BCH 101 6
advertisement

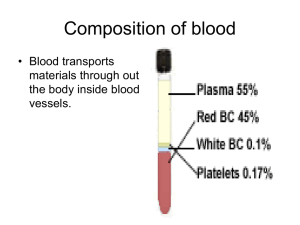
Primary Structure of Proteins The primary structure of peptides and proteins refers to the linear number and order of the amino acids present. The convention for the designation of the order of amino acids is that the N-terminal end (i.e. the end bearing the residue with the free α-amino group) is to the left (and the number 1 amino acid) and the C-terminal end (i.e. the end with the residue containing a free αcarboxyl group) is to the right. Secondary Structure in Proteins The ordered array of amino acids in a protein confer regular conformational forms upon that protein. These conformations constitute the secondary structures of a protein. In general proteins fold into two broad classes of structure termed, globular proteins or fibrous proteins. Globular proteins are compactly folded and coiled, whereas, fibrous proteins are more filamentous or elongated. It is the partial double-bond character of the peptide bond that defines the conformations a polypeptide chain may assume. Within a single protein different regions of the polypeptide chain may assume different conformations determined by the primary sequence of the amino acids. The α-Helix The α-helix is a common secondary structure encountered in proteins of the globular class. The formation of the α-helix is spontaneous and is stabilized by H-bonding between amide nitrogens and carbonyl carbons of peptide bonds spaced four residues apart. This orientation of H-bonding produces a helical coiling of the peptide backbone such that the R-groups lie on the exterior of the helix and perpendicular to its axis. Typical α-Helix Not all amino acids favor the formation of the (α-helix due to steric constraints of the R-groups. Amino acids such as A, D, E, I, L and M favor the formation of α-helices, whereas, G and P favor disruption of the helix. This is particularly true for P since it is a pyrrolidine based imino acid (HN=) whose structure significantly restricts movement about the peptide bond in which it is present, thereby, interfering with extension of the helix. The disruption of the helix is important as it introduces additional folding of the polypeptide backbone to allow the formation of globular proteins. β-Sheets Whereas an α-helix is composed of a single linear array of helically disposed amino acids, β-sheets are composed of 2 or more different regions of stretches of at least 5-10 amino acids. The folding and alignment of stretches of the polypeptide backbone aside one another to form β-sheets is stabilized by H-bonding between amide nitrogens and carbonyl carbons. However, the H-bonding residues are present in adjacently opposed stretches of the polypetide backbone as opposed to a linearly contiguous region of the backbone in the α-helix. β-sheets are said to be pleated. This is due to positioning of the α-carbons of the peptide bond which alternates above and below the plane of the sheet. β-sheets are either parallel or antiparallel. In parallel sheets adjacent peptide chains proceed in the same direction (i.e. the direction of N-terminal to C-terminal ends is the same), whereas, in antiparallel sheets adjacent chains are aligned in opposite directions. β-sheets can be depicted in ball and stick format or as ribbons in certain protein formats. Ball and Stick Representation of a βSheet Ribbon Depiction of βSheet Super-Secondary Structure Some proteins contain an ordered organization of secondary structures that form distinct functional domains or structural motifs. Examples include the helix-turn-helix domain of bacterial proteins that regulate transcription and the leucine zipper, helix-loop-helix and zinc finger domains of eukaryotic transcriptional regulators. These domains are termed super-secondary structures. Tertiary Structure of Proteins Tertiary structure refers to the complete three-dimensional structure of the polypeptide units of a given protein. Included in this description is the spatial relationship of different secondary structures to one another within a polypeptide chain and how these secondary structures themselves fold into the three-dimensional form of the protein. Secondary structures of proteins often constitute distinct domains. Therefore, tertiary structure also describes the relationship of different domains to one another within a protein. The interactions of different domains is governed by several forces: These include hydrogen bonding, hydrophobic interactions, electrostatic interactions and van der Waals forces. Forces Controlling Protein Structure Hydrogen Bonding: Polypeptides contain numerous proton donors and acceptors both in their backbone and in the R-groups of the amino acids. The environment in which