK cell responses. This mechanism may be particularly useful to HCV during viremia and in the liver, where viral titers are high and NK cell defense is important. Conclusion Viruses have evolved numerous mechanisms to evade immune responses. Although many are used to avoid multiple immunologic effector cells, several are specifically directed at NK cells. Viral evasion strategies targeting NK cells can be divided into five mechanistic categories: ligation of inhibitory class I receptors on NK cells by virus-encoded MHC class I homologs; virus-induced regulation of class I protein expression resulting in selective expression or up-regulation of class I molecules that can bind NK cell class I inhibitory receptors; interference with NK cell–activating receptor function caused by virus-mediated inhibition of the expression of the corresponding ligand, of their signaling in infected host cells or of the production of virus encoded antagonists of NK cell– activating receptors; inhibition of NK cell activation, trafficking, or both, resulting from viral modulation of cytokine–chemokine networks or virus-encoded cytokine or chemokine homologs that prevent NK cell function; and direct inhibitory effects of viruses on NK cells, including infection of NK cells and ligation of non-class I NK cell inhibitory receptors by viral envelope proteins. These multiple mechanisms highlight the importance of NK cells in defense against viral infections. In particular, certain viruses, including herpesviruses, appear to be especially adept in their avoidance of NK cells, consistent with the crucial role of NK cells in the control of these viruses. Further study of viral evasion mechanisms will provide useful models to elucidate NK cell biology as well as to provide therapeutic targets to enhance host advantage during infection. References 1. Biassoni, R. et al. Human natural killer cell receptors and co-receptors. Immunol. Rev. 181, 203-214 (2001). | Article | PubMed | ISI | 2. Smith, H.R., Idris, A.H. & Yokoyama, W.M. Murine natural killer cell activation receptors. Immunol. Rev. 181, 115-125 (2001). | Article | PubMed | ISI | 3. Mandelboim, O. et al. Recognition of haemagglutinins on virus-infected cells by NKp46 activates lysis by human NK cells. Nature 409, 1055-1060 (2001). | Article | PubMed | ISI | 4. Arase, H., Mocarski, E.S., Campbell, A.E., Hill, A.B. & Lanier, L.L. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296, 1323-1326 (2002). | Article | PubMed | ISI | 5. Smith, H.R. et al. Recognition of a virus-encoded ligand by a natural killer cell activation receptor. Proc. Natl. Acad. Sci. USA 99, 8826-8831 (2002). | PubMed | ISI | 6. Melero, I. et al. Signaling through the LFA-1 leucocyte integrin actively regulates intercellular adhesion and tumor necrosis factor- production in natural killer cells. Eur. J. Immunol. 23, 18591865 (1993). | PubMed | ISI | 7. Boles, K.S., Stepp, S.E., Bennett, M., Kumar, V. & Mathew, P.A. 2B4 (CD244) and CS1: novel members of the CD2 subset of the immunoglobulin superfamily molecules expressed on natural killer cells and other leukocytes. Immunol. Rev. 181, 234-249 (2001). | Article | PubMed | ISI | 8. Biron, C.A., Nguyen, K.B., Pien, G.C., Cousens, L.P. & Salazar-Mather, T.P. Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu. Rev. Immunol. 17, 189-220 (1999). | Article | PubMed | ISI | 9. Biron, C.A., Byron, K.S. & Sullivan, J.L. Severe herpesvirus infections in an adolescent without natural killer cells. N. Engl. J. Med. 320, 1731-1735 (1989). | PubMed | ISI | 10. Ballas, Z.K., Turner, J.M., Turner, D.A., Goetzman, E.A. & Kemp, J.D. A patient with simultaneous absence of "classical" natural killer cells (CD3-, CD16+, and NKH1+) and expansion of CD3+, CD4-, CD8-, NKH1+ subset. J. Allergy Clin. Immunol. 85, 453-459 (1990). | PubMed | ISI | 11. Wendland, T., Herren, S., Yawalkar, N., Cerny, A. & Pichler, W.J. Strong and TCR response in a patient with disseminated mycobacterium avium infection and lack of NK cells and monocytopenia. Immunol. Lett. 72, 75-82 (2000). | Article | PubMed | ISI | 12. Beck, S. & Barrell, B.G. Human cytomegalovirus encodes a glycoprotein homologous to MHC class-I antigens. Nature 331, 269-272 (1988). | PubMed | ISI | 13. Reyburn, H.T. et al. The class I MHC homologue of human cytomegalovirus inhibits attack by natural killer cells. Nature 386, 514-517 (1997). | PubMed | ISI | 14. Braud, V.M. et al. HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 391, 795-799 (1998). | Article | PubMed | ISI | 15. Tomasec, P. et al. Surface expression of HLA-E, an inhibitor of natural killer cells, enhanced by human cytomegalovirus gpUL40. Science 287, 1031 (2000). | Article | PubMed | ISI | 16. Leong, C.C. et al. Modulation of natural killer cell cytotoxicity in human cytomegalovirus infection: the role of endogenous class I major histocompatibility complex and a viral class I homolog. J. Exp. Med. 187, 1681-1687 (1998). | Article | PubMed | ISI | 17. Cosman, D. et al. A novel immunoglobulin superfamily receptor for cellular and viral MHC class I molecules. Immunity 7, 273-282 (1997). | PubMed | ISI | 18. Farrell, H.E. et al. Inhibition of natural killer cells by a cytomegalovirus MHC class I homologue in vivo. Nature 386, 510-514 (1997). | PubMed | ISI | 19. Kubota, A., Kubota, S., Farrell, H.E., Davis-Poynter, N. & Takei, F. Inhibition of NK cells by murine CMV-encoded class I MHC homologue m144. Cell. Immunol. 191, 145-151 (1999). | Article | PubMed | ISI | 20. Cretney, E. et al. m144, a murine cytomegalovirus (MCMV)-encoded major histocompatibility complex class I homologue, confers tumor resistance to natural killer cell-mediated rejection. J. Exp. Med. 190, 435-444 (1999). | Article | PubMed | ISI | 21. Kloover, J.S., Grauls, G.E., Blok, M.J., Vink, C. & Bruggeman, C.A. A rat cytomegalovirus strain with a disruption of the r144 MHC class I-like gene is attenuated in the acute phase of infection in neonatal rats. Arch. Virol. 147, 813-824 (2002). | PubMed | ISI | 22. Senkevich, T.G. & Moss, B. Domain structure, intracellular trafficking, and 2-microglobulin binding of a major histocompatibility complex class I homolog encoded by molluscum contagiosum virus. Virology 250, 397-407 (1998). | Article | PubMed | ISI | 23. Tortorella, D., Gewurz, B.E., Furman, M.H., Schust, D.J. & Ploegh, H.L. Viral subversion of the immune system. Annu. Rev. Immunol. 18, 861-926 (2000). | Article | PubMed | ISI | 24. Gewurz, B.E., Wang, E.W., Tortorella, D., Schust, D.J. & Ploegh, H.L. Human cytomegalovirus US2 endoplasmic reticulum-lumenal domain dictates association with major histocompatibility complex class I in a locus-specific manner. J. Virol. 75, 5197-5204 (2001). | Article | PubMed | ISI | 25. Machold, R.P., Wiertz, E.J., Jones, T.R. & Ploegh, H.L. The HCMV gene products US11 and US2 differ in their ability to attack allelic forms of murine major histocompatibility complex (MHC) class I heavy chains. J. Exp. Med. 185, 363-366 (1997). | Article | PubMed | ISI | 26. Schust, D.J., Tortorella, D., Seebach, J., Phan, C. & Ploegh, H.L. Trophoblast class I major histocompatibility complex (MHC) products are resistant to rapid degradation imposed by the human cytomegalovirus (HCMV) gene products US2 and US11. J. Exp. Med. 188, 497-503 (1998). | Article | PubMed | ISI | 27. Lopez-Botet, M., Llano, M. & Ortega, M. Human cytomegalovirus and natural killer-mediated surveillance of HLA class I expression: a paradigm of host-pathogen adaptation. Immunol. Rev. 181, 193-202 (2001). | Article | PubMed | ISI | 28. Jun, Y. et al. Human cytomegalovirus gene products US3 and US6 down-regulate trophoblast class I MHC molecules. J. Immunol. 164, 805-811 (2000). | PubMed | ISI | 29. Park, B. et al. The MHC class I homolog of human cytomegalovirus is resistant to down-regulation mediated by the unique short region protein (US)2, US3, US6, and US11 gene products. J. Immunol. 168, 3464-3469 (2002). | PubMed | ISI | 30. Kavanagh, D.G., Gold, M.C., Wagner, M., Koszinowski, U.H. & Hill, A.B. The multiple immuneevasion genes of murine cytomegalovirus are not redundant: m4 and m152 inhibit antigen presentation in a complementary and cooperative fashion. J. Exp. Med. 194, 967-978 (2001). | Article | PubMed | ISI | 31. Cohen, G.B. et al. The selective downregulation of class I major histocompatibility complex proteins by HIV-1 protects HIV-infected cells from NK cells. Immunity 10, 661-671 (1999). | PubMed | ISI | 32. Le Gall, S. et al. Nef interacts with the mu subunit of clathrin adaptor complexes and reveals a cryptic sorting signal in MHC I molecules. Immunity 8, 483-495 (1998). | PubMed | ISI | 33. Swigut, T., Iafrate, A.J., Muench, J., Kirchhoff, F. & Skowronski, J. Simian and human immunodeficiency virus Nef proteins use different surfaces to downregulate class I major histocompatibility complex antigen expression. J. Virol. 74, 5691-5701 (2000). | Article | PubMed | ISI | 34. Coscoy, L., Sanchez, D.J. & Ganem, D. A novel class of herpesvirus-encoded membrane-bound E3 ubiquitin ligases regulates endocytosis of proteins involved in immune recognition. J. Cell Biol. 155, 1265-1273 (2001). | Article | PubMed | ISI | 35. Ishido, S., Wang, C., Lee, B.S., Cohen, G.B. & Jung, J.U. Downregulation of major histocompatibility complex class I molecules by Kaposi's sarcoma-associated herpesvirus K3 and K5 proteins. J. Virol. 74, 5300-5309 (2000). | Article | PubMed | ISI | 36. Ishido, S. et al. Inhibition of natural killer cell-mediated cytotoxicity by Kaposi's sarcoma-associated herpesvirus K5 protein. Immunity 13, 365-374 (2000). | PubMed | ISI | 37. Ulbrecht, M. et al. Cutting edge: the human cytomegalovirus UL40 gene product contains a ligand for HLA-E and prevents NK cell-mediated lysis. J. Immunol. 164, 5019-5022 (2000). | PubMed | ISI | 38. Braud, V., Jones, E.Y. & McMichael, A. The human major histocompatibility complex class Ib molecule HLA-E binds signal sequence-derived peptides with primary anchor residues at positions 2 and 9. Eur. J. Immunol. 27, 1164-1169 (1997). | PubMed | ISI | 39. Wang, E.C. et al. UL40-mediated NK evasion during productive infection with human cytomegalovirus. Proc. Natl. Acad. Sci. USA 99, 7570-7575 (2002). | Article | PubMed | ISI | 40. Pietra, G. et al. Natural killer cells lyse autologous herpes simplex virus infected targets using cytolytic mechanisms distributed clonotypically. J. Med. Virol. 62, 354-363 (2000). | Article | PubMed | ISI | 41. Huard, B. & Fruh, K. A role for MHC class I down-regulation in NK cell lysis of herpes virus-infected cells. Eur. J. Immunol. 30, 509-515 (2000). | Article | PubMed | ISI | 42. Fletcher, J.M., Prentice, H.G. & Grundy, J.E. Natural killer cell lysis of cytomegalovirus (CMV)infected cells correlates with virally induced changes in cell surface lymphocyte function-associated antigen-3 (LFA-3) expression and not with the CMV-induced down-regulation of cell surface class I HLA. J. Immunol. 161, 2365-2374 (1998). | PubMed | ISI | 43. Krmpotic, A. et al. MCMV glycoprotein gp40 confers virus resistance to CD8+ T cells and NK cells in vivo. Nat. Immunol. 3, 529-535 (2002). | Article | PubMed | ISI | 44. Coscoy, L. & Ganem, D. A viral protein that selectively downregulates ICAM-1 and B7-2 and modulates T cell costimulation. J. Clin. Invest. 107, 1599-1606 (2001). | PubMed | ISI | 45. Zheng, Z.Y. & Zucker-Franklin, D. Apparent ineffectiveness of natural killer cells vis-a-vis retrovirusinfected targets. J. Immunol. 148, 3679-3685 (1992). | PubMed | ISI | 46. Sutherland, C.L., Chalupny, N.J. & Cosman, D. The UL16-binding proteins, a novel family of MHC class I-related ligands for NKG2D, activate natural killer cell functions. Immunol. Rev. 181, 185-192 (2001). | Article | PubMed | ISI | 47. Kubin, M. et al. ULBP1, 2, 3: novel MHC class I-related molecules that bind to human cytomegalovirus glycoprotein UL16, activate NK cells. Eur. J. Immunol. 31, 1428-1437 (2001). | Article | PubMed | ISI | 48. Cosman, D. et al. ULBPs, novel MHC class I-related molecules, bind to CMV glycoprotein UL16 and stimulate NK cytotoxicity through the NKG2D receptor. Immunity 14, 123-133 (2001). | PubMed | ISI | 49. Zocchi, M.R., Rubartelli, A., Morgavi, P. & Poggi, A. HIV-1 Tat inhibits human natural killer cell function by blocking L-type calcium channels. J. Immunol. 161, 2938-2943 (1998). | PubMed | ISI | 50. Poggi, A. et al. NK cell activation by dendritic cells is dependent on LFA-1-mediated induction of calcium-calmodulin kinase II: inhibition by HIV-1 Tat C-terminal domain. J. Immunol. 168, 95-101 (2002). | PubMed | ISI | 51. Lalani, A.S., Barrett, J.W. & McFadden, G. Modulating chemokines: more lessons from viruses. Immunol. Today 21, 100-106 (2000). | Article | PubMed | ISI | 52. D'Andrea, A. et al. Interleukin 10 (IL-10) inhibits human lymphocyte interferon production by suppressing natural killer cell stimulatory factor/IL-12 synthesis in accessory cells. J. Exp. Med. 178, 1041-1048 (1993). | PubMed | ISI | 53. Fleming, P. et al. The murine cytomegalovirus chemokine homolog, m131/129, is a determinant of viral pathogenicity. J. Virol. 73, 6800-6809 (1999). | PubMed | ISI | 54. Saederup, N., Aguirre, S.A., Sparer, T.E., Bouley, D.M. & Mocarski, E.S. Murine cytomegalovirus CC chemokine homolog MCK-2 (m131-129) is a determinant of dissemination that increases inflammation at initial sites of infection. J. Virol. 75, 9966-9976 (2001). | Article | PubMed | ISI | 55. Kledal, T.N. et al. A broad-spectrum chemokine antagonist encoded by Kaposi's sarcomaassociated herpesvirus. Science 277, 1656-1659 (1997). | Article | PubMed | ISI | 56. Inngjerdingen, M., Damaj, B. & Maghazachi, A.A. Expression and regulation of chemokine receptors in human natural killer cells. Blood 97, 367-375 (2001). | Article | PubMed | ISI | 57. Luttichau, H.R. et al. A highly selective CC chemokine receptor (CCR) 8 antagonist encoded by the poxvirus molluscum contagiosum. J. Exp. Med. 191, 171-180 (2000). | Article | PubMed | ISI | 58. Kotenko, S.V., Saccani, S., Izotova, L.S., Mirochnitchenko, O.V. & Pestka, S. Human cytomegalovirus harbors its own unique IL-10 homolog (cmvIL-10). Proc. Natl Acad. Sci. USA 97, 1695-1700 (2000). | Article | PubMed | ISI | 59. Moore, K.W. et al. Homology of cytokine synthesis inhibitory factor (IL-10) to the Epstein-Barr virus gene BCRFI. Science 248, 1230-1234 (1990). | PubMed | ISI | 60. Born, T.L. et al. A poxvirus protein that binds to and inactivates IL-18, and inhibits NK cell response. J. Immunol. 164, 3246-3254 (2000). | PubMed | ISI | 61. Senkevich, T.G. et al. Genome sequence of a human tumorigenic poxvirus: prediction of specific host response-evasion genes. Science 273, 813-816 (1996). | PubMed | ISI | 62. Senkevich, T.G., Koonin, E.V., Bugert, J.J., Darai, G. & Moss, B. The genome of molluscum contagiosum virus: analysis and comparison with other poxviruses. Virology 233, 19-42 (1997). | Article | PubMed | ISI | 63. Xiang, Y. & Moss, B. IL-18 binding and inhibition of interferon induction by human poxvirusencoded proteins. Proc. Natl Acad. Sci. USA 96, 11537-11542 (1999). | Article | PubMed | ISI | 64. Lee, S.J. et al. Both E6 and E7 oncoproteins of human papillomavirus 16 inhibit IL-18-induced IFNproduction in human peripheral blood mononuclear and NK cells. J. Immunol. 167, 497-504 (2001). | PubMed | ISI | 65. Cho, Y.S. et al. Down modulation of IL-18 expression by human papillomavirus type 16 E6 oncogene via binding to IL-18. FEBS Lett. 501, 139-145 (2001). | Article | PubMed | ISI | 66. Parry, C.M. et al. A broad spectrum secreted chemokine binding protein encoded by a herpesvirus. J. Exp. Med. 191, 573-578 (2000). | Article | PubMed | ISI | 67. Alcami, A., Symons, J.A., Collins, P.D., Williams, T.J. & Smith, G.L. Blockade of chemokine activity by a soluble chemokine binding protein from vaccinia virus. J. Immunol. 160, 624-633 (1998). | PubMed | ISI | 68. Chehimi, J. et al. In vitro infection of natural killer cells with different human immunodeficiency virus type 1 isolates. J. Virol. 65, 1812-1822 (1991). | PubMed | ISI | 69. York, I.A. & Johnson, D.C. Direct contact with herpes simplex virus-infected cells results in inhibition of lymphokine-activated killer cells because of cell-to-cell spread of virus. J. Infect. Dis. 168, 11271132 (1993). | PubMed | ISI | 70. Wack, A. et al. Binding of the hepatitis C virus envelope protein E2 to CD81 provides a costimulatory signal for human T cells. Eur. J. Immunol. 31, 166-175 (2001). | Article | PubMed | ISI | 71. Crotta, S. et al. Inhibition of natural killer cells through engagement of CD81 by the major hepatitis C virus envelope protein. J. Exp. Med. 195, 35-41 (2002). | Article | PubMed | ISI | 72. Tseng, C.T. & Klimpel, G.R. Binding of the hepatitis C virus envelope protein E2 to CD81 inhibits natural killer cell functions. J. Exp. Med. 195, 4349 (2002). Figure 1. Viral mechanisms for evading NK cells. The strategies by which viruses evade NK cells fall into five categories and are depicted in the interaction between a virus infected target cell (left) and an NK cell (right). (a) NK cells can be inhibited by a viral MHC class I homolog with structural similarity to endogenous host class I that binds to inhibitory class I receptors on NK cells. (b) Viruses can inhibit expression of HLA-A and HLA-B, resulting in a relative increase in HLA-C and HLA-E on the surface of the target cell; these inhibit NK cells through the class I inhibitory receptors CD94-NKG2A and KIR, respectively. Alternatively, viral gene expression can result in selectively increased expression of HLA-E, which inhibits NK cells through CD94-NKG2A. (c) Virus-encoded proteins can function as cytokine binding proteins that block the action of NK cell activating cytokines. In addition, viruses can produce homologs, or increase host production of cytokines that inhibit NK cells. (d) NK cell activities can also be avoided by decreased expression of NK cell–activating ligands in virus-infected target cells, which prevent signal transduction via NK cell–activating receptors. To achieve the same end, viruses can encode antagonists of the activating receptor–ligand interaction. (e) Viruses can also directly inhibit NK cells by infecting them or using envelope proteins to ligate NK cell inhibitory receptors. Proteins outlined in red are virally encoded. Each mechanism corresponds to the similarly numbered section of the text where additional details and examples are provided. Table 1. Viral mechanisms of evading NK cell responses Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 1013 - 1018 (2002) doi:10.1038/ni1102-1013 © Nature America, Inc. <> To kill or be killed: viral evasion of apoptosis Chris A. Benedict, Paula S. Norris & Carl F. Ware Division of Molecular Immunology, La Jolla Institute for Allergy and Immunology, 10355 Science Center Drive, San Diego, CA 92121, USA. Correspondence should be addressed to C F Ware cware@liai.org In the struggle between virus and host, control over the cell's death machinery is crucial for survival. Viruses are obligatory intracellular parasites and, as such, must modulate apoptotic pathways to control the lifespan of their host in order to complete their replication cycle. Many of the counter-assaults mounted by the immune system incorporate activation of the apoptotic pathway—particularly by members of the tumor necrosis factor cytokine family—as a mechanism to restrict viral replication. Thus, apoptosis serves as a powerful selective pressure for the virus to evade. However, for the host, success is harsh and potentially costly, as apoptosis often contributes to pathogenesis. Here we examine some of the molecular mechanisms by which viruses manipulate the apoptotic machinery to their advantage and how we (as vertebrates) have evolved and learned to cope with viral evasion. The apoptotic machinery Apoptosis results from a collapse of cellular infrastructure through regulated internal proteolytic digestion, which leads to cytoskeletal disintegration, metabolic derangement and genomic fragmentation. Members of the cytosolic caspase family of proteinases (cysteine-based, aspartatedirected) form "the engine" of the apoptotic pathway1 (Fig. 1). The caspases (11 in total) represent one of more than 20 distinct components involved in initiation, execution and regulatory phases of the pathway, which indicates the extensive regulation this process has engendered over time. The apoptotic machinery shows sensitivity to a variety of agents by coordinating signals initiated by both internal sensors (intrinsic pathway, mitochondria-dependent) and external stimuli (extrinsic pathway, death receptor–mediated). When triggered, internal sensors (for example, p53) can initiate processes that result in the ultimate loss of mitochondrial integrity and apoptosis. Signals from the internal sensors are propagated to the mitochondria via pro-apoptotic Bcl-2 subfamily members (BH3 only), such as Bid, to oligomerize Bax and Bak, which are outer mitochondrial membrane proteins that promote the release of cytochrome c. Cytochrome c oligomerizes Apaf1 and recruits pro-caspase-9 (forming the apoptosome), which results in proteolytic conversion of pro-caspase-9 to an active enzyme. Caspase-9 then converts pro-caspase-3 to its active form, which, with other executioner caspases, such as caspase-7, then cleaves key substrates in the cell to orchestrate the cell's fatal collapse. High resolution image and legend (67K) Figure 1. Molecular pathways of apoptosis. The intrinsic apoptotic pathway is initiated by internal sensors that monitor cellular stresses such as viral infection via activation of BH3 domain–only members of the Bcl-2 family. Activated BH3-only proteins are thought to mediate the assembly of pro-apoptotic members of the Bcl-2 family (Bax, Bak, Bcl-rambo, Bok) into hetero-oligomeric "pores" in the outer membrane of the mitochondria; this results in the release of factors such as cytochrome c, Smac (also known as Diablo) and Omi (also known as HtrA2) into the cytoplasm. The loss of mitochondrial membrane integrity can be blocked by the anti-apoptotic Bcl-2 family members Bcl-2, Bcl-xL, Bcl-w, Mcl-1 and Bcl-B. Release of cytochrome c promotes formation of the apoptosome, which contains Apaf-1 and pro-caspase-9. Autocatalytic activation of caspase-9 initiates the effector caspase cascade, which includes caspase-2, -3, -6 and/or -7. Caspase activation is negatively regulated by the IAPs, which are counter-balanced by the release of pro-apoptotic Smac and Omi from the mitochondria. The extrinsic pathway of apoptosis is triggered by TNF family death ligands binding to their cognate death receptors. Via their DDs, multimerized receptors interact with the DDs of adaptor proteins such as FADD. These adaptor proteins also contain DEDs that facilitate their binding to procaspase-8 and/or pro-caspase-10 to form the DISC. As part of the DISC, the pro-caspases are cleaved into their active forms and initiate the intrinsic pathway of apoptosis by cleaving Bid into tBid and activate the effector caspase cascade (caspase-3 is shown). In contrast, the extrinsic pathway starts with members of the tumor necrosis factor (TNF) superfamily of death receptors transmitting external signals provided by immune effector cells to the virus-infected cell. Innate effector cells—for example, natural killer (NK) cells and dendritic cells (DCs)—can mount rapid antiviral responses through direct detection of viral products by pattern recognition receptors—such as binding of double-stranded RNA (dsRNA) by Toll-like receptor 3 (TLR3)—or by up-regulating death receptor ligands—for example, Fas ligand (FasL), TNF receptor– related apoptosis-inducing ligand (TRAIL) and TNF2. Later during adaptive immunity, these same death ligands are produced by antigen-specific cytotoxic T cells (CTLs). The lethal program set in motion by these ligands mandates that their expression is transient and highly regulated at the transcriptional and post-translational steps. Upon binding TNF-family ligands, death receptors recruit adaptors and initiator caspases in a stepwise sequence based on specific interaction domains and form the death-inducing signaling complex (DISC) at the plasma membrane. In the case of Fas, the cytosolic adaptor FADD (Fas-associated death domain– containing protein) contains two interaction motifs: a death domain (DD) that associates with the homologous structure in Fas and a death effector domain (DED) that interacts with a homologous DED in pro-caspase-8 or pro-caspase-10. The proximal positioning of pro-caspase-8 and procaspase-10 in the DISC is thought to lead to autocatalysis and conversion to an active enzyme. Activated caspase-8 or caspase-10 can then directly convert pro-caspase-3 to its active form, completing the initiation phase of the pathway. There is cross-talk between the death receptor pathways and the mitochrondria-dependent arm through cleavage of the BH3-only protein Bid by caspase-8. In addition, the cytotoxic granule–associated proteinase granzyme B can bypass caspase-8 and, upon delivery by CTLs and NK effector cells, can directly cleave and activate caspase-3. Caspase-3 and other executioner caspases cleave numerous substrates, such as ICAD (inhibitor of caspase-activated DNase), which leads to genome fragmentation, collapse of the cell and preparation of cellular remnants for phagocytosis. Thus, vertebrates have evolved several distinct strategies to initiate apoptosis that lead to a common execution phase of the pathway. All of the key pro-apoptotic components are preformed in the cell, enabling the pathway to respond rapidly to apoptosis-inducing signals. In contrast, the cellular regulatory elements of the death pathway often require new gene expression. The transcription factor NF- B controls several genes, including those of the Bcl-2 and inhibitors of apoptosis (IAP) families, whose functions regulate apoptosis and promote cell survival3. This is especially pertinent when a virus has compromised the biosynthetic capacity of the host cell: death becomes the default outcome and, predictably, viral counter-strategies have evolved that mimic the key cellular regulatory elements. The key regulatory steps in the cell death pathway targeted by viral pathogens include control over the expression of internal sensors, cytochrome c release by Bcl-2 family members, inhibition of caspases and regulation of death receptor signaling. Indeed, the discovery of several of the key regulatory points in the apoptotic pathway have emerged directly from studies of the death-escape mechanisms used by viruses. Blinding the sensors The tumor suppressor p53 limits cellular proliferation by inducing apoptosis or cell cycle arrest in response to cellular stresses and is intimately linked to cancer development. p53-deficient mice are more prone to certain viral infections, indicating a role beyond tumor suppression4. Some of the genes whose transcription is stimulated by p53 include those that encode the death receptors Fas and TRAIL receptor 2 (TRAILR2), although the importance of their up-regulation by p53 in triggering apoptosis is unclear. Additionally, p53 induces the transcription of genes such as Bax, Bak1, Bbc3 and Pmaip1 whose products are involved in death signal propagation through the mitochondria5-9. p53 represses transcription of the anti-apoptotic protein Bcl-2, an antagonist of Bax and Bak 10. Viruses can also disrupt apoptosis by inactivating p53. The SV40 large T antigen binds to p53 and sequesters it in an inactive complex11, 12. The human papillomavirus E6 protein and adenovirus E1B-55K protein, in concert with E4orf6, promote ubiquitination and degradation of p53, albeit via different mechanisms13-16. Additionally, the pX protein encoded by hepatitis B virus complexes with p53 and inhibits p53-mediated transcriptional activation as well as p53-dependent apoptosis17. Mimicking Bcl-2 Viral encoded orthologs of the anti-apoptotic regulator Bcl-2 are a widely used immune evasion strategy (Fig. 2). A pertinent example is adenovirus E1B-19K, which is similar both in sequence and function to Bcl-218, 19. Unlike cell death signaled through cell-surface receptors, p53-mediated apoptosis in response to adenovirus infection does not require the cleavage of Bid into t-Bid to achieve Bax-Bak oligomerization and the subsequent release of cytochrome c. Instead, p53 stimulates a conformational change in Bax that is required for Bax-Bak interaction. These events are blocked by adenovirus E1B-19K as a result of its binding to Bak and the abrogation of Bax-Bak oligomerization20. E1B-19K also affects the apoptotic process signaled via TNF at the level of Bax and Bak activation. Treatment of cells with TNF produces a death signal that results in the cleavage of Bid to t-Bid followed by the recruitment of monomeric, pro-apoptotic Bax into a 500-kD protein complex and the release of cytochrome c from the mitochondria. In the case of TNF-mediated apoptosis, E1B-19K appears to interact primarily with Bax, inhibiting oligomerization and the subsequent release of cytochrome c21. High resolution image and legend (61K) apoptotic pathway. Figure 2. Viral regulation of the intrinsic Cellular sensors, such as p53, that detect stress and initiate the apoptotic process are inactivated by the proteins adenovirus E1B-55K and human papillomavirus E6. Several viruses encode orthologs of Bcl-2 family proteins that antagonize their pro-apoptotic activity. Several viral strategies target the caspases. The vIAPs contain conserved BIR domains and their expression may shift the equilibrium of Smac to favor cell survival. Non-BIR–containing proteins, such as p35 and the serpins (including CrmA), also inhibit caspase activation. Oncogenic human herpesviruses use Bcl-2 orthologs to block mitochondrial release of cytochrome c. This is also true for Epstein-Barr virus (EBV), which encodes two Bcl-2 orthologs (BHRF1 and BALF-122, 23), and Kaposi's sarcoma–associated -herpesvirus (KSHV), which expresses KSbcl-224, 25. Mouse -herpesvirus MHV-68, a virus that serves as a model for human EBV infection, encodes a Bcl-2 ortholog (MHVBcl-2) that protects against TNF-mediated apoptosis in cell culture. In addition, MHVBcl-2 is important for chronic infection, as demonstrated by the impaired virulence of MHV-68 virus lacking the Bcl-2 ortholog in interferon- (IFN- )–deficient mice26. In contrast, the human cytomegalovirus (CMV) UL37 gene product vMIA shares no sequence homology to Bcl-2; however, it resides in the mitochondria and appears to be functionally similar to Bcl-2, as it associates with the adenine nucleotide translocator and inhibits Fas-mediated apoptosis27. Other viral proteins inhibit apoptosis by modulating Bcl-2 family members at the transcriptional level or via post-translational modification. The human T cell leukemia virus type 1 (HTLV-1) Tax protein transcriptionally activates the Bcl-xL promoter while repressing transcription of Bax28. HIV-1 Nef mediates phosphorylation of the pro-apoptotic Bad protein, abrogating its activity and suppressing apoptosis in T cells29. Similarly, the U(S)3 protein kinase encoded by herpes simplex virus 1 (HSV1) mediates a post-translational modification of Bad and blocks its cleavage and subsequent activation of apoptosis30. Caspase regulation Caspases play a central role in apoptosis and are regulated in several ways 31-33. The enzymatic activity of caspases is inhibited by a conserved family of inhibitor of apoptosis proteins (IAPs) 34, 35 that were originally defined in baculovirus based on the suppression of apoptosis and presence of a zinc-binding motif called a BIR (baculoviral IAP repeat) 36. To date, eight cellular IAPs have been identified that regulate both the effector and initiator caspases37. For example, XIAP, c-IAP1, c-IAP2 and ML-IAP (livin) target the initiator caspase, caspase-9, and the effector caspases, caspase-3 and caspase-7. XIAP, c-IAP1 and c-IAP2 contain three BIR domains, each with a different function. The third BIR domain (BIR3) inhibits the activity of processed caspase-9, whereas the linker region between BIR1 and BIR2 abrogates the activity of caspase-3 and caspase-7. An IAP-ortholog strategy is used infrequently in mammalian viruses, with the exception of African swine fever virus, which encodes a viral IAP (vIAP) that does not contribute to virulence 38. In mammals, the serpin CrmA—which is derived from cowpox and is present in most poxviruses—also inhibits several caspases, likely through covalent modification of caspase-8, and blocks or delays apoptosis in response to TNF and Fas signaling or CTLs39-41. Also, CrmA inhibits caspase-1, a critical processing enzyme for the inflammatory cytokine interleukin 1 (IL-1 ). Modulation of TNFR signaling Several members of the TNF receptor (TNFR) superfamily, including Fas, TNFR1 and TRAILR2, are potent inducers of apoptosis42. Not surprisingly many viruses specifically target these cytokine receptors (Fig. 3). Neutralization of TNF by soluble decoy receptors was one of the first-described evasion tactics, as shown by the secreted TNFR2 ortholog expressed by Shope fibroma virus (rabbit poxvirus)43. Several TNFR orthologs have been identified in the genomes of lepri- and orthopoxviruses, including smallpox, which indicates its impact on the success of poxviruses 44. The T2 protein of myxoma virus is a dimeric, high-affinity binding protein for TNF and virulence is attenuated when it is deleted from the viral genome45. Interestingly, the TNFR decoys in vaccinia, the attenuated form of smallpox, are mutated. Additionally, the poxviral TNFR ortholog T2 exists in an intracellular form that is required to inhibit apoptosis of lymphocytes 46. Another TNFR ortholog is found in avian leukocytosis virus, which encodes an ortholog of TRAILR2 that serves as a virus entry factor47. Additionally, human CMV (but not mouse CMV) contains a TNFR ortholog encoded by the UL144 orf, although its functional significance remains obscure 48. High resolution image and legend (52K) apoptotic pathway. Figure 3. Viral regulation of the extrinsic Poxviruses block signaling via TNF and lymphotoxin by producing soluble decoy receptors (for example, myxoma virus T2 protein). vFLIPs contain two DEDs that interact with the homologous DED of FADD and pro-caspase-8 or pro-caspase-10, blocking caspase activation and inhibiting death receptor–induced apoptosis. The human CMV protein vICA binds and inhibits pro-caspase-8 activation, but lacks a DED. The EBV protein LMP1 self-aggregates and engages TRAF and TRADD molecules, activating "anti-apoptotic" NF- B– and Jnk-dependent pathways. A heterocomplex of proteins encoded by the E3 region of adenovirus (E3-10.4K, E3-14.5K and E36.7K) facilitates the removal of the death receptors Fas, TRAILR1 and TRAILR2 from the surface of infected cells. Consequently, receptors accumulate in late endocytic compartments, and cells are desensitized to killing. Herpes simplex virus 1, through its envelope glycoprotein D, uses a TNFR family member, herpesvirus entry mediator (HVEM), to gain access to the lymphoid compartment, where it can induce apoptosis of T cells49 and block maturation of antigen-presenting DCs50. The major B cell– transforming protein in EBV, LMP1, behaves like a constitutively activated CD40 by engaging TRAFs and TNFR1-associated DD protein (TRADD), which are adaptors used by TNFR to activate the transcription of anti-apoptotic genes through NF- B and c-Jun NH2-terminal kinase (Jnk)dependent signaling pathways. In a transgenic model, LMP1 expression prevents B cells from localizing to the follicle, thus protecting cells harboring latent virus from interactions with T cells 51. Whereas poxviruses inhibit TNF ligand-receptor interactions by deploying a soluble receptor smoke screen, a distinct strategy is used by adenoviruses. The E3 region encodes several proteins that sweep the cell surface clear of the death-inducing receptors Fas, TRAILR1 and TRAILR2. The E3 proteins responsible for modulating cell surface amounts of Fas are E3-10.4K and E3-14.5K52, 53, which localize to various cellular membrane compartments, including the plasma membrane, as a heteromeric complex54. However, the ability of adenovirus to modulate TRAILR2 absolutely requires a third E3 protein, E3-6.7K, in addition to E3-10.4K and E3-14.5K55; this highlights the complexity of the mechanisms used by adenoviruses to inhibit signaling by these death receptors. Upon E3induced down-regulation, death-inducing receptors accumulate in late endocytic compartments, resulting in the desensitization of infected cells to killing by FasL and TRAIL53, 55. Prevailing wisdom is that the E3-10.4K–E3-14.5K–E3-6.7K complex pirates the cellular endocytic compartments that direct membrane protein trafficking. A similar strategy is used by the HIV Nef protein, which down-modulates CD4 and major histocompatibility complex (MHC) class I from the cell surface by cross-linking these proteins to the cellular endocytic machinery56. As a consequence of Nef's actions, the membrane forms of TNF and the related cytokine LIGHT show sustained expression on the surface of T cells, potentially contributing to the cytopathic effects of HIV on the T cell compartment57. The substantial death of bystander (uninfected) lymphocytes during HIV infection may stimulate lymphopoiesis, possibly by homeostatic control mechanisms, which could provide a pool of dividing (but not HIV-specific) T cells for HIV genome replication58. Viruses can down-modulate or enhance