Supplementary Methods - Word file (104 KB )
advertisement

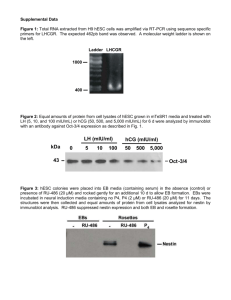
Supplementary method Human embryonic stem cell cultures: The hESC lines, H1 and H9 1, were obtained from WiCell Research Institute (Madison, WI). HESCs (passages 29-60) were grown on mouse embryonic fibroblasts (MEFs), which were inactivated with irradiation (60 Gy) or mitomycin C (1 mg/ml), in hESC medium containing DMEM/F12, 20% knockout serum replacement (KSR), 2 mM Lglutamine, 0.1 mM nonessential amino acid (all from Invitrogen), 0.1 mM mercaptoethanol (Sigma), and 4 ng/ml human FGF-2 (R&D Systems). The feeder-free culture on Matrigel (BD Biosciences) with MEF-conditioned medium (CM) was performed as described 2. MEF-CM was filtered through a 0.22 m sterile membrane and stored at >–20oC. To maintain hESCs in a long-term undifferentiated status, hESCs were cultured at ~500 hESC colonies per 100-mm dish at 37°C and 5% CO2. We found that the plating density of MEFs had a significant effect on the differentiation of hESCs into CD34+ cells. We identified the optimal MEFs plating density to be ~1x10 4 cells/cm2 on a gelatin-coated dish (5 to 6 x 105 cells/100-mm). MEFs were used up to passage 3 for hESC cultures. MEF conditioned medium (MEF-CM) with 4 ng/ml of human FGF-2 (hFGF-2, R&D Systems or PeproTech) was used on the first day of hESC passage. MEF-CM and hESC medium (1:1) was used on the second day of hESC culture in the presence of 4 ng/ml of hFGF-2. From the third day onward, the culture medium was changed daily with hESC media. Differentiation of hESCs: To induce 3D embryoid bodies (EBs), the hESC colonies were treated with 2 mg/ml dispase for 15 minutes at 37oC to loosen colonies and the colonies were transferred into ultra low-attachment plates (Corning Incorporated) for EB formation. EBs were differentiated in hESC differentiation medium consisting of IMDM, 15% defined-FBS (Hyclone), 450 M monothioglycerol (Sigma), 2 mM L-glutamine, 50 U/ml penicillin, 50 g/ml streptomycin, and 0.1 mM non-essential amino acid for 10 days with half of the medium changed every 2-3 days. 1 For 2D monolayer differentiation, undifferentiated hESCs were cultured at a higher density (~1200 hESC colonies/100-mm dish) on MEFs for 7 days. At day 0 of differentiation, the culture medium was changed to hESC differentiation medium and the medium was changed every 2-3 days for 10 days. Three batches of defined-FBS from Hyclone were tested (Lot# ARA25685, ARA25663, and AQL25247). Similar CD34 and CD31 expression was obtained from these 3 batches of defined-FBS. For serum-free differentiation, 15% defined-FBS in hESC differentiation medium was replaced with either 20% BIT 9500 (StemCell Tech) or KSR, and supplemented with VEGF (50 ng/ml), bFGF (50 ng/ml), and BMP-4 (50 ng/ml). Isolation of CD34+ progenitor cells: A recent study demonstrated that hESCs differentiate into cells of hematopoietic and endothelial lineage at ~ day 10 3. After 10 days of differentiation in either 2D or 3D culture, the single-cell suspensions were made from differentiated hESCs by treatment with 2 mg/ml collagenase B (Roche) for 10 minutes at 37oC. The cells were dissociated by gentle pipetting, and passaged through 70 and 40 m cell strainers (BD Biosciences). To minimize the loss of cells, the strainers were rinsed extensively with PBS/1%FBS or an additional strainer was used. CD34+ cells were isolated from differentiated hESCs by using MACS MicroBeads columns or AutoMACS (Miltenyi Biotec), according to the manufacturer’s instructions. PBS/0.5% BSA (without EDTA) was used as the buffer for cell isolation. In some experiments, a Dead Cell Removal Kit (Miltenyi Biotec) was used to remove dead cells prior to CD34 isolation. AutoMACS