proteins are found also contains the ample H-bond donors and acceptors of the water molecule. H-bonding, therefore, occurs not only within and between polypeptide chains but with the surrounding aqueous medium. Hydrophobic Forces: Proteins are composed of amino acids that contain either hydrophilic or hydrophobic R-groups. It is the nature of the interaction of the different Rgroups with the aqueous environment that plays the major role in shaping protein structure. The spontaneous folded state of globular proteins is a reflection of a balance between the opposing energetics of H-bonding between hydrophilic R-groups and the aqueous environment and the repulsion from the aqueous environment by the hydrophobic R-groups. The hydrophobicity of certain amino acid R-groups tends to drive them away from the exterior of proteins and into the interior. This driving force restricts the available conformations into which a protein may fold. Electrostatic Forces: Electrostatic forces are mainly of three types; charge-charge, charge-dipole and dipole-dipole. Typical charge-charge interactions that favor protein folding are those between oppositely charged R-groups such as K or R and D or E. A substantial component of the energy involved in protein folding is chargedipole interactions. This refers to the interaction of ionized R-groups of amino acids with the dipole of the water molecule. The slight dipole moment that exist in the polar R-groups of amino acid also influences their interaction with water. It is, therefore, understandable that the majority of the amino acids found on the exterior surfaces of globular proteins contain charged or polar Rgroups. van der Waals Forces: There are both attractive and repulsive van der Waals forces that control protein folding. Attractive van der Waals forces involve the interactions among induced dipoles that arise from fluctuations in the charge densities that occur between adjacent uncharged non-bonded atoms. Repulsive van der Waals forces involve the interactions that occur when uncharged non-bonded atoms come very close together but do not induce dipoles. The repulsion is the result of the electron-electron repulsion that occurs as two clouds of electrons begin to overlap. Although van der Waals forces are extremely weak, relative to other forces governing conformation, it is the huge number of such interactions that occur in large protein molecules that make them significant to the folding of proteins. Quaternary Structure Many proteins contain 2 or more different polypeptide chains that are held in association by the same non-covalent forces that stabilize the tertiary structures of proteins. Proteins with multiple polypetide chains are oligomeric proteins. The structure formed by monomer-monomer interaction in an oligomeric protein is known as quaternary structure. Oligomeric proteins can be composed of multiple identical polypeptide chains or multiple distinct polypeptide chains. Proteins with identical subunits are termed homo-oligomers. Proteins containing several distinct polypeptide chains are termed hetero-oligomers. Hemoglobin, the oxygen carrying protein of the blood, contains two α and two β subunits arranged with a quaternary structure in the form, α 2β2. Hemoglobin is, therefore, a hetero-oligomeric protein. Structure of Hemoglobin Complex Protein Structures Proteins also are found to be covalently conjugated with carbohydrates. These modifications occur following the synthesis (translation) of proteins and are, therefore, termed post-translational modifications. These forms of modification impart specialized functions upon the resultant proteins. Proteins covalently associated with carbohydrates are termed glycoproteins. Glycoproteins are of two classes, N-linked and O-linked, referring to the site of covalent attachment of the sugar moieties. N-linked sugars are attached to the amide nitrogen of the R-group of asparagine; O-linked sugars are attached to the hydroxyl groups of either serine or threonine and occasionally to the hydroxyl group of the modified amino acid, hydroxylysine. There are extremely important glycoproteins found on the surface of erythrocytes. It is the variability in the composition of the carbohydrate portions of many glycoproteins and glycolipids of erythrocytes that determines blood group specificities. There are at least 100 blood group determinants, most of which are due to carbohydrate differences. The most common blood groups, A, B, and O, are specified by the activity of specific gene products whose activities are to incorporate distinct sugar groups onto RBC membrane glycoshpingolipids as well as secreted glycoproteins. Structural complexes involving protein associated with lipid via noncovalent