expression of death receptors and ligands to their own advantage. The loss of death receptors may function to protect infected cells from cytolysis by CTLs or NK cells, which express FasL or TRAIL upon activation59. In addition, this down-regulation may help inhibit apoptosis mediated by neighboring cells that are induced to express death receptor ligands upon infection. Induction of TRAIL has been observed in HCMV-50, 60 and reovirus-infected61 cells and of FasL by HSV in T cells and HCMV in DCs49, 50. It has even been proposed that this induction of FasL and TRAIL is another viral immune evasion tactic, through the killing of infiltrating host CTLs and DCs49, 50. IFNs induce TRAIL in various cell types60, 62, 63; this suggests that a possible mechanism for up-regulation of TRAIL could be via production of IFNs by the virus-infected cell. Several viruses have evolved a different strategy for blocking death receptor signaling at the level of DISC assembly; they do this through blockade of caspase-8 and caspase-10 processing. The viral FLICE (caspase-8) inhibitory proteins (FLIPs) contain DEDs, but lack caspase activity, and are present in the genomes of various -herpesviruses, including equine herpesvirus 2 (EHV-2), herpesvirus saimiri (HVS), KSHV, bovine herpesvirus 4 (BHV-4) and moluscum contagiosum virus (MCV)64, 65. The cellular ortholog of vFLIP was subsequently cloned after identification of the vFLIPs65 and exists in both a 26-kD short (cFLIPS) and 55-kD long (cFLIPL) form generated by alternative splicing. cFLIPS is essentially the cellular ortholog of vFLIP and encodes two DEDs, whereas cFLIPL encodes an additional COOH-terminal domain with high homology to caspase-8 and caspase-10. Both isoforms of cFLIP are recruited to the DISC and inhibit death receptor– induced apoptosis; however, their mechanisms of action are slightly different: cFLIP S completely inhibits proteolytic processing of caspase-8 (similar to vFLIP) and cFLIPL allows partial caspase-8 processing66. The HCMV UL36 gene product vICA also associates with caspase-8 and blocks its activation, but shows no sequence identity to its proposed cellular orthologs, the FLIPs67. vFLIPs interact with adaptor proteins that regulate the expression of NF- B, including TNFRassociated factor 2 (TRAF2), receptor-interacting protein (RIP), NF- B–inducing kinase (NIK) and the inhibitor of B-kinase 2 (IKK2)68, which indicates that the vFLIPs may also regulate activation of transcription factors important in inhibiting apoptosis. Death taxes Cell death comes at a potentially large cost for the host. However, it is becoming appreciated that IFN-dependent nonapoptotic mechanisms can result in the successful attenuation of viral spread 69. IFNs play a critical role in mounting innate responses to viral infection. IFNs are typically considered nonapoptotic but, in some circumstances, can promote apoptosis of virus-infected cells and are especially potent when they act in concert with TNF-related ligands. IFNs signal via the Janus protein kinase–signal transducers and activators of transcription pathway (Jak-STAT)70. Well studied IFN-inducible signaling pathways include the RNA-dependent protein kinase (PKR)71 and the 2',5-oligoadenylate and RNase-L systems72. Upon interaction with virus-derived dsRNA, PKR is activated and can inhibit host-cell translation via phosphorylation of elongation initiation-factor 2 (eIF2 )73. More recently, it has been shown that PKR has additional activities, including the modulation of NF- B74 and promotion of apoptosis by functional enhancement of the tumor suppressor genes p53 and IFN response factor 1 (IRF-1)75. The regulation of NF- B by PKR places this protein at a critical crossroad in the coordination of apoptotic signals, potentially through the NF- B–dependent induction of FasL and IFN-induced TRAIL. Several noncytolytic antiviral programs activated by IFNs can arrest the viral life cycle at different steps, thereby attenuating virus spread and limiting the infection. However, IFNs and their response genes are themselves targeted by many viruses76. Replication-competent HCMV specifically inhibits induction of IFN- transcription by an unknown mechanism 77 that may involve inhibition of IRF-3 activation78. This blockade of IFN- transcription is overridden by signaling via TNFR1 or the lymphotoxin receptor (LT R), but not by Fas or TRAIL receptors79. Induction of IFN- by these two TRAF-adapting receptors is NF- B–dependent, but occurs only in HCMV-infected cells. Therefore, host and virus factors cooperate to induce IFN- , establishing a state of détente in which the host cell survives and the viral genome persists, but cannot produce new virions. This differs from the case of hepatitis B, where IFN signaling results in protease-dependent clearance of the viral genome69. These two examples highlight the virus- and cell type–specific issues often seen with IFN's antiviral action. The diversity of strategies used by viruses to modulate the apoptotic pathway is as varied as there are viruses. In addition, for each virus, these strategies are integrated into a wider scheme that manipulates other aspects of host defenses. For example, herpesviruses have large DNA genomes that have accumulated an extensive repertoire of immune-evasion tactics that target both the afferent (for example, antigen recognition) and effector phases (apoptosis) of host defenses80. The cumulative effect of these mechanisms is thought to contribute to the ability of herpesviruses to sustain life-long infection. As a cautionary note, nearly all the data gathered to date are on clinically important viruses, which unfortunately often lack comparable animal models with which we can assess the role of immune-modulatory genes in viral pathogenesis. Indeed, there are significant differences in the molecular mechanisms used to modulate immune function by mouse CMV compared with those of human CMV. It is also important to recognize that a particular viral evasion strategy may act differently in a non-native host compared to in tissue culture, which also contributes to the difficulty in defining mechanisms of action. Nonetheless, the elucidation of evasion strategies directed at apoptotic mechanisms can provide deeper insight into the host-virus relationship that hopefully will yield better vaccine strategies. References 1. Green, D.R. Apoptotic pathways: paper wraps stone blunts scissors. Cell 102, 1-4 (2000). | PubMed | ISI | 2. Medzhitov, R. Toll-like receptors and innate immunity. Nature Rev. Immunol. 1, 135-145 (2001). | Article | PubMed | 3. Karin, M. & Lin, A. NF- B at the crossroads of life and death. Nature Immunol. 3, 221-227 (2002). | Article | PubMed | ISI | 4. Harvey, M. et al. Spontaneous and carcinogen-induced tumorigenesis in p53-deficient mice. Nature Genet. 5, 225-229 (1993). | PubMed | ISI | 5. Kannan, K. et al. DNA microarrays identification of primary and secondary target genes regulated by p53. Oncogene 20, 2225-2234 (2001). | Article | PubMed | ISI | 6. Miyashita, T. & Reed, J.C. Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 80, 293-299 (1995). | PubMed | ISI | 7. Nakano, K. & Vousden, K.H. PUMA, a novel proapoptotic gene, is induced by p53. Mol. Cell 7, 683-694 (2001). | PubMed | ISI | 8. Oda, E. et al. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53induced apoptosis. Science 288, 1053-1058 (2000). | Article | PubMed | ISI | 9. Wu, G.S. et al. KILLER/DR5 is a DNA damage-inducible p53-regulated death receptor gene. Nature Genet. 17, 141-143 (1997). | PubMed | ISI | 10. Gross, A., McDonnell, J.M. & Korsmeyer, S.J. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 13, 1899-1911 (1999). | PubMed | ISI | 11. Lane, D.P. & Crawford, L.V. T antigen is bound to a host protein in SV40-transformed cells. Nature 278, 261-263 (1979). | PubMed | ISI | 12. Linzer, D.I. & Levine, A.J. Characterization of a 54K dalton cellular SV40 tumor antigen present in SV40-transformed cells and uninfected embryonal carcinoma cells. Cell 17, 43-52 (1979). | PubMed | ISI | 13. Steegenga, W.T., Riteco, N., Jochemsen, A.G., Fallaux, F.J. & Bos, J.L. The large E1B protein together with the E4orf6 protein target p53 for active degradation in adenovirus infected cells. Oncogene 16, 349-357 (1998). | Article | PubMed | ISI | 14. Querido, E. et al. Degradation of p53 by adenovirus E4orf6 and E1B55K proteins occurs via a novel mechanism involving a Cullin-containing complex. Genes Dev. 15, 3104-3117 (2001). | Article | PubMed | ISI | 15. Scheffner, M., Werness, B.A., Huibregtse, J.M., Levine, A.J. & Howley, P.M. The E6 oncoprotein encoded by human papillomavirus types 16 and 18 promotes the degradation of p53. Cell 63, 1129-1136 (1990). | PubMed | ISI | 16. Werness, B.A., Levine, A.J. & Howley, P.M. Association of human papillomavirus types 16 and 18 E6 proteins with p53. Science 248, 76-79 (1990). | PubMed | ISI | 17. Wang, X.W. et al. Abrogation of p53-induced apoptosis by the hepatitis B virus X gene. Cancer Res. 55, 6012-6016 (1995). | PubMed | ISI | 18. White, E., Cipriani, R., Sabbatini, P. & Denton, A. Adenovirus E1B 19-kilodalton protein overcomes the cytotoxicity of E1A proteins. J. Virol. 65, 2968-2978 (1991). | PubMed | ISI | 19. White, E. Regulation of apoptosis by adenovirus E1A and E1B oncogenes. Semin. Virol. 8, 505-513 (1998). | Article | ISI | 20. Henry, H., Thomas, A., Shen, Y. & White, E. Regulation of the mitochondrial checkpoint in p53-mediated apoptosis confers resistance to cell death. Oncogene 21, 748-760 (2002). | Article | PubMed | ISI | 21. Sundararajan, R. & White, E. E1B 19K blocks Bax oligomerization and tumor necrosis factor - mediated apoptosis. J. Virol. 75, 7506-7516 (2001). | Article | PubMed | ISI | 22. Henderson, S. et al. Epstein-Barr virus-coded BHRF1 protein, a viral homologue of Bcl-2, protects human B cells from programmed cell death. Proc. Natl. Acad. Sci. USA 90, 8479-8483 (1993). | PubMed | ISI | 23. Marshall, W.L., Datta, R., Hanify, K., Teng, E. & Finberg, R.W. U937 cells overexpressing bclxL are resistant to human immunodeficiency virus-1-induced apoptosis and human immunodeficiency virus-1 replication. Virology 256, 1-7 (1999). | Article | PubMed | ISI | 24. Sarid, R., Sato, T., Bohenzky, R.A., Russo, J.J. & Chang, Y. Kaposi's sarcoma-associated herpesvirus encodes a functional bcl-2 homologue. Nature Med. 3, 293-298 (1997). | PubMed | ISI | 25. Cheng, E.H. et al. A Bcl-2 homolog encoded by Kaposi sarcoma-associated virus, human herpesvirus 8, inhibits apoptosis but does not heterodimerize with Bax or Bak. Proc. Natl. Acad. Sci. USA 94, 690-694 (1997). | Article | PubMed | ISI | 26. Gangappa, S., van Dyk, L.F., Jewett, T.J., Speck, S.H. & Virgin, H.W. Identification of the in vivo role of a viral bcl-2. J. Exp. Med. 195, 931-940 (2002). | Article | PubMed | ISI | 27. Goldmacher, V.S. et al. A cytomegalovirus-encoded mitochondria-localized inhibitor of apoptosis structurally unrelated to Bcl-2. Proc. Natl. Acad. Sci. USA 96, 12536-12541 (1999). | Article | PubMed | ISI | 28. Tsukahara, T. et al. Induction of Bcl-xL expression by human T-cell leukemia virus type 1 Tax through NF- B in apoptosis-resistant T-cell transfectants with Tax. J. Virol. 73, 7981-7987 (1999). | PubMed | ISI | 29. Wolf, D. et al. HIV-1 Nef associated PAK and PI3-kinases stimulate Akt-independent Badphosphorylation to induce anti-apoptotic signals. Nature Med. 7, 1217-1224 (2001). | Article | PubMed | ISI | 30. Munger, J. & Roizman, B. The US3 protein kinase of herpes simplex virus 1 mediates the posttranslational modification of BAD and prevents BAD-induced programmed cell death in the absence of other viral proteins. Proc. Natl. Acad. Sci. USA 98, 10410-10415 (2001). | Article | PubMed | ISI | 31. Earnshaw, W.C., Martins, L.M. & Kaufmann, S.H. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu. Rev. Biochem. 68, 383-424 (1999). | Article | PubMed | ISI | 32. Huang, H. et al. The inhibitor of apoptosis, cIAP2, functions as a ubiquitin-protein ligase and promotes in vitro monoubiquitination of caspases 3 and 7. J. Biol. Chem. 275, 26661-26664 (2000). | PubMed | ISI | 33. Suzuki, Y., Nakabayashi, Y. & Takahashi, R. Ubiquitin-protein ligase activity of X-linked inhibitor of apoptosis protein promotes proteasomal degradation of caspase-3 and enhances its anti-apoptotic effect in Fas-induced cell death. Proc. Natl. Acad. Sci. USA 98, 8662-8667 (2001). | Article | PubMed | ISI | 34. Deveraux, Q.L. & Reed, J.C. IAP family proteins-suppressors of apoptosis. Genes Dev. 13, 239-252 (1999). | PubMed | ISI | 35. Hay, B.A. Understanding IAP function and regulation: a view from Drosophila. Cell Death Differ. 7, 1045-1056 (2000). | Article | PubMed | ISI | 36. Clem, R.J. Baculoviruses and apoptosis: the good, the bad, and the ugly. Cell Death Differ. 8, 137-143 (2001). | Article | PubMed | ISI | 37. Shi, Y. Mechanisms of caspase activation and inhibition during apoptosis. Mol. Cell 9, 459-470 (2002). | PubMed | ISI | 38. Neilan, J.G. et al. An African swine fever virus gene with similarity to the proto-oncogene bcl-2 and the Epstein-Barr virus gene BHRF1. J. Virol. 67, 4391-4394 (1993). | PubMed | ISI | 39. Miura, M., Friedlander, R.M. & Yuan, J. Tumor necrosis factor-induced apoptosis is mediated by a CrmA-sensitive cell death pathway. Proc. Natl. Acad. Sci. USA 92, 8318-8322 (1995). | PubMed | ISI | 40. Talley, A.K. et al. Tumor necrosis factor -induced apoptosis in human neuronal cells: protection by the antioxidant N-acetylcysteine and the genes bcl-2 and crmA. Mol. Cell. Biol. 15, 2359-2366 (1995). | PubMed | ISI | 41. Tewari, M. & Dixit, V.M. Fas- and tumor necrosis factor-induced apoptosis is inhibited by the poxvirus crmA gene product. J. Biol. Chem. 270, 3255-3260 (1995). | Article | PubMed | ISI | 42. Wallach, D. et al. Tumor necrosis factor receptor and Fas signaling mechanisms. Annu. Rev. Immunol. 17, 331-367 (1999). | Article | PubMed | ISI | 43. Smith, C.A. et al. A receptor for tumor necrosis factor defines an unusual family of cellular and viral proteins. Science 248, 1019-1024 (1990). | PubMed | ISI | 44. Reading, P.C., Khanna, A. & Smith, G.L. Vaccinia virus CrmE encodes a soluble and cell surface tumor necrosis factor receptor that contributes to virus virulence. Virology 292, 285-298 (2002). | Article | PubMed | ISI | 45. Upton, C., Macen, J., Schreiber, M. & McFadden, G. Myxoma virus expresses a secreted protein with homology to the tumor necrosis factor receptor gene family that contributes to viral virulence. Virology 184, 370-382 (1991). | PubMed | ISI | 46. Schreiber, M., Sedger, L. & McFadden, G. Distinct domains of M-T2, the myxoma virus tumor necrosis factor (TNF) receptor homolog, mediate extracellular TNF binding and intracellular apoptosis inhibition. J. Virol. 71, 2171-2181 (1997). | PubMed | ISI | 47. Brojatsch, J., Naughton, J., Rolls, M.M., Zingler, K. & Young, J.A. CAR1, a TNFR-related protein, is a cellular receptor for cytopathic avian leukosis-sarcoma viruses and mediates apoptosis. Cell 87, 845-855 (1996). | PubMed | ISI | 48. Benedict, C. et al. Cutting edge: A novel viral TNF receptor superfamily member in virulent strains of human cytomegalovirus. J. Immunol. 126, 6967-6970 (1999). 49. Raftery, M.J. et al. Herpes simplex virus type 1 infection of activated cytotoxic T cells: Induction of fratricide as a mechanism of viral immune evasion. J. Exp. Med. 190, 1103-1104 (1999). | Article | PubMed | ISI | 50. Raftery, M.J. et al. Targeting the function of mature dendritic cells by human cytomegalovirus: a multilayered viral defense strategy. Immunity 15, 997-1009 (2001). | PubMed | ISI | 51. Uchida, J. et al. Mimicry of CD40 signals by Epstein-Barr virus LMP1 in B lymphocyte responses. Science 286, 300-303 (1999). | Article | PubMed | ISI | 52. Shisler, J., Yang, C., Walter, B., Ware, C. & Gooding, L. The adenovirus E3-10. 4K/14. 5K complex mediates loss of cell surface fas (CD95) and resistance to fas-induced apoptosis. J. Virol. 71, 8299-8306 (1997). | PubMed | ISI | 53. Tollefson, A.E. et al. Forced degradation of Fas inhibits apoptosis in adenovirus-infected cells. Nature 392, 726-730 (1998). | Article | PubMed | ISI | 54. Stewart, A.R., Tollefson, A.E., Krajcsi, P., Yei, S.P. & Wold, W.S. The adenovirus E3 10.4K and 14.5K proteins, which function to prevent cytolysis by tumor necrosis factor and to downregulate the epidermal growth factor receptor, are localized in the plasma membrane. J. Virol. 69, 172-181 (1995). | PubMed | ISI | 55. Benedict, C.A. et al. Three adenovirus E3 proteins cooperate to evade apoptosis by tumor necrosis factor-related apoptosis-inducing ligand receptor-1 and -2. J. Biol. Chem. 276, 3270-3278 (2001). | Article | PubMed | ISI | 56. Piguet, V., Schwartz, O., Le Gall, S. & Trono, D. The downregulation of CD4 and MHC-I by primate lentiviruses: a paradigm for the modulation of cell surface receptors. Immunol. Rev. 168, 51-63 (1999). | PubMed | ISI | 57. Lama, J. & Ware, C.F. Human immunodeficiency virus type 1 Nef mediates sustained membrane expression of tumor necrosis factor and the related cytokine LIGHT on activated T cells. J. Virol. 74, 9396-9402 (2000). | Article | PubMed | ISI | 58. Overbaugh, J. & Bangham, C.R.M. Selection forces and constraints on retroviral sequence variation. Science 292, 1106-1109 (2001). | Article | PubMed | ISI | 59. Ashkenazi, A. & Dixit, V.M. Apoptosis control by death and decoy receptors. Curr. Opin. Cell. Biol. 11, 255-260 (1999). | Article | PubMed | ISI | 60. Sedger, L.M. et al. IFN- mediates a novel antiviral activity through dynamic modulation of TRAIL and TRAIL receptor expression. J. Immunol. 163, 920-926 (1999). | PubMed | ISI | 61. Vidalain, P.O. et al. Measles virus induces functional TRAIL production by human dendritic cells. J. Virol. 74, 556-559 (2000). | PubMed | ISI | 62. Kayagaki, N. et al. Expression and function of TNF-related apoptosis-inducing ligand on murine activated NK cells. J. Immunol. 163, 1906-1913 (1999). | PubMed | ISI | 63. Fanger, N.A., Maliszewski, C.R., Schooley, K. & Griffith, T.S. Human dendritic cells mediate cellular apoptosis via tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). J. Exp. Med. 190, 1155-1164 (1999). | Article | PubMed | ISI | 64. Bertin, J. et al. Death effector domain-containing herpesvirus and poxvirus proteins inhibit both Fas- and TNFR1-induced apoptosis. Proc. Natl. Acad. Sci. USA 94, 1172-1176 (1997). | Article | PubMed | ISI | 65. Thome, M. & Tschopp, J. Regulation of lymphocyte proliferation and death by FLIP. Nature Rev. Immunol. 1, 50-58 (2001). | Article | PubMed | 66. Krueger, A., Schmitz, I., Baumann, S., Krammer, P.H. & Kirchhoff, S. Cellular FLICE-inhibitory protein splice variants inhibit different steps of caspase-8 activation at the CD95 death-inducing signaling complex. J. Biol. Chem. 276, 20633-20640 (2001). | Article | PubMed | ISI | 67. Skaletskaya, A. et al. A cytomegalovirus-encoded inhibitor of apoptosis that suppresses caspase-8 activation. Proc. Natl. Acad. Sci. USA 7829-7834 (2001). | Article | 68. Chaudhary, P.M., Jasmin, A., Eby, M.T. & Hood, L. Modulation of the NF- B pathway by virally encoded death effector domains-containing proteins. Oncogene 18, 5738-5746 (1999). | Article | PubMed | ISI | 69. Guidotti, L.G. & Chisari, F.V. Noncytolytic control of viral infections by the innate and adaptive immune response. Annu. Rev. Immunol. 19, 65-91 (2001). | Article | PubMed | ISI | 70. Darnell, J.E. Jr. Studies of IFN-induced transcriptional activation uncover the Jak-Stat pathway. J. Interferon Cytokine Res. 18, 549-554 (1998). | PubMed | ISI | 71. Tan, S.L. & Katze, M.G. The emerging role of the interferon-induced PKR protein kinase as an apoptotic effector: a new face of death? J. Interferon Cytokine Res. 19, 543-554 (1999). | Article | PubMed | ISI | 72. Castelli, J.C. et al. A study of the interferon antiviral mechanism: apoptosis activation by the 25A system. J. Exp. Med. 186, 967-972 (1997). | Article | PubMed | ISI | 73. Meurs, E. et al. Molecular cloning and characterization of the human double-stranded RNAactivated protein kinase induced by interferon. Cell 62, 379-390 (1990). | PubMed | ISI | 74. Chu, W.M. et al. JNK2 and IKK are required for activating the innate response to viral infection. Immunity 11, 721-731 (1999). | PubMed | ISI | 75. Jagus, R., Joshi, B. & Barber, G.N. PKR, apoptosis and cancer. Int. J. Biochem. Cell. Biol. 31, 123-138 (1999). | Article | PubMed | ISI | 76. Barber, G.N. Host defense, viruses and apoptosis. Cell Death Differ. 8, 113-126 (2001). | Article | PubMed | ISI | 77. Browne, E.P., Wing, B., Coleman, D. & Shenk, T. Altered cellular mRNA levels in human cytomegalovirus-infected fibroblasts: viral block to the accumulation of antiviral mRNAs. J. Virol. 75, 12319-12330 (2001). | Article | PubMed | ISI | 78. Preston, C.M., Harman, A.N. & Nicholl, M.J. Activation of interferon response factor-3 in human cells infected with herpes simplex virus type 1 or human cytomegalovirus. J. Virol. 75, 8909-8916 (2001). | Article | PubMed | ISI | 79. Benedict, C.A. et al. Lymphotoxins and cytomegalovirus cooperatively induce interferon- , establishing host-virus détente. Immunity 15, 617-626 (2001). | PubMed | ISI | 80. Tortorella, D., Gewurz, B.E., Furman, M.H., Schust, D.J. & Ploegh, H.L. Viral subversion of the immune system. Annu. Rev. Immunol. 18, 861-926 (2000). | Article | PubMed | ISI | Figure 1. Molecular pathways of apoptosis. The intrinsic apoptotic pathway is initiated by internal sensors that monitor cellular stresses such as viral infection via activation of BH3 domain–only members of the Bcl-2 family. Activated BH3-only proteins are thought to mediate the assembly of pro-apoptotic members of the Bcl2 family (Bax, Bak, Bcl-rambo, Bok) into hetero-oligomeric "pores" in the outer membrane of the mitochondria; this results in the release of factors such as cytochrome c, Smac (also known as Diablo) and Omi (also known as HtrA2) into the cytoplasm. The loss of mitochondrial membrane integrity can be blocked by the anti-apoptotic Bcl-2 family members Bcl-2, Bcl-xL, Bcl-w, Mcl-1 and Bcl-B. Release of cytochrome c promotes formation of the apoptosome, which contains Apaf-1 and pro-caspase-9. Autocatalytic activation of caspase-9 initiates the effector caspase cascade, which includes caspase-2, -3, -6 and/or -7. Caspase activation is negatively regulated by the IAPs, which are counter-balanced by the release of proapoptotic Smac and Omi from the mitochondria. The extrinsic pathway of apoptosis is triggered by TNF family death ligands binding to their cognate death receptors. Via their DDs, multimerized receptors interact with the DDs of adaptor proteins such as FADD. These adaptor proteins also contain DEDs that facilitate their binding to pro-caspase-8 and/or pro-caspase-10 to form the DISC. As part of the DISC, the pro-caspases are cleaved into their active forms and initiate the intrinsic pathway of apoptosis by cleaving Bid into t-Bid and activate the effector caspase cascade (caspase-3 is shown). Figure 2. Viral regulation of the intrinsic apoptotic pathway. Cellular sensors, such as p53, that detect stress and initiate the apoptotic process are inactivated by the proteins adenovirus E1B-55K and human papillomavirus E6. Several viruses encode orthologs of Bcl-2 family proteins that antagonize their pro-apoptotic activity. Several viral strategies target the caspases. The vIAPs contain conserved BIR domains and their expression may shift the equilibrium of Smac to favor cell survival. Non-BIR–containing proteins, such as p35 and the serpins (including CrmA), also inhibit caspase activation. Figure 3. Viral regulation of the extrinsic apoptotic pathway. Poxviruses block signaling via TNF and lymphotoxin by producing soluble decoy receptors (for example, myxoma virus T2 protein). vFLIPs contain two DEDs that interact with the homologous DED of FADD and pro-caspase-8 or pro-caspase-10, blocking caspase activation and inhibiting death receptor–induced apoptosis. The human CMV protein vICA binds and inhibits pro-caspase-8 activation, but lacks a DED. The EBV protein LMP1 self-aggregates and engages TRAF and TRADD molecules, activating "anti-apoptotic" NF- B– and Jnk-dependent pathways. A heterocomplex of proteins encoded by the E3 region of adenovirus (E3-10.4K, E314.5K and E3-6.7K) facilitates the removal of the death receptors Fas, TRAILR1 and TRAILR2 from the surface of infected cells. Consequently, receptors accumulate in late endocytic compartments, and cells are desensitized to killing. Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 1019 - 1025 (2002) doi:10.1038/ni1102-1019 © Nature America, Inc. <> Viral interference with antigen presentation Jonathan W. Yewdell1 & Ann B. Hill2 1 2 Laboratory of Viral Diseases, National Institute of Allergy and Infectious Diseases, Bethesda, MD, USA. Department of Molecular Microbiology and Immunology, Oregon Health and Science University, Portland, OR, USA. Correspondence should be addressed to J W Yewdell jyewdell@nih.gov CD8+ T cells play an important role in immunity to viruses. Just how important these cells are is demonstrated by the evolution of viral strategies for blocking the generation or display of peptide–major histocompatibility complex class I complexes on the surfaces of virusinfected cells. Here, we focus on viral interference with antigen presentation; in particular we consider the importance (and difficulty) of establishing the evolutionary significance (that is, the ability to enhance viral transmission) of viral gene products that interfere with antigen presentation in vitro. Viruses are the ultimate obligate intracellular parasites. Lacking virtually all the machinery necessary for their own replication, they consist of a fragment of nucleic acid (encoding anywhere from less than ten to several hundred proteins) enclosed in a protein or proteolipid shell with just enough of the right polymerases to initiate gene expression ("bad news wrapped in protein", according to Peter Medawar). For viruses to survive in nature, they must devise a means to be transmitted between hosts. This, not dissemination within a host, is their sole evolutionary selection factor, although dissemination is usually a prerequisite for transmission. At a bare minimum, viruses must encode information to enable regeneration of their structural proteins. Because hosts are generally hostile to sharing their resources with viruses, viruses need to be a bit cleverer than this. Virus versus host: role of CD8+ T cells Through various mechanisms, host cells may sense the presence of a virus and attempt to trigger apoptosis to preclude viral replication. Viruses can counter these attempts (see Review by Ware and colleagues in this issue). The presence of unusual nucleic acids produced in the early stages of viral replication (for example, double-stranded RNA) triggers protein kinase R (PKR), which induces an antiviral state in the cells and synthesis and release of interferons (IFNs). IFNs signal the presence of virus to the cellular immune system, whose initial response consists of natural killer (NK) cells. Some viruses have devised means for evading NK cell recognition (see Review by Strominger and colleagues in this issue). The second wave of the cellular immune counterattack consists of CD8 + T cells, which are activated via the presentation of viral antigens by professional antigen-presenting cells (APCs). The contribution of CD8+ T cells to antiviral immunity has been extensively demonstrated in mouse model systems. Starting with experiments in the late 1960s, T cells (which were later shown to be CD8+) were demonstrated to be responsible for the recovery of mice from acute mousepox (ectromelia) infection1-3. Subsequently, we have learned that CD8+ T cells are important in mouse immunity to many viruses. CD8+ T cells exert antiviral effects via the localized secretion of molecules in close vicinity to the virus-infected APC (professional or not). Many CD8+ T cells kill APCs by releasing perforin and granzymes (if APCs express Fas, engagement by Fas ligand on CD8+ T cells can also induce lysis). In addition, nearly all CD8+ T cells secrete IFN- and tumor necrosis factor- (TNF- ) (a small percentage of cells secrete IFN- only), which induces a potent antiviral state in cells. It is important to emphasize that CD8+ T cells are but one of many weapons deployed by the immune system to combat viruses. NK cells, CD4+ T cells and antibody all participate in antiviral immunity. Mice deficient in CD8+ T cell responses effectively handle infections with many viruses, although they do succumb to others, such as ectromelia. Direct demonstration of the role of CD8+ T cells in human antiviral immunity is more difficult to establish. On the one hand, adoptively transferred virus-specific CD8+ T cells are effective against human cytomegalovirus (HCMV) 4, HIV15 and Epstein-Barr virus (EBV)6. On the other hand, T cell–deficient individuals seem to do quite well with common viral infections. There is a single hereditary condition that selectively interferes with CD8+ T cell induction: absence of functional transporter associated with antigen processing (TAP) (described below). Curiously, afflicted individuals suffer from bacterial, not viral, infections 7. These patients show only a partial reduction in CD8+ T cells, however, and the residual CD8+ T cell activity may be sufficient to handle common viral infections. Evolutionary selection for individual elements of the immune system may be punctuated: for example, the sporadic appearance of a potentially lethal pathogen would be sufficient to maintain an immune effector mechanism that reduces its lethality. It is plausible, for example, that variola virus (the agent of smallpox) would display a greatly increased mortality rate in TAP-deficient individuals. Perhaps the best evidence for the importance of CD8+ T cells in human immunity to viruses is the lengths that some human viruses have gone to interfere with antigen presentation, which is the principal focus of our review. Antigen-presentation primer To enhance nonspecialists' comprehension of the area of antigen processing and presentation we shall briefly discuss how viral proteins are processed for recognition by virus-specific CD8+ T cells8, 9. The specificity of CD8+ T cells is conferred by the clonally restricted T cell receptor (TCR) that interacts with residues from both major histocompatibility complex (MHC) class I molecules and an oligopeptide (>90% are 8–11 amino acids in length) encoded by a viral gene. Peptides are predominantly generated from the byproducts of proteasomal degradation (Fig. 1). Most of the substrates consist of defective ribosomal products (DRiPs) that are degraded within 30 min of their synthesis10. This process enables the rapid recognition of viral proteins early in infections, when viral proteins represent a tiny fraction of total cell proteins. Peptides are transported into the endoplasmic reticulum (ER) by the TAP protein. Here, MHC class I molecules are folded through the concerted actions of general purpose molecular chaperones working with a dedicated chaperone (tapasin) that tethers MHC class I to TAP. Upon peptide binding, MHC class I molecules dissociate from TAP, leave the ER and make their way to the plasma membrane via the Golgi complex. High resolution image and legend (78K) Figure 1. The classical MHC class I pathway is depicted with reference to viral interfering proteins. Oligopeptides (red circle) are derived from DRiPs through the action of proteasomes. Nascent MHC class I molecules—consisting of a heavy chain and 2-microglobulin ( 2M)—bind to TAP via tapasin. Peptide binding releases MHC class I to the cell surface. VIPRs interfere with this process at multiple steps. (1) EBV EBNA-1 contains a sequence that renders it resistant to proteasomal degradation. HCMV IE is phosphorylated by another viral protein, preventing generation of the immunodominant peptide epitope. (2A) HSV ICP47 and a BHV1 protein bind to the cytosolic side of TAP and prevent peptide translocation. (2B) HCMV US6 binds to TAP in the ER lumen and prevents peptide translocation. (2C) Several VIPRs bind to MHC class I in the ER, retaining it and/or interfering with the function of the peptide-loading complex. The proteins include AdE319K (retains MHC class I and also prevents tapasin-mediated docking with TAP); HCMV US3 (binds MHC class I but dissociates, the mechanism of MHC class I retention is not clear but may involve repeated rebinding by newly synthesized US3). MCMV m4/gp34 forms extensive complexes with MHC class I in the ER. (3) HCMV US2 and US11 and MHV-68 K3 bind MHC class I in the ER and induce retrotranslocation for degradation by the proteasome. For US11, ubiquitylation of heavy chain cytoplasmic tail, possibly after retrograde translocation, has been described. MHV-68 K3 ubiquitylates the tail before translocation. HIV-1 Vpu also induces degradation of newly synthesized MHC class I—probably via retrograde translocation and proteasome degradation—although, unlike US2 and US11, proteasome activity is required for retrograde translocation. (4) MCMV m152/gp40 causes MHC class I to be retained in the ER cis-Golgi complex intermediate compartment (ERGIC). The fact that m152, itself a distant MHC class I homolog, can reduce cell surface expression of H60 as well as classical MHC class I (in an allomorph-specific manner) makes its poorly understood mechanism of action of particular interest. Only the lumenal domain of m152/gp40 is required for the retention of MHC class I. Binding of m152/gp40 to MHC class I has not been demonstrated, and m152/gp40 itself has a default pathway of export to and degradation in the lysosome. (5) Several VIPRs remove MHC class I either from the Golgi or the cell surface. MCMV m6/gp48 contains a lysosomal targeting di-leucine motif in its tail; it binds directly to MHC class I and redirects it to the lysosome. HIV and SIV Nef use the PACS-1 (phosphofurin acidic cluster sorting protein 1)–sorting protein to remove MHC class I from the cell surface and sequester it in the trans-Golgi network. KSHV K3 and K5 ubiquitylate the MHC class I tail and target MHC class I to lysosomes in a TG101dependent manner. K5 also targets ICAM-1 (intercellular adhesion molecule 1) and B7-2. (6) MCMV m4/gp34 is found associated with MHC class I at the cell surface and inhibits CTL recognition, but cause and effect have not been firmly linked. As peptide-MHC (pMHC) class I complexes accumulate at the cell surface, they have a greater chance of triggering activation by CD8+ T cells with a cognate receptor. The half-life of cell surface complexes depends on the koff of the bound peptide; immunogenic peptides generally dissociate with half-lives in the range of hours to hundreds of hours. There is no simple relationship between complex stability or abundance and the magnitude of the cognate CD8 + T cell response11. It is important to distinguish naïve CD8+ T cells from armed effector CD8+ T cells. Naïve CD8+ T cells can only be activated by cells expressing the proper costimulatory molecules, that is, by professional APCs. Dendritic cells (DCs) are believed to be the principal APCs for activating naïve CD8+ T cells, although the evidence is largely circumstantial. Effector CD8 + T cells have no such limitation and require only recognition of a cognate complex for activation, although expression of adhesion molecules can decrease the number of complexes required. Effector CD8 + T cells circulate "ready to kill" with preformed perforin and granzymes, but only express IFN- and TNFupon activation by interaction with a virus-infected APC. Viral strategies for circumventing CD8+ T cells In principal, viruses can thwart CD8+ T cell function by interfering with the activation of naïve cells, CD8+ T cells trafficking to infected cells, antigen presentation to effector CD8+ T cells or CD8+ T cell–mediated effects on virus-infected cells. For viruses that are capable of infecting CD8+ T cells, destroying responding CD8+ T cells would seem to be a simple strategy. This mechanism has been described in vitro (but not demonstrated in vivo) for herpes simplex virus (HSV)12. Viruses with this ability are highly unusual, which is interesting, inasmuch as it suggests that hosts are capable of modulating the antiviral state in a cell type–specific manner, with CD8+ T cells being particularly difficult to infect. Blocking activation of naïve CD8+ T cells can be achieved by preventing APC antigen presentation. Many viruses are capable of infecting DCs, and there have been a number of reports that viruses interfere with DC-mediated CD8+ T cells activation in vitro13-16. This is an area, however, with a potentially enormous gap between in vitro phenomenology and in vivo reality. There is no certain relationship between DCs propagated ex vivo and immature DCs residing in the tissue or activated DCs that have migrated to draining lymph nodes once activated by viral infection. Viral infections generally result in a gross alteration of host cell functions over time. The conditions of infection in vivo and in vitro can differ in terms of the number of viral particles that initiate the infection. Even if a virus-induced decline in antigen presentation measured in vitro accurately reflects the in vivo situation, there could be sufficient residual presentation to enable CD8 + T cell activation in vivo. For example, whereas the antigen-presenting function of DCs in