was able to give higher number of CD34+cells than manual-selection. As determined by flow cytometry, the purity of isolated CD34+ cells was generally 60-80% at a single column, and > 95% after the second column. Endothelial and hematopoietic differentiation: For expansion and differentiation of endothelial cells, isolated CD34+ cells were seeded on gelatin-coated wells (1.5 to 2x104 cells/cm2) in EGM-2MV medium (Cambrex).The majority of cells (~65%) were adherent. The adherent cells reached to ~90% confluence after 7-10 day culture. We found that the attachment of CD34+ cells was greater on 2 collagen I-coated wells (BD labware) than on gelatin-coated wells. However, gelatin has the advantage of being considerable less expensive than collagen I-coated wells. In some experiments, the CD34+ cells were cultured in hESC differentiation medium containing the endothelial growth factors, hVEGF165 (50ng/ml) and FGF-2 (5 ng/ml )(R&D Systems or PeproTech). After 7-10 days of incubation, the adherent cells were harvested by trypsin-treatment and used for analyses. We noticed that the hES-ECs have low proliferative capacity. Longer culture could result in the outgrowth of other cell types besides endothelial cells. For further expansion and differentiation of hematopoietic cells, isolated CD34+ cells were cultured in 24-well plates (4 to 5 x104 cells/well in 0.5 ml) in hESC differentiation medium containing 15% defined-FBS (Hyclone), 450 M monothioglycerol (Sigma), 2 mM L-glutamine, 50 U/ml penicillin, 50 g/ml streptomycin, and 0.1 mM non-essential amino acid, and in the presence or absence of SCF (100 ng/ml) and Flt-3 ligand (100 ng/ml). In some experiments, the CD34+ cells were cultured in EGM-2MV in the presence of SCF (100 ng/ml) and Flt-3 ligand (100 ng/ml). After 2 to 3 weeks of culture, the suspension cells were analyzed. Flow cytometry and immunostaining: The cells were prepared in PBS containing1% FBS or 0.5% BSA, and were labeled for 15-30 minutes at 4oC with a combination of monoclonal antibodies (mAbs): CD31-PE (clone WM-59), CD34-APC (clone 581), CD45-FITC CD45-FITC (clone HI30) (all from BD PharMingen), and KDR-PE (clone 89106) (R&D system). We noticed that the titrations of the specific antibodies have consequence on FACS results. In general, we used 1:50 dilution for specific antibodies, and 1:200 for IgG control. The samples were analyzed by a FACSCalisbur (Becton Dickson). Data analyses were performed using CellQuest and FlowJo software. For immunostaining, the cells were fixed with methanol for 5 minutes at -20oC or with 4% paraformaldehyde in PBS at room temperature for 15 minutes. The fixed cells were incubated with 4% goat serum for 30 minutes to block nonspecific binding, and stained for 1 hour with the primary antibodies: SSEA-1, SSEA-4, TRA-1-60, and TRA-1-81 (all from Chemicon International Inc), CD31 (Dako), and VE-cadherin (BD Pharmingen), 3 respectively. The cells were then incubated for 30 minutes with either FITC-conjugated rat anti-mouse secondary antibody (BD PharMingen) or Cy3-conjugated donkey antimouse secondary antibody (Jackson ImmunoResearch Laboratories). For the LDL uptake assay, the cells were incubated with 10 ug/ml of diI-acetylated low-density lipoprotein (Dil-LDL, Molecular Probes) for at least 4 hours. After washing twice with PBS, the cells were examined under a fluorescence microscope. Matrigel assay: The assay performed essentially as previously described 4, 5. Twenty four-well plates were coated with 200 μl/well Matrigel matrix (BD Biosciences) at room temperature for more than 30 minutes. The hESC-derived endothelial cells (5 x 104 cells) were trypsinized and replated onto Matrigel plates in differentiation medium at 37C in 5% CO2. The