interactions are termed lipoproteins. The distinct roles of lipoproteins are described on the linked page. Their major function in the body is to aid in the storage transport of lipid and cholesterol. Clinical Significances This discussion is not intended to be a complete review of all disorders that result from defects in protein structure and function. Visit the Inborn Errors page for a more complete listing of diseases related to abnormal proteins and also click on the links to the specific examples below for more information. The substitution of a hydrophobic amino acid (V) for an acidic amino acid (E) in the β-chain of hemoglobin results in sickle cell anemia (HbS). This change of a single amino acid alters the structure of hemoglobin molecules in such a way that the deoxygenated proteins polymerize and precipitate within the erythrocyte, leading to their characteristic sickle shape. Collagens are the most abundant proteins in the body. Alterations in collagen structure arising from abnormal genes or abnormal processing of collagen proteins results in numerous diseases, including Larsen syndrome, scurvy, osteogenesis imperfecta and Ehlers-Danlos syndrome. Ehlers-Danlos syndrome is actually the name associated with at least ten distinct disorders that are biochemically and clinically distinct yet all manifest structural weakness in connective tissue as a result of defective collagen structure. Osteogenesis imperfecta also encompasses more than one disorder. At least four biochemically and clinically distinguishable maladies have been identified as osteogenesis imperfecta, all of which are characterized by multiple fractures and resultant bone deformities. Marfan syndrome manifests itself as a disorder of the connective tissue and was originally believed to be the result of abnormal collagens. However, recent evidence has shown that Marfan syndrome results from mutations in the extracellular protein, fibrillin, which is an integral constituent of the noncollagenous microfibrils of the extracellular matrix. Several forms of familial hypercholesterolemia are the result of genetic defects in the gene encoding the receptor for low-density lipoprotein (LDL). These defects result in the synthesis of abnormal LDL receptors that are incapable of binding to LDLs, or that bind LDLs but the receptor/LDL complexes are not properly internalized and degraded. The outcome is an elevation in serum cholesterol levels and increased propensity toward the development of atherosclerosis. A number of proteins can contribute to cellular transformation and carcinogenesis when their basic structure is disrupted by mutations in their genes. These genes are termed proto-oncogenes. For some of these proteins, all that is required to convert them to the oncogenic form is a single amino acid substitution. The cellular gene, RAS, is observed to sustain single amino acid substitutions at positions 12 or 61 with high frequency in colon carcinomas. Mutations in RAS are most frequently observed genetic alterations in colon cancer. Myoglobin Myoglobin and hemoglobin are hemeproteins whose physiological importance is principally related to their ability to bind molecular oxygen. Myoglobin is a monomeric heme protein found mainly in muscle tissue where it serves as an intracellular storage site for oxygen. During periods of oxygen deprivation oxymyoglobin releases its bound oxygen which is then used for metabolic purposes. The tertiary structure of myoglobin is that of a typical water soluble globular protein. Its secondary structure is unusual in that it contains a very high proportion (75%) of α-helical secondary structure. A myoglobin polypeptide is comprised of 8 separate right handed α-helices, designated A through H, that are connected by short non helical regions. Amino acid R-groups packed into the interior of the molecule are predominantly hydrophobic in character while those exposed on the surface of the molecule are generally hydrophilic, thus making the molecule relatively water soluble. Structure of Myoglobin with Heme Each myoglobin molecule contains one heme prosthetic group inserted into a hydrophobic cleft in the protein. Each heme residue contains one central coordinately bound iron atom that is normally in the Fe 2+, or ferrous, oxidation state. The oxygen carried by hemeproteins is bound directly to the ferrous iron atom of the heme prosthetic group. Oxidation of the iron to the Fe3+, ferric, oxidation state renders the molecule incapable of normal oxygen binding. Hydrophobic interactions between the tetrapyrrole ring and hydrophobic amino acid R groups on the interior of the cleft in the protein strongly stabilize the heme protein conjugate. In addition a nitrogen atom from a