vitro is adversely affected by infection with vaccinia virus (VV)15, VV-infected DCs can be visualized presenting antigens to CD8+ T cells in lymph nodes; this provides the most direct evidence, to date, that DCs function as APCs in viral infections17. Numerous poxviruses and herpesviruses encode molecules with known or likely effects on chemokine function. As chemokines direct trafficking of CD8+ T cells to the sites of inflammation, these could influence the effectiveness of CD8+ T cells. A secreted chemokine ligand homolog, murine herpes virus 68 (MHV-68) M3, has been implicated as affecting CD8+ T cell function18. Many viruses induce an anti-apoptotic state that should hinder the cytolytic activity of CD8+ T cells. Resistance to the effects of IFN- and TNF- is also a common viral strategy19, 20. Viruses block the presentation of endogenous antigens by APCs by expressing VIPRs (viral proteins interfering with antigen presentation, pronounced "viper", with an etymological nod to herpesviruses). Numerous VIPRs have been described, and they target virtually all steps in the antigen-processing and -presentation pathway (Fig. 1). This appears to be the principal means by which viruses interfere with CD8+ T cell function, and it is certainly the best characterized. For these reasons we will focus on VIPRs, but not their cell biology and mechanisms of function, which have been the subject of detailed reviews19-21. Instead, we will deal primarily with the in vivo function of VIPRs, a topic ripe for review. Cross-priming: a hurdle to VIPR function Viruses that block CD8+ T cell activation by expressing VIPRs in APCs face a potentially insurmountable hurdle in cross-priming22. In this process, DCs (and macrophages) internalize antigen from infected cells and reprocess it in the cytosol or endosomal compartment for presentation on their own MHC class I molecules. The nature of the cross-priming material is unknown at present, but it is likely to represent a mixture of phagocytosed intact dying cells and cell debris containing intact and partially degraded proteins, some complexed with sundry molecular chaperones. Because, by definition, cross-priming APCs are not infected, viruses can only impose global restrictions on antigen presentation by releasing soluble inhibitory factors from infected cells. In lieu of this, the best they can do is to evolve proteins that cannot be processed by cross-priming, but even this appears to be rather difficult. EBV nuclear antigen 1 (EBNA-1) is not processed and presented by infected cells due to its indigestibility by proteasomes23, yet EBNA-1–specific CD8+ T cells appear to be generated in vivo by cross-priming24. Cross-priming is a robust process that may have evolved in response to viral interference with CD8 + T cell activation. There is now solid evidence for cross-priming in a number of viral systems25-28. Ultimately, the most compelling evidence for the effectiveness of cross-priming may be the vigorous responses to viruses that are loaded with genes encoding proteins capable of interfering with the generation of pMHC class I complexes. A murine CMV (MCMV) determinant whose presentation was completely abrogated by m152 in vitro was equally immunodominant in mice infected with wildtype virus or m152-deficient viruses29. This strongly suggests that cross-priming contributes to the generation of CD8+ T cells specific for this determinant. Similarly, the enigma that CD8+ T cells specific for immediate early protein (IE) are readily generated in HCMV-infected individuals when antigen presentation of IE is efficiently prevented in HCMV-infected cells30 is probably explained by cross-priming. Cross-priming can occur by either the classical cytosolic processing route or via the alternative endosomal pathway whose details are just beginning to appear from the mists 31. An important question regarding the endosomal route is the extent to which the peptides it generates overlap with those generated by the cytosolic route, particularly because distinct proteases are involved. Herpesviruses: a VIPR catalog Although known VIPRs are encoded by retroviruses and adenoviruses, herpesviruses are the clear champions in the evolution of VIPRs. Herpesviruses encode proteins that interfere with virtually every step of antigen processing and presentation (Fig. 1); indeed, individual family members encode multiple VIPRs (HCMV encodes at least four). It seems worthwhile to pause to consider the characteristics of this family of viruses that are so hell-bent on interfering with CD8+ T cell function. Herpesviruses are large double-stranded DNA viruses that replicate their genomes with high fidelity compared to RNA viruses (for example, HIV or influenza virus). Herpesviruses fall into three subfamilies. -herpesviruses are neurotropic: HSV and varicella zoster (VZV, the cause of chickenpox and shingles) are the prototypes. -herpesviruses are ubiquitous, highly speciesspecific and cause minimal or no disease in immunocompetent hosts. They include CMVs and human herpesvirus 6 (HHV6) and HHV7. -herpesviruses fall into two subgroups: the 1 prototype is EBV (the cause of infectious mononucleosis, Burkitt's lymphoma and nasopharyngeal carcinoma) and the 2 viruses include Kaposi's sarcoma herpesvirus (KSHV) and the important model virus MHV-68. Unique among the herpesviruses, -herpesviruses encode genes that are only expressed during latency. They also encode oncogenes and are associated with malignancy. Herpesviruses are the most ancient known mammalian viruses and are extremely successful in evolutionary terms. Many family members infect a high percentage of individuals of their host species for the lifetime of the individual. Although the cell types they infect are diverse, herpesviruses share the ability to establish latent infection. True viral latency means that the virus can exist in host cells without reproducing itself, in contrast to persistence, when basal viral replication continues (the distinction is imperfect). Herpesviruses reactivate and replicate in a fully immune host, which enables their transmission to children, the next generation of hosts. Herpesviruses live on the edge: they depend on immune control for their host's (and hence their own) survival, yet must impair immunity to avoid eradication. Severely immunocompromised patients often succumb to reactivated herpesvirus infections (especially CMV). The stability of the host-virus relationship shows that this is a robust and well-buffered equilibrium, only rarely does the host or the virus gain the advantage. This equilibrium is generally maintained only when herpesviruses infect their natural host. For example, the monkey virus herpes B virus causes an unapparent infection in monkeys but kills 70% of the people who are infected. Dissecting the complex relationship between herpesviruses and the immune system depends on mouse models, where it is possible to genetically alter both virus and host. The best characterized mouse herpesviruses are MCMV, a pathogen that is ubiquitous in wild mice, and MHV-68, originally isolated from voles but nevertheless able to infect and establish latent infection in laboratory mice. Immune control of MCMV is complex. Both type I IFN and IFN- and NK cells play major roles in containing primary infection32, 33, yet acute infection is invariably fatal in the absence of B and T cell responses. Antibodies, NK cells, CD8+ T cells and CD4+ T cells all contribute to preventing dissemination of reactivated latent virus. Reactivation in antibody-deficient mice requires depleting any two of these three effector cell populations34. For the 2 herpesvirus MHV-68 the situation is complicated further by a biphasic acute infection and limited understanding of the genetic program of latent infection. Again, all components of the immune response are involved in host defense 35. After intranasal infection, MHV-68 replicates acutely in the lungs and disseminates to the spleen. This first phase of infection is controlled primarily by type I IFN, with some help from CD4 + and CD8+ T cells. A second (mononucleosis-like) phase of the infection ensues, characterized by a massive CD4+ T cell–dependent splenic B cell expansion and increase in viral load. Various viral transcripts can be detected, but infectious virus cannot be recovered, so virus at this stage is considered latent. This phase is gradually controlled by antibodies and both T cell subsets. Finally, a poorly characterized true latency is established, during which depleting both T cell subsets does not lead to virus reactivation. Acute viruses: VIPR-free? Viruses that are transmitted via acute infections are not known to encode VIPRs. This cannot be easily dismissed as evolutionary incompetence. HSV-encoded ICP47 is the "poster" protein for how easy blocking presentation can be. This highly effective VIPR comprises only 87 amino acids (even a 32-residue fragment is highly active). It would be trivial for even small viruses with severe nucleic acid packaging constraints to encode something similar (this could easily be accommodated in an overlapping reading frame, requiring no increase in genome size). This suggests that blocking antigen presentation to CD8+ T cells is useful only under a highly restricted set of circumstances. Perhaps blocking MHC class I expression greatly sensitizes cells for NK recognition, and the costs of countering both CD8+ T cells and NK cells are just too steep for acute viruses. Or perhaps very acute viruses are transmitted too rapidly for CD8+ T cells to have an impact on their evolution. MHC class II VIPRs Interference with the MHC class II antigen-processing pathway has been described for several herpesviruses36-38. As with cross-priming of CD8+ T cells, MHC class II VIPRs are not expected to block the presentation of exogenous antigens to CD4+ T cells, as—by definition—the APC is not infected and will not therefore express the VIPR. Thus, as with MHC class I VIPRs, class II VIPRs would have to act in vivo by interfering with CD4+ T cell–mediated clearance of virus-infected cells. For viruses whose transmission entails infection of MHC class II–expressing cells, this is certainly possible, but the physiological relevance of MHC class II VIPRs remains to be established. Establishing VIPR function Physiological relevance is, of course, the ultimate measure of VIPR function. Demonstrating that a viral protein can interfere with some aspect of antigen presentation or CD8+ T cell activation in vitro is not synonymous with equivalent in vivo function. There are examples of individual VIPRs that interfere with the function of numerous host proteins in cultured cells, and it is possible (even likely) that some of the interactions are not important (admittedly, disproving something is difficult). The degree of skepticism regarding the relevance of in vitro findings should reflect the deviation from natural circumstances, for example, when transfection is used to achieve expression that vastly exceeds that seen in infected cells39. Such approaches reveal potential functions, but eventually must be reinforced by evidence obtained from animal experiments. We do not intend to demean the value of in vitro studies. Some VIPRs act with such efficiency in cultured cells (interference with TAP function by HSV ICP47 protein is the best example), as to make it exceedingly unlikely that the interaction is insignificant. Establishing VIPR function in vivo is never easy. For human viruses, often the best that can be achieved is demonstrating interference with CTL recognition of virus-infected cells in vitro. This has been shown for some VIPRs, notably HIV Nef40 and HCMV pp6530. With few exceptions, human viruses do not naturally infect other species, so it is simply not possible to rigorously study their VIPRs under natural circumstances. VIPRs can demonstrate similar interactions with homologous targets from different species: for example, AdE319K and HCMV US2 and US11 down-regulate mouse MHC class I, but HSV ICP47 does not. When a VIPR does function in mice, it may be possible to establish an in vivo function for a given gene product from a human virus, but again caution is necessary. No matter how the VIPR is expressed, the conditions of expression will differ from the natural situation in a manner that can profoundly affect its function. Variables include the amount of expression, cell type and a lack of other appropriate viral gene products. This basically limits thorough investigation of VIPRs to those expressed by natural animal pathogens. Given the vast superiority of mice as an experimental system, this puts the focus on mouse viruses, although the importance of the AIDS epidemic has made the rhesus macaque model of SIV an important exception. The basic rules for establishing the in vivo function of VIPRs (and other immunomodulatory proteins) were spelled out in a report by Koszinowski and colleagues 41 (extending a previously pioneered approach to demonstrating the function of a viral complement–interfering protein42). First, disabling the gene reduces the fitness of the mutant virus in vivo without affecting its ability to replicate in tissue culture. Ideally, fitness is defined by transmission. This is technically difficult to determine (for some viruses, well nigh impossible), so viral titers are taken as a surrogate. Second, reinserting the gene into the mutant virus (generating a "rescuant") restores reproductive fitness. This step is needed to ensure that other alterations in the mutant virus inadvertently introduced during its generation are not responsible for the phenotype. Small viruses can be completely sequenced, but this remains impractical for herpesviruses and other large viruses. Third, the fitness of the mutant virus is restored by interfering with CD8+ T cell function, either with an appropriate genetically deficient mouse (TAP-deficient, for example) or by treating mice with a CD8+ T cell– depleting antibody. Just as Koch's postulates (KPI) for demonstrating the cause of transmissible diseases cannot constitute proof (in the mathematical sense of the word) that a given agent is responsible for a given disease, fulfilling Koszinowski's postulates (KPII) does not guarantee that a gene product evolved to perform the proposed function. At the very least, the following caveats need to be considered. Even the simple proposition that removing a gene should reduce fitness and reveal its function is problematical. If there is true redundancy in either the viral genes or the host defense mechanisms (or in both) such an effect may be difficult to demonstrate. Multifunctional viral genes (which are common) raise obvious problems. Here the researcher must attempt to make mutants in which the putative immune evasion phenotype is lost while other functions are maintained (this presumes that the other functions are known). Excluding extraneous effects due to inadvertent genetic alterations is usually tidily dealt with by making resucants, but even this can be misinterpreted. If disruption of the targeted gene disrupts expression or control of an adjacent or unappreciated overlapping open-reading frame (ORF) that contributes to the phenotype, restoring the original genetic sequence will also restore the phenotype. Replacing the gene elsewhere in the genome is theoretically a better control, but rarely done. An alternative is to make multiple independent mutants, disrupting the gene in different ways (for example, mutating the promoter or the ORF) and showing that they have the same phenotype. Equalizing the fitness of the mutant and wild-type virus by deleting the putatively targeted host mechanism (so called "genetic complementation") is an intellectually appealing and essential part of KPII. Yet interpreting such experiments can be fraught with hazards. The first comes from making qualitative interpretations of an outcome that may result from an unrelated quantitative advantage. That is, if the balance between virus and host is such that CD8+ T cells achieve good but imperfect control, in theory, any (that is, VIPR-unrelated) reduction in viral fitness could tip the balance and enable CD8+ T cells to achieve complete control. Another problem is that genes may have unappreciated targets whose function contributes to the phenotype examined. The MCMV class I homolog m144 is assumed to act as a ligand for inhibitory NK receptors. Indeed, deleting m144 reduced virus fitness, the rescuant showed wild-type fitness and the mutant and wild-type fitnesses were equalized by NK cell depletion43. However, the appreciation that the ligand for the homologous HCMV class I protein UL18 is in fact a leukocyte Ig-like receptor (LIR) expressed more on macrophages than NK cells suggests other interpretations for the data. For example, m144 may inhibit MCMV-induced macrophage or DC activation, reducing cytokine secretion and hence the NK cell response. This scenario shows that our interpretation of KPII relies on current models through whose smoky glass we interpret the data. Despite these caveats, KPII remains the essential means for addressing the in vivo function of viral gene products. We now have a handful of examples of its application. The first application of KPII to establish the function of a VIPR was the seminal study that showed MCMV lacking m152 replicated less efficiently than wild-type or rescuant virus 7 days after infection, a difference that was abolished by depleting CD8+ T cells41. Although welcome as the first demonstration of VIPR function in vivo, the real surprise of this article was just how modest the effect was (usually less than a tenfold difference). This raises an important question: do VIPRs generally exert such a modest effect? Or is there something that we are missing about m152? Indeed, m152 had an even more profound effect on the activity on NK cells and hence on viral titers at day 3, due to its inhibition of the NK activating ligand H-6044. So the "true" evolutionary function of m152 is difficult to establish. The most important message from m152 may be that evolution favors complexity and that we need to interpret experiments with this in mind. MHV-68 lacking the M3 chemokine-binding protein replicated acutely in lung epithelium and initially seeded the spleen to almost the same extent as wild-type virus. However, in contrast to wild-type virus, the mutant virus did not cause B cell expansion in the spleen with its concomitant amplification of latent viral load unless CD8+ T cells were depleted18. The interpretation favored by the authors—that M3 prevents chemokine-driven CD8+ T cells trafficking and impairs their ability to control latent virus—is reasonable. The difficulty in interpreting this type of experiment (see above) is underscored by the fact that an independent M3 virus carrying a deletion had little, if any, latency deficit45. MHV-68 K3 interferes directly with CD8+ T cell recognition of infected cells46. The K3-deficient mutant, like the M3-deficient mutant, was primarily impaired during the second mononucleosis-like phase of infection, which was attributed to more efficient CD8+ T cell function47. These two mutants suggest CD8+ T cells must be disabled for splenic amplification of the latent virus pool and, therefore, that MHV-68 VIPRs are important for this phase. It is not known how closely infection of mice mimics infection of the natural host (voles); consequently, the real contribution made by VIPRs to the fitness (that is, transmission) of 2 herpesviruses may be difficult to determine in this model. The role of MHC class I down-regulation by Nef has been investigated in the SIV model48. Three rhesus macaques were infected with SIV with a Nef that was modified (Y223F) to disable its VIPR activity without affecting its myriad other functions. Four weeks after infection, the majority of viruses recovered from each animal had reverted to wild-type Nef. Although this suggests that MHC class I down-regulation has a strong influence on viral fitness, further evidence is needed to fulfill KPII. Does reversion occur in CD8+ T cell–depleted animals? Is SIV replication compromised by the introduction of a mutation that is more difficult to revert? These examples represent the vanguard of what we hope will be a serious long-term effort to assess the in vivo function of VIPRs and other immunomodulatory proteins. Although the complexity of the immune system makes such studies difficult, they should provide a bounty of findings that will enliven the enterprise and deepen our insight into the general workings of the immune system. Escape mutants Viruses have another potential means for avoiding recognition by CD8+ T cells. RNA viruses have extremely high mutation rates, which means that they exist in nature as a diverse genetic mix. Of course, most mutations are harmful, and in the absence of selective pressure the average genetic composition of the population tends to remain fairly stable, reflecting the optimal gene function. However, these viruses have the potential to rapidly decrease the immunogenicity of their gene products. This is not terribly difficult due to the number of filters in place that limit the potential immunogenicity of peptides. Consequently, the chance that any given peptide in a protein will be immunogenic with any given MHC class I allomorph is 1/200011. Highly artificial model systems have shown that CD8+ T cells can select for antigenic escape mutants in mice. Clear evidence has been presented for the selection of escape variants in SIV and HIV infections49-51. Individual escape mutants may enable a virus to persist in one host and hence improve transmission to others. MHC polymorphism, however, limits the advantage in a new host, which is likely to possess an MHC allomorph that binds to different peptides. Thus, RNA viruses profit from their ability to rapidly generate new variants per se, rather than generate any given escape variant. Indeed, preventing escape variants is thought to be the primary evolutionary selection factor for MHC polymorphism (ask any viral immunologist). In theory, viruses could avoid MHC polymorphism by creating proteins that are antigenically identical to the host. This would enable viral avoidance of antibody recognition as well. Despite the high hopes of immunologists, if viruses exploit mimicry, they do so subtly and infrequently; viral proteins are usually only distantly related to host proteins, even those with similar functions. Horizons We will end our review by considering what we have learned from our fascination with VIPRs and what might be gleaned from further studies. Following the long tradition of viral gene products, VIPRs have contributed to our understanding of basic cellular biological processes. An early contribution came from the first-defined VIPR, AdE3-19K, which led to the identification of ERretention motifs in the cytosolic domains of membrane proteins 52. More recently, HCMV US2 and US11 greatly facilitated studies in mammalian cells of retro-translocation of ER proteins into the cytosol. Overall, however, our understanding of the classical MHC class I pathway has enabled us to understand the mechanistic actions of VIPRs rather than vice versa. VIPRs may yet teach us something about antigen presentation. Two classes of VIPRs (HCMV US1153 and the K3 from KSHV and MHV-6854) ubiquitylate MHC class I heavy chain cytosolic domains to target it for destruction, either in the ER or a post-medial Golgi complex compartment. The lysine ubiquitylated through the action of K3 is highly conserved among MHC class I allomorphs, which suggests that this process has a normal counterpart, perhaps identifying MHC class I molecules lacking peptides for internalization and degradation. No doubt this will soon be sorted out. VIPRs may also fulfill their promise and become powerful tools for sorting out the contributions of direct and cross-presentation in activating naïve virus-specific CD8+ T cells28. Occurring simultaneously with a great leap in deciphering the molecular basis for NK cell recognition, the study of herpesvirus immune evasion has played an important role in the expansion of our concept of the extended family of MHC class I molecules and their cognate receptors. It is likely that when the dust settles, the very definition of the MHC class I family and its role in immunobiology will have to be reconsidered. The HCMV MHC class I homolog UL18 seemed tailor made as an NK cell decoy, but identification of a cellular ligand led to the discovery of an entire new family of killer cell inhibitory receptor (KIR)-like genes, the LIRs55. The LIRs also recognize classical MHC class I gene products, but are expressed more by macrophages and B cells than by NK cells or T cells. The functions of the HCMV LIR ligand56 and the LIRs expressed by myeloid cells are a complete mystery. Meanwhile, NK cell biologists have extended the concept of "class I–like" by identifying ligands for the activating NK receptor NKG2D. An HCMV gene, UL16, played an important role in this story57. Although homology of some of these ligands to class I molecules is so low as to strain credulity, it seems sufficient for the MCMV VIPR m152, which targets both classical MHC class I and a gene product that hardly looks like one (H-60)44. Extending the boundaries still further, the MHC class I family has recently accepted another cohort of relatives, with the identification of another MCMV gene, m157, as the ligand for the activating NK receptor Ly49H58, 59. m157 has distant secondary structural homology to MHC class I, as do a handful of other MCMV genes, including m15259 (and L. Lanier, personal communication). This story remains to be written, but it is clear that VIPRs are providing crucial insights into both the identities and functions of members the far-flung MHC class I family. With the possible exception of MHV-68 K3, we lack a clear demonstration that VIPRs are necessary for viral persistence via interfering with CD8+ T cell recognition of infected cells. The odds still favor the possibility that this property is a selective factor driving VIPR evolution. However, our broadened awareness of the biology of the extended MHC class I family and its ligands forces us to consider broader hypotheses for the interest of viruses in MHC class I molecules. It would be an ironic twist in the VIPR story if interference with antigen presentation to CD8+ T cells turns out to be only a minor part of their complete job description. After all, snakes enjoy a varied diet. References 1. Blanden, R.V. Mechanisms of recovery from a generalized viral infection: mousepox. I. The effects of anti-thymocyte serum. J. Exp. Med. 132, 1035-1054 (1970). | PubMed | ISI | 2. Blanden, R.V. Mechanisms of recovery from a generalized viral infection: mousepox. II. Passive transfer of recovery mechanisms with immune lymphoid cells. J. Exp. Med. 133, 10741089 (1971). | PubMed | ISI | 3. Blanden, R.V. Mechanisms of recovery from a generalized viral infection: mousepox. 3. Regression infectious foci. J. Exp. Med. 133, 1090-1104 (1971). | PubMed | ISI | 4. Walter, E.A. et al. Reconstitution of cellular immunity against cytomegalovirus in recipients of allogeneic bone marrow by transfer of T-cell clones from the donor. N. Eng. J. Med. 333, 10381044 (1995). | Article | ISI | 5. Brodie, S.J. et al. In vivo migration and function of transferred HIV-1-specific cytotoxic T cells. Nature Med. 5, 34-41 (1999). | Article | PubMed | ISI | 6. Khanna, R. et al. Activation and adoptive transfer of Epstein-Barr virus-specific cytotoxic T cells in solid organ transplant patients with post-transplant lymphoproliferative disease. Proc. Natl. Acad. Sci. USA 96, 10391-10396 (1999). | Article | PubMed | ISI | 7. Gadola, S.D., Moins-Teisserenc, H.T., Trowsdale, J., Gross, W.L. & Cerundolo, V. TAP deficiency syndrome. Clin. Exp. Immunol. 121, 173-178 (2000). | Article | PubMed | ISI | 8. Rock, K.L. & Goldberg, A.L. Degradation of cell proteins and the generation of MHC class Ipresented peptides. Annu. Rev. Immunol. 17, 739-779 (1999). | Article | PubMed | ISI | 9. Pamer, E. & Cresswell, P. Mechanisms of MHC class I-restricted antigen processing. Annu. Rev. Immunol. 16, 323-358 (1998). | Article | PubMed | ISI | 10. Yewdell, J.W., Schubert, U. & Bennink, J.R. At the crossroads of cell biology and immunology: DRiPs and other sources of peptide ligands for MHC class I molecules. J. Cell Sci. 114, 845-851 (2001). | PubMed | ISI | 11. Yewdell, J.W. & Bennink, J.R. Immunodominance in major histocompatibility complex class Irestricted T lymphocyte responses. Annu. Rev. Immunol. 17, 51-88 (1999). | Article | PubMed | ISI | 12. Posavad, C.M., Newton, J.J. & Rosenthal, K.L. Infection and inhibition of human cytotoxic T lymphocytes by herpes simplex virus. J. Virol. 68, 4072-4074 (1994). | PubMed | ISI | 13. Andrews, D.M., Andoniou, C.E., Granucci, F., Ricciardi-Castagnoli, P. & Degli-Esposti, M.A. Infection of dendritic cells by murine cytomegalovirus induces functional paralysis. Nature Immunol. 2, 1077-1084 (2001). | Article | PubMed | ISI | 14. Moutaftsi, M., Mehl, A.M., Borysiewicz, L.K. & Tabi, Z. Human cytomegalovirus inhibits maturation and impairs function of monocyte-derived dendritic cells. Blood 99, 2913-2921 (2002). | Article | PubMed | ISI | 15. Engelmayer, J. et al. Vaccinia virus inhibits the maturation of human dendritic cells: a novel mechanism of immune evasion. J. Immunol. 163, 6762-6768 (1999). | PubMed | ISI | 16. Salio, M., Cella, M., Suter, M. & Lanzavecchia, A. Inhibition of dendritic cell maturation by herpes simplex virus. Eur. J. Immunol. 29, 3245-3253 (1999). | Article | PubMed | ISI | 17. Norbury, C.C., Malide, D., Gibbs, J.S., Bennink, J.R. & Yewdell, J.W. Visualizing priming of virus-specific CD8+ T cells by infected dendritic cells in vivo. Nature Immunol. 3, 265-271 (2002). | Article | PubMed | ISI | 18. Bridgeman, A., Stevenson, P.G., Simas, J.P. & Efstathiou, S. A secreted chemokine binding protein encoded by murine herpesvirus-68 is necessary for the establishment of a normal latent load. J. Exp. Med. 194, 301-312 (2001). | Article | PubMed | ISI | 19. Tortorella, D., Gewurz, B.E., Furman, M.H., Schust, D.J. & Ploegh, H.L. Viral subversion of the immune system. Annu. Rev. Immunol. 18, 861-926 (2000). | Article | PubMed | ISI | 20. Alcami, A. & Koszinowski, U.H. Viral mechanisms of immune evasion. Immunol. Today 21, 447-55 (2000). | Article | PubMed | ISI | 21. Yewdell, J.W. & Bennink, J.R. Mechanisms of viral interference with MHC class I antigen processing and presentation. Annu. Rev. Cell Dev. Bio. 15, 579-606 (1999). | Article | ISI | 22. Bevan, M.J. Cross-priming for a secondary cytotoxic response to minor H antigens with H-2 congenic cells which do not cross-react in the cytotoxic assay. J. Exp. Med. 143, 1283-1288 (1976). | PubMed | ISI | 23. Levitskaya, J. et al. Inhibition of antigen processing by the internal repeat region of the Epstein-Barr virus nuclear antigen-1. Nature 375, 685-688 (1995). | PubMed | ISI | 24. Blake, N. et al. Human CD8+ T cell responses to EBV EBNA1: HLA class I presentation of the (Gly-Ala)-containing protein requires exogenous processing. Immunity 7, 791-802 (1997). | PubMed | ISI | 25. Sigal, L.J. & Rock, K.L. Bone marrow-derived antigen-presenting cells are required for the generation of cytotoxic T lymphocyte responses to viruses and use transporter associated with antigen presentation (TAP)-dependent and - independent pathways of antigen presentation. J. Exp. Med. 192, 1143-1150 (2000). | Article | PubMed | ISI | 26. Prasad, S.A., Norbury, C.C., Chen, W., Bennink, J.R. & Yewdell, J.W. Cutting edge: recombinant adenoviruses induce CD8 T cell responses to an inserted protein whose expression is limited to nonimmune cells. J. Immunol. 166, 4809-4812 (2001). | PubMed | ISI | 27. Norbury, C.C. et al. Multiple antigen-specific processing pathways for activating naive CD8+ T cells in vivo. J. Immunol. 166, 4355-4362 (2001). | PubMed | ISI | 28. Basta, S., Chen, W., Bennink, J.R. & Yewdell, J.W. Inhibitory effects of cytomegalovirus proteins US2 and US11 point to contributions from direct priming and cross-priming in induction of vaccinia virus-specific CD8+ T cells. J. Immunol. 168, 5403-5408 (2002). | PubMed | ISI | 29. Gold, M.C. et al. The murine cytomegalovirus immunodomulatory gene m152 prevents recognition of infected cells by M45-specific CTL, but does not alter the immunodominance of the M45-specific CD8 T cell response in vivo. J. Immunol. 169, 359-365 (2002). | PubMed | ISI | 30. Gilbert, M.J., Riddell, S.R., Plachter, B. & Greenberg, P.D. Cytomegalovirus selectively blocks antigen processing and presentation of its immediate-early gene product. Nature 383, 720-722 (1996). | PubMed | ISI | 31. Reimann, J. & Schirmbeck, R. Alternative pathways for processing exogenous and endogenous antigens that can generate peptides for MHC class I-restricted presentation. Immunol. Rev. 172, 131-152 (1999). | PubMed | ISI | 32. Orange, J.S. & Biron, C.A. Characterization of early IL-12, IFN- , and TNF effects on antiviral state and NK cell responses during murine cytomegalovirus infection. J. Immunol. 156, 4746-4756 (1996). | PubMed | ISI | 33. Orange, J.S. & Biron, C.A. An absolute and restricted requirement for IL-12 in natural killer cell IFN- production and antiviral defense. Studies of natural killer and T cell responses in contrasting viral infections. J. Immunol. 156, 1138-1142 (1996). | PubMed | ISI | 34. Polic, B. et al. Hierarchical and redundant lymphocyte subset control precludes cytomegalovirus replication during latent infection. J. Exp. Med. 188, 1047-1054 (1998). | Article | PubMed | ISI | 35. Doherty, P.C., Christensen, J.P., Belz, G.T., Stevenson, P.G. & Sangster, M.Y. Dissecting the host response to a -herpesvirus. Phil. Trans. R. Soc. Lond. B 356, 581-593 (2001). | ISI | 36. Redpath, S., Angulo, A., Gascoigne, N.R. & Ghazal, P. Murine cytomegalovirus infection down-regulates MHC class II expression on macrophages by induction of IL-10. J. Immunol. 162, 6701-6707 (1999). | PubMed | ISI | 37. Tomazin, R. et al. Cytomegalovirus US2 destroys two components of the MHC class II pathway, preventing recognition by CD4+ T cells. Nature Med. 5, 1039-1043 (1999). | Article | PubMed | ISI | 38. Cebulla, C.M. et al. Human cytomegalovirus disrupts constitutive MHC class II expression. J. Immunol. 169, 167-176 (2002). | PubMed | ISI | 39. Liu, X., Schrager, J.A., Lange, G.D. & Marsh, J.W. HIV Nef-mediated cellular phenotypes are differentially expressed as a function of intracellular Nef concentrations. J. Biol. Chem. 276, 3276332770 (2001). | Article | PubMed | ISI | 40. Collins, K.L., Chen, B.K., Kalams, S.A., Walker, B.D. & Baltimore, D. HIV-1 Nef protein protects infected primary cells against killing by cytotoxic T lymphocytes. Nature 391, 397-401 (1998). | Article | PubMed | ISI | 41. Krmpotic, A. et al. The immunoevasive function encoded by the mouse cytomegalovirus gene m152 protects the virus against T cell control in vivo. J. Exp. Med. 190, 1285-1296 (1999). | Article | PubMed | ISI | 42. Lubinski, J.M. et al. Herpes simplex virus type 1 glycoprotein gC mediates immune evasion in vivo. J. Virol. 72, 8257-8263 (1998). | PubMed | ISI | 43. Farrell, H.E. et al. Inhibition of natural killer cells by a cytomegalovirus MHC class I homologue in vivo. Nature 386, 510-514 (1997). | PubMed | ISI | 44. Krmpotic, A. et al. MCMV glycoprotein gp40 confers virus resistance to CD8+ T cells and NK cells in vivo. Nature Immunol. 6, 529-535 (2002). | Article | 45. van Berkl, V. et al. Critical role for a high-affinity chemokine-binding protein in -herpesvirusinduced lethal meningitis. J. Clin. Invest. 109, 905-914 (2002). | Article | PubMed | ISI | 46. Stevenson, P.G., Efstathiou, S., Doherty, P.C. & Lehner, P.J. Inhibition of MHC class Irestricted antigen presentation by 2-herpesviruses. Proc. Natl. Acad. Sci. USA 97, 8455-8460 (2000). | Article | PubMed | ISI | 47. Stevenson, P.G. et al. K3-mediated evasion of CD8+ T cells aids amplification of a latent herpesvirus. Nature Immunol. 3, 733-740 (2002). | Article | PubMed | ISI | 48. Munch, J., Stolte, N., Fuchs, D., Stahl-Hennig, C. & Kirchhoff, F. Efficient MHC class I major histocompatibility complex down-regulation by simian immunodeficiency virus Nef is associated with a strong selective advantage in infected rhesus macaques. J. Virol. 75, 10532-10536 (2001). | Article | PubMed | ISI | 49. Allen, T.M. et al. Tat-specific cytotoxic T lymphocytes select for SIV escape variants during resolution of primary viraemia. Nature 407, 386-390 (2000). | Article | PubMed | ISI | 50. Moore, C. et al. Evidence of HIV adaptation to HLA-restricted immune responses at a population level. Science 296, 1439-1443 (2002). | Article | PubMed | ISI | 51. Goulder, P.J. et al. Evolution and transmission of stable CTL escape mutations in HIV infection. Nature 412, 334-338 (2001). | Article | PubMed | ISI | 52. Paabo, S., Bhat, B.M., Wold, W.S.M. & Peterson, P.A. A short sequence in the COOHterminus makes an adenovirus membrane glycoprotein a resident of the endoplasmic reticulum. Cell 50, 311-317 (1987). | PubMed | ISI | 53. Shamu, C.E., Story, C.M., Rapoport, T.A. & Ploegh, H.L. The pathway of US11-dependent degradation of MHC class I heavy chains involves a ubiquitin-conjugated intermediate. J. Cell Biol. 147, 45-58 (1999). | Article | PubMed | ISI | 54. Hewitt, E.W. et al. Ubiquitylation of MHC class I by the K3 viral protein signals internalization and TSG101-dependent degradation. EMBO J. 21, 2418-2429 (2002). | Article | PubMed | ISI | 55. Cosman, D. et al. A novel immunoglobulin superfamily receptor for cellular and viral MHC class I molecules. Immunity 7, 273-282 (1997). | PubMed | ISI | 56. Cosman, D., Fanger, N. & Borges, L. Human cytomegalovirus, MHC class I and inhibitory signalling receptors: more questions than answers. Immunol. Rev. 168, 177-185 (1999). | PubMed | ISI | 57. Cosman, D. et al. ULBPs, novel MHC class I-related molecules, bind to CMV glycoprotein UL16 and stimulate NK cytotoxicity through the NKG2D receptor. Immunity 14, 123-133 (2001). | PubMed | ISI | 58. Arase, H., Mocarski, E.S., Campbell, A.E., Hill, A.B. & Lanier, L.L. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296, 1323-1326 (2002). | Article | PubMed | ISI | 59. Smith, H. et al. Recognition of a virus-encoded ligand by a natural killer cell activation receptor. Proc. Natl. Acad. Sci. USA 19, 8826-8831 (2002). 60. Levitskaya, J., Sharipo, A., Leonchiks, A., Ciechanover, A. & Masucci, M.G. Inhibition of ubiquitin/proteasome-dependent protein degradation by the Gly-Ala repeat domain of the EpsteinBarr virus nuclear antigen 1. Proc. Natl. Acad. Sci. USA 94, 12616-12621 (1997). | Article | PubMed | ISI | 61. Hill, A. et al. Herpes simplex virus turns off the TAP to evade host immunity. Nature 375, 411415 (1995). | PubMed | ISI | 62. Fruh, K. et al. A viral inhibitor of peptide transporters for antigen presentation. Nature 375, 415-418 (1995). | PubMed | ISI | 63. Hinkley, S., Hill, A.B. & Srikumaran, S. Bovine herpesvirus-1 infection affects the peptide transport activity in bovine cells. Virus Res. 53, 91-96 (1998). | Article | PubMed | ISI | 64. Ahn, K. et al. The ER-luminal domain of the HCMV glycoprotein US6 inhibits peptide translocation by TAP. Immunity 6, 613-621 (1997). | PubMed | ISI | 65. Bennett, E.M., Bennink, J.R., Yewdell, J.W. & Brodsky, F.M. Cutting edge: adenovirus E19 has two mechanisms for affecting class I MHC expression. J. Immunol. 162, 5049-5052 (1999). | PubMed | ISI | 66. Gruhler, A., Peterson, P.A. & Fruh, K. Human cytomegalovirus immediate early glycoprotein US3 retains MHC class I molecules by transient association. Traffic 1, 318-325 (2000). | Article | PubMed | ISI | 67. Jones, T.R. et al. Human cytomegalovirus US3 impairs transport and maturation of major histocompatibility complex class I heavy chains. Proc. Natl. Acad. Sci. USA 93, 11327-11333 (1996). | Article | PubMed | ISI | 68. Kavanagh, D.G., Koszinowski, U.H. & Hill, A.B. The murine cytomegalovirus immune evasion protein m4/gp34 forms biochemically distinct complexes with class I MHC at the cell surface and in a pre-golgi compartment. J. Immunol. 167, 3894-3902 (2001). | PubMed | ISI | 69. Wiertz, E.J.H. et al. The human cytomegalovirus US11 gene product dislocates MHC class I heavy chains from the endoplasmic reticulum to the cytosol. Cell 84, 769-779 (1996). | PubMed | ISI | 70. Wiertz, E.J.H.J. et al. Sec61-mediated transfer of a membrane protein from the endoplasmic reticulum to the proteasome for destruction. Nature 384, 432-438 (1996). | PubMed | ISI | 71. Boname, J.M. & Stevenson, P.G. MHC class I ubiquitination by a viral PHD/LAP finger protein. Immunity 15, 627-636 (2001). | PubMed | ISI | 72. Kerkau, T. et al. The human immunodeficiency virus type-1 (HIV-1) Vpu protein interferes with an early step in the biosynthesis of MHC class I molecules. J. Exp. Med. 185, 1295-1305 (1997). | Article | PubMed | ISI | 73. Schubert, U. et al. CD4 glycoprotein degradation induced by human immunodeficiency virus type 1 Vpu protein requires the function of proteasomes and the ubiquitin-conjugating pathway. J. Virol. 72, 2280-2288 (1998). | PubMed | ISI | 74. Ziegler, H. et al. A mouse cytomegalovirus glycoprotein retains MHC class I complexes in the ERGIC/cis-Golgi compartments. Immunity 6, 57-66 (1997). | PubMed | ISI | 75. Kavanagh, D.G., Gold, M.C., Wagner, M., Koszinowski, U.H. & Hill, A.B. The Multiple immuneevasion genes of murine cytomegalovirus are not redundant. M4 and m152 inhibit antigen presentation in a complementary and cooperative fashion. J. Exp. Med. 194, 967-978 (2001). | Article | PubMed | ISI | 76. Ziegler, H., Muranyi, W., Burgert, H.G., Kremmer, E. & Koszinowski, U.H. The luminal part of the murine cytomegalovirus glycoprotein gp40 catalyzes the retention of MHC class I molecules. EMBO J. 19, 870-881 (2000). | Article | PubMed | ISI | 77. Reusch, U. et al. A cytomegalovirus glycoprotein re-routes MHC class I complexes to lysosomes for degradation. EMBO J. 18, 1081-1091 (1999). | Article | PubMed | ISI | 78. Piguet, V. et al. HIV-1 Nef protein binds to the cellular protein PACS-1 to downregulate class I major histocompatibility complexes. Nature Cell Biol. 2, 163-167 (2000). | Article | PubMed | ISI | 79. Ishido, S. et al. Inhibition of natural killer cell-mediated cytotoxicity by Kaposi's sarcomaassociated herpesvirus K5 protein. Immunity 13, 365-374 (2000). | PubMed | ISI | 80. Kleijnen, M.F. et al. A mouse cytomegalovirus glycoprotein, gp34, forms a complex with folded class I MHC molecules in the ER, which is not retained but is transported to the cell surface. EMBO J. 16, 685-694 (1997). | Article | PubMed | ISI | Figure 1. The classical MHC class I pathway is depicted with reference to viral interfering proteins. Oligopeptides (red circle) are derived from DRiPs through the action of proteasomes. Nascent MHC class I molecules—consisting of a heavy chain and 2-microglobulin ( 2M)—bind to TAP via tapasin. Peptide binding releases MHC class I to the cell surface. VIPRs interfere with this process at multiple steps. (1) EBV EBNA-1 contains a sequence that renders it resistant to proteasomal degradation23, 60. HCMV IE is phosphorylated by another viral protein, preventing generation of the immunodominant peptide epitope30. (2A) HSV ICP4761, 62 and a BHV1 protein63 bind to the cytosolic side of TAP and prevent peptide translocation. (2B) HCMV US6 binds to TAP in the ER lumen and prevents peptide translocation64. (2C) Several VIPRs bind to MHC class I in the ER, retaining it and/or interfering with the function of the peptide-loading complex. The proteins include AdE319K (retains MHC class I and also prevents tapasin-mediated docking with TAP65); HCMV US3 (binds MHC class I but dissociates, the mechanism of MHC class I retention is not clear but may involve repeated rebinding by newly synthesized US3)66, 67. MCMV m4/gp34 forms extensive complexes with MHC class I in the ER68. (3) HCMV US2 and US11 and MHV-68 K3 bind MHC class I in the ER and induce retrotranslocation for degradation by the proteasome69, 70. For US11, ubiquitylation of heavy chain cytoplasmic tail, possibly after retrograde translocation, has been described53. MHV-68 K3 ubiquitylates the tail before translocation71. HIV-1 Vpu also induces degradation of newly synthesized MHC class I72—probably via retrograde translocation and proteasome degradation— although, unlike US2 and US11, proteasome activity is required for retrograde translocation73. (4) MCMV m152/gp40 causes MHC class I to be retained in the ER cis-Golgi complex intermediate compartment (ERGIC)74. The fact that m152, itself a distant MHC class I homolog, can reduce cell surface expression of H-60 as well as classical MHC class I (in an allomorph-specific manner75) makes its poorly understood mechanism of action of particular interest. Only the lumenal domain of m152/gp40 is required for the retention of MHC class I 76. Binding of m152/gp40 to MHC class I has not been demonstrated, and m152/gp40 itself has a default pathway of export to and degradation in the lysosome74. (5) Several VIPRs remove MHC class I either from the Golgi or the cell surface. MCMV m6/gp48 contains a lysosomal targeting di-leucine motif in its tail; it binds directly to MHC class I and redirects it to the lysosome77. HIV and SIV Nef use the PACS-1 (phosphofurin acidic cluster sorting protein 1)–sorting protein to remove MHC class I from the cell surface and sequester it in the trans-Golgi network78. KSHV K3 and K5 ubiquitylate the MHC class I tail and target MHC class I to lysosomes in a TG101-dependent manner54. K5 also targets ICAM-1 (intercellular adhesion molecule 1) and B7-279. (6) MCMV m4/gp34 is found associated with MHC class I at the cell surface 80 and inhibits CTL recognition75, but cause and effect have not been firmly linked. Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 1026 - 1032 (2002) doi:10.1038/ni1102-1026 © Nature America, Inc. <> Chronic bacterial infections: living with unwanted guests Douglas Young, Tracy Hussell & Gordon Dougan Centre for Molecular Microbiology and Infection, Imperial College of Science, Technology and Medicine, London SW7 2AZ, UK. Correspondence should be addressed to G Dougan g.dougan@ic.ac.uk Some bacterial pathogens can establish life-long chronic infections in their hosts. Persistence is normally established after an acute infection period involving activation of both the innate and acquired immune systems. Bacteria have evolved specific pathogenic mechanisms and harbor sets of genes that contribute to the establishment of a persistent lifestyle that leads to chronic infection. Persistent bacterial infection may involve occupation of a particular tissue type or organ or modification of the intracellular environment within eukaryotic cells. Bacteria appear to adapt their immediate environment to favor survival and may hijack essential immunoregulatory mechanisms designed to minimize immune pathology or the inappropriate activation of immune effectors. Most encounters with pathogenic bacteria lead to an acute period of subclinical infection and, occasionally, clinical symptoms. If the infected host survives the encounter, the acute infection is usually fully resolved by elimination of the invading bacteria. However, in a minority of cases, small numbers of bacteria survive and cause persistent and sometimes life-long infections. The persistence of bacteria in the face of innate and primed acquired immunity means that an immune status quo has been established after the acute response. This situation is intriguing because we know that the innate immune system has evolved Toll and other pattern-recognition receptors to interact with common bacteria-specific macromolecules, such as lipopolysaccharide (LPS) 1 and CpG-containing motifs. Although bacteria can modify the structure of some Toll-binding components (for example, Salmonella and other bacteria will modify their lipid A composition under different growth conditions2) they cannot live without these classes of macromolecules and, consequently, mechanisms are likely to be operating during persistent infection to moderate or modify innate immunity. Before discussing particular types of persistent bacterial infections, it is worth considering some general mechanisms that could contribute to the persistent lifestyle (Table 1). High resolution image and legend (90K) Persistence and commensal bacteria Sophisticated regulatory controls are built into the mammalian immune system to prevent the activation of inappropriate immune responses and minimize the potential for immune damage 3. Consequently, mounting an effective response to pathogens is compromised by the needs of the host to prevent immune-mediated damage. This is most apparent at the mucosal surfaces of the body. Commensal colonization provides a model for persistent infections and may provide insights into how some microbes survive. Neonates are colonized by bacteria very shortly after birth and there is a rapid onset of immune activation driven by the newly colonized microbes that leads to both local and systemic humoral and cellular responses against these bacteria. The generation of significant antibacterial responses to the normal flora may go on throughout the life of an individual. Evidence suggests that commensals such as Bacteroides fragilis in the gut4 and Neisseria meningitidis in the upper respiratory tract5 undergo significant antigenic variation in their surface antigens, such as polysaccharide capsules, which is almost certainly immune-driven. Bacterial colonization of the intestine is a required signal for the normal development and regulation of the mucosal immune system, as germ-free animals show impaired development of Peyer's patches and other mucosal immune tissues. Also, certain knockout mice that show defects in regulatory cytokines (including interleukin 2 (IL-2)6 and IL-107, 8), key components of the signaling pathways9-11 or maintenance of mucosal integrity12, 13 will develop inflamed bowels if housed under normal laboratory conditions, but will develop less bowel inflammation in a germ-free environment or when receiving antibiotics14. Thus, the mucosal immune system consistently encounters a substantial number of bacterial antigens. How, then, is the balance maintained between this exposure that could drive immune activation, the need to prevent immunological damage and the requirement for eliminating pathogens? A simple explanation often put forward is that antigens from commensals are rapidly eliminated from mucosa-associated tissues by local effectors15. The mucosal immune system has developed specialized noninflammatory mechanisms for eliminating antigens. For example, dimeric immunoglobulin A (IgA) is secreted by mucosal tissues and prevents pathogen entry by agglutination (immune-exclusion). The T cell repertoire at resting mucosal sites in some species, such as laboratory-housed mice, is skewed towards a T helper type 2 (T H2) phenotype16. The IL-4 and IL-5 produced by these T cells, together with transforming growth factor- (TGF- ), contribute to classical IgA class switching. A T cell–independent IgA production mechanism that targets commensal bacteria has also been identified17. Commensal bacteria reside mainly in the lumen of the intestine behind a glycocalyx barrier that separates them from enterocytes. This location may limit their potential to activate inflammatory responses18. In addition, epithelial cells and intraepithelial lymphocytes may have a higher threshold for responding to cues from bacteria. The down-regulation of receptors such as Toll-like receptor 4 (TLR4) on epithelial cells, gut macrophages and T cells serve to limit the inappropriate response to commensal LPS 19, and this may be true of other molecules that respond to bacterial components 20. The down-regulated, antigen-specific responses by mucosal T cells, although most likely designed to prevent clonal Table 1. Some m expansion of T cells specific for dietary and commensal antigens, could also assist in the bacterial persistence process. Pathogenic bacteria that can cross the glycocalyx and bind enterocytes or epithelial cells directly, such as pathogenic Escherichia coli and Helicobacter pylori, can trigger IL-8 and chemokine production that leads to inflammation. This inflammation is often associated with the recruitment of neutrophils and bacterial elimination, although in the case of H. pylori some bacteria may be resident at sites in the stomach not available to the effector cells and will remain as commensals or pathogens. In some H. pylori–infected individuals, this balance between commensalism and binding leads to chronic gastritis or even, in some individuals, stomach cancer. Evidence suggests that individuals become colonized for life by particular H. pylori strain types and that only a percentage of these infected individuals will go on to manifest clinical disease. This particular situation vividly illustrates the close relationship between colonization and disease at mucosal surfaces. Dysregulation of the TH1-TH2 balance at the mucosa can lead to disease. Induction of TH1 interferon- (IFN- )–secreting T cells at mucosal surfaces is associated with ulcerative colitis, inflammatory bowel disease, gastritis, pneumonia and bronchiolitis 21. Mechanisms responsible for the breakdown of mucosal immune homeostasis are unclear, but it is thought that genetically susceptible individuals over-respond to antigens from commensal and possibly pathogenic bacteria. Coupled with modified innate immune responses, it is likely that a balance involving a cascade of signals including the action of regulatory T cells secreting IL-10 and/or TGF- 22 and the activation state of dendritic and other antigen-presenting cells is critical15. Many Peyer's patch–associated mucosal dendritic cells are of the myeloid lineage and a combination of the pattern of costimulatory molecules expressed on their surface and their exposure to anti-inflammatory signals such as IL-10 and prostaglandin E2 may modify their ability to become activated or their migrational targeting 23. It is clear that T cell activation is critical for controlling mucosal immune responses and specialized T regulatory cells have been identified22, 24. Specific T cells are recruited to the mucosa through mucosal homing addressins such as the 4 7 or E 7 integrins and the expression of specific chemokine receptors such as CCR925. Such mechanisms clearly control the population of lymphocytes resident at mucosal sites. It appears that a cascade of signals may be required to fully activate mucosal immune responses. Even studies with simple mucosal adjuvants such as cholera toxin or the E. coli heat-labile toxin suggest that multiple factors in addition to binding are needed for full adjuvant activation26. This situation is even more complex in that the threshold needed to activate mucosal cells may be markedly different between species. For example, cells from the human Peyer's patch constitutively secrete IL-12 and show high activation of the signal transducers and activators of transcription factor 4 (STAT4) pathway27. Although we do not know all the mechanisms involved, it is clear that the normal flora is in some form of stasis with the mucosal immune system thus limiting pathology. S. enterica Typhi, adapted to systemic persistence Many bacterial pathogens cause a localized and acute infection in the intestine. A typical example is Salmonella enterica serovar Typhimurium, which causes invasive gastroenteritis and diarrhea in humans and cattle. S. enterica Typhimurium bacteria do not normally pass beyond the mesenteric lymph nodes in significant numbers except in immunocompromised individuals, such as those infected with HIV. In contrast to S. enterica Typhimurium, S. enterica Typhi is a close relative that routinely causes systemic infection (typhoid) that involves colonization of the reticuloendothelial system, including spleen and bone marrow (Fig. 1). Certain individuals infected with S. enterica Typhi become life-long carriers, periodically secreting high amounts of bacteria in their stools. Others will relapse to typhoid disease with the same S. enterica Typhi strain several months after the initial infection (1–5% of antibiotic-treated individuals), which suggests the presence of a persistent reservoir of bacteria in these individuals28. Short-term persistence (lasting several months) by S. enterica Typhi may involve colonization of immature immune cells in the bone marrow29, whereas longer term carriage is associated with infection of the gall bladder from where bacteria can be directly shed into the intestine via the bile duct. We know little about how bacteria adapt from being gut pathogens to derivatives capable of persistence in deeper tissues. However, the genomes of representative S. enterica Typhi30 and S. enterica Typhimurium31 have been published and comparison reveals some differences. S. enterica Typhi and S. enterica Typhimurium differ in about 10% of their genes. This difference also includes mutations in over 200 S. enterica Typhi genes, 145 of which are apparently intact in S. enterica Typhimurium. These S. enterica Typhi pseudogenes include mutations in 7 of 12 bacterial attachment factors (fimbrial operons)32 as well as mutations in genes involved in fecal shedding33 or modifying the intracellular lifestyle34 of Salmonella (for example, sopA, sopD2, sopE2, sseJ, cigR and misL). Pseudogene accumulation has been associated with other pathogens such as Yersinia pestis (plague bacilli)35 that have moved towards a systemic lifestyle as well as those that have an obligate intracellular lifestyle such as Mycobacterium leprae36. In the case of S. enterica Typhi, the loss of multiple adhesive determinants may preferentially target the pathogen to particular cell types such as dendritic cells 37 or CD18+ cells38 capable of delivering the bacteria to the systemic system while avoiding nonspecific targeting to epithelial cells, which leads to local gut inflammation. S. enterica Typhi may also modify the intracellular environment in a different manner to S. enterica Typhimurium (one consequence of which is host restriction to humans). In a direct comparison of analogous attenuated S. enterica Typhi– and S. enterica Typhimurium–based vaccines in human volunteers, the S. enterica Typhi vaccine was less efficient in colonizing the bowel and inducing inflammatory molecules, such as C-reactive protein, compared to S. enterica Typhimurium39. Poor colonization of the bowel by S. enterica Typhi may normally be compensated for by gall bladder–shedding to maintain transmission between hosts. Although the gall bladder is the principle site for life-long colonization of humans, virtually nothing is known about the mechanisms of persistence. High resolution image and legend (69K) Figure 1. Schematic representation of a model for persistence in human typhoid involving S. enterica Typhi. On the left of the figure, the acute infection (human gastroenteritis) mediated by S. enterica Typhimurium involves bacteria replicating freely in the intestine lumen and using multiple attachment factors (represented by multicolored tips or fimbriae). S. enterica Typhimurium targets both enterocytes and M cells for invasion, but is stopped at the mesenteric lymph nodes. Neutrophils are quickly attracted to the invasion site and inflammation follows, leading to diarrhea. S. enterica Typhi has dispensed with many attachment and shedding factors and may preferentially target a limited number of host cell types that favor dissemination to deeper tissues. S. enterica Typhi can persist in the bone marrow for extended periods and in the gall bladder for life. T, T cell; B, B cell. Although the genetics and immunology of S. enterica persistence has been studied in the mouse and could shed light on other persistent Salmonella infections, for example in the chicken or cattle, care should be taken in extrapolating data from the mouse model to human typhoid. Mutations that affect long-term Salmonella persistence in the mouse have been identified40. Indeed, it is possible to generate mutant S. enterica Typhimurium derivatives (such as aroA-purA double mutants) that persist for many months, even in susceptible mouse strains 41. Sublethal infection can also be established in genetically resistant mice and can be used to explore the immunology of persistence. Early bacterial clearance is influenced by factors such as the LPS type of the bacteria and by complement factors such as C1q42. Both antibodies and B cells also play critical roles in controlling infection by preventing initial colonization43. In mice, S. enterica Typhimurium targets the M cells and is subsequently taken into macrophages, polymorphs and dendritic cells. Salmonella bacteria may ultimately reside in particular macrophage types such as the red pulp macrophages of the spleen44. In the liver, Kupfer cells are initially targeted and infection foci are normally controlled by granuloma formation. Bacterial growth rate in macrophages is regulated in mice by such factors as Nramp1, although no evidence is available for a role of Nramp1 in human typhoid 45. Several inflammatory cytokines such as IL-12, tumor necrosis factor(TNF) and IL-18 are also key controlling factors. However, ultimate clearance of bacteria from tissues is dependent on T cell activity with CD4+ T cells playing a central role46, 47. Mice deficient in T cells (nu/nu-/-) or costimulatory molecules (CD28-/-) are more susceptible to S. enterica Typhimurium and T cell receptor (TCR)–deficient mice clear infection poorly at later time points 48-50. CD8+ T cells play a less important role46 and the contribution47 of TCR T cells in Salmonella control has yet to be completely resolved. The pivotal role played by CD4+ T cells in pathogen control is clearly observed in major histocompatibility complex (MHC) class II–deficient mice, which show impaired clearance of Salmonella in both mouse and human typhoid51. Tuberculosis initial evasion Mycobacteria are perhaps the first infectious agents to spring to mind in connection with chronic or persistent infections. Tuberculosis infection is initiated when Mycobacterium tuberculosis bacteria are inhaled deep into the alveoli of the lung, and persistence depends on the ability of the bacteria to resist the antimicrobial activities of resident alveolar macrophages (Fig. 2). A key step for bacteria to maintain persistence is interference with the process of phagosome maturation together with an inherent ability to survive under adverse conditions. By subverting the normal program by which phagosomes mature through the endocytic pathway, pathogenic mycobacteria are able to persist in a nonacidified compartment with the characteristics of an early endosome52. The immature phagosome continues to interact with recycling endosomes but excludes the vacuolar proton ATPase and retains the small GTPase Rab5 in preference to Rab7, which is found on mature phagosomes. Phagosomal arrest is a feature of live organisms; killed mycobacteria are rapidly transferred to conventional acidified phagolysosomes. Mechanisms underlying phagosomal arrest have yet to be identified and might include secretion of a low molecular weight metabolite by viable bacteria, expression of an enzyme capable of modifying a host component involved in regulation of endosomal interactions or modification of the phagosomal membrane by lipids present on the mycobacterial outer surface. Mycobacteria characteristically express a complex assortment of lipids and glycolipids53, with a major portion of the genome of M. tuberculosis encoding for protein important in lipid metabolism 54. Lipid components—including the long-chain fatty dialcohol phthiocerol dimycocerosate—form a waxy coating around the bacteria and make an important contribution to their inherent resistance to desiccation and exposure to osmotic imbalance and extremes of pH55. In combination with several enzymes capable of detoxifying active oxygen and nitrogen radicals56, the inherent resistance conferred by the cell wall structure makes mycobacteria a challenging target for the effector mechanisms of host macrophages. The ability of M. tuberculosis to negotiate this first phase of infection is lost after the mutation of genes involved in nutrition 57 and in cell wall structure55. During this initial stage of infection, mycobacteria spread from the lung and have the ability to establish infection in most parts of the body. High resolution image and legend (64K) tuberculosis. Figure 2. Persistent infection with M. (a) Infecting mycobacteria taken up by alveolar macrophages in the lung resist killing by subverting phagosome maturation and by the protective effect of their lipid-rich cell wall. (b) Inflammatory signaling in response to mycobacterial components results in the recruitment of TH1 T cells. T cell– mediated activation of macrophages enhances their ability to control mycobacteria. (c) Remaining viable mycobacteria are sequestered within a granuloma made up of macrophages and a variety of T cell subsets. (d) M. tuberculosis is able to persist in an asymptomatic form within the host over many decades. It is likely that mycobacteria are held in a nondividing or slowly dividing state in open lesions or in closed lesions walled-off by fibrosis. (e) Reduced immunity is associated with reactivation disease. Lack of immunosurveillance may stimulate renewed replication of mycobacteria in open lesions. For mycobacteria in closed lesions, liquefaction and breakdown in the absence of immune surveillance will result in disease. Once the disease process is underway, immunopathology contributes to tissue damage and ultimately efficient aerosol transmission to a new host. Containment by adaptive immune responses The antimycobacterial activity of macrophages is enhanced by T cell stimulation. Phagosomal arrest is reversed in macrophages that have been activated by IFN- before mycobacterial infection58, and this interface between the innate and acquired arms of the immune response is central to the control of mycobacterial disease. Genetic defects in the IL-12– or IFN- –activation pathways severely compromise the ability to control mycobacterial infection in both humans59 and mice60. Mycobacteria express a range of ligands that trigger signaling through pattern recognition receptors on host phagocytes61, 62, resulting in NF- B–mediated release of pro-inflammatory signals including TNF- and IL-12. This response may be reduced in the case of live M. tuberculosis63, but infection provokes a potent TH1 cell–mediated immune response. Activation of the adaptive immune response results in arrest of mycobacterial replication. The duration of the initial phase of bacterial muliplication depends on previous exposure. Priming by Bacillus Calmette Guerin (BCG) vaccination establishes a circulating pool of memory T cells that are rapidly recruited to the site of infection, limiting the extent of initial replication. In contrast, the requirement for T cell priming (presumably mediated by dendritic cells migrating to adjacent lymph nodes) in naïve hosts delays the response, thus extending the initial phase of infection. CD4 + T cells recognizing antigens presented on MHC class II molecules are central to this initial phase of bacterial containment, whereas CD8+ T cells may assume greater importance at the later stages of infection 60. In addition, T cells respond vigorously to phosphorylated ligands from mycobacteria 64 and may make a further contribution to the pool of IFN- , as may T cells restricted by CD165. CD1-restricted cells recognize lipid and glycolipid antigens that are exported from the mycobacterial phagosome 66 and may be available, therefore, at an early stage of infection. Lipoproteins also leave the live phagosome and, by gaining access to MHC class I processing pathways, may contribute to the set of antigens available for recognition by CD8+ T cells67. In addition to IFN- –mediated activation of macrophages, the cytotoxic function of T cells may contribute to control of the infection by releasing mycobacteria from cells that are refractory to activation and perhaps also by delivery of antimicrobial peptides68. The process of cell recruitment and activation results in sequestration of the infection within a granuloma. The lesion may then be further walled-off by fibrosis. At this late stage of the infection the balance seems to lie in favor of the host. In addition to falling back on their inherent cell wall defense mechanisms, several strategies have been suggested to contribute to mycobacterial survival in the face of this aggressive immune attack. Mycobacteria can interfere with T cell recognition of infected macrophages by reducing antigen presentation and by induction of suppressive cytokines. The expression of MHC class II molecules is down-regulated in infected cells, for example, by mechanisms that include signaling through TLRs69. High IL-6 secretion is associated with reduced T cell proliferation, and IL-10 and TGFmay play a role in suppressing T cell responses and macrophage activation. Sensitive assay systems demonstrate the presence of T H2 cytokines, IL-4 and IL-5, during active tuberculosis, and these may further contribute to suppression of macrophage activation 70. M. tuberculosis infection has also been associated with a reduction in the ability of macrophages to respond to IFNsignaling71 and with promotion of apoptosis. In this complex set of responses, it is hard to distinguish active microbe-mediated suppression from natural regulatory pathways associated with the immune homeostasis that is required to prevent pathological consequences of chronic macrophage activation. Persistent infection in tuberculosis Whether immunity to the initial infection is limited by pathogen-mediated suppression or by hostmediated regulation, it results in an opportunity for survival of a population of viable M. tuberculosis. This incomplete elimination, seeding the potential for future reactivation disease, is central to the pathogenesis of tuberculosis. Despite the obvious clinical importance, there are formidable gaps in our understanding of the frequency, location and physiological status of persistent populations of M. tuberculosis. Some idea of the extent of persistent human infection can be obtained by post mortem examination of tissues from asymptomatic individuals that died from causes unlinked to tuberculosis. Positive culture rates of 40–50% have been recorded in lung samples from individuals who have lived in high-incidence areas72. Old lesions characterized by extensive fibrosis and calcification yielded positive cultures less frequently than more open lesions. One extensive study recorded the presence of viable mycobacteria in areas of the lung free from granulomatous lesions 73. A recent polymerase chain reaction–based study supported the conclusion that persistent M. tuberculosis is not confined to histological lesions74. Studies of resected lung lesions from patients with active tuberculosis also showed the existence of distinct bacterial populations. Mycobacteria in open lesions were found to be actively replicating, whereas those in closed lesions were nondividing and yielded cultures only after prolonged in vitro incubation75. Experimental animal models have been used to analyze the biology of persistent infection. The simplest model is based on the natural immune-mediated control of a low-dose infection, generally in C57BL/6 mice. After the initial phase of mycobacterial multiplication, the bacterial load stabilizes at around one-million organisms in the lung. This is maintained for approximately one year, with the mice eventually succumbing to progressive pathology in the lung. The absence of substantial accumulation of dead organisms suggests that the bacteria in this model are replicating either slowly or not at all76, with a thermoresistant phenotype similar to that seen in stationary phase cultures77. This model has been exploited in the characterization of mutant strains of M. tuberculosis78, 79. Distinct from the auxotrophic and cell wall mutants discussed above that fail to multiply during the initial phase of infection, a second class of mutants that lacks a functional isocitrate gene matches the wild-type during the initial phase of infection, but show defective persistence78. Isocitrate lyase is particularly important for the utilization of fatty acids as a carbon source, and the behavior of the mutant suggests that lipids are a predominant source of nutrition during persistent infection. A regulatory mutation that results in constitutive overexpression of heatshock proteins also shows a persistence defect. In this case it is proposed that the effect is mediated through enhanced immune interactions79. A third class of mutant, defective in a twocomponent regulator resembling the phoPQ system of Salmonella, is restricted in its initial replication but is able to persist in infected tissues80. A second model for persistence involves infection of mice and incomplete treatment with isoniazid and pyrazinamide. At the end of the treatment, no bacteria are detected by conventional microbiological culture of tissue homogenates, although active infection becomes evident after immunosuppression81. This system, referred to as the "Cornell" model, may mimic the phenotype observed in closed lesions from human lungs and is of particular importance in investigating mycobacterial populations that persist during chemotherapy. A constant bacterial load during persistence could represent a nondividing population or a balance between active replication and immune killing. From the fragmentary information available, it seems likely that persistent M. tuberculosis divide only very slowly, if at all. The consequent production of only low amounts of new antigen is probably crucial in ensuring that the immune system tolerates the presence of the persistent infection. It is also likely that at least a portion of the persistent bacteria is physically screened from immune surveillance within closed lesions. The regulatory process involved in shutdown of mycobacterial cell division remains to be defined. The simplest hypothesis is that this resembles entry into stationary phase as a result of nutrient deprivation within the intracellular, or intra-granulomatous, microenvironment. Anoxia has been extensively investigated as a potential physiological stimulus82, and the resulting transcriptional changes have been characterized by whole genome microarray83. Of particular interest is the induction of expression of a protein belonging to the -crystallin or low molecular weight heat shock protein family. The -crystallin protein is a prominent target of B and T cell responses to tuberculosis and is essential for mycobacterial