structures were photographed under phase-contrast microscope (Nikon) after 16 hours of incubation. Tissue engineered blood vessels: To generate hES cell-derived endothelial cells, human ES cell-derived CD34+ cells were cultured in either EGM-2 medium or differentiation medium with 50 ng/ml rhVEGF and 5 ng/ml rhFGF-2 for 7 to 10 days. One million endothelial cells and 2x10 5 10T1/2 cells were suspended in 1 ml solution of rat-tail type 1 collagen (1.5 mg/ml) (BD Biosciences, Bedford, MA) and human plasma fibronectin (90 mg/ml) (Sigma) in 25 mM Hepes (Sigma) buffered EGM medium at 4°C. The pH was adjusted to 7.4 by using 1N NaOH (Fisher Science, NJ). The cell suspension was pipetted into 12-well plates (Falcon) and warmed to 37°C for 30 minutes to allow polymerization of collagen. Each solidified gel construct was covered by one ml of warmed EGM medium. After one day of culture in 5% CO2, a skin puncher was applied to create circular disk-shape pieces of the construct (4-mm diameter), and they were implanted into the cranial windows in SCID mice 6, 7. Multiphoton laser-scanning microscopy was used to visualize and quantify the morphological changes of EGFP-expressing hESC-derived endothelial cells. The perfused vessels were highlighted by tail vein injection of 1% 4 tetramethylrhodamine-labeled dextran (MW 2000,000), indicating the formation of functional engineered vessels 6, 7 RT-PCR analysis Total RNAs were isolated using Trizol (Invitrogen). To eliminate DNA contamination, the RNA samples were treated with DNase (Invitrogen) before the reverse transcription (RT) reaction (SuperScript II RNase H- Reverse Transcriptase, Invitrogen). The sequences of oligonucleotide primers used for PCR are listed. Oct-4 (F) AGGGCAAGCGATCAAGCA Oct-4 (R) GGAAAGGGACCGAGGAGTA CD31 (F) CCCAGCCCAGGATTTCTTAT CD31 (R) ACCGCAGGATCATTTGAGTT GATA-2 (F) GACGACAACCACCACCTTATG GATA-2 (R) GACTTAAAGGTGGGAGGTGTC VE-cad (F) CAGCCCAAAGTGTGTGAGAA VE-cad (R) TGTGATGTTGGCCGTGTTAT CD34 (F) GCCATTCAGCAAGACAACAC CD34 (R) AAGGGTTGGGCGTAAGAGAT KDR (F) GGGAAAGCATCGAAGTCTCA KDR (R) CGTCCTCCTTCCTCACTCTG vWF (F) TCGGGCTTCACTTACGTTCT vWF (R) CCTTCACTCGGACACACTCA CD105 (F) GCCAGCATTGTCTCACTTCA CD105 (R) GGCACACTTTGTCTGGATCA Nanog (F) ACTAACATGAGTGTGGATCC Nanog (R) TCATCTTCACACGTCTTCAG -SMA (F) CTGTTCCAGCCATCCTTCAT -SMA (R) CGGCTTCATCGTATTCCTGT EphB4 (F) GCCCATCATCATGTCTGTTTCCA EphB4 (R) CACCAACTACCGCCCTTTTCAC ephrinB2 (F) TGAGTGGGTGCGTGGTAT 5 ephrinB2 (R) GGAGAGGTTGGGGTGATG LYVE-1 (F) TGGAGATGGATTCGTGGTC LYVE-1 (R) GGGCAGGTATTGTAGAGTAAGG Flt-4 (F) AGTTTGTGGAGGGAAAGAATAAGA Flt-4 (R) TGGACAGGTTGAGGCGGTA CD133 (F) AGCACTCTATACCAAAGCGTCAA CD133 (R) CTCCCATACTTCTTAGTTTCCTCAA Tie-2 (F) AGACCAGCACGTTGATGTGA Tie-2 (R) TGGGTTGCTTGACCCTATGT 1. 2. 3. 4. 5. 6. 7. Thomson, J.A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145-1147. (1998). Xu, C. et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol 19, 971-974. (2001). Wang, L. et al. Endothelial and hematopoietic cell fate of human embryonic stem cells originates from primitive endothelium with hemangioblastic properties. Immunity 21, 3141 (2004). Albini, A. et al. Inhibition of angiogenesis and vascular tumor growth by interferonproducing cells: A gene therapy approach. Am J Pathol 156, 1381-1393 (2000). Bompais, H. et al. Human endothelial cells derived from circulating progenitors display specific functional properties compared with mature vessel wall endothelial cells. Blood 103, 2577-2584 (2004). Koike, N. et al. Tissue engineering: creation of long-lasting blood vessels. Nature 428, 138-139 (2004). Yuan, F. et al. Vascular permeability and microcirculation of gliomas and mammary carcinomas transplanted in rat and mouse cranial windows. Cancer Res 54, 4564-4568 (1994). 6