FTIR_plant_soil_hopper
advertisement

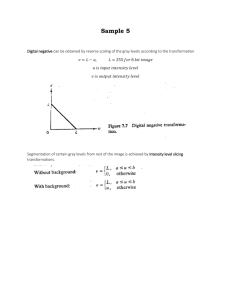
Plant FTIR Results Plant FTIR 1.10 a 1.00 b 0.90 c 0.80 absorbance f d g e h 0.70 i j 0.60 k l 0.50 m 0.40 0.30 3900.00 3400.00 PRP-8 LEAF 2900.00 2400.00 PRP-8 VEIN 1900.00 PRP-8 ROOT 1400.00 PRP-11 GREEN 900.00 PRP-12 BROWN 0.20 400.00 Table 1 Interpretation of Plant FTIR profile Peak Wavenumber Description broad, strong intensity Possible functional groups O-H Stretching(3350±150);NH2 Stretching(3300±100) CH3 Stretching(2962±10),CH2 Stretching(2926±10), C-H Stretching(2961) -COOH Overtone (2500±300),-COOH Stretching (3000±500) a 3370-3570 b 2900-2960 sharp peak c 2850-2870 sharp peak d 2352&2364 e 1727 f 1608-1670 medium intensity, sharp with saw tooth g 1465 medium intensity, sharp with saw tooth CH2 bend(1465) h 1383 medium intensity, sharp CH3 bend (1375±10) i 1180 medium intensity j 1000-1100 weak k 904 l 671 m 435 medium intensity, sharp with two peaks medium intensity, sharp with saw tooth weak medium intensity, sharp medium intensity with saw tooth CO2 C=O (1727) aldehyde H2O bending(1650±30);C=C Stetching (1640±20,1670±10); Aromatic Ring Stretching (1950±10); Comments Since it's broad and strong intensity, this is due to O-H bond rather than NH2 bond This part is generally accept as acid dimer or group as -COOH in cellulose study Source Wang et al 2006 Wang et al 2006 the dip between two peaks could be well explained by the CO2 For PRP-8 root, this peak is more obvious which could be explained by less cellulose oxidation noise Saw tooth are highly possible products of cellulose (H-C-OH) partial oxidation (16171745). Meanwhile, same peak in the leaf sample (PRP-8) are smoother. Water should also be considered as a possible results Possible part of cellulose, also more evident for stem and root samples instead of PRP-8 leaf sample This peak is most obvious in the leaf sample. The saw tooth in the rest samples could possible be both xyloglucan(1371) and cellulose(1362) J.Løjewska et al 2005 M.Kačuráková et al 2002 M.Kačuráková et al 2002 Most likely CH3 bending. C-O Stretching, CCstretching(1115,1100,1075,1060,1042,1030,10 15) =CH2 bending(909) Aromatic bending(690±20) S-S Stretch (430-500) Literature ascribes small peaks in this range as cellulose, pectin, xyloglucan This peak could only be explained as a standard cellulose peak according to literatures (894), also it is most obvious on the brown sample Most likely C6, very sharp peak is found at Leaf Sample Most possible Aryl disulfides, sharp peak is found at the green sample M.Kačuráková et al 2002 Zhong et al 2000 Wang et al 2006 Coates 2000 Soil FTIR Results Soil FTIR - Norm alized 1 0.9 b c n 0.7 l h d e f 0.6 m 0.5 g j 0.4 0.3 k 0.2 0.1 0 4000 3500 15 3000 2500 16 2000 Wavelength (cm-1) 17 1500 1000 18 0 500 7 Absorbance a 0.8 i Table 2 Interpretation of Soil FTIR profile Peak Wavenumber Description Possible functional groups O-H stretching region (3800-3400 for clay mineral), O-H stretching region (3800-3400 for clay mineral) a 3700 sharp peak b 3622 sharp peak c 3464 broad, strong intensity O-H , N-H c 2935 tiny broad C-H (3150-2850) e 2372 & 2347 f g 2011 1901 tiny saw tooth peaks weak weak h 1655 medium intensity i 1450 & 1400 weak j 1099-1034 k 800 l 696 m 540 n 472 sharp & strongest peak medium intensity, saw tooth medium intensity, sharp medium intensity, sharp strong intensity, sharp Comments Source Stuart Bands due to Si-O-O-OH