survival in macrophages. An alternative hypothesis would be that pathogenic mycobacteria have evolved a developmental program that generates some spore-like form specifically adapted for persistence. Although unusual forms of mycobacteria have been reported in studies of infected tissues, these observations have yet to find any counterpart in postgenomic analysis. Reactivation disease in tuberculosis It is estimated that one in ten individuals who mount an effective immune response to an initial encounter with M. tuberculosis will nevertheless develop clinical tuberculosis in later life. Disease may result from reactivation of the initial infection or from a subsequent reinfection; the relative prevalence of the two pathways is unknown and probably differs between geographical areas. Whether the source of bacteria is endogenous or exogenous, it remains to be explained why there is a failure of an initially protective immune response. This is most readily understood in the case of HIV infection. At the early stage of progression to AIDS, reduced CD4 counts are associated with markedly enhanced susceptibility to tuberculosis; individuals coinfected with HIV have a 10% annual risk of disease. Similarly, the reduced T cell immunity that occurs during immune senescence in elderly populations and transient immunosuppression caused by poor nutrition or alcohol consumption probably contributes to an increased risk of tuberculosis. Therapy for autoimmune disease that reduces TNF- activity also increases the risk of tuberculosis84. This occurs earlier in females than in males, suggesting an endocrine influence. Other infections may also be an important trigger for disease in endemic areas. Helminth infections that induce a strong systemic TH2 response might compromise TH1 antimycobacterial immunity, for example. Alternatively, repeated exposure to environmental mycobacteria may alter the kinetics of T cell turnover, effectively exhausting the original antimycobacterial memory pool. A third potential mechanism involves the induction of immune tolerance as a result of prolonged exposure to antigen in the absence of an appropriate second signal required for T cell activation. This is an attractive explanation for the highly specific TH1 anergy seen in lepromatous leprosy patients. Finally, it may be that an immune response sufficient to control a low dose infection is simply overwhelmed by a subsequent high dose exposure. This might occur as a result of intimate contact with a particularly infectious individual or from breakdown of an initially contained lesion. Most patients with active tuberculosis mount a strong immune response to mycobacterial antigens. The response (assessed by T cell proliferation or IFN- production, for example) is generally lower in patients than in healthy exposed individuals, but again it is predominantly T H1. Once immune containment has been lost, however, the TH1 response contributes to pathology rather than to protection85. As the immune system responds to challenge with an escalating concentration of mycobacterial antigen, the resulting inflammatory response causes localized tissue destruction, increasing the extent of mycobacterial multiplication and subsequent transmission. Thus, success of M. tuberculosis as a pathogen depends on its ability to withstand initial attack by the immune response and also on its ability to induce the immune-mediated tissue destruction required for efficient transmission. Elimination of persistent M. tuberculosis infection Elimination of persistent infection represents an important strategy for control of the associated diseases. Preventive therapy of individuals with latent M. tuberculosis infection significantly reduces the risk of subsequent disease. Although this intervention is routinely applied in the United States, it requires a robust infrastructure for prolonged administration of potentially toxic drugs, with a risk that misuse will contribute to drug resistance. Drugs targeted specifically against the nonreplicating or slowly replicating phenotypes implicated in persistence would enhance the feasibility of widespread preventive therapy. Despite the obvious pitfalls in extrapolating from mice to humans, the persistence mutants identified in animal models present potential leads for the development of such interventions. It is attractive to envisage an immune-based strategy for elimination of persistent infection. This could be based on boosting T H1 T cell responses that are induced during the initial infection but may wane with time or on artificial induction of additional responses directed to antigens that are under-represented in the natural response. An alternative strategy would be to enhance existing T cell responses, for example, by stimulating second signal responses in infected cells. A caveat concerning immune control of latent tuberculosis is the need to avoid triggering of the immunopathological manifestations characteristic of active disease. Other issues Modification of the intravacuole environment is a key feature of other persistent bacterial pathogens, including Salmonella34, Brucella and Chlamydia species. A modified intracellular environment could clearly favor persistence and evasion of immune responses through reduced surface antigen presentation or the control of apoptotic pathways. Brucella species use a type IV secretion system to avoid phagosomal fusion with the lysosomes and survive within an acidified intracellular compartment86, 87. Brucella accumulates inside macrophages where the type IV secretion system senses an increasing acidic environment and maintains a Brucella-adapted compartment. Successful replication by Chlamydia pneumoniae also involves inhibition of phagolysosomal fusion, although the mechanisms used by C. pneumoniae are unknown. With the use of pH-sensitive probes it has been shown that C. pneumoniae circumvents the host endocytic pathway and inhabits a nonacidic vacuole 88. This vacuole does not express the late endosomal marker cation-independent mannose-6-phosphate receptor or lysosomal-associated membrane protein 1 (LAMP-1) and LAMP-2 and CD63. A set of chlamydial proteins (Inc proteins) has been mapped to the surface membrane containing chlamydial inclusions. It is thought that these proteins may anchor the pathogen to the inclusion membrane, allowing it to occupy this distinct intracellular environment. The intracellular growth rate of C. pneumoniae slows dramatically during chronic infection, leading to attenuated production of new elementary bodies, appearance of morphologically aberrant reticulate bodies and altered expression of several chlamydial genes. Using infected HEp-2 cells, one group has shown that IFN- treatment reduced the expression of genes involved in cell division but maintained the transcription of genes involved in chlamydial DNA replication89. In order for C. pneumoniae to persist in the host, it would be an advantage for the infected cell to be long-lived. C. pneumoniae is capable of interfering with host cell apoptosis induced by staurosporine and CD95 death receptor–signaling90. This, together with inhibition of phagolysosomal fusion, may in part explain the ability of these bacteria to cause chronic infections in humans. For bacteria to survive long-term inside cells they must be able to adapt to a lifestyle involving slow replication or even dormancy. At present, little is known about how bacterial growth rate is regulated in vivo, although mycobacteria are well adapted to grow slowly. Antigenic variation is also a strategy used by several bacterial pathogens to evade the humoral immune system and persist in the host. Some bacteria such as Borrelia burgdorferi91 and Neisseria gonorrhea92 encode sophisticated genetic systems and gene families to increase the diversity of surface-located proteins in a similar manner to eukaryotic trypanosomes. Although the well characterized antigens of M. tuberculosis show no sequence diversity, it is speculated that two large gene families identified by genome analysis might provide a source of mycobacterial antigenic variation54, 93. Unlike the related spirochete B. burgdorferi, Treponema pallidum—the etiological agent of syphilis—apparently has a limited genetic ability to vary antigenic composition 94. The mechanisms of T. pallidum persistence remain a mystery. Although several hypotheses have been put forward to explain persistence, such as lack of expression of surface proteins, infection of nonprofessional antigen-presenting cells95 or coating in host antigens, our understanding of the immunology of T. pallidum persistence is almost nonexistent. Indeed, we know very little about the detailed mechanisms of how bacteria evade immunosurveillance on a long-term basis in the host. If we are to improve our therapeutic approaches to dealing with microbes such as M. tuberculosis, the gathering of more knowledge would be useful. References 1. Hijjar, A.M., Ernst, R.K., Tsai, J.H., Wilson, C.B. & Miller, S.I. Human Toll-like receptor 4 recognizes host-specific LPS modifications. Nature Immunol. 3, 354-359 (2002). | Article | PubMed | 2. Guo, L. et al. Regulation of lipid A modifications by Salmonella typhimurium virulence genes phoP-phoQ. Science 276, 250-253 (1997). | Article | PubMed | ISI | 3. Stockinger, B., Barthlott, T. & Kassiotis, G. T cell regulation: a special job or everyone's responsibility? Nature Immunol. 2, 757-758 (2001). | Article | PubMed | ISI | 4. Krinos, C.M., Coyne, M.J., Weinacht, K.G., Tzianabos, A.O., Kasper, D.L. & Comstock, L.E. Extensive surface diversity of a commensal microorganism by multiple DNA inversions. Nature 414, 555-558 (2001). | Article | PubMed | ISI | 5. Virji, M., Makepeace, K., Peak, I.R., Ferguson, D.J. & Moxon, E.R. Pathogenic mechanisms of Neisseria meningitides. Ann. NY Acad. Sci. 797, 273-276 (1996). | PubMed | ISI | 6. Ma, A., Datta, M., Margosian, E., Chen, J. & Horak, I. T cells, but not B cells, are required for bowel inflammation in interleukin 2-deficient mice. J. Exp. Med. 182, 1567-1572 (1995). | PubMed | ISI | 7. Kuhn, R., Lohler, J., Rennick, D., Rajewsky, K. & Muller, W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell 75, 263-274 (1993). | PubMed | ISI | 8. Sellon, R.K. et al. Resident enteric bacteria are necessary for development of spontaneous colitis and immune system activation in interleukin-10-deficient mice. Infect. Immun. 66, 5224-5231 (1998). | PubMed | ISI | 9. Simpson, S.J. et al. T cell-mediated pathology in two models of experimental colitis depends predominantly on the interleukin 12/signal transducer and activator of transcription (Stat)-4 pathway, but is not conditional on interferon expression by T cells. J. Exp. Med. 187, 1225-1234 (1998). | Article | PubMed | ISI | 10. Takeda, K. et al. Enhanced Th1 activity and development of chronic enterocolitis in mice devoid of Stat3 in macrophages and neutrophils. Immunity 10, 39-49 (1999). | PubMed | ISI | 11. Strober, W., Nakamura, K. & Kitani, A. The SAMP1/Yit mouse: another step closer to modeling human inflammatory bowel disease. J. Clin. Invest. 107, 667-670 (2001). | PubMed | ISI | 12. Panwala, C.M., Jones, J.C. & Viney, J.L. A novel model of inflammatory bowel disease: mice deficient for the multiple drug resistance gene, mdr1a, spontaneously develop colitis. J. Immunol. 161, 5733-5744 (1998). | PubMed | ISI | 13. Hermiston, M.L. & Gordon, J.I. Inflammatory bowel disease and adenomas in mice expressing a dominant negative N-cadherin. Science 270, 1203-1207 (1995). | PubMed | ISI | 14. Madsen, K.L. et al. Antibiotic therapy attenuates colitis in interleukin 10 gene-deficient mice. Gastroenterology 118, 1094-1105 (2000). | PubMed | ISI | 15. Nagler-Anderson, C. Man the barrier! Strategic defences in the intestinal mucosa. Nature Rev. Immunol. 1, 59-67 (2001). | Article | PubMed | 16. Strobel, S. & Mowat, A.M. Immune responses to dietary antigens: oral tolerance. Immunol. Today 19, 173-181 (1998). | Article | PubMed | ISI | 17. Macpherson, A.J. et al. IgA production without or chain expression in developing B cells. Nature Immunol. 2, 625-631 (2001). | Article | PubMed | ISI | 18. Hooper, L.V. & Gordon, J.I. Commensal host-bacterial relationships in the gut. Science 292, 1115-1118 (2001). | Article | PubMed | ISI | 19. Cario, E. & Podolsky, D.K. Differential alteration in intestinal epithelial cell expression of tolllike receptor 3 (TLR3) and TLR4 in inflammatory bowel disease. Infect. Immun. 68, 7010-7017 (2000). | Article | PubMed | ISI | 20. Smith, P.D. et al. Intestinal macrophages lack CD14 and CD89 and consequently are downregulated for LPS- and IgA-mediated activities. J. Immunol. 167, 2651-2656 (2001). | PubMed | ISI | 21. Blumberg, R.S., Saubermann, L.J. & Strober, W. Animal models of mucosal inflammation and their relation to human inflammatory bowel disease. Curr. Opin. Immunol. 11, 648-656 (1999). | Article | PubMed | ISI | 22. Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737-742 (1997). | Article | PubMed | ISI | 23. Newberry, R.D., McDonough, J.S., Stenson, W.F. & Lorenz, R.G. Spontaneous and continuous cyclooxygenase-2-dependent prostaglandin E2 production by stromal cells in the murine small intestine lamina propria: directing the tone of the intestinal immune response. J. Immunol. 166, 4465-4472 (2001). | PubMed | ISI | 24. Maloy, K.J. & Powrie, F. Regulatory T cells in the control of immune pathology. Nature Immunol. 2, 816-822 (2001). | Article | PubMed | ISI | 25. Zabel, B.A. et al. Human G protein-coupled receptor GPR-9-6/CC chemokine receptor 9 is selectively expressed on intestinal homing T lymphocytes, mucosal lymphocytes, and thymocytes and is required for thymus-expressed chemokine-mediated chemotaxis. J. Exp. Med. 190, 12411256 (1999). | Article | PubMed | ISI | 26. Rappuoli, R., Pizza, M., Douce, G. & Dougan, G. Structure and mucosal adjuvanticity of cholera and Escherichia coli heat-labile enterotoxins. Immunol. Today 20, 493-500 (1999). | Article | PubMed | ISI | 27. Macdonald, T.T. & Monteleone, G. IL-12 and Th1 immune responses in human Peyer's patches. Trends. Immunol 22, 244-247 (2001). | Article | PubMed | ISI | 28. Wain, J. et al. Molecular typing of multiple-antibiotic-resistant Salmonella enterica serovar Typhi from Vietnam: application to acute and relapse cases of typhoid fever. J. Clin. Microbiol. 37, 2466-2472 (1999). | PubMed | ISI | 29. Wain, J. et al. Quantitation of bacteria in bone marrow from patients with typhoid fever: relationship between counts and clinical features. J. Clin. Microbiol. 39, 1571-1576 (2001). | Article | PubMed | ISI | 30. Parkhill, J. et al. genome sequence of a multiple drug resistant Salmonella enterica serovar Typhi CT18. Nature 413, 848-852 (2001). | Article | PubMed | ISI | 31. McClelland, M. et al. Complete genome sequence of Salmonella enterica serovar Typhimurium LT2. Nature 413, 852-856 (2001). | Article | PubMed | ISI | 32. Townsend, S.M. et al. Salmonella enterica serovar Typhi possesses a unique repertoire of fimbrial gene sequences. Infect. Immun. 69, 2894-2901 (2001). | Article | PubMed | ISI | 33. Kingsley, R.A., Santos, R.L.K.A.M., Adams, L.G. & Baumler, A.J. Salmonella enteritica serotype Typhimurium ShdA is an outer membrane fibronectin-binding protein that is expressed in the intestine. Mol. Microbiol. 43, 895-905 (2002). | Article | PubMed | ISI | 34. Hughes, E.A. & Galan, J.E. Immune response to Salmonella: location, location, location? Immunity 16, 325-328 (2002). | PubMed | ISI | 35. Parkhill, J. et al. Genome sequence of Yersinia pestis, the causative agent of plague. Nature 413, 523-527 (2001). | Article | PubMed | ISI | 36. Cole, S.T. et al. Massive gene decay in the leprosy bacillus. Nature 409, 1007-1011 (2001). | Article | PubMed | ISI | 37. Rescigno, M. et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunol. 2, 361-367 (2001). | Article | PubMed | ISI | 38. Vazquez-Torres, A. et al. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature 401, 804-808 (1999). | Article | PubMed | ISI | 39. Hindle, Z. et al. Characterisation in volunteers of Salmonella enteritica derivatives harboring defined aroC and SPI-2 type III secretion system (ssaV) mutations. Infect. Immun. 70, 3457-3467 (2002). | Article | PubMed | ISI | 40. Clements, M.O. et al. Polynucleotide phosphorylase is a global regulator of virulence and persistency in Salmonella enterica. Proc. Natl. Acad. Sci. USA 99, 8784-8789 (2002). | Article | PubMed | ISI | 41. O'Callaghan, D., Maskell, D., Liew, F.Y., Easmon, C.S. & Dougan, G. Characterization of aromatic- and purine-dependent Salmonella typhimurium: attention, persistence, and ability to induce protective immunity in BALB/c mice. Infect. Immun. 56, 419-423 (1988). | PubMed | ISI | 42. Warren, J. et al. Increased susceptibility of C1q-deficient mice to Salmonella enterica serovar Typhimurium infection. Infect. Immun. 70, 551-557 (2002). | Article | PubMed | ISI | 43. Mastroeni, P., Simmons, C., Fowler, R., Hormaeche, C.E. & Dougan, G. Igh-6-/- (B-celldeficient) mice fail to mount solid acquired resistance to oral challenge with virulent Salmonella enterica serovar typhimurium and show impaired Th1 T-cell responses to Salmonella antigens. Infect. Immun. 68, 46-53 (2000). | PubMed | ISI | 44. Salcedo, S.P., Noursadeghi, M., Cohen, J. & Holden, D.W. Intracellular replication of Salmonella typhimurium strains in specific subsets of splenic macrophages in vivo. Cell Microbiol. 3, 587-597 (2001). | Article | PubMed | ISI | 45. Dunstan, S.J. et al. fever and genetic polymorphisms at the natural resistance-associated macrophage protein 1. J. Infect. Dis. 183, 1156-1160 (2001). | Article | PubMed | ISI | 46. Hess, J., Ladel, C., Miko, D. & Kaufmann, S.H. Salmonella typhimurium aroA- infection in gene-targeted immunodeficient mice: major role of CD4+ TCRcells and IFN- in bacterial clearance independent of intracellular location. J. Immunol. 156, 3321-3326 (1996). | PubMed | ISI | 47. Nauciel, C. Role of CD4+ T cells and T-independent mechanisms in acquired resistance to Salmonella typhimurium infection. J. Immunol. 145, 1265-1269 (1990). | PubMed | ISI | 48. O'Brien, A.D. & Metcalf, E.S. Control of early Salmonella typhimurium growth in innately Salmonella-resistant mice does not require functional T lymphocytes. J. Immunol. 129, 1349-1351 (1982). | PubMed | ISI | 49. Mittrucker, H.W., Kohler, A., Mak, T.W. & Kaufmann, S.H. Critical role of CD28 in protective immunity against Salmonella typhimurium. J. Immunol. 163, 6769-6776 (1999). | PubMed | ISI | 50. McSorley, S.J. & Jenkins, M.K. Antibody is required for protection against virulent but not attenuated Salmonella enterica serovar typhimurium. Infect. Immun. 68, 3344-3348 (2000). | Article | PubMed | ISI | 51. Dunstan, S.J. et al. Genes of the class II and class III major histocompatibility complex are associated with typhoid fever in Vietnam. J. Infect. Dis. 183, 261-268 (2001). | Article | PubMed | ISI | 52. Russell, D.G. Mycobacterium tuberculosis: here today, and here tomorrow. Nature Rev. Mol. Cell Biol. 2, 569-577 (2001). | Article | PubMed | ISI | 53. Brennan, P.J. & Nikaido, H. The envelope of mycobacteria. Annu. Rev. Biochem. 64, 29-63 (1995). | Article | PubMed | ISI | 54. Cole, S.T. et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393, 537-544 (1998). | Article | PubMed | ISI | 55. Barry, C.E. Interpreting cell wall 'virulence factors' of Mycobacterium tuberculosis. Trends. Microbiol. 9, 237-241 (2001). | Article | PubMed | ISI | 56. Bryk, R., Lima, C.D., Erdjument-Bromage, H., Tempst, P. & Nathan, C. Metabolic enzymes of mycobacteria linked to antioxidant defense by a thioredoxin-like protein. Science 295, 1073-1077 (2002). | Article | PubMed | ISI | 57. Hondalus, M.K. et al. Attenuation of and protection induced by a leucine auxotroph of Mycobacterium tuberculosis. Infect. Immun. 68, 2888-2898 (2000). | Article | PubMed | ISI | 58. Via, L.E. et al. Effects of cytokines on mycobacterial phagosome maturation. J. Cell Sci. 111, 897-905 (1998). | PubMed | ISI | 59. Casanova, J.L. & Abel, L. Genetic dissection of immunity to mycobacteria: the human model. Annu. Rev. Immunol. 20, 581-620 (2002). | Article | PubMed | ISI | 60. Flynn, J.L. & Chan, J. Immunology of tuberculosis. Annu. Rev. Immunol. 19, 93-129 (1902). | Article | 61. Brightbill, H.D. et al. Host defense mechanisms triggered by microbial lipoproteins through tolllike receptors. Science 285, 732-736 (1999). | Article | PubMed | ISI | 62. Underhill, D.M., Ozinsky, A., Smith, K.D. & Aderem, A. Toll-like receptor-2 mediates mycobacteria-induced proinflammatory signaling in macrophages. Proc. Natl. Acad. Sci. USA 96, 14459-14463 (1999). | Article | PubMed | ISI | 63. Nau, G.J. et al. Human macrophage activation programs induced by bacterial pathogens. Proc. Natl. Acad. Sci. USA 99, 1503-1508 (2002). | Article | PubMed | ISI | 64. Constant, P. et al. Stimulation of human T cells by nonpeptidic mycobacterial ligands. Science 264, 267-270 (1994). | PubMed | ISI | 65. Ulrichs, T. & Porcelli, S.A. CD1 proteins: targets of T cell recognition in innate and adaptive immunity. Rev. Immunogenet. 2, 416-432 (2000). | PubMed | 66. Schaible, U.E., Hagens, K., Fischer, K., Collins, H.L. & Kaufmann, S.H. Intersection of group I CD1 molecules and mycobacteria in different intracellular compartments of dendritic cells. J. Immunol. 164, 4843-4852 (2000). | PubMed | ISI | 67. Neyrolles, O. et al. Lipoprotein access to MHC class I presentation during infection of murine macrophages with live mycobacteria. J. Immunol. 166, 447-457 (2001). | PubMed | ISI | 68. Stenger, S. et al. Differential effects of cytolytic T cell subsets on intracellular infection. Science 276, 1684-1687 (1997). | Article | PubMed | ISI | 69. Noss, E.H. et al. Toll-like receptor 2-dependent inhibition of macrophage class II MHC expression and antigen processing by 19-kDa lipoprotein of Mycobacterium tuberculosis. J. Immunol. 167, 910-918 (2001). | PubMed | ISI | 70. Seah, G.T., Scott, G.M. & Rook, G.A. Type 2 cytokine gene activation and its relationship to extent of disease in patients with tuberculosis. J. Infect. Dis. 181, 385-389 (2000). | Article | PubMed | ISI | 71. Ting, L.M., Kim, A.C., Cattamanchi, A. & Ernst, J.D. Mycobacterium tuberculosis inhibits IFNtranscriptional responses without inhibiting activation of STAT1. J. Immunol. 163, 3898-3906 (1999). | PubMed | ISI | 72. Rich, A.R. The Pathogenesis of Tuberculosis (Blackwell Scientific Publications, Oxford, 2002). 73. Opie, E.L. & Aronson, J.D. Tubercle bacilli in latent tuberculosis lesions and in lung tissue without tuberculosis lesions. Arch. Pathol. 4, 1-21 (1927). 74. Hernandez-Pando, R. et al. Persistence of DNA from Mycobacterium tuberculosis in superficially normal lung tissue during latent infection. Lancet 356, 2133-2138 (2000). | Article | PubMed | ISI | 75. Vandiviere, H.M., Loring, W.E., Melvin, I. & Willis, S. The treated pulmonary lesion and its tubercule bacillus. II The death and the resurrection. Am. J. Med. Sci. 232, 30-37 (1956). 76. Rees, R.J. & Hart, P.D. Analysis of the host-parasite equilibrium in chronic murine tuberculosis by total and viable bacillary counts. Br. J. Exp. Pathol. 42, 83-88 (1961). | ISI | 77. Wallace, J.G. The heat resistance of tubercule in the lungs of infected mice. Am. Rev. Respir. Dis. 83, 866-871 (1961). | ISI | 78. McKinney, J.D. et al. Persistence of Mycobacterium tuberculosis in macrophages and mice requires the glyoxylate shunt enzyme isocitrate lyase. Nature 406, 735-738 (2000). | Article | PubMed | ISI | 79. Stewart, G.R. et al. Overexpression of heat-shock proteins reduces survival of Mycobacterium tuberculosis in the chronic phase of infection. Nature Med. 7, 732-737 (2001). | Article | PubMed | ISI | 80. Perez, E. et al. An essential role for phoP in Mycobacterium tuberculosis virulence. Mol. Microbiol. 41, 179-187 (2001). | Article | PubMed | ISI | 81. McCune, R.M., Feldmann, F.M., Lambert, H.P. & McDermott, W. Microbial persistence. I. The capacity of tubercle bacilli to survive sterilization in mouse tissues. J. Exp. Med. 123, 445-468 (1966). | PubMed | ISI | 82. Wayne, L.G. & Sohaskey, C.D. Nonreplicating persistence of Mycobacterium tuberculosis. Annu. Rev. Microbiol. 55, 139-163 (1902). | Article | 83. Sherman, D.R. et al. Regulation of the Mycobacterium tuberculosis hypoxic response gene encoding -crystallin. Proc. Natl. Acad. Sci. USA 98, 7534-7539 (2001). | Article | PubMed | ISI | 84. Keane, J. et al. Tuberculosis associated with infliximab, a tumor necrosis factor -neutralizing agent. N. Engl. J. Med. 345, 1098-1104 (2001). | Article | PubMed | ISI | 85. Moreira, A.L. et al. Mycobacterial antigens exacerbate disease manifestations in Mycobacterium tuberculosis-infected mice. Infect. Immun. 70, 2100-2107 (2002). | Article | PubMed | ISI | 86. O'Callaghan, D. et al. A homologue of the Agrobacterium tumefaciens VirB and Bordetella pertussis Ptl type IV secretion systems is essential for intracellular survival of Brucella suis. Mol. Microbiol. 33, 1210-1220 (1999). | PubMed | ISI | 87. Boschiroli, M.L. et al. The Brucella suis virB operon is induced intracellularly in macrophages. Proc. Natl. Acad. Sci. USA 99, 1544-1549 (2002). | Article | PubMed | ISI | 88. Boulton, I.C. & Gray-Owen, S.D. Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes. Nature Immunol. 3, 229-236 (2002). | Article | PubMed | ISI | 89. Byrne, G.I. et al. Chlamydia pneumoniae expresses genes required for DNA replication but not cytokinesis during persistent infection of HEp-2 cells. Infect. Immun. 69, 5423-5429 (2001). | Article | PubMed | ISI | 90. Fischer, S.F., Schwarz, C., Vier, J. & Hacker, G. Characterization of antiapoptotic activities of Chlamydia pneumoniae in human cells. Infect. Immun. 69, 7121-7129 (2001). | Article | PubMed | ISI | 91. Fraser, C.M. et al. Genomic sequence of a Lyme disease spirochaete, Borrelia burgdorferi. Nature 390, 580-586 (1997). | Article | PubMed | ISI | 92. Merz, A.J. & So, M. Interactions of pathogenic neisseriae with epithelial cell membranes. Annu. Rev. Cell Dev. Biol. 16, 423-457 (2000). | Article | PubMed | ISI | 93. Brennan, M.J. & Delogu, G. The PE multigene family: a 'molecular mantra' for mycobacteria. Trends. Microbiol. 10, 246-249 (2002). | Article | PubMed | ISI | 94. Porcella, S.F. & Schwan, T.G. Borrelia burgdorferi and Treponema pallidum: a comparison of functional genomics, environmental adaptations, and pathogenic mechanisms. J. Clin. Invest. 107, 651-656 (2001). | PubMed | ISI | 95. Sansonetti, P. Phagocytosis of bacterial pathogens: implications in the host response. Semin. Immunol. 13, 381-390 (2001). | Article | PubMed | ISI | 96. Galyov, E.E. et al. A secreted effector protein of Salmonella dublin is translocated into eukaryotic cells and mediates inflammation and fluid secretion in infected ileal mucosa. Mol. Microbiol. 25, 903-912 (1997). | PubMed | ISI | Acknowledgments. Supported by The Wellcome Trust (G. D. and D. Y.) and the Medical Research Council (T. H.). Table 1. Some mechanisms of increasing persistence used by pathogenic bacteria Figure 1. Schematic representation of a model for persistence in human typhoid involving S. enterica Typhi. On the left of the figure, the acute infection (human gastroenteritis) mediated by S. enterica Typhimurium involves bacteria replicating freely in the intestine lumen and using multiple attachment factors (represented by multicolored tips or fimbriae). S. enterica Typhimurium targets both enterocytes and M cells for invasion, but is stopped at the mesenteric lymph nodes. Neutrophils are quickly attracted to the invasion site96 and inflammation follows39, leading to diarrhea. S. enterica Typhi has dispensed with many attachment32 and shedding factors33 and may preferentially target a limited number of host cell types that favor dissemination to deeper tissues. S. enterica Typhi can persist in the bone marrow for extended periods and in the gall bladder for life. T, T cell; B, B cell. Figure 2. Persistent infection with M. tuberculosis. (a) Infecting mycobacteria taken up by alveolar macrophages in the lung resist killing by subverting phagosome maturation and by the protective effect of their lipid-rich cell wall. (b) Inflammatory signaling in response to mycobacterial components results in the recruitment of TH1 T cells. T cell–mediated activation of macrophages enhances their ability to control mycobacteria. (c) Remaining viable mycobacteria are sequestered within a granuloma made up of macrophages and a variety of T cell subsets. (d) M. tuberculosis is able to persist in an asymptomatic form within the host over many decades. It is likely that mycobacteria are held in a nondividing or slowly dividing state in open lesions or in closed lesions walled-off by fibrosis. (e) Reduced immunity is associated with reactivation disease. Lack of immunosurveillance may stimulate renewed replication of mycobacteria in open lesions. For mycobacteria in closed lesions, liquefaction and breakdown in the absence of immune surveillance will result in disease. Once the disease process is underway, immunopathology contributes to tissue damage and ultimately efficient aerosol transmission to a new host. Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 1033 - 1040 (2002) doi:10.1038/ni1102-1033 © Nature America, Inc. <> Bacterial strategies for overcoming host innate and adaptive immune responses Mathias W. Hornef1, Mary Jo Wick2, Mikael Rhen1 & Staffan Normark1 1 2 Microbiology and Tumor Biology Center (MTC), Karolinska Institutet, Nobelsväg 16, SE-17177 Stockholm, Sweden. Department of Clinical Immunology, University of Göteborg, Guldhedsgatan 10, SE-41346 Göteborg, Sweden. Correspondence should be addressed to S Normark staffan.normark@smi.ki.se In higher organisms a variety of host defense mechanisms control the resident microflora and, in most cases, effectively prevent invasive microbial disease. However, it appears that microbial organisms have coevolved with their hosts to overcome protective host barriers and, in selected cases, actually take advantage of innate host responses. Many microbial pathogens avoid host recognition or dampen the subsequent immune activation through sophisticated interactions with host responses, but some pathogens benefit from the stimulation of inflammatory reactions. This review will describe the spectrum of strategies used by microbes to avoid or provoke activation of the host's immune response as well as our current understanding of the role this immunomodulatory interference plays during microbial pathogenesis. Like all other higher organisms, humans have evolved in the continuous presence of various microbes. In fact, many body surfaces are densely populated by what we call the "normal microflora", which is mainly constituted of a variety of commensal bacteria, such as Bacteroides thetaiotamicron and Lactobacillus species. These bacteria are harmless and even beneficial under normal circumstances, but may cause local or systemic inflammatory disease if the integrity of the hosts' surface is disturbed. On the other hand, pathogenic bacteria are able to invade sterile body sites, proliferate and cause substantial tissue damage or systemic inflammation, such as is seen after infection with Shigella dysenteriae or Mycobacterium tuberculosis. The success of many pathogens relies on their ability to circumvent, resist or counteract host defense mechanisms; yet some bacteria also provoke activation of the immune system, which ultimately leads to disruption of the epithelial barrier and bacterial invasion. Consequently, pathogens have provided many examples of how to avoid and manipulate host responses. In this review, we will discuss how pathogens mechanistically avoid recognition by the immune system, and how they interfere—or possibly could interfere—with innate or adaptive immune responses, should recognition occur. We will examine the various steps that take place during the course of infection, starting with microbial attachment and colonization of host surfaces, which can eventually lead to invasion of the epithelial cell layer and penetration to the subepithelial tissue, and finally facilitate systemic dissemination of the microbes. We will discuss the strategies these microbes use to modify or circumvent the host defense mechanisms that come into play during this process, including recognition by surface immune receptors, secretion of antimicrobial effector molecules, internalization and degradation by phagocytes and activation of the humoral as well as the cellular immune systems. Attachment to and colonization of body surfaces Bacteria are excluded from the host tissue by anatomical barriers that consist of the skin and mucous membranes. The integrity of the mucosal surfaces is protected by active removal of bacteria, for example by the acid environment of the stomach, the ciliary movement in the upper respiratory tract and the continuous flushing with urine of the lower urinary tract. Thus, motility and attachment factors (so-called adhesins) found in most pathogenic bacteria are essential for approaching cellular surfaces and withstanding mechanical removal. For example, lack of expression of the major attachment factor toxin-coregulated pili (TCP) in Vibrio cholerae, significantly reduces the severity of bacteria-induced diarrhea in humans1. Alternatively, the secretion of bacterial toxins impairs protective functions and facilitates colonization. For example, Bordetella pertussis, the agent that causes whooping cough, paralyses the ciliary clearance function of the respiratory tract via the release of cell wall constituents that induce nitric oxide– mediated ciliostasis2. Biofilm formation by the opportunistic pathogens Pseudomonas aeruginosa and Staphylococcus epidermidis or the production of a protective bacterial extracellular matrix ("curli" surface fibers) by Escherichia coli shield bacteria from the hostile environment and might facilitate resistance against the host surface protective mechanisms 3. Finally, the presence of an extensive resident microflora represents yet another means of effectively protecting the host's mucosal surfaces; this is illustrated by infection with the opportunistic pathogen Clostridium difficile—the agent that causes pseudomembranous colitis—in patients undergoing antibiotic treatment, which results in disturbance of the enteric microflora. Thus, colonization by pathogens in the presence of a resident flora requires successful strategies that enable invading microbes to successfully compete for nutritional and spatial resources and displace commensal organisms from the microbial niche. Evasion of immune recognition by mucosal surfaces Besides acting as mechanical barriers, vulnerable mucosal membranes are covered with an array of soluble opsonizing factors, such as antibodies, that immobilize and remove approaching bacteria. Bacteria counter this with the proteolytic degradation of secretory immunoglobulin. This method of evasion is used particularly by bacteria that colonize the upper respiratory tract; Haemophilus influenzae—an important causative agent of respiratory tract infections—is one example of a microbe that uses such mechanisms to prevent opsonization and Fc receptor–mediated phagocytosis. Many types of epithelial cells have the intrinsic ability to sense the presence of microbial organisms and respond specifically through the identification of conserved components of these microbes. These microbial structures are termed pathogen-associated molecular patterns (PAMPs) and include parts of the bacterial cell envelope, such as lipopolysaccharide (LPS), peptidoglycan and bacterial DNA. Recognition of microbial structures by host cells relies on diverse families of genome-encoded receptors that allow detection of infectious nonself particles and provide signals that activate the defense mechanisms4. One group of membrane receptors, the tolllike receptors (TLRs), has attracted substantial attention due to their role in cellular signaling and their importance during initiation of the adaptive immune response5. The most effective strategy for avoiding innate recognition could involve steric shielding or modification of exposed PAMPs. In fact, host-like bacterial capsular structures have long been recognized as important virulence factors. Also, various LPS species from different commensal as well as pathogenic bacteria show some variance in the capacity to induce cytokine synthesis. Multiple alterations in the structure of Salmonella LPS decrease the microbe's potential to provoke innate immune responses 6. However, due to the pivotal role played by most PAMPs in essential bacterial cell functions as well as structure, major modifications might well decrease the viability and fitness of the bacterial intruders. Bacterial flagellin, which is recognized by TLR5, might represent an exception; flagellin shows in many Gram-negative bacteria as a result of phase and antigenic variation7. Also, although it is an important virulence factor for many bacteria (for example V. cholerae), it appears that flagellar expression is not an essential contributor to the pathogenicity of the prominent enteric pathogen Salmonella enterica serovar Typhimurium 8. The cellular process of pattern molecule recognition is only beginning to be understood. In addition to the need for soluble as well as membrane-bound accessory proteins such as LPS-binding protein (LBP), CD14 and MD-2, the cellular localization of a given TLR