histidine R group located above the plane of the heme ring is coordinated with the iron atom further stabilizing the interaction between the heme and the protein. In oxymyoglobin the remaining bonding site on the iron atom (the 6th coordinate position) is occupied by the oxygen, whose binding is stabilized by a second histidine residue. Carbon monoxide also binds coordinately to heme iron atoms in a manner similar to that of oxygen, but the binding of carbon monoxide to heme is much stronger than that of oxygen. The preferential binding of carbon monoxide to heme iron is largely responsible for the asphyxiation that results from carbon monoxide poisoning. Hemoglobin Adult hemoglobin is a [α(2):β(2)] tetrameric hemeprotein found in erythrocytes where it is responsible for binding oxygen in the lung and transporting the bound oxygen throughout the body where it is used in aerobic metabolic pathways. Structure of Hemoglobin For a description of the different types of hemoglobin tetramers see the section below on Hemoglobin Genes. Each subunit of a hemoglobin tetramer has a heme prosthetic group identical to that described for myoglobin. The common peptide subunits are designated α, β, γ and δ which are arranged into the most commonly occurring functional hemoglobins. Although the secondary and tertiary structure of various hemoglobin subunits are similar, reflecting extensive homology in amino acid composition, the variations in amino acid composition that do exist impart marked differences in hemoglobin's oxygen carrying properties. In addition, the quaternary structure of hemoglobin leads to physiologically important allosteric interactions between the subunits, a property lacking in monomeric myoglobin which is otherwise very similar to the α-subunit of hemoglobin. Comparison of the oxygen binding properties of myoglobin and hemoglobin illustrate the allosteric properties of hemoglobin that results from its quaternary structure and differentiate hemoglobin's oxygen binding properties from that of myoglobin. The curve of oxygen binding to hemoglobin is sigmoidal typical of allosteric proteins in which the substrate, in this case oxygen, is a positive homotropic effector. When oxygen binds to the first subunit of deoxyhemoglobin it increases the affinity of the remaining subunits for oxygen. As additional oxygen is bound to the second and third subunits oxygen binding is further, incrementally, strengthened, so that at the oxygen tension in lung alveoli, hemoglobin is fully saturated with oxygen. As oxyhemoglobin circulates to deoxygenated tissue, oxygen is incrementally unloaded and the affinity of hemoglobin for oxygen is reduced. Thus at the lowest oxygen tensions found in very active tissues the binding affinity of hemoglobin for oxygen is very low allowing maximal delivery of oxygen to the tissue. In contrast the oxygen binding curve for myoglobin is hyperbolic in character indicating the absence of allosteric interactions in this process. The allosteric oxygen binding properties of hemoglobin arise directly from the interaction of oxygen with the iron atom of the heme prosthetic groups and the resultant effects of these interactions on the quaternary structure of the protein. When oxygen binds to an iron atom of deoxyhemoglobin it pulls the iron atom into the plane of the heme. Since the iron is also bound to histidine F8, this residue is also pulled toward the plane of the heme ring. The conformational change at histidine F8 is transmitted throughout the peptide backbone resulting in a significant change in tertiary structure of the entire subunit. Conformational changes at the subunit surface lead to a new set of binding interactions between adjacent subunits. The latter changes include disruption of salt bridges and formation of new hydrogen bonds and new hydrophobic interactions, all of which contribute to the new quaternary structure. The latter changes in subunit interaction are transmitted, from the surface, to the heme binding pocket of a second deoxy subunit and result in easier access of oxygen to the iron atom of the second heme and thus a greater affinity of the hemoglobin molecule for a second oxygen molecule. The tertiary configuration of low affinity, deoxygenated hemoglobin (Hb) is known as the taut (T) state. Conversely, the quaternary structure of the fully oxygenated high affinity form of hemoglobin (HbO2) is known as the relaxed (R) state. In the context of the affinity of hemoglobin for oxygen there are four primary regulators, each of which has a negative impact. These are CO2, hydrogen ion (H+), chloride ion (Cl–), and 2,3-bisphosphoglycerate (2,3BPG, or also just BPG). Some older texts abbreviate 2,3BPG as DPB. Although they can influence O