vibration. Since it's broad and strong intensity, this is due to O-H bond rather than N-H bond. The peak is below 3000, so it is an aliphatic CH vibration. Medium intensity absorptions at 1450 and 1375 cm-1 will indicate -CH3 bend stretching. CO2 bend CO2 bend C=C (1680-1600 for aromatic and alkenes); C=O vibrations (1680-1630 for amide), C=N (1690-1630) and also of N-H bend (1650-1475); water absorbance (~ 1640) Some soil literature assigned this to C=O vibrations of carboxylates and aromatic. Vibrations involving most polar bonds, such as C=O and O-H have the most intense IR absorptions. This peak has medium intensity and most likely due to water. C-H, alkenes, -CH3 (bend, 1450 and 1375), -CH2 (bend, at 1465), Most likely CH3 bending. Si-O vibration of clay minerals Consistent with FTIR spectra of soil in the literature NH2 wagging and twisting, =C-H bend, alkenes N-C=O bend for secondary amides C-C=O bend for secondary amides, SiO3-2 Intense absorption at 460-475 corresponds to SiO3-2 vibration. In the literature, bands at 800,780,650,590,530 and 470 are attributed to inorganic materials, such as clay and quartz minerals. Gerzabek et al 2006, Lojewska et al. 2005 Gerzabeck et al. 2006 Stuart Grasshopper Soil FTIR Results Grasshopper FTIR 0.65 a 0.60 e b 0.55 f absorbance c 0.50 g h d 0.45 i j k 0.40 l 0.35 m 0.30 0.25 3900.00 3400.00 2900.00 2400.00 1900.00 wavenumber 1400.00 900.00 0.20 400.00 Table 3 Interpretation of Grasshopper FTIR profile Peak Wavenumber a 3430 b 2967 c 2933 d 2346&2360 Description broad, strong intensity medium intensity medium intensity medium intensity with two peaks sharp Possible functional groups O-H Stretching(3350±150);NH2 Stretching(3300±100) CH3 Stretching(2962±10) CH2 Stretching(2926±10) CO2 H2O bending(1650±30);N-H bending(15901650), =N-H bending,(1550-1660),C=C Stretching(1640±20),β sheet e 1650 f 1556 g 1471 h 1407 i 1251 j 1182 weak CH3 bending k 1097 weak C-O Stretching; C-C Stretching(1150±25, 1125±25) l 682 m 418 medium intensity with saw tooth medium intensity with saw tooth medium intensity medium intensity with saw tooth medium intensity, sharp medium intensity with saw tooth Aromatic nitro compounds (1485-1555); Aliphatic nitro compounds(1540-1560); Carboxylate acid salt(1550-1610) Comments Since it's broad and strong intensity, this is due to O-H bond rather than NH2 bond Hydrocarbons have major absorption bands at 2800–3000 Hydrocarbons have major absorption bands at 2800–3000 Source Gibbs 2002 Gibbs 2003 the dip between two peaks could be well explained by the CO2 Possible the influence of water , if water influence is not considered, this peak should be Amide I (1600-1700), literature points out 1655 should be Amide I which is mainly composition of Chitin Literature shows no peaks around 15001600 is ascribed to Chitin Iconomidou et al 2001 Iconomidou et al 2002; Coates 2000 Methylene C-H bending(1445-1485);Aromatic ring stretching(1450-1510) Ammonium ion(1390-1430) not a peak of Amine or Amide C-N stretching(1250-1340);P=O stretching(1250-1350) this peak should be Amide III (1230-1320), also mainly composition of Chitin Coates 2000 Same peak is also found in plant samples, so here we would not ascribe it into Amine and Amide group Coates 2000 Aromatic bending(690±20) Most likely C6,same peak found in plant samples Coates 2000 S-S Stretching (430-500) Most possible Aryl disulfides, same peak found in plant samples Coates 2000