seems to be highly specific. For example, TLR2 is situated on the plasma membrane of macrophages and stays bound to its ligands—such as yeast zymosan—even after internalization in the phagosome9. In order for hypomethylated CpG motifs—a characteristic feature of bacterial DNA—to be recognized, endocytosis must occur so that the cell can signal through the intracellularly located receptor TLR910. TLR4 is found on the surface of macrophages but in the Golgi apparatus of intestinal epithelial cells, colocalized with its internalized ligand LPS11. These examples demonstrate the complexity of TLR-mediated recognition processes, which involve ligand internalization, cell traffic and fusion of subcellular compartments. Although the exact relationship between ligand localization and TLR-mediated signaling has not been determined, the possibility exists that microbes inhibit or delay recognition by interference with membrane and vesicular trafficking. Alternatively, because expression of recognition receptors seems to be organ-specific, recognition might be avoided through the selection of certain favorable anatomical sites for colonization and invasion12. In contrast to the avoidance of immune recognition, some microbial pathogens, under certain conditions, enhance immune-activation and pro-inflammatory responses by producing maximally stimulatory pattern molecules. For example, S. dysenteriae, which causes bacillary dysentery in humans, contains two copies of the msbB gene; one of these genes is located on the virulence plasmid. The msbB gene product is involved in the biosynthetic pathway of lipid A, the immunostimulatory part of the LPS molecule. Deletion of the msbB gene in E. coli leads to the production of hypoacylated lipid A with strongly decreased pro-inflammatory activity. The second msbB gene encoded by Shigella might be used to ensure complete acylation of lipid A and generate maximal stimulatory LPS. Cell activation is required to induce intestinal leukocyte infiltration followed by disruption of the enteric mucosal layer, which facilitates bacterial invasion13. As this example demonstrates, inflammation during the early course of infection might, under certain conditions, be advantageous. In contrast, long-term microbial colonization requires that cellular stimulation and activation of host defenses are avoided. This point is illustrated by Helicobacter pylori, which colonizes the human gastric mucosa and causes chronic infections in a large percentage of the human population. H. pylori activates stomach epithelial cells in a process that is mainly dependent on proteins encoded by the CagA pathogenicity island 14. After prolonged colonization, part of the bacterial population in the stomach tends to delete cag genes15. This may reflect a need to reduce the inflammatory response as soon as microbial colonization is established. In addition, a global modulation of virulence gene expression is associated with the transition from acute to chronic infection of mice with S. enterica Typhimurium16. In contrast, isolates of P. aeruginosa that chronically colonize the lungs of patients with the inherited disease cystic fibrosis continue to produce highly stimulatory LPS17. Biofilm formation and low susceptibility to host defense molecules (such as antimicrobial peptides and complement) might provide sufficient protection to allow P. aeruginosa to persist in the face of ongoing inflammation, which enhances the supply of nutrients. Recognition via host receptor molecules eventually leads to the activation of signal transduction cascades—including recruitment of adaptor molecules, tyrosine phosphorylation and activation of transcription factors—and subsequent activation of defense responses such as chemokine release and antimicrobial peptide production. An alternative immune-evasion strategy might interfere with cellular signaling during the stages that follow actual recognition. However, regarding microbial interference with TLR-induced signaling, only one example—that used by the vaccinia virus—has been described18. Possibly, disruption of immediate TLR-mediated signaling in host cells requires a pace that simply is not easily achieved. The alternative, then, would be to interfere with downstream signaling events. Active suppression, at the molecular level, of an induced pro-inflammatory immune response is demonstrated by S. enterica serovar Pullorum, the agent that causes fowl typhoid. In contrast to the well studied serovar Typhimurium—which causes inflammatory gastroenteritis in humans, and high secretion of pro-inflammatory cytokines in polarized human intestinal epithelial cells—S. enterica Pullorum produces only a minimal cellular response. More strikingly S. enterica Pullorum can suppress the pro-inflammatory activation of a subsequent exposure to S. enterica Typhimurium through active inhibition of I B ubiquitination19. Inhibition of cellular activation by commensal or pathogenic microbes may therefore represent a strategy with which gastrointestinal mucosal tolerance to pro-inflammatory stimuli can be maintained and host defenses avoided. Microbial strategies for the manipulation or avoidance of surface defense mechanisms of the host epithelial barrier are illustrated (Fig. 1). High resolution image and legend (75K) epithelial defense mechanisms. Figure 1. Strategies for bacterial escape from Prevention of opsonization (1) is required to facilitate colonization of host surfaces (2). Toxin secretion can paralyze the host's defenses (3) and disrupt its mucosal integrity (4). Microbial recognition and host responses—such as the secretion of antimicrobial peptides (5) or chemokine production (6)—can be impaired by modification of pattern molecule presentation or interference with intracellular signaling or cell trafficking (8). Microbe-induced self-uptake (7) and escape from the phagosome along with inhibition of intracellular recognition (9) or persistence in modified endosomes (10) can then impede removal by host defense mechanisms. Green, host responses; orange, bacterial components and interference with host defense strategies. Resistance to antibacterial effectors on epithelial surfaces In addition to their ability to attract professional immune cells, the epithelial body surfaces themselves provide effective innate antimicrobial defense. A large variety of antimicrobial peptides protect the inner and outer surfaces of most multicellular organisms against environmental microbial pathogens. These locally secreted short peptides are highly resistant to enzymatic degradation and show a net positive charge, which facilitates their binding to prokaryotic cell surfaces. Antimicrobial peptide–induced bacterial killing involves attachment and integration of the peptide into the surface of the invading prokaryote and subsequent disturbance of membrane integrity20. A whole spectrum of adaptive mechanisms used by bacteria lowers susceptibility to antimicrobial peptides expressed by the host. Although they are considered relatively resistant to enzymatic digestion, degradation of at least some linear antimicrobial peptides by bacterial proteases has been reported 21, 22, and active transport of peptides out of the bacterial cytoplasm also occurs 23. Some bacteria degrade extracellular matrix, and the resulting fragments bind to antimicrobial peptides and abolish their efficacy24. Bacterial membranes are much less susceptible to antimicrobial peptides than artificial membranes25. This might be explained by the fact that the negatively charged membranes of many bacteria are modified by the addition of positively charged residues. Staphylococcus aureus, the dominant causative agent of purulent wound infections, modifies its principal membrane lipid, phosphatidylglycerol, with lysine26 and adds D-alanine to teichoic acid27. Both changes reduce the net negative charge of the membrane. Similarly, under certain circumstances Gram-negative bacteria modify the structure of their LPS so they become less susceptible to antimicrobial killing. For example, S. enterica Typhimurium can form hepta-acylated lipid A (via the addition of palmitate by the bacterial protein PagP), add phosphate and phosphoethanolamine to the core polysaccharide and modify lipid A phosphate groups with ethanolamine and aminoarabinose. These alterations decrease the susceptibility of microbes to -helical antimicrobial peptides or the cyclic polypeptide polymyxin6, 28. Adaptation to antimicrobial peptides seems to play a critical role in microbial virulence, as 11 out of 12 S. enterica Typhimurium mutants with decreased susceptibility to host peptides, showed reduced virulence in vivo 29. Also, the mechanism used by the virulence factor encoded by the S. enterica Typhimurium mig14 gene was recently identified: mig-14 mutants showed enhanced susceptibility to antimicrobial peptides30. Finally, S. aureus that was unable to modify phosphatidylglycerol with L-lysin and thereby reduce its negatively charged surface membrane showed attenuated virulence in mice26. Thus, bacteria have evolved a number of mechanisms in order to adapt to surrounding antimicrobial peptides, and these mechanisms appear to be important in the expression of full virulence. However, high concentrations of animicrobial peptides at vulnerable body sites in vivo do nevertheless impede microbial colonization and growth. Why do bacteria not attempt full resistance to antimicrobial peptides? One explanation may lie in the high costs to microbial organisms that the development and expression of resistance engender. The diverse and highly specific biological functions of the microbial membrane might preclude modifications that allow resistance to the membrane-disturbing activity of antimicrobial peptides in the interest of preserving the functional and structural integrity of the microbial cell 20. For example, a strain of Streptococcus pyogenes that is resistant to the murine antimicrobial peptide Cramp shows growth inhibition in enriched culture medium 31. Therefore, the importance of decreased susceptibility to antimicrobial peptides may, in most cases, lie in the competition with resident microbial organisms for nutrients and space rather than resistance to the hosts' immune defense. Resistance-enhancing changes to LPS structure in Salmonella are tightly regulated by the PhoPPhoQ and PmrA-PmrB two-component signal transduction systems, which are central regulators of bacterial virulence6, 32. The ability to monitor the environment and accordingly modify the cell wall structure might allow the organism to adapt to specific requirements during infection and therefore minimize the accompanying high metabolic costs. Another explanation for the lack of emergence of resistance to antimicrobial peptides is the simultaneous production of a variety of different peptides at most body sites. The simultaneous use of different antimicrobial substances significantly impairs the development of microbial resistance, as is illustrated by the modern multiple drug regimens prescribed to treat tuberculosis or HIV infection. A recent genetic analysis has identified a large number of potential antimicrobial peptides in vertebrates, which further increases the quantity and diversity of molecules identified to date 33. Accordingly, gene-deletion strategies to prevent the expression of a single antimicrobial peptide have so far failed to reveal a clear phenotype of enhanced susceptibility to infection. (The only exception is a report on mice deficient in Cramp, the only murine member of the cathelicidin gene family, which showed enhanced susceptibility to skin infection with S. pyogenes 31.) In contrast, mice lacking the whole group of enteric -defensins—as a result of deletion of the proteolytic enzyme matrilysin—showed an increased susceptibility to orally administered S. enterica Typhimurium; this demonstrates the importance of enteric antimicrobial peptides in host defense 34. A third reason for the lack of fully resistant bacteria may be the fact that the activity of antimicrobial peptides seems to be highly regulated both at the transcriptional level and through enzymatic processing and secretion35, 36. Consequently, interference with antimicrobial peptide regulation seems to represent another microbial strategy for avoiding killing37, 38. In addition, the continuous presence of high peptide concentrations is restricted to defined and particularly vulnerable body sites such as the mouth, airways and intestinal crypts (where the intestinal epithelium regenerates) as a means of controlling the distribution of the normal flora 39, 40. The situation in the intestinal crypts illustrates this scenario well. These small, gland-like appendices (with a volume of 4–6 l) contain high concentrations of antimicrobial peptides (estimated to be of the order of grams per liter) that are produced by the Paneth cells at the lower end of the crypts and which effectively inhibit bacterial entry into the crypts and protect the site of epithelial regeneration 20, 40. Diffusion into the comparatively large intestinal lumen, absorption by the mucus overlying the epithelium and consumption through the abundant intestinal microflora results in a peptide concentration below that required to inhibit bacterial growth. Therefore, restricted secretion of antimicrobial peptides might help to avoid the development of microbial resistance by minimizing the selective pressure on the surrounding resident flora. Strategies for invading and crossing the epithelium Invasion of the epithelial layer provides protection from surface defense molecules. For example, S. enterica Typhimurium invades epithelial cells using a mechanism by which it induces its own uptake. The microbe uses a syringe-like transfer apparatus—termed a type III secretion system—to transfer two bacterial products, SopE and SopE2, directly from the bacterial cytoplasm into the eukaryotic host cell. Both proteins act as nucleotide exchange factors that activate central regulators of the actin cytoskeleton, the small GTP-binding proteins CDC42 and Rac, and induce subsequent engulfment of the bacterium 41, 42. However, activation of these proteins also stimulates nuclear responses through the transcription factors NF- B and AP-1, ultimately leading to the secretion of pro-inflammatory cytokines and attraction of professional phagocytes. This immune stimulation is counteracted by yet another translocated bacterial protein, SptP, which quenches the activated GTP-binding proteins involved and thereby limits cell activation42. Similarly, uropathogenic E. coli invade the bladder epithelium and thereby avoid clearance by surface host defense mechanisms43. Internalized Shigella flexneri—which has a similar clinical profile to S. dysenteriae— produces and secretes IpaB, which mediates lysis of the phagosome and allows the bacterium to escape into the cytoplasmic space44. Actin nucleation and polymerization initiated by the bacterial protein IcsA—which is located at the rear pole of the bacterium—enables S. flexneri to move through the cytoplasm and enter neighboring cells, facilitating microbe evasion of activated immune reponses45. One might assume that the cytosolic location provides optimal protection from immune recognition and response. However, even the cytosol seems to be equipped to detect the presence of bacterial pattern molecules, such as LPS, mediated by members of the nonobese diabetic (NOD) protein family leading to a pro-inflammatory cellular response46. Mutations in the human NOD2 gene, which are involved in cytosolic recognition of LPA, are associated with Crohns' disease, an inflammatory bowel disease of unknown etiology47, 48. Direct penetration of the skin is found with vector-born microbial diseases. In the case of Lyme disease, the protective skin barrier is transversed by the bite of a tick. The tick translocates Borrelia burgdorferi directly to the subepithelial space, where the bacteria initiate systemic infection. Infection with the spirochete Leptospira in an example of active transcutaneous migration, as this bacterium has the exceptional ability to actively penetrate the skin without the aid of any vector. Other bacteria such as S. pyogenes or Clostridium perfringens, both prominent causative agents of soft tissue infections, bypass the epithelial barrier via pre-existing injuries and use enzymatic degradation of the host's extracellular matrix, toxin-mediated cell destruction or induction of programmed cell death to spread themselves through intact tissue49. Yet another important mode of entry, bypassing the intestinal epithelial barrier—used, for example, by Salmonella and Shigella— occurs through a specialized cell type: the M cell. M cells overlay the Peyer's patches in the small intestine and can translocate luminal antigens (and even intact bacteria) to the basolateral side of the epithelia for uptake and recognition by the underlying cells of the immune system 50. However, once they are beyond this entry point, bacteria must defend themselves against resident professional immune cells. Escape from phagocyte responses Upon arrival at the subepithelial space, bacteria encounter locally resident as well as newly infiltrated professional phagocytic cells that are attracted by the chemokine response of the overlying epithelial cells. The strategies used by bacteria to overcome this additional defense barrier are shown (Fig. 2) Phagocytes are equipped with a number of receptors that detect the presence of invading microbes and bind opsonized microbial surfaces. Membrane-bound scavenger receptors, lectins, Fc receptors and complement receptors as well as signaling through TLRs may cooperate to determine the ultimate cellular response. This may lead to phagocyte maturation, activation of antimicrobial substances and secretion of pro-inflammatory cytokines, as well as phagocytosis and microbial degradation. High resolution image and legend (60K) Figure 2. Bacterial defense against phagocytes. Bacterial defense strategies against phagocyte engulfment include the induction of programmed cell death (1) as well as inhibition of uptake (2) by translocated effector proteins. Effector proteins can also be used to down-regulate other host cell nuclear responses (3). Should phagocytosis occur, bacteria can escape from the endosome into the host cell cytosol (4) or interfere with endosomal trafficking as well as maturation of the phagosome (5) and the subcellular localization of defense factors (6). Consequently, bacteria use a variety of strategies to avoid engulfment and degradation by phagocytes and facilitate proliferation and spread among host tissues 51. Examples are the inhibition of phagocytosis by capsule formation or toxin-mediated cellular destruction and necrosis. In contrast, induction of apoptosis avoids the release of pro-inflammatory signals49. Host-induced apoptosis of lung epithelial cells during infection with P. aeruginosa plays an important role in reducing leukocyte infiltration and maintaining the essential function of the lung: the oxygenation of blood52. In contrast, Salmonella and Shigella both actively stimulate pro-apoptotic pathways in order to paralyze phagocytic defense: SipB from S. enterica Typhimurium and the similar IpaB from S. flexneri are translocated via a type III secretion apparatus into the host cytosol. These proteins bind to caspase-1, which activates downstream caspases and induces apoptosis53, 54. The observation that caspase-1–deficient mice are resistant to infection with wild-type Salmonella suggests that this mechanism may contribute to the pathogenesis of this bacterium 55. Yersinia enterocolitica YopP (like its homolog Yersinia pseudotuberculosis YopJ) can also inhibit anti-apoptotic signals via the repression of NF- B activation as well as stimulation of pro-apoptotic signals through LPS-mediated activation of the TLR4 pathway56. Y. enterocolitica and Y. pseudotuberculosis—which both cause enterocolitis and abdominal lymphadenitis—can inhibit phagocytosis by the translocation of bacterial mediators that specifically disorganize the host cell cytoskeleton preventing bacterial uptake by macrophages and polymorphonuclear leukocytes. Bacterial YopE RhoGAP activity promotes the disruption of actin filaments by interaction with the Rho GTPases Rac, Rho and CDC42. YopH destabilizes focal adhesion via dephosphorylation of the adapter protein p130Cas and inhibits phagocytosis that is mediated by Fc receptors and complement receptors57, 58. Once internalization has occurred, some bacteria—such as the food-born pathogen Listeria monocytogenes, which is responsible for serious infections in immunocompromised individuals—manage to survive, persist and even proliferate in host phagocytes. To avoid degradation in the phagolysosome, L. monocytogenes is able to escape into the host cell cytosol by means of a bacterial toxin, listeriolysin, which disrupts the endosomal membrane59. Other pathogens such as Salmonella are able to manipulate endosomal trafficking and recruit defense factors to the maturing vacuole60. S. enterica Typhimurium, for example, is able to reduce the recruitment of NADPH oxidase and inducible nitric oxide synthase (iNOS) to the vacuole through interference with vacuolar trafficking, thereby preventing oxygen radical production and bacterial killing in macrophages61-63. The fact that many different Salmonella mutants that are able to down-regulate host iNOS activity could be isolated in a screen of macrophage-adapted bacteria suggests that Salmonella use several strategies for interfering with the host NO response60. And like many other bacteria, Salmonella is able to detoxify oxygen radicals enzymatically64. M. tuberculosis inhibits phagosomal maturation by depleting H+ ATPase molecules from the vacuolar membrane65. This leads to reduced acidification and allows intracellular survival and growth. Resistance to humoral defense mechanisms Successful escape by microbes from internalization by phagocytes opens the way for systemic spread in the host via the blood or lymph vessels. However, the limited supply of essential nutrients such as iron requires a high degree of adaptation to this environment. This is illustrated by the example of Yersinia, which carries genes that encode high-affinity uptake systems for ferric iron. In addition, bacteria will encounter humoral defenses. Soluble factors such as the C-reactive protein (CRP), mannan-binding lectin (MBL) and serum amyloid protein (SAP) are produced by the liver and function as opsonins. CRP and MBL also act as alternative recognition molecules for the antibody-independent activation of complement by binding to C1q, the activator of the classical complement activation pathway. Both S. pyogenes and Streptococcus pneumoniae possess surface structures that bind the complement regulatory component factor H66, 67. Factor H binding consequently promotes complement factor I–mediated degradation of C3b deposited on the bacterial surface and inhibits the release of chemotactic molecules, such as C5a and C3a, as well as formation of the membrane attack complex. Additionally, certain bacteria express proteases that degrade C1q, C3, C4 and C5-C968. As we mentioned before, intracellular persistence and proliferation, such as that seen with S. enterica Typhimurium, represents an opposite yet similarly effective strategy for avoiding the limited growth factors as well as soluble humoral defense molecules. Bacterial interference with cytokine secretion The innate immune system is clearly critical in the early control of bacterial replication and successful eradication of an infection. It is also linked to the adaptive immune response, which helps clear the infection and builds specific immunity with a memory component. Activation of the adaptive response occurs through cytokine secretion, antigenic processing and presentation as well as proliferation and differentiation of effector cells. Secretion of cytokines—particularly by effector T cells—killing of cells harboring intracellular pathogens by cells with cytolytic activity—such as CD8+ T cells—and antibody production by B cells all then contribute to controlling bacterial infections. Examples of the strategies bacteria use to deal with this complex defense network are shown (Fig. 3). High resolution image and legend (57K) the adaptive immune response. Figure 3. Bacterial defense strategies against Strategies include the induction of immunosuppressive cytokines, such as IL-10, IL-6 and TGF(1); inhibition of pro-inflammatory cytokine production; and surface expression of costimulatory molecules such as CD86 (2) by antigen presenting cells (APC). Interference with bacterial uptake (3), phagosome maturation (4) and antigen processing (5) as well as MHC class I and II expression (6) also lead to diminished antigen presentation. Inhibiting tyrosine phosphorylation of the T and B cell receptors (7) and activating the inhibitory CEACAM1 receptor on T cells (8) further decreases effector cell function. Certain bacteria can also induce regulatory T cells (formerly called suppressor T cells) that dampen the immune response (9) or induce T cell apoptosis by enhancing FasL expression on T cells (10). The production of pro-inflammatory cytokines such as tumor necrosis factor- (TNF- ), interleukin 1 (IL-1), IL-8 and IL-12 by host cells upon sensing bacterial products is crucial in the innate and adaptive immune responses to infection. These cytokines play a role in enhancing the bactericidal capacity of phagocytes, recruiting additional innate cell populations to sites of infection, inducing dendritic cell maturation and directing the ensuing specific immune response to the invading microbes. Some bacterial pathogens have evolved mechanisms for modulating cytokine production by host cells, which modifies the host's subsequent immune response. Mycobacteria provide a good example of bacterial manipulation of the cytokine response. These bacteria can induce the production of anti-inflammatory cytokines, which dampen the immune response. Mycobacteria-infected macrophages produce IL-6, which inhibits T cell activation69, as well as the potent immunosuppressive cytokines IL-1070 and transforming growth factor- (TGF)71. IL-10 is immunosuppressive in several ways72, including the inhibition of macrophage activation and production of reactive oxygen and nitrogen intermediates, suppression of inflammatory cytokine production as well as down-regulation of the production of molecules important in triggering specific immunity (for example the major histocompatibility complex (MHC) class II antigen presentation complex and the costimulatory molecule CD86). Mycobacterium-induced production of immunosuppressive cytokines may also contribute to the generation of regulatory T cells, also called T suppressor cells, that down-regulate immune activation. For example, aerosol treatment of mice with killed Mycobacterium vaccae induces regulatory T cells that prevent airway inflammation in an IL-10 and TGF- –dependent manner73. Similarly, B. pertussis exploits IL-10 in order to downregulate the host immune response. Bordetella filamentous hemagglutinin (FHA) induces IL-10 production by dendritic cells. This induces naïve T cells to develop into regulatory cells that suppress interferon- (IFN- ) production by antigen-specific T cells74. Also the LcrV protein produced by Y. enterocolitica induces macrophages to secrete IL-10, which, in turn, suppresses TNF- production75. Thus, bacterial exploitation of host cell capacity to produce immunosuppressive cytokines, particularly IL-10, provides an effective means for invading microbes to modulate host defense mechanisms and evade immune recognition. Certain bacteria have evolved mechanisms for interfering with the signal transduction pathways important in regulating expression of cytokines and other proteins involved in inflammation. For example, YopP from Y. enterocolitica and YopJ from Y. pseudotuberculosis inhibit NF- B and MAPK (mitogen-activated protein kinase) signal transduction pathways 76-78. Thus, Yersinia avoids the detrimental effects of pro-inflammatory cytokines secretion by suppressing TNF, IL-1 and IL-8 production. In contrast to Yersinia inhibition of NF- B activation, the intracellular pathogen L. monocytogenes activates this transcription factor as a potential means of increasing its pathogenicity79, 80. Listeria-mediated NF- B activation of endothelial cells results in increased expression of the adhesion molecules intercellular adhesion molecule 1 (ICAM-1) and E-selectin, and secretion of IL-8 and macrophage chemoattractant protein 1 (MCP-1)80. This attracts circulating phagocytes and promotes diapedesis and tissue infiltration. This "Trojan horse" mechanism directs Listeria-infected phagocytes to the subendothelial space, facilitating tissue spread and bacterial dissemination. Bacterial interference with antigen presentation Interfering with antigen processing and presentation is another strategy used by bacterial pathogens to prevent stimulation of an adaptive immune response. For example, S. enterica Typhimurium mutants that constitutively express the phoP-phoQ regulatory locus, which is important for survival in macrophages and bacterial virulence, are inefficiently processed by macrophages for MHC class II presentation81, 82. Similarly, the vacuolating toxin VacA produced by H. pylori diminishes the capacity of antigen-presenting cells to degrade internalized antigens83. Also, M. tuberculosis shows several strategies for suppressing antigen presentation and T cell activation, including inhibition of phagosomal maturation (see above) and sequestration of mycobacterial antigens from molecules required for T cell stimulation, such as MHC class II presentation84-86. Mycobacteria also down-regulate surface expression of MHC class II and CD1 and interfere with the presentation of antigens by MHC class II molecules 87-90. Mycobacteria inhibit the transcription of IFN- –responsive genes, including the master regulator for MHC class II expression, the class II transactivator (CIITA)89, 91. As MHC class II expression by resting macrophages is very low and IFN- is a potent inducer of MHC class II on these cells, the capacity of Mycobacteria to inhibit MHC class II expression by interfering with IFN- –mediated signaling pathways provides a potent means for dampening critical CD4+ T cell responses to this bacterium. Chlamydia trachomatis, a sexually transmitted pathogen that causes urogenital tract and ocular infections, also inhibits surface expression of MHC molecules on infected cells 92, 93. Like Mycobacteria, C. trachomatis inhibits IFN- –inducible MHC class II expression by interfering with CIITA activation. The mechanism used by Chlamydia to inhibit activation of CIITA involves degrading the upstream stimulatory factor 1 (USF-1) which is required for IFN- –mediated CIITA induction and, thus, IFN- –inducible MHC class II expression92. In addition, C. trachomatis suppresses both constitutive and IFN- –inducible MHC class I expression on infected cells by degrading the transcription factor regulatory factor X 5 (RFX5) in addition to degrading USF-193. As regulatory factor RFX5, a key component of the RFX transcription complex, is required for both constitutive and IFN- –inducible MHC class I expression and the RFX complex is required for MHC class II transcription94, the ability of C. trachomatis to degrade these transcription factors provides an effective means of blocking adaptive immunity. Inhibiting T and B cell effector functions Some bacteria interfere with the capacity of T and B cells—the effector cells of the adaptive immune system—to carry out their functions. For example, H. pylori Cag pathogenicity island-encoded genes induce Fas ligand (FasL) expression on T cells and mediate apoptosis 95. YopH from Y. pseudotuberculosis also suppresses antigen-specific T cell activation and IL-2 production by inhibiting tyrosine phosphorylation of components of the T cell receptor 96. YopH also inhibits tyrosine phosphorylation of components of the B cell receptor and suppresses up-regulation of the costimulatory molecule CD86 after B cell receptor engagement with antigen96. The YopHdependent inhibition of signaling cascades associated with antigen receptor engagement is an additional immune evasion strategy to the above-mentioned capacity of YopH to impair bacterial internalization97. Another bacterium that exploits the host receptor signal transduction machinery in order to modulate immunity is Neisseria gonorrhoeae, a sexually transmitted pathogen that causes urogenital infection. One class of the multiallelic, phase-variable Neisseria OPA proteins—which bind various ligands and mediate uptake by host cells—binds members of the CD66 receptor family, also known as carcinoembryonic antigen–related cellular adhesion molecule (CEACAM) family. CECAM1 is the only CEACAM molecule expressed on human lymphocytes, and the presence of an immunoreceptor tyrosine-based inhibitory motif (ITIM) in its cytoplasmic tail highlights its role as a coinhibitory receptor on CEACAM1+ cells. N. gonorroheae expressing a CEACAM1-binding OPA protein inhibits the activation and proliferation of CD4+ T cells stimulated by ligation of the T cell receptor98. This inhibitory effect was associated with increased recruitment of two tyrosine phosphatases, SHP-1 and SHP-2, that are critical to the inhibitory function of ITIMcontaining receptors98. Thus, the capacity of N. gonorrohea to inhibit T cell activation in addition to antigenic variation of surface proteins including OPAs99 likely contributes to the poor specific immune response observed in Neisseria-infected individuals. Our current knowledge of the strategies used by bacteria to interfere with innate and adaptive immunity and escape host defenses is largely incomplete. Nevertheless, the diverse disciplines of immunology, microbiology, infectious diseases and cell biology have contributed much to the exciting progress we have made in our knowledge over recent years. These studies have also revealed the complex interplay between microbial pathogens and higher organisms. However, when the role played by the host's normal microbial flora is included in the analysis, the complexity of bacteria-host interactions is even greater. How does the host differentiate between its responses to pathogens and commensals? One explanation is that the mucosal linings are tolerant to microbes at locations colonized by the normal flora, and that innate responses are induced only after bacterial intrusion beyond these barriers. However, even in the absence of pathogenic microorganisms, host defense mechanisms are required to maintain the integrity of the anatomical barrier against the resident microbial flora. The need for continuous vigilance is illustrated by mice that are deficient in the production of bactericidal oxygen and nitrogen intermediates (gp91phox-/-NOS2-/-). Such mice spontaneously develop massive abscesses that are caused by the normal flora of the intestine, respiratory tract and skin100. On the other hand, down-regulation of pro-inflammatory responses as well as enhancement of the intestinal barrier function appear to represent important functions of the normal microbial flora. The intestinal commensal Bacteroides thetaiotamicron protects host cells from complement-mediated cytotoxicity via the up-regulation of DAF (decay-accelerating factor), a central regulator of complement deposition on nucleated cells, and simultaneous enhancement of cutaneous repair and barrier functions101. Therefore, one might assume that commensals, like pathogens, express factors that directly or indirectly interfere with immune defense. However, the extent to which commensal microbes apply similar strategies remains an important question that needs to be addressed. Another important factor that should be taken into account is the heterogeneity of the host population: the genetic polymorphisms of receptor or effector molecules, and also the diversity of environmental conditions including the constitution of the resident microflora. Upcoming studies will undoubtedly reveal further surprising details of the intriguing relationship between bacteria and host immunity, and will hopefully provide us with the knowledge to improve the prevention and treatment of infectious diseases in the near future. References 1. Tacket, C.O. et al. Investigation of the roles of toxin-coregulated pili and mannose-sensitive hemagglutinin pili in the pathogenesis of Vibrio cholera O139 infection. Infect. Immun. 66, 692-695 (1998). | PubMed | ISI | 2. Flak, T.A. & Goldman, W.E. Signalling and cellular specificity of airway nitric oxide production in pertussis. Cell. Microbiol. 1, 51-60 (1999). | Article | PubMed | ISI | 3. Chapman, M.R. et al. Role of Escherichia coli curli operons in directing amyloid fiber formation. Science 295, 851-855 (2002). | Article | PubMed | ISI | 4. Janeaway, C.A. Jr. & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197-216 (2002). | Article | PubMed | ISI | 5. Medzhitov, R., Preston-Hurlburt, P. & Janeway, C.A. Jr. A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature 388, 394-397 (1997). | Article | PubMed | ISI | 6. Guo, L. et al. Regulation of lipid A modifications by Salmonella typhimurium virulence genes phoP-phoQ. Science 276, 250-253 (1997). | Article | PubMed | ISI | 7. Hayashi, F. et al. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 410, 1099-1103 (2001). | Article | PubMed | ISI | 8. Schmitt, C.K. et al. Absence of all components of the flagellar export and synthesis machinery differentially alters virulence of Salmonella enterica serovar Typhimurium in models of typhoid fever, survival in macrophages, tissue culture invasiveness, and calf enterocolitis. Infect Immun. 69, 5619-5625 (2001). | Article | PubMed | ISI | 9. Underhill, D.M. et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401, 811-815 (1999). | Article | PubMed | ISI | 10. Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740-745 (2000). | Article | PubMed | ISI | 11. Hornef, M.W., Frisan, T., Vandewalle, A., Normark, S. & Richter-Dahlfors, A. Toll-like receptor 4 resides in the Golgi apparatus and colocalizes with internalized lipopolysaccharide in intestinal epithelial cells. J. Exp. Med. 195, 559-570 (2002). | Article | PubMed | ISI | 12. Backhed, F., Soderhall, M., Ekman, P., Normark, S. & Richter-Dahlfors, A. Induction of innate immune responses by Escherichia coli and purified lipopolysaccharide correlate with organ- and cell-specific expression of Toll-like receptors within the human urinary tract. Cell. Microbiol. 3, 153158 (2001). | Article | PubMed | ISI | 13. D'Hauteville, H. et al. Two msbB genes encoding maximal acylation of lipid A are required for invasive Shigella flexneri to mediate inflammatory rupture and destruction of the intestinal epithelium. J. Immunol. 168, 5240-5251 (2002). | PubMed | ISI | 14. Fischer, W. et al. Systematic mutagenesis of the Helicobacter pylori cag pathogenicity island: essential genes for CagA translocation in host cells and induction of interleukin-8. Mol. Microbiol. 42, 1337-1348 (2001). | Article | PubMed | ISI | 15. Akopyants, N.S. et al. Analyses of the cag pathogenicity island of Helicobacter pylori. Mol. Microbiol. 28, 37-53 (1998). | Article | PubMed | ISI | 16. Clements, M.O. et al. Polynucleotide phosphorylase is a global regulator of virulence and persistency in Salmonella enterica. Proc. Natl. Acad. Sci. USA 99, 8784-8789 (2002). | Article | PubMed | ISI | 17. Hajjar, A.M., Ernst, R.K., Tsai, J.H., Wilson, C.B. & Miller, S.I. Human Toll-like receptor 4 recognizes host-specific LPS modifications. Nature Immunol. 3, 354-359 (2002). | Article | PubMed | ISI | 18. Bowie, A. et al. A46R and A52R from vaccinia virus are antagonists of host IL-1 and toll-like receptor signaling. Proc. Natl. Acad. Sci. USA 97, 10162-10167 (2000). | Article | PubMed | ISI | 19. Neish, A.S. et al. Prokaryotic regulation of epithelial responses by inhibition of I B ubiquitination. Science 289, 1560-1563 (2000). | Article | PubMed | ISI | 20. Zasloff, M. Antimicriobial peptides of multicellular organisms. Nature 415, 389-394 (2002). | Article | PubMed | ISI | 21. Guina, T., Yi, E.C., Wang, H., Hackett, M. & Miller, S.I. A PhoP-regulated outer membrane protease of Salmonella enterica serovar typhimurium promotes resistance to -helical antimicobial peptides. J. Bacteriol. 182, 4077-4086 (2000). | Article | PubMed | ISI | 22. Stumpe, S., Schmid, R., Stephens, D.L., Georgiou, G. & Bakker, E.P. Identification of OmpT as the protease that hydrolyses the antimicrobial peptide protamine before it enters growing cells of Escherichia coli. J. Bacteriol. 180, 4002-4006 (1998). | PubMed | ISI | 23. Shafer, W.M., Qu, X.-D., Waring, A.J. & Lehrer, R.I. Modulation of Neisseria gonorrhoeae susceptibility to vertebrate antibacterial peptides due to a member of the resistance/nodulation/division efflux pump family. Proc. Natl. Acad. Sci. USA 95, 1829-1833 (1998). | Article | PubMed | ISI | 24. Schmidtchen, A., Frick, I.-M. & Björck, L. Dermatan sulphate is released by proteinases of common pathogenic bacteria and inactivates antibacterial -defensins. Mol. Microbiol. 39, 708-713 (2001). | Article | PubMed | ISI | 25. Matsuzaki, K., Fukui, M., Fujii, N. & Miyajima, K. Interactions of an antimicrobial peptide, tachyplesin I, with lipid membranes. Biochem. Biophys. Acta 1070, 259-264 (1991). | ISI | 26. Peschel, A., Vuong, C., Otto, M. & Gotz, F. The D-alanine residues of Staphylococcus aureus teichoic acids alter the susceptibility to vancomycin and the activity of autolytic enzymes. Antimicrob. Agents Chemother. 44, 2845-2847 (2000). | Article | PubMed | ISI | 27. Peschel, A. et al. Staphylococcus aureus resistance to human defensins and evasion of neutrophil killing via the novel virulence factor mprF is based on modification of membrane lipids with L-lysine J. Exp. Med. 193, 1067-1076 (2001). | Article | PubMed | ISI | 28. Guo, L. et al. Lipid A acylation and bacterial resistance against vertebrate antimicrobial peptides. Cell 95, 189-198 (1998). | PubMed | ISI | 29. Groisman, E.A., Parra-Lopez, C., Salcedo, M., Lipps, C.J. & Heffron, F. Resistance to host antimicrobial peptides is necessary for Salmonella virulence. Proc. Natl. Acad. Sci. USA 89, 11939-11943 (1992). | PubMed | ISI | 30. Brodsky, I.E., Ernst, R.K., Miller, S.I. & Falkow, S. mig-14 is a Salmonella gene that plays a role in bacterial resistance to antimicrobial peptides. J. Bacteriol. 184, 3203-3213 (2002). | Article | PubMed | ISI | 31. Nizet, V. et al. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature 414, 454-457 (2001). | Article | PubMed | ISI | 32. Gunn, J.S., Ryan, S.S., Van Velkinburgh, J.C., Ernst, R.K. & Miller, S.I. Genetic and functional analysis of a PmrA-PmrB-regulated locus necessary for lipopolysaccharide modification, antimicrobial peptide resistance, and oral virulence of Salmonella enterica serovar typhimurium. Infect. Immun. 68, 6139-6146 (2000). | Article | PubMed | ISI | 33. Schutte, B.C. et al. Discovery of five conserved -defensin gene clusters using a computational search strategy. Proc. Natl. Acad. Sci. USA 99, 2129-2133 (2002). | Article | PubMed | ISI | 34. Wilson, C.L. et al. Regulation of intestinal -defensin activation by the metalloproteinase matrilysin in innate host defense. Science 286, 113-117 (1999). | Article | PubMed | ISI | 35. Ghosh, D. et al. Paneth cell trypsin is the processing enzyme for human defensin-5. Nature Immunol. 3, 583-590 (2002). | Article | PubMed | ISI | 36. Diamond, G., Kaiser, V., Rhodes, J., Russell, J.P. & Bevins, C.L. Transcriptional regulation of -defensin gene expression in tracheal epithelial cells. Infect. Immun. 68, 113-119 (2000). | PubMed | ISI | 37. Islam, D. et al. Downregulation of bactericidal peptides in enteric infections: a novel immune escape mechanism with bacterial DNA as a potential regulator. Nature Med. 7, 180-185 (2001). | Article | PubMed | ISI | 38. Lindmark, H. et al. Enteric bacteria counteract lipopolysaccharide induction of antimicrobial peptide genes. J. Immunol. 167, 6920-6923 (2001). | PubMed | ISI | 39. Dale, B.A. et al. Localized antimicrobial peptide expression in human gingiva. J. Periodontal. Res. 36, 285-294 (2001). | Article | PubMed | ISI | 40. Ayabe, T. et al. Secretion of microbicidal -defensins by intestinal Paneth cells in response to bacteria. Nature Immunol. 1, 113-118 (2000). | Article | PubMed | ISI | 41. Friebel, A. et al. SopE and SopE2 from Salmonella typhimurium activate different sets of RhoGTPases of the host cell. J. Biol. Chem. 276, 34035-34040 (2001). | Article | PubMed | ISI | 42. Galan, J.E. & Zhou, D. Striking a balance: modulation of the actin cytoskeleton by Salmonella. Proc. Natl. Acad. Sci. USA 97, 8754-8761 (2000). | Article | PubMed | ISI | 43. Mulvey, M.A. et al. Induction and evasion of host defenses by type 1-piliated uropathogenic Escherichia coli. Science 282, 1494-1497 (1998). | Article | PubMed | ISI | 44. High, N., Mounier, J., Prevost, M.C. & Sansonetti, P.J. IpaB of Shigella flexneri causes entry into epithelial cells and escape from the phagocytic vacuole. EMBO J. 11, 1991-1999 (1992). | PubMed | ISI | 45. Goldberg, M.B. & Theriot, J.A. Shigella flexneri surface protein IcsA is sufficient to direct actin-based motility. Proc. Natl. Acad. Sci. USA 92, 6572-6576 (1995). | PubMed | ISI | 46. Girardin, S.E. et al. CARD4/Nod1 mediates NF- B and JNK activation by invasive Shigella flexneri. EMBO Rep. 2, 736-742 (2001). | PubMed | ISI | 47. Hugot, J.P. et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature 411, 599-603 (2001). | Article | PubMed | ISI | 48. Ogura, Y. et al. A frameshift mutation in NOD2 associated with susceptibility to Crohn's disease. Nature 411, 603-606 (2001). | Article | PubMed | ISI | 49. Weinrauch, Y. & Zychlinsky, A. The induction of apoptosis by bacterial pathogens. Annu. Rev. Microbiol. 53, 155-187 (1999). | Article | PubMed | ISI | 50. Jones, B.D., Ghori, N. & Falkow, S. Salmonella typhimurium initiates murine infection by penetrating and destroying the specialized epithelial M cells of the Peyer's patches. J. Exp. Med. 180, 15-23 (1994). | PubMed | ISI | 51. Underhill, D.M. & Ozinsky, A. Phagocytosis of microbes: complexity in action. Annu. Rev. Immunol. 20, 825-852 (2002). | Article | PubMed | ISI | 52. Grassme, H. et al. CD95/CD95 ligand interactions on epithelial cells in host defense to Pseudomonas aeruginosa. Science 290, 527-530 (2000). | Article | PubMed | ISI | 53. Hersh, D. et al. The Salmonella invasin SipB induces macrophage apoptosis by binding to caspase-1. Proc. Natl. Acad. Sci. USA 96, 2396-2401 (1999). | Article | PubMed | ISI | 54. Zychlinsky, A. et al. IpaB mediates macrophage apoptosis induced by Shigella flexneri. Mol. Microbiol. 11, 619-627 (1994). | PubMed | ISI | 55. Monack, D.M. et al. Salmonella exploits caspase-1 to colonize Peyer's patches in a murine typhoid model. J. Exp. Med. 192, 249-258 (2000). | Article | PubMed | ISI | 56. Ruckdeschel, K., Mannel, O. & Schrottner, P. Divergence of apoptosis-inducing and preventing signals in bacteria-faced macrophages through myeloid differentiation factor 88 and IL1 receptor-associated kinase members. J. Immunol. 168, 4601-4611 (2002). | PubMed | ISI | 57. Black, D.S. & Bliska, J.B. The RhoGAP activity of the Yersinia pseudotuberculosis cytotoxin YopE is required for antiphagocytic function and virulence. Mol. Microbiol. 37, 515-527 (2000). | Article | PubMed | ISI | 58. Fallman, M. et al. Yersinia pseudotuberculosis inhibits Fc receptor-mediated phagocytosis in J774 cells. Infect. Immun. 63, 3117-3124 (1995). | PubMed | ISI | 59. Dramsi, S. & Cossart, P. Listeriolysin O: a genuine cytolysin optimized for an intracellular parasite. J. Cell. Biol. 156, 943-946 (2002). | Article | PubMed | ISI | 60. Eriksson, S. et al. Salmonella typhimurium mutants that downregulate phagocyte nitric oxide production. Cell. Microbiol. 2, 239-250 (2000). | Article | PubMed | ISI | 61. Uchiya, K. et al. A Salmonella virulence protein that inhibits cellular trafficking. EMBO J. 18, 3924-3933 (1999). | Article | PubMed | ISI | 62. Vazquez-Torres, A. et al. Salmonella pathogenicity island 2-dependent evasion of the phagocyte NADPH oxidase. Science 287, 1655-1658 (2000). | Article | PubMed | ISI | 63. Chakravortty, D., Hansen-Wester, I. & Hensel, M. Salmonella pathogenicity island 2 mediates protection of intracellular Salmonella from reactive nitrogen intermediates. J. Exp. Med. 195, 11551166 (2002). | Article | PubMed | ISI | 64. Fang, F.C. et al. Virulent Salmonella typhimurium has two periplasmic Cu, Zn-superoxide dismutases. Proc. Natl. Acad. Sci. USA 96, 7502-7507 (1999). | Article | PubMed | ISI | 65. Sturgill-Koszycki, S. et al. Lack of acidification in Mycobacterium phagosomes produced by exclusion of the vesicular proton-ATPase. Science 263, 678-681 (1994). | PubMed | ISI | 66. Horstmann, R.D., Sievertsen, H.J., Knobloch, J. & Fischetti, V.A. Antiphagocytic activity of streptococcal M protein: selective binding of complement control protein factor H. Proc. Natl. Acad. Sci. USA 85, 1657-1661 (1988). | PubMed | ISI | 67. Brown, E.J., Joiner, K.A., Gaither, T.A., Hammer, C.H. & Frank, M.M. The interaction of C3b bound to pneumococci with factor H ( 1H globulin), factor I (C3b/C4b inactivator), and properdin factor B of the human complement system. J. Immunol. 131, 409-415 (1983). | PubMed | ISI | 68. Wurzner, R. Evasion of pathogens by avoiding recognition or eradication by complement, in part via molecular mimicry. Mol. Immunol. 36, 249-60 (1999). | Article | PubMed | ISI | 69. van Heyningen, T.K., Collins, H.L. & Russell, D.G. IL-6 produced by macrophages infected with Mycobacterium species suppresses T cell responses. J. Immunol. 158, 330-337 (1997). | PubMed | ISI | 70. Giacomini, E. et al. Infection of human macrophages and dendritic cells with Mycobacterium tuberculosis induces a differential cytokine gene expression that modulates T cell response. J. Immunol. 166, 7033-7041 (2001). | PubMed | ISI | 71. Toossi, Z., Gogate, P., Shiratsuchi, H., Young, T. & Ellner, J.J. Enhanced production of TGFby blood monocytes from patients with active tuberculosis and presence of TGF- in tuberculosis granulamotous lung lesions. J. Immunol. 154, 465-473 (1995). | PubMed | ISI | 72. Moore, K.W., O'Garra, A., de Waal Malefyt, R., Vieira, P. & Mosmann, T.R. Interleukin-10. Annu. Rev. Immunol. 11, 165-190 (1993). | Article | PubMed | ISI | 73. Zuany-Amorim, C. et al. Suppression of airway eosinophilia by killed Mycobacterium vaccaeinduced allergen-specific regulatory T-cells. Nature Med. 8, 625-629 (2002). | Article | PubMed | ISI | 74. McGuirk, P., McCann, C. & Mills, K.H.G. Pathogen-specific T regulatory 1 cells induced in the respiratory tract by a bacterial molecule that stimulates interleukin 10 production by dendritic cells: a novel strategy for evasion of protective T helper type 1 responses by Bordetella pertussis. J. Exp. Med. 195, 221-231 (2002). | Article | PubMed | ISI | 75. Sing, A., Roggenkamp, A., Geiger, A.M. & Heesemann, J. Yersinia enterocolitica evasion of the host innate immune response by V antigen-induced IL-10 production of macrophages is abrogated in IL-10-deficient mice. J. Immunol. 168, 1315-1321 (2002). | PubMed | ISI | 76. Ruckdeschel, K. et al. Yersinia enterocolitica impairs activation of transcription factor NF- B: involvement in the induction of programmed cell death and in the suppression of the macrophage tumor necrosis factor production. J. Exp. Med. 187, 1069-1079 (1998). | Article | PubMed | ISI | 77. Schesser, K. et al. The YopJ locus is required for Yersinia-mediated inhibition of NF- B activation and cytokine expression: YopJ contains a eukaryotic SH2-like domain that is essential for its repressive activity. Mol. Microbiol. 28, 1067-1079 (1998). | PubMed | ISI | 78. Orth, K. et al. Inhibition of the mitogen-activated protein kinase kinase superfamily by a Yersinia effector. Science 285, 1920-1923 (1999). | Article | PubMed | ISI | 79. Kayal, S. et al. Listeriolysin O-dependent activation of endothelial cells during infection with Listeria monocytogenes: activation of NF- B and upregulation of adhesion molecules and chemokines. Mol. Microbiol. 31, 1709-1722 (1999). | PubMed | ISI | 80. Kayal, S. et al. Listeriolysin O secreted by Listeria monocytogenes induces NF- B signalling by activating the I B kinase complex. Mol. Microbiol. 44, 1407-1419 (2002). | Article | PubMed | ISI | 81. Wick, M.J., Harding, C.V., Twesten, N.J., Normark, S.J. & Pfeifer, J.D. The phoP locus influences processing and presentation of Salmonella typhimurium antigens by activated macrophages. Mol. Microbiol. 16, 465-476 (1995). | PubMed | ISI | 82. Niedergang, F., Sirard, J.-C., Blanc, C.T. & Kraehenbuhl, J.-P. Entry and survival of Salmonella typhimurium in dendritic cells and presentation of recombinant antigens do not require macrophage-specific virulence factors. Proc. Natl. Acad. Sci. USA 97, 14650-14655 (2000). | Article | PubMed | ISI | 83. Molinari, M. et al. Selective inhibition of Ii-dependent antigen presentation by Helicobacter pylori toxin VacA. J. Exp. Med. 187, 135-140 (1998). | Article | PubMed | ISI | 84. Ferrari, G., Naito, M., Langen, H. & Pieters, J. A coat protein on phagosomes involved in the intracellular survival of mycobacteria. Cell 97, 435-447 (1999). | PubMed | ISI | 85. Ullrich, H.-J., Beatty, W.L. & Russell, D.G. Interaction of Mycobacterium avium-containing phagosomes with the antigen presentation pathway. J. Immunol. 165, 6073-6080 (2000). | PubMed | ISI | 86. Ramachandra, L., Noss, E., Boom, H.W. & Harding, C.V. Processing of Mycobacterium tuberculosis antigen 85B involves intraphagosomal formation of peptide-major histocompatibility class II complexes and is inibited by live bacilli that decrease phagosome maturation. J. Exp. Med. 194, 1421-1432 (2001). | Article | PubMed | ISI | 87. Stenger, S., Niazi, K.R. & Modlin, R.L. Down-regulation of CD1 on antigen-presenting cells by infection with Mycobacterium tuberculosis. J. Immunol. 161, 3582-3588 (1998). | PubMed | ISI | 88. Hmama, Z., Gabathuler, R., Jefferies, W.A., de Jong, G. & Reiner, N.E. Attenuation of HLADR expression by mononuclear phagocytes infected with Mycobacterium tuberculosis is related to intracellular sequestration of immature class II heterodimers. J. Immunol. 161, 4882-4893 (1998). | PubMed | ISI | 89. Wojciechowski, W., DeSanctis, J., Skamene, E. & Radzioch, D. Attenuation of MHC class II expression in macrophages infected with Mycobacteirum bovis Bacillus Calmette-Geurin involves class II transactivator and depends on the Nramp1 gene. J. Immunol. 163, 2688-2696 (1999). | PubMed | ISI | 90. Noss, E.H. et al. Toll-like receptor 2-dependent inhibition of macrophage class II MHC expression and antigen processing by 19-kDa lipoprotein of Mycobacterium tuberculosis. J. Immunol. 167, 910-918 (2001). | PubMed | ISI | 91. Ting, L.-M., Kim, A.C., Cattamanchi, A. & Ernst, J.D. Mycobacterium tuberculosis inhibits IFN- transcriptional responses without inhibiting activation of STAT1. J. Immunol. 163, 3898-3906 (1999). | PubMed | ISI | 92. Zhong, G., Fan, T. & Liu, L. Chlamydia inhibits interferon- -inducible major histocompatibility complex class II expression by degradation of upstream stimulatory factor 1. J. Exp. Med. 189, 1931-1937 (1999). | Article | PubMed | ISI | 93. Zhong, G., Liu, L., Fan, T., Fan, P. & Ji, H. Degradation of transcription factor RFX5 during the inhibition of both constitutive and interferon -inducible major histocompatibility complex class I expression in Chlamydia-infected cells. J. Exp. Med. 191, 1525-2534 (2000). | Article | PubMed | ISI | 94. van den Elsen, P.J., Peijnenburg, A., van Eggermond, M.C. & Gobin, S.J. Shared regulatory elements in the promoters of MHC class I and class II genes. Immunol. Today 19, 308-312 (1998). | Article | PubMed | ISI | 95. Wang, J. et al. Negative selection of T cells by Helicobacter pylori as a model for bacterial strain selection by immune evasion. J. Immunol. 167, 926-934 (2001). | PubMed | ISI | 96. Yao, T., Mecsas, J., Healy, J.I., Falkow, S. & Chien, Y.-H. Suppression of T and B lymphocyte activation by a Yersinia pseudotuberculosis virulence factor, YopH. J. Exp. Med. 190, 1343-1350 (1999). | Article | PubMed | ISI | 97. Persson, C., Carballeira, N., Wolf-Watz, H. & Fällman, M. The PTPase YopH inhibits uptake of Yersinia, tyrosine phosphorylation of p130Cas and FAK, and the associated accumulation of these proteins in peripheral focal adhesions. EMBO J. 16, 2307-2318 (1997). | Article | PubMed | ISI | 98. Boulton, I.C. & Gray-Owen, S.D. Neisserial binding to CEACAM1 arrests the activation and proliferation of CD4+ T lymphocytes. Nature Immunol. 3, 229-236 (2002). | Article | PubMed | ISI | 99. Nassif, X., Pujol, C., Morand, P. & Eugene, E. Interactions of pathogenic Neisseria with host cells. Is it possible to assemble the puzzle? Mol. Microbiol. 32, 1124-1132 (1999). | PubMed | ISI | 100. Shiloh, M.U. et al. Phenotype of mice and macrophages deficient in both phagocytic oxidase and inducible nitric oxide synthase. Immunity 10, 29-38 (1999). | PubMed | ISI | 101. Hooper, L.V. et al. Molecular analysis of commensal host-microbial relationships in the intestine. Science 291, 881-884 (2001). | Article | PubMed | ISI | Acknowledgments. Supported by grants from the Deutsche Forschungsgemeinschaft and the Karolinska Institutet (to M. W. H.); the Swedish Medical Research Council (to M.-J. W., M. R. and S. N.), the Foundation for Strategic Research (to M. R.) and the Swedish Cancer Foundation (to S. N.). Figure 1. Strategies for bacterial escape from epithelial defense mechanisms. Prevention of opsonization (1) is required to facilitate colonization of host surfaces (2). Toxin secretion can paralyze the host's defenses (3) and disrupt its mucosal integrity (4). Microbial recognition and host responses—such as the secretion of antimicrobial peptides (5) or chemokine production (6)—can be impaired by modification of pattern molecule presentation or interference with intracellular signaling or cell trafficking (8). Microbe-induced self-uptake (7) and escape from the phagosome along with inhibition of intracellular recognition (9) or persistence in modified endosomes (10) can then impede removal by host defense mechanisms. Green, host responses; orange, bacterial components and interference with host defense strategies. Figure 2. Bacterial defense against phagocytes. Bacterial defense strategies against phagocyte engulfment include the induction of programmed cell death (1) as well as inhibition of uptake (2) by translocated effector proteins. Effector proteins can also be used to down-regulate other host cell nuclear responses (3). Should phagocytosis occur, bacteria can escape from the endosome into the host cell cytosol (4) or interfere with endosomal trafficking as well as maturation of the phagosome (5) and the subcellular localization of defense factors (6). Figure 3. Bacterial defense strategies against the adaptive immune response. Strategies include the induction of immunosuppressive cytokines, such as IL-10, IL6 and TGF- (1); inhibition of pro-inflammatory cytokine production; and surface expression of costimulatory molecules such as CD86 (2) by antigen presenting cells (APC). Interference with bacterial uptake (3), phagosome maturation (4) and antigen processing (5) as well as MHC class I and II expression (6) also lead to diminished antigen presentation. Inhibiting tyrosine phosphorylation of the T and B cell receptors (7) and activating the inhibitory CEACAM1 receptor on T cells (8) further decreases effector cell function. Certain bacteria can also induce regulatory T cells (formerly called suppressor T cells) that dampen the immune response (9) or induce T cell apoptosis by enhancing FasL expression on T cells (10). Focus on Immune Evasion Volume 3 No 11 November 2002 Review Nature Immunology 3, 1041 - 1047 (2002) doi:10.1038/ni1102-1041 © Nature America, Inc. <> Evasion of innate immunity by parasitic protozoa David Sacks & Alan Sher Laboratory of Parasitic Diseases, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD 20892, USA. Correspondence should be addressed to D Sacks dsacks@nih.gov Parasitic protozoa are a major cause of global infectious disease. These eukaryotic pathogens have evolved with the vertebrate immune system and typically produce longlasting chronic infections. A critical step in their host interaction is the evasion of innate immune defenses. The ability to avoid attack by humoral effector mechanisms, such as complement lysis, is of particular importance to extracellular parasites, whereas intracellular protozoa must resist killing by lysosomal enzymes and toxic metabolites. They do so by remodeling the phagosomal compartments in which they reside and by interfering with signaling pathways that lead to cellular activation. In addition, there is growing evidence that protozoan pathogens modify the antigen-presenting and immunoregulatory functions of dendritic cells, a process that facilitates their evasion of both innate and adaptive immunity. Parasitic protozoa are unicellular eukaryotic pathogens that dwell inside host cells and/or in extracellular fluids. These organisms typically produce long-lasting chronic infections in order to maximize their opportunities for successful transmission by contact with their intermediate hosts or release into the environment. Their success as parasites depends on a series of intricate and highly evolved host adaptations that enable them to evade destruction by the immune system. Protozoan infections are a major global health problem, affecting over half a billion people world wide. Several of the diseases they induce (such as malaria, African trypanosomiasis and visceral leishmaniasis) represent major causes of mortality and morbidity in tropical countries and, as such, are important impediments to economic development. No standardized vaccines exist for preventing any of the protozoan infections of humans, a situation attributed both to the complexity of the pathogens involved and their sophisticated strategies for evading host immune responses 1. Protozoan parasites are highly adept at escaping the effects of adaptive humoral and cellular immunity. Perhaps the simplest solution for evading antibody responses is the adoption of an intracellular life style, as is done by Leishmania, Trypanosoma cruzi, Toxoplasma gondii and the liver and blood stages of malaria. Another major strategy—antigenic variation—protects extracellular protozoa such as African trypanosomes2 and Giardia3 and even malaria parasites4, which express their antigens on the surface of infected erythrocytes, from immune recognition. In addition, there is abundant evidence that protozoan infections actively regulate adaptive T cell responses, resulting in suppressed effector functions 1. A striking example of this phenomenon is the recent demonstration that Leishmania major actively induces interleukin 10 (IL-10)–producing CD25+ T regulatory cells to prevent complete clearance of the parasite5. Although parasitic protozoa have provided some of the best studied paradigms of evasion of antibody- and T cell–mediated immunity by pathogens, a series of equally important adaptations occur during the initial establishment of infection, when parasitic invaders confront the innate immune system. The innate host defenses that the major blood and tissue protozoan pathogens of humans must penetrate, modify or avoid vary according to their mode of entry and the primary host tissue encountered (Table 1). These innate defenses include the epithelial barrier of the skin, the alternative complement cascade and other lytic serum components, lysosomal hydrolases and toxic oxygen metabolites of mononuclear phagocytes and the antigen-presentation and immunoregulatory functions of dendritic cells (DCs), which provide a crucial link with the adaptive immune response6. That protozoa have evolved specific mechanisms to evade these defenses is dramatically shown by the rapid destruction of preinfective vector-derived life cycle stages when artificially inoculated into mammalian hosts. These developmental adaptations, which enable blood and tissue protozoa to "run the gauntlet" of the vertebrate innate response, are the focus of our review. High resolution image and legend (31K) humans Table 1. The major protozoan pathogens of Evasion of humoral innate defenses Innate resistance to protozoa is mediated, in part, by pre-existing soluble factors that can potentially recognize and destroy invading parasites or target them for killing by effector cells. The alternative pathway of complement activation provides a first line of defense against extracellular parasites that must be subverted for infection to proceed. For example, whereas the epimastigote stage of T. cruzi found in insect vectors is susceptible to the alternative complement pathway, infective metacyclic and blood-stream trypomastigotes are resistant7. In this case, evasion of complement appears to be due to trypomastigote expression of a 160-kD glycoprotein (gp160), which is a homolog of the host complement-regulatory protein decay-accelerating factor (DAF)8. Like DAF, gp160 can bind to C3b and C4b and inhibit the uptake of subsequent members of the complement cascade, thus preventing convertase formation and lysis of the parasite. Importantly, whereas complementsensitive epimastigotes fail to express gp160, epimastigotes transfected with gp160 are resistant to complement-mediated lysis9. Another interesting strategy is deployed by Leishmania species, for which the infective insect stage is transiently exposed to potentially lethal serum components after inoculation by pool-feeding phlebotomine vectors. Leishmania evade complement-mediated lysis while using complement activation as a mechanism for targeting host cells. When insect-stage procyclic promastigotes develop into infective metacyclic forms, their membrane is altered to prevent insertion of the lytic C5b-C9 membrane attack complex (MAC)10. This correlates with their expression of a modified surface lipophosphoglycan (LPG) that is approximately twice as long as the form on procyclic promastigotes, and which may act as a barrier for insertion of the MAC into the parasite surface membrane11. A further developmental change that occurs during generation of metacyclics is enhanced synthesis of the surface proteinase gp63, which can cleave C3b to the inactive iC3b form, thus preventing deposition of the lytic C5b-C9 complex12. However, iC3b also opsonizes the parasites for phagocytosis through the complement receptors CR3 and CR1, thereby targeting the parasite to the macrophage, its host cell of choice13. Conclusive evidence that LPG and gp63 are virulence factors has emerged from studies with L. major null-mutants that do not express these molecules; in each case these mutants were highly sensitive to complement-mediated lysis and showed reduced virulence in BALB/c mice14, 15. In addition to complement, protozoa must evade other soluble mediators of innate immunity in order to establish a foothold in the host. Perhaps the best studied are the primate-specific trypanosome lysis factors (TLFs)16, which contribute to the resistance of humans to infection with Trypanosoma brucei, an important pathogen of livestock. Biochemical analysis of the activity present in human serum revealed that high-density lipoproteins are part of the substance that mediates cytolysis of the parasite, and initial studies demonstrated that this complex, TLF1, is composed of several common apolipoproteins as well as a haptoglobin-related protein (Hpr)17, 18. A second cytolytic complex, TLF2, has also been identified that shares many of the components of TLF1 but contains a distinct immunoglobulin M component and a lower lipid content19. Although the final lytic event has not been delineated, TLF must undergo receptor-mediated uptake and enter an intracellular acidic compartment for cytotoxicity to occur18. Whereas T. brucei is sensitive to TLF-mediated killing, the species that infect humans—T. brucei gambiense and T. brucei rhodesiense—are both refractory. This resistance is associated with a block in TLF endocytosis and retention of the complex in a structure known as the flagellar pocket16. The refractoriness of certain T. brucei rhodensiese strains to lysis also correlates with the expression of a serum resistance–associated (SRA) gene that is equivalent to truncated variant surface glycoprotein20. Importantly, transfection of SRA from T. brucei rhodesiense into T. brucei confers resistance to lysis by human serum; this argues that its expression may have been a critical step in the adaptation of an ancestral parasite for infection of primates21. Interestingly, antibodies raised against a homolog of SRA localize to the flagellar pocket, suggesting a possible role for the protein in TLF retention22. Remodeling host-cell compartments Parasitic protozoa that are adapted to an intracellular lifestyle must resist the antimicrobial mechanisms that can be induced in phagocytic and even nonphagoctyic host cells. Insofar as the acidified hydrolytic environment of host cell lysosomes represents the heart of the defensive machinery of many nucleated cells, the ability of acid-labile protease-sensitive parasites to avoid or modify this compartment is likely to be one key to their survival (Fig. 1). T. gondii resides in a phagosome that restricts its fusion with host endosomes and lysosomes. Toxoplasma actively penetrates both phagocytic and nonphagocytic cells, propelled by an actin-myosin–dependent gliding motility23. In the process, it establishes a nonfusigenic compartment, called the parasitophorous vacuole (PV), that lacks integral membrane proteins of host cell origin, but is extensively modified by secreted parasite proteins24, 25. This remodeling seems to be crucial to the inhibition of PV acidification and lysosome fusion, as these events proceed normally after uptake of dead or opsonized parasites, which are internalized via classical receptor-mediated phagocytosis. High resolution image and legend (64K) Figure 1. Remodeling of macrophage intracellular compartments by parasitic protozoa. T. cruzi trypomastigotes enter the macrophage by inducing the recruitment of lysosomes to the plasma membrane; they only transiently reside in the parasitophorous vacuole before escape into the cytoplasm via secretion of a pore-forming molecule, termed Tc-TOX (yellow). T. gondii tachyzoites actively invade the cell and remodel a parasitophorous vacuole membrane (blue) that contains secreted parasite proteins but excludes host proteins that would normally promote phagosome maturation, thereby preventing lysosome fusion. Leishmania metacyclic promastigotes are taken up by receptor-mediated phagocytosis; phagosome maturation may be transiently inhibited by LPG (green), which becomes incorporated into the phagosome membrane. The replicating amastigote stage ultimately resides within a phagolysosome where they survive via production of cell-surface and secreted glycoconjugates, including GIPLS and proteophosphoglycan (PPG) (green). T. cruzi trypomastigotes also actively invade mammalian cells but, unlike T. gondii, their cell entry is dependent not on the propulsive properties of the parasite, but on their ability to subvert a Ca 2+regulated lysosomal exocytic pathway23. The membrane of the parasitophorous vacuole is, therefore, derived from the membrane of lysosomes, and the vacuole itself remains acidic and potentially fusigenic with other lysosomes. T. cruzi growth and development cannot be sustained within this compartment; it depends instead on the ability of the parasite to escape rapidly into the cytosol. Exit from the vacuole is mediated by a parasite-secreted molecule, Tc-TOX, which has membrane pore-forming activity at acidic pH and is facilitated by a trans-sialidase present on the trypomastigote surface26, 27. Interestingly, T. cruzi is unable to invade cells lacking transforming growth factor- (TGF- ) receptors I or II28; this suggests that triggering this signaling pathway, perhaps by some parasite TNF- homolog, might be needed to deactivate host cells during the early stages of infection. Leishmania parasites lack the machinery necessary for active invasion and are confined instead to professional phagocytes, mainly macrophages, with some exceptions (for example, fibroblasts, DCs and neutrophils)29. The uptake of Leishmania by macrophages proceeds via conventional receptormediated phagocytosis that involves a diversity of opsonic or pattern-recognition receptors (for example, CR3, CR1 and mannose fucose receptors)30 that are used depending on the species and stage of parasite and the presence or absence of fresh serum. Leishmania metacyclic promastigotes and amastigotes do not seem to remodel the phagosome in a major way because it rapidly fuses (within 30 min) with late endosomes or lysosomes 31, generating a parasitophorous vacuole that maintains an acidic pH and hydrolytic activity. Using macrophage cell lines and unselected promastigotes, researchers have shown that LPG-repeating units can transiently inhibit phagosome maturation and that this delay may be necessary to allow sufficient time for promastigotes to differentiate into more hydrolase-resistant amastigotes32, 33. These findings are consistent with the attenuation of L. major promastigote survival within macrophages after infection with targeted null mutants lacking LPG14. The general significance of the transient inhibition of phagosome-endosome fusion has been questioned, however, for two reasons. First, Leishmania mexicana promastigote mutants deficient in LPG or other phosphoglycan-containing molecules survive equally well as wild-type organisms within macrophages34, and second, there is an absence of direct data to indicate that mature promastigotes are any less hydrolase-resistant than amastigotes. That amastigotes are remarkably robust is clear: the parasite does not escape from the vacuole, but manages to withstand the low pH and onslaught of acid hydrolases, presumably by producing an abundance of cell surface and secreted glyconjugates that protect the cell from proteolytic damage. As the L. mexicana phosphoglycan–deficient mutants were able to survive normally, it is likely that this species displays different or redundant virulence determinants. It is possible, for example, that the huge parasitophorous vacuole that is formed after infection with parasites of the L. mexicana complex might effectively dilute the hydrolases to which the parasite is exposed within the vacuole and obviate the need for certain surface or secreted glycans. Inhibition of host cell signaling pathways Macrophages possess primary defense mechanisms—including activation of macrophage oxidative metabolism and synthesis and release of arachidonic acid metabolites—that are induced by the attachment and engulfment of microbial agents. The major source of reactive oxygen intermediates (ROIs) in macrophages is the NADPH oxidase, a multicomponent enzyme that catalyzes the transfer of electrons from NADPH to molecular oxygen, resulting in the production of superoxide and hydrogen peroxide35. It is generally believed that activation of protein kinase C (PKC) and protein tyrosine kinases (PTKs) are the two critical events involved in regulating phagocyte functions in response to a variety of extracellular stimuli. In vitro studies involving human cells have shown that macrophage functions, including phagocytosis and ROI generation, are severely impaired after uptake of an insoluble degraded host hemoglobin, called hemozoin, generated during blood-stage malarial infection36 (Fig. 2). These dysfunctions were attributed to inhibition of PKC translocation and activation in the hemozoin-loaded cells37. Early studies suggested that Leishmania parasites also avoid triggering the oxidative burst by actively inhibiting PKC activation in macrophages38, 39. Inhibition of PKC-mediated protein phosphorylation was observed with purified LPG40, which might act either as competitive inhibitor of the PKC activator DAG and/or by altering the physical properties of the bilayer to inhibit PKC membrane translocation. These findings are again consistent with impaired intracellular survival of the L. major LPG–deficient mutant compared to the mutant in which LPG expression was restored, although effects on specific signaling events were not directly compared14. High resolution image and legend (75K) pathways. Figure 2. Inhibition of macrophage signaling Inhibition of pathways by malaria-generated hemozoin or infection with Leishmania or T. gondii. Hemozoin and Leishmania impair the oxidative burst associated with phagocytosis by inhibiting the PKC activation required for assembly of the NADPH oxidase complex in its active form. Leishmania parasites also inhibit IFN- – or CD40L-induced PTK-dependent signaling involved in IL-12 production by activation of the cellular phosphatase SHP-1 that inhibits Jak2 and STAT1 phosphorylation (P). Toxoplasma inhibits LPS-induced cytokine responses by inhibiting nuclear translocation of NF- B and possibly phosphorylated STAT1. In addition to their innate microbicidal responses, macrophages can initiate the host activation cascade by presenting antigens and costimulatory molecules and by providing regulatory cytokines to T cells. A number of studies suggest mechanisms by which intracellular protozoa can interfere with the immune-initiation functions of macrophages. One of the more consistent and striking dysfunctions observed in macrophages infected with protozoan parasites is their inability to produce IL-12, which—as the main physiological inducer of interferon- (IFN- ) and T helper type 1 (TH1) cell differentiation—is an essential cytokine for the development of acquired resistance to most intracellular pathogens. In addition, excess IL-12 production can lead to severe tissue damage and even mortality, a consequence that may also be detrimental to the parasite in blocking its transmission cycle. It is no wonder then that protozoa have evolved a series of intricate and often redundant mechanisms for regulating IL-12 production by antigen-presenting cells, especially macrophages, which are the major potential source of the cytokine stimulated during early infection. Infectious stages of Leishmania do not merely avoid IL-12 induction, they actively and selectively inhibit it, leaving other pro-inflammatory cytokine or chemokine response pathways relatively intact41. Whereas the glycophosphatidylinositol (GPI) anchors of many parasites are potent macrophage activators, Leishmania LPG and glycoinositol phospholipid (GIPL) inhibit IL-12p40 transcription while failing to suppress TNF gene expression42. Although the receptors and signaling pathways involved in this selective inhibition of IL-12 synthesis have yet to be identified, suppression of NF- B activity does not appear to be involved. Because many IL-12 agonists that are inhibited in Leishmania-infected macrophages—including lipopolysaccharide (LPS), CD40 ligand (CD40L) and especially IFN- —signal primarily through protein-tyrosine kinases, the observation that Janus kinase–signal transducers and activators of transcription (Jak-STAT) signaling pathways are also inhibited in Leishmania-infected cells seems especially relevant to the defective IL-12 response43. Defective phosphorylation of Jak2 is attributed to rapid activation of a cytoplasmic protein tyrosine phosphatase (PTP), SHP-144 (Fig. 2). SHP-1 is necessary for L. major survival insofar as SHP-1–deficient motheaten mice do not produce lesions and their macrophages fail to support infection in vitro45. Because specific ligation of the macrophage receptors CR1 and CR3 also leads to selective inhibition of IL-12 production—which, at least in the case of CR3, is associated with impaired tyrosine phosphorylation of STAT146—it is likely that host cell PTPs are rapidly induced by CR3 ligation during attachment and uptake of serum-exposed C3-opsonized parasites. Given that Jak-STAT signaling pathways are involved in a broad range of macrophage responses, including induction of most pro-inflammatory cytokines, it is difficult to understand how Leishmania infection maintains such a selective effect on IL-12. One possible explanation lies in two observations: first, STAT1 regulates IFN consensus-binding protein (ICSBP) induction and—of the cytokines assayed—only activation of the IL-12p40 promoter appears to require ICSBP, and second, ICSBP knock-out mice (on resistant background) are susceptible to L. major infection47. The involvement of redundant STAT1-independent induction pathways for tumor necrosis factor(TNF- ), IL-1 and inducible nitric oxide synthase (iNOS) has been described (see below) and suggests a basis for the maintenance of these responses in Leishmania-infected cells. A similar mechanism may account for the down-regulation of IL-12p40 gene expression in mouse macrophages after uptake of Plasmodium berghei–infected erythrocytes48, which appear to be selective for IL-12 also and do not appear to be due to impaired NF- B activation. The NF- B family of transcription factors is an evolutionarily conserved group of proteins that are important in the regulation of numerous genes involved in innate and adaptive immunity—including those encoding IL-12, IFN- , TNF- , iNOS and adhesion molecules—as well as those involved in cell proliferation and survival. Studies in T. gondii49, 50 have demonstrated the ability of intracellular protozoa to actively interfere with the NF- B–activation pathway in macrophages. In T. gondii– infected cells, despite rapid I B phosphorylation and degradation, NF- B fails to translocate to the nucleus. In contrast to Leishmania, T. gondii–induced response defects are more generalized and include both IL-12 and TNF- . With the use of virulent strains of T. gondii, the inhibition of NF- B translocation in macrophages and its effect on iNOS expression was shown to be dependent on HS70 expression by the parasite that might impede nuclear transport by competing for access to nuclear pore complexes51. Disrupted nuclear transport of phosphorylated STAT has also been reported for T. gondii–infected macrophages and is thought to inhibit Jak-STAT–dependent MHC class II expression51 (Fig. 2). In addition to being directly suppressed by the parasite within infected cells, macrophage IL-12 production and effector functions can also be held in check by the down-modulatory cytokines IL-10 and TGF- , which can themselves be up-regulated by the parasites or their products. Striking evidence in support of the importance of IL-10–dependent down-regulation of IL-12 production for both host and parasite comes from studies in IL-10–deficient mice infected with either T. gondii or T. cruzi. These animals display dysregulated IL-12 production and die of cytokine-associated tissue damage while showing decreased parasite burdens 52, 53. Similarly, low amounts of active TGFproduction by splenic mononuclear cells are associated with a lethal outcome in rodent malaria, and anti–TGF- treatment transformed a normally resolving infection into a lethal one due to overproduction of pathogenic pro-inflammatory cytokines54. With respect to Leishmania, there is as yet no evidence that deficient production of these anti-inflammatory mediators is responsible for severe clinical outcomes. In contrast, there is ample evidence to suggest that overproduction of these cytokines contribute to uncontrolled parasite growth and nonhealing infections. Both IL-10 and TGF- are produced by murine macrophages after Leishmania infection in vitro, and both promote intramacrophage replication and are important factors for determining in vivo susceptibility55, 56. Finally, parasitic protozoa have evolved strategies to down-modulate signaling pathways leading to host cell apoptosis, thereby prolonging the life of the host cell and their own intracellular survival 57. Induced apoptotic pathways in macrophages infected with either Leishmania donovani promastigotes58 or T. gondii tachyzoites59 were strongly inhibited, perhaps by parasite induced upregulation of Bcl-2 homologs. Interestingly, the pro-apoptotic effects that T. cruzi infection has on both CD4+ and CD8+ T cells indirectly enhanced parasite growth, as the binding of apoptotic lymphocytes to vitronectin receptors on macrophages triggered TGF- production and a burst of parasite replication in infected cells60. Manipulation of DC function To the extent that pathogenic protozoa have learned to condition their initial encounter with the innate immune system as a means of manipulating the subsequent adaptive immune response, it has been important to extend these observations to effects on "professional" antigen-presenting cells. In contrast to primary targets of infection, such as macrophages, DCs may be temporally removed from encounter with parasites or their products and may respond distinctively. For example, whereas IL-12 production is actively suppressed in macrophages infected with L. major or T. gondii, DCs actively produce IL-12p40 in response to the same parasites, both in vitro and in vivo61-65. It should be noted that in each case the parasite infection alone was not sufficient for high IL-12p70 production in vitro and that endogenous agonists, such as CD40L or IFN- , were necessary as costimuli. The different outcomes of protozoan encounter on IL-12 production by macrophages versus DCs is consistent with the delayed, albeit effective, immune response to these infections that is ultimately achieved. Apart from the obvious parasite advantage that delayed onset of immunity brings to bear, suppression of IL-12 production by infected macrophages may again represent a coadaptation that dampens their local activation while protecting the host from cytokine shock. For those parasitic infections associated with T cell control mechanisms that are not simply delayed, but are persistently compromised, impaired maturation and function of appropriate DC subsets might be more expected (Table 2). A particularly interesting example is the inhibition of DC maturation by the malaria parasite Plasmodium falciparum66. Malaria-infected erythrocytes bind to the surface of myeloid DCs in vitro and markedly suppress the normal up-regulation of major histocompatibility complex (MHC) class II molecules, adhesion molecules (for example, ICAM-1) and costimulatory molecules (CD83 and CD86) on the cells after stimulation with LPS. The resulting DCs were severely impaired in their capacity to induce allogenic as well as antigen-specific primary and secondary T cell responses67. The receptors on the surface of DCs mediating this inhibitory effect are CD36 and CD51, and their artificial ligation mimicked the suppression induced by infected erythrocytes68. Interestingly, the same receptors are also involved in the recognition of apoptotic cells and, when incubated with DCs, such cells trigger reduced secretion of IL-12 and increased production of IL-10 by these antigen-presenting cells. The major parasite ligand for CD36 binding in infected erythrocytes is thought to be a conserved domain of P. falciparum erythrocyte membrane protein 1 (PfEMP1), a molecule that undergoes antigenic variation. Thus, a nonadherent parasite line, which does not express PfEMP1 antigens on the red cell surface, failed to inhibit DC maturation. High resolution image and legend (36K) Table 2. Impairment of DC function by parasitic protozoa The relevance of these observations to acute disease is indicated by the finding that the percentage of HLA-DR+ DCs was significantly lower in children with severe or mild malaria compared to healthy controls68. At present there is no direct evidence that impaired DC maturation in children with acute malaria is CD36-mediated or that this event is at all necessary for successful malaria infection. Nevertheless, it is intriguing that infected erythrocytes with high CD36 binding affinity are more frequently observed in isolates from patients with mild as opposed to severe disease 69, 70. It suggests that adhesion of infected erythrocytes to CD36 may suppress the pro-inflammatory response to the parasite, an outcome that would favor both host and parasite. Infection of monocyte-derived human DCs with T. cruzi trypomastigotes also inhibits LPS-induced secretion of pro-inflammatory cytokines—including IL-12 and TNF- —as well as HLA-DR and CD40 up-regulation71. Similar effects were observed when human DCs were exposed to T. cruzi– derived GIPL or to only the ceramide portion of the compound72, although nonphysiological high concentrations of both molecules were used in this in vitro study. With respect to Leishmania, the spontaneous migration of murine splenic DCs or Langerhans cells (LCs) was inhibited by L. major promastigote–conditioned media or L. major LPGs, respectively; this suggested that their ability to transport antigen to or within lymphoid tissue might be impaired during infection73, 74. These effects need to be interpreted, however, in the context of other studies (referred to above). These studies involved infection of murine DCs with viable L. major promastigotes or amastigotes; the infected cells up-regulated MHC class II and costimulatory molecules, produced IL-12p40 and down regulated E-cadherin, which indicated that they were competent for antigen transport and presentation61, 75. Uptake of Leishmania amazonensis amastigotes or metacyclic promastigotes by mouse bone marrow–derived DCs (BMDCs) also induced up-regulated expression of MHC class II, CD40, CD80 and CD86 but, in contrast to L. major, the infected DCs produced IL-4 and no IL-1276. This suggested that the DCs were conditioned by the parasite to prime the pathogenic T H2 cells that dominate the response in vivo. Studies involving yet other Leishmania species suggest a more passive strategy for immune evasion, in which their interactions with antigen-presenting cells appear to proceed in a relatively silent fashion. Uptake of L. mexicana by mouse BMDCs was not sufficient to activate them in vitro, although their maturation response to other stimuli was not impaired77. A similar species restriction in Leishmania-induced DC activation was observed after uptake of metacyclic promastigotes by human myeloid DCs, which were efficiently primed for CD40L-induced ILp70 secretion by L. major, but not by L. tropica or L. donovani78. These observations may be significant because whereas L. major produces self-limiting infections that are controlled in the skin, the other species mentioned are capable of disseminating to the viscera or to other cutaneous sites. Given the well described polymorphisms in the surface and secreted glycan structures that are expressed by different Leishmania species79, it will be interesting to determine whether they account—at least in part—for the differences in innate response patterns and the spectrum of clinical disease. From the above discussion it would appear that parasites carefully regulate the induction of IL-12 from DCs during early infection as a means of determining the character of the adaptive immune response that eventually decides their fate in the host. In addition, there is emerging evidence for the existence of distinct parasite-triggered pathways for regulating DC IL-12 production once it has been initiated. One such mechanism has been identified by studies of IL-12 synthesis by murine splenic DCs after injection of a soluble extract of T. gondii tachyzoites (STAg). The response was short-lived and could not be recalled by a second injection of STAg for a period of 1 week after initial priming80. This "paralysis" of the DC IL-12 response induced by STAg does not require IL-10 but instead appears to depend on the induction of lipoxin A4 (LXA4), a product of arachadonic metabolism81. This eicosonoid appears to function by suppressing CCR5 expression, a chemokine receptor involved in IL-12 stimulation by STAg. The same mechanism of IL-12 suppression is likely to operate during natural infection because T. gondii–infected mice with a defect in an enzyme (5LO) required for LXA4 show excess IL-12 production and, in common with infected IL-10–deficient mice53, succumb to cytokine shock and show decreased parasite burdens (J. Aliberti et al., unpublished data). Taken together, these findings suggest that once IL-12 production by DCs has been induced and TH1 responses initiated, synthesis of the cytokine is actively down-regulated, probably to the joint benefit of both host and parasite. Whether analogous mechanisms suppress the IL-12 responses of DCs to other protozoa remains to be determined. Concluding comments In learning to evade innate host defenses, protozoan parasites appear to have mastered the intricacies of both cell biology and cellular immunology. As such, they can teach us important lessons about both fields of biology as well as their points of intersection. In particular, the strategies used by these pathogens for remodeling cellular compartments offer insights into the dynamics of host membranes and intracellular trafficking. At the same time there is much to be learned about host-signaling pathways and immune-response initiation and polarization by studying the mechanisms used by protozoa to suppress or divert immune responses. Because innate defenses (for example, lysis by complement or TLF) have the potential to block infection, the mechanisms used to evade them are potential "Achilles' heels", and offer newly identified and largely unexploited targets for vaccine and/or pharmacologic intervention. References 1. Pearce, E.J., Scott, P.A. & Sher, A. in Fundamental immunology (ed. Paul, W.) 1271-1294 (Lippincott-Raven, Philadelphia, 1999). 2. Borst, P. et al. Antigenic variation in trypanosomes. Arch. Med. Res. 27, 379-388 (1996). | PubMed | ISI | 3. Nash, T.E. Surface antigenic variation in Giardia lamblia. Mol. Microbiol. 45, 585-590 (2002). | Article | PubMed | ISI | 4. Kyes, S., Horrocks, P. & Newbold, C. Antigenic variation at the infected red cell surface in malaria. Annu. Rev. Microbiol. 55, 673-707 (2001). | Article | PubMed | ISI | 5. Belkaid, Y. et al. The role of interleukin (IL)-10 in the persistence of Leishmania major in the skin after healing and the therapeutic potential of anti-IL-10 receptor antibody for sterile cure. J. Exp. Med. 194, 1497-506 (2001). | Article | PubMed | ISI | 6. Hunter, C. & Sher, A. in Immunology of Infectious Diseases (eds. Kaufman, S., Sher, A. & Ahmed, R.) 111-126 (ASM Press, Washington DC, 2001). 7. Joiner, K.A. Complement evasion by bacteria and parasites. Annu. Rev. Microbiol. 42, 201230 (1988). | Article | PubMed | ISI | 8. Norris, K.A., Bradt, B., Cooper, N.R. & So, M. Characterization of a Trypanosoma cruzi C3 binding protein with functional and genetic similarities to the human complement regulatory protein, decay-accelerating factor. J. Immunol. 147, 2240-2247 (1991). | PubMed | ISI | 9. Norris, K.A. Stable transfection of Trypanosoma cruzi epimastigotes with the trypomastigotespecific complement regulatory protein cDNA confers complement resistance. Infect. Immun. 66, 2460-2465 (1998). | PubMed | ISI | 10. Puentes, S.M., Da Silva, R.P., Sacks, D.L., Hammer, C.H. & Joiner, K.A. Serum resistance of metacyclic stage Leishmania major promastigotes is due to release of C5b-9. J. Immunol. 145, 4311-4316 (1990). | PubMed | ISI | 11. McConville, M.J., Turco, S.J., Ferguson, M.A. & Sacks, D.L. Developmental modification of lipophosphoglycan during the differentiation of Leishmania major promastigotes to an infectious stage. EMBO J. 11, 3593-3600 (1992). | PubMed | ISI | 12. Brittingham, A. et al. Role of the Leishmania surface protease gp63 in complement fixation, cell adhesion, and resistance to complement-mediated lysis. J. Immunol. 155, 3102-3111 (1995). | PubMed | ISI | 13. Mosser, D.M. & Edelson, P.J. The mouse macrophage receptor for C3bi (CR3) is a major mechanism in the phagocytosis of Leishmania promastigotes. J. Immunol. 135, 2785-2789 (1985). | PubMed | ISI | 14. Spath, G.F. et al. Lipophosphoglycan is a virulence factor distinct from related glycoconjugates in the protozoan parasite Leishmania major. Proc. Natl. Acad. Sci. USA 97, 9258-9263 (2000). | Article | PubMed | ISI | 15. Joshi, P.B., Kelly, B.L., Kamhawi, S., Sacks, D.L. & McMaster, W.R. Targeted gene deletion in Leishmania major identifies leishmanolysin (GP63) as a virulence factor. Mol. Biochem. Parasitol. 120, 33-40 (2002). | Article | PubMed | ISI | 16. Raper, J., Portela, M.P., Lugli, E., Frevert, U. & Tomlinson, S. Trypanosome lytic factors: novel mediators of human innate immunity. Curr. Opin. Microbiol. 4, 402-408 (2001). | Article | PubMed | ISI | 17. Hajduk, S.L. et al. Lysis of Trypanosoma brucei by a toxic subspecies of human high density lipoprotein. J. Biol. Chem. 264, 5210-5217 (1989). | PubMed | ISI | 18. Smith, A.B., Esko, J.D. & Hajduk, S.L. Killing of trypanosomes by the human haptoglobinrelated protein. Science 268, 284-286 (1995). | PubMed | ISI | 19. Raper, J., Fung, R., Ghiso, J., Nussenzweig, V. & Tomlinson, S. Characterization of a novel trypanosome lytic factor from human serum. Infect. Immun. 67, 1910-1916 (1999). | PubMed | ISI | 20. De Greef, C. & Hamers, R. The serum resistance-associated (SRA) gene of Trypanosoma brucei rhodesiense encodes a variant surface glycoprotein-like protein. Mol. Biochem. Parasitol. 68, 277-284 (1994). | PubMed | ISI | 21. Xong, H.V. et al. A VSG expression site-associated gene confers resistance to human serum in Trypanosoma rhodesiense. Cell 95, 839-846 (1998). | PubMed | ISI | 22. Milner, J.D. & Hajduk, S.L. Expression and localization of serum resistance associated protein in Trypanosoma brucei rhodesiense. Mol. Biochem. Parasitol. 104, 271-283 (1999). | Article | PubMed | ISI | 23. Sibley, L.D. & Andrews, N.W. Cell invasion by un-palatable parasites. Traffic 1, 100-106 (2000). | Article | PubMed | ISI | 24. Mordue, D.G., Desai, N., Dustin, M. & Sibley, L.D. Invasion by Toxoplasma gondii establishes a moving junction that selectively excludes host cell plasma membrane proteins on the basis of their membrane anchoring. J. Exp. Med. 190, 1783-1792 (1999). | Article | PubMed | ISI | 25. Lingelbach, K. & Joiner, K.A. The parasitophorous vacuole membrane surrounding Plasmodium and Toxoplasma: an unusual compartment in infected cells. J. Cell Sci. 111, 14671475 (1998). | PubMed | ISI | 26. Andrews, N.W., Abrams, C.K., Slatin, S.L. & Griffiths, G.A T. cruzi-secreted protein immunologically related to the complement component C9: evidence for membrane pore-forming activity at low pH. Cell 61, 1277-1287 (1990). | PubMed | ISI | 27. Hall, B.F., Webster, P., Ma, A.K., Joiner, K.A. & Andrews, N.W. Desialylation of lysosomal membrane glycoproteins by Trypanosoma cruzi: a role for the surface neuraminidase in facilitating parasite entry into the host cell cytoplasm. J. Exp. Med. 176, 313-325 (1992). | PubMed | ISI | 28. Ming, M., Ewen, M.E. & Pereira, M.E. Trypanosome invasion of mammalian cells requires activation of the TGF- signaling pathway. Cell 82, 287-296 (1995). | PubMed | ISI | 29. Rittig, M.G. & Bogdan, C. Leishmania-host-cell interaction: complexities and alternative views. Parasitol. Today 16, 292-297 (2000). | Article | PubMed | ISI | 30. Alexander, J. & Russell, D.G. The interaction of Leishmania species with macrophages. Adv. Parasitol. 31, 175-254 (1992). | PubMed | ISI | 31. Courret, N. et al. Biogenesis of Leishmania-harbouring parasitophorous vacuoles following phagocytosis of the metacyclic promastigote or amastigote stages of the parasites. J. Cell Sci. 115, 2303-2316 (2002). | PubMed | ISI | 32. Desjardins, M. & Descoteaux, A. Inhibition of phagolysosomal biogenesis by the Leishmania lipophosphoglycan. J. Exp. Med. 185, 2061-2068 (1997). | Article | PubMed | ISI | 33. Dermine, J.F., Scianimanico, S., Prive, C., Descoteaux, A. & Desjardins, M. Leishmania promastigotes require lipophosphoglycan to actively modulate the fusion properties of phagosomes at an early step of phagocytosis. Cell Microbiol. 2, 115-126 (2000). | Article | PubMed | ISI | 34. Ilg, T., Demar, M. & Harbecke, D. Phosphoglycan repeat-deficient Leishmania mexicana parasites remain infectious to macrophages and mice. J. Biol. Chem. 276, 4988-4997 (2001). | Article | PubMed | ISI | 35. Nathan, C. & Shiloh, M.U. Reactive oxygen and nitrogen intermediates in the relationship between mammalian hosts and microbial pathogens. Proc. Natl. Acad. Sci. USA 97, 8841-8848 (2000). | Article | PubMed | ISI | 36. Schwarzer, E. et al. Impairment of macrophage functions after ingestion of Plasmodium falciparum-infected erythrocytes or isolated malarial pigment. J. Exp. Med. 176, 1033-1041 (1992). | PubMed | ISI | 37. Schwarzer, E., Turrini, F., Giribaldi, G., Cappadoro, M. & Arese, P. Phagocytosis of P. falciparum malarial pigment hemozoin by human monocytes inactivates monocyte protein kinase C. Biochim. Biophys. Acta 1181, 51-54 (1993). | PubMed | ISI | 38. Moore, K.J., Labrecque, S. & Matlashewski, G. Alteration of Leishmania donovani infection levels by selective impairment of macrophage signal transduction. J. Immunol. 150, 4457-4465 (1993). | PubMed | ISI | 39. Olivier, M., Brownsey, R.W. & Reiner, N.E. Defective stimulus-response coupling in human monocytes infected with Leishmania donovani is associated with altered activation and translocation of protein kinase C. Proc. Natl. Acad. Sci. USA 89, 7481-7485 (1992). | PubMed | ISI | 40. Descoteaux, A., Matlashewski, G. & Turco, S.J. Inhibition of macrophage protein kinase Cmediated protein phosphorylation by Leishmania donovani lipophosphoglycan. J. Immunol. 149, 3008-3015 (1992). | PubMed | ISI | 41. McDowell, M.A. & Sacks, D.L. Inhibition of host cell signal transduction by Leishmania: observations relevant to the selective impairment of IL-12 responses. Curr. Opin. Microbiol. 2, 438443 (1999). | Article | PubMed | ISI | 42. Piedrafita, D. et al. Regulation of macrophage IL-12 synthesis by Leishmania phosphoglycans. Eur. J. Immunol. 29, 235-244 (1999). | Article | PubMed | ISI | 43. Nandan, D. & Reiner, N.E. Attenuation of interferon-induced tyrosine phosphorylation in mononuclear phagocytes infected with Leishmania donovani: selective inhibition of signaling through Janus kinases and Stat1. Infect. Immun. 63, 4495-4500 (1995). | PubMed | ISI | 44. Blanchette, J., Racette, N., Faure, R., Siminovitch, K.A. & Olivier, M. Leishmania-induced increases in activation of macrophage SHP-1 tyrosine phosphatase are associated with impaired IFN- -triggered Jak2 activation. Eur J. Immunol. 29, 3737-3744 (1999). | Article | PubMed | ISI | 45. Forget, G. et al. Role of host phosphotyrosine phosphatase SHP-1 in the development of murine Leishmaniasis. Eur. J. Immunol. 31, 3185-3196 (2001). | Article | PubMed | ISI | 46. Marth, T. & Kelsall, B.L. Regulation of interleukin-12 by complement receptor 3 signaling. J. Exp. Med. 185, 1987-1995 (1997). | Article | PubMed | ISI | 47. Giese, N.A. et al. Interferon (IFN) consensus sequence-binding protein, a transcription factor of the IFN regulatory factor family, regulates immune responses in vivo through control of interleukin 12 expression. J. Exp. Med. 186, 1535-1546 (1997). | Article | PubMed | ISI | 48. Xu, X. et al. Down-regulation of IL-12 p40 gene in Plasmodium berghei-infected mice. J. Immunol. 167, 235-241 (2001). | PubMed | ISI | 49. Butcher, B.A., Kim, L., Johnson, P.F. & Denkers, E.Y. Toxoplasma gondii tachyzoites inhibit proinflammatory cytokine induction in infected macrophages by preventing nuclear translocation of the transcription factor NF- B. J. Immunol. 167, 2193-2201 (2001). | PubMed | ISI | 50. Shapira, S., Speirs, K., Gerstein, A., Caamano, J. & Hunter, C.A. Suppression of NF- B activation by infection with Toxoplasma gondii. J. Infect. Dis. 185 (Suppl.) 66-72 (2002). 51. Dobbin, A., Smith, N.C. & Jonson, A.M. Heat shock protein 70 is a potential virulence factor in murine Toxoplasma infection via immunomodulation of host NF- B and nitric oxide. J. Immunol. 169, 958-965 (2002). | PubMed | 52. Neyer, L.E. et al. Role of interleukin-10 in regulation of T-cell-dependent and T-cellindependent mechanisms of resistance to Toxoplasma gondii. Infect. Immun. 65, 1675-1682 (1997). | PubMed | ISI | 53. Gazzinelli, R.T. et al. In the absence of endogenous IL-10, mice acutely infected with Toxoplasma gondii succumb to a lethal immune response dependent on CD4+ T cells and accompanied by overproduction of IL-12, IFN- and TNF- . J. Immunol. 157, 798-805 (1996). | PubMed | ISI | 54. Omer, F.M., Kurtzhals, J.A. & Riley, E.M. Maintaining the immunological balance in parasitic infections: a role for TGF- ? Parasitol. Today 16, 18-23 (2000). | Article | PubMed | ISI | 55. Kane, M.M. & Mosser, D.M. The role of IL-10 in promoting disease progression in Leishmaniasis. J. Immunol. 166, 1141-1147 (2001). | PubMed | ISI | 56. Barral, A. et al. Transforming growth factor as a virulence mechanism for Leishmania braziliensis. Proc. Natl. Acad. Sci. USA 90, 3442-3446 (1993). | PubMed | ISI | 57. Luder, C.G., Gross, U. & Lopes, M.F. Intracellular protozoan parasites and apoptosis: diverse strategies to modulate parasite-host interactions. Trends Parasitol. 17, 480-486 (2001). | Article | PubMed | ISI | 58. Moore, K.J., Turco, S.J. & Matlashewski, G. Leishmania donovani infection enhances macrophage viability in the absence of exogenous growth factor. J. Leukoc. Biol. 55, 91-98 (1994). | PubMed | ISI | 59. Nash, P.B. et al. Toxoplasma gondii-infected cells are resistant to multiple inducers of apoptosis. J. Immunol. 160, 1824-1830 (1998). | PubMed | ISI | 60. Freire-de-Lima, C.G. et al. Uptake of apoptotic cells drives the growth of a pathogenic trypanosome in macrophages. Nature 403, 199-203 (2000). | Article | PubMed | ISI | 61. von Stebut, E., Belkaid, Y., Jakob, T., Sacks, D.L. & Udey, M.C. Uptake of Leishmania major amastigotes results in activation and interleukin 12 release from murine skin-derived dendritic cells: implications for the initiation of anti-Leishmania immunity. J. Exp. Med. 188, 1547-1552 (1998). | Article | PubMed | ISI | 62. Marovich, M.A., McDowell, M.A., Thomas, E.K. & Nutman, T.B. IL-12p70 production by Leishmania major-harboring human dendritic cells is a CD40/CD40 ligand-dependent process. J. Immunol. 164, 5858-5865 (2000). | PubMed | ISI | 63. Gorak, P.M., Engwerda, C.R. & Kaye, P.M. Dendritic cells, but not macrophages, produce IL12 immediately following Leishmania donovani infection. Eur. J. Immunol. 28, 687-695 (1998). | Article | PubMed | ISI | 64. Subauste, C.S. & Wessendarp, M. Human dendritic cells discriminate between viable and killed Toxoplasma gondii tachyzoites: dendritic cell activation after infection with viable parasites results in CD28 and CD40 ligand signaling that controls IL-12-dependent and -independent T cell production of IFN- . J. Immunol. 165, 1498-505 (2000). | PubMed | ISI | 65. Scanga, C.A. et al. Cutting edge: MyD88 is required for resistance to Toxoplasma gondii infection and regulates parasite-induced IL-12 production by dendritic cells. J. Immunol. 168, 59976001 (2002). | PubMed | ISI | 66. Urban, B.C. & Roberts, D.J. Malaria, monocytes, macrophages and myeloid dendritic cells: sticking of infected erythrocytes switches off host cells. Curr. Opin. Immunol. 14, 458-465 (2002). | Article | PubMed | ISI | 67. Urban, B.C. et al. Plasmodium falciparum-infected erythrocytes modulate the maturation of dendritic cells. Nature 400, 73-77 (1999). | Article | PubMed | ISI | 68. Urban, B.C., Willcox, N. & Roberts, D.J. A role for CD36 in the regulation of dendritic cell function. Proc. Natl. Acad. Sci. USA 98, 8750-8755 (2001). | Article | PubMed | ISI | 69. Newbold, C. et al. Receptor-specific adhesion and clinical disease in Plasmodium falciparum. Am. J. Trop. Med. Hyg. 57, 389-398 (1997). | PubMed | ISI | 70. Rogerson, S.J. et al. Cytoadherence characteristics of Plasmodium falciparum-infected erythrocytes from Malawian children with severe and uncomplicated malaria. Am. J. Trop. Med. Hyg. 61, 467-472 (1999). | PubMed | ISI | 71. Van Overtvelt, L. et al. Trypanosoma cruzi infects human dendritic cells and prevents their maturation: inhibition of cytokines, HLA-DR, and costimulatory molecules. Infect. Immun. 67, 40334040 (1999). | PubMed | ISI | 72. Brodskyn, C. et al. Glycoinositolphospholipids from Trypanosoma cruzi interfere with macrophages and dendritic cell responses. Infect. Immun. 70, 3736-3743 (2002). | Article | PubMed | ISI | 73. Jebbari, H., Stagg, A.J., Davidson, R.N. & Knight, S.C. Leishmania major promastigotes inhibit dendritic cell motility in vitro. Infect. Immun. 70, 1023-1026 (2002). | Article | PubMed | ISI | 74. Ponte-Sucre, A., Heise, D. & Moll, H. Leishmania major lipophosphoglycan modulates the phenotype and inhibits migration of murine Langerhans cells. Immunology 104, 462-467 (2001). | Article | PubMed | ISI | 75. Konecny, P. et al. Murine dendritic cells internalize Leishmania major promastigotes, produce IL-12 p40 and stimulate primary T cell proliferation in vitro. Eur. J. Immunol. 29, 1803-1811 (1999). | Article | PubMed | ISI | 76. Qi, H., Popov, V. & Soong, L. Leishmania amazonensis-dendritic cell interactions in vitro and the priming of parasite-specific CD4+ T cells in vivo. J. Immunol. 167, 4534-4542 (2001). | PubMed | ISI | 77. Bennett, C.L., Misslitz, A., Colledge, L., Aebischer, T. & Blackburn, C.C. Silent infection of bone marrow-derived dendritic cells by Leishmania mexicana amastigotes. Eur. J. Immunol. 31, 876-883 (2001). | Article | PubMed | ISI | 78. McDowell, M.A., Marovich, M., Lira, R., Braun, M. & Sacks, D. Leishmania priming of human dendritic cells for CD40 ligand-induced interleukin-12p70 secretion is strain and species dependent. Infect. Immun. 70, 3994-4001 (2002). | Article | PubMed | ISI | 79. Turco, S.J., Spath, G.F. & Beverley, S.M. Is lipophosphoglycan a virulence factor? A surprising diversity between Leishmania species. Trends Parasitol. 17, 223-226 (2001). | Article | PubMed | ISI | 80. Reis e Sousa, C. et al. Paralysis of dendritic cell IL-12 production by microbial products prevents infection-induced immunopathology. Immunity 11, 637-647 (1999). | PubMed | 81. Aliberti, J., Hieny, S., Reis e Sousa, C., Serhan, C.N. & Sher, A. Lipoxin-mediated inhibition of IL-12 production by DCs: a mechanism for regulation of microbial immunity. Nature Immunol. 3, 76-82 (2002). | Article | PubMed | ISI | Figure 1. Remodeling of macrophage intracellular compartments by parasitic protozoa. T. cruzi trypomastigotes enter the macrophage by inducing the recruitment of lysosomes to the plasma membrane; they only transiently reside in the parasitophorous vacuole before escape into the cytoplasm via secretion of a pore-forming molecule, termed Tc-TOX (yellow). T. gondii tachyzoites actively invade the cell and remodel a parasitophorous vacuole membrane (blue) that contains secreted parasite proteins but excludes host proteins that would normally promote phagosome maturation, thereby preventing lysosome fusion. Leishmania metacyclic promastigotes are taken up by receptor-mediated phagocytosis; phagosome maturation may be transiently inhibited by LPG (green), which becomes incorporated into the phagosome membrane. The replicating amastigote stage ultimately resides within a phagolysosome where they survive via production of cell-surface and secreted glycoconjugates, including GIPLS and proteophosphoglycan (PPG) (green). Figure 2. Inhibition of macrophage signaling pathways. Inhibition of pathways by malaria-generated hemozoin or infection with Leishmania or T. gondii. Hemozoin and Leishmania impair the oxidative burst associated with phagocytosis by inhibiting the PKC activation required for assembly of the NADPH oxidase complex in its active form. Leishmania parasites also inhibit IFN- – or CD40Linduced PTK-dependent signaling involved in IL-12 production by activation of the cellular phosphatase SHP-1 that inhibits Jak2 and STAT1 phosphorylation (P). Toxoplasma inhibits LPS-induced cytokine responses by inhibiting nuclear translocation of NF- B and possibly phosphorylated STAT1. Table 2. Impairment of DC function by parasitic protozoa To return to the article, close this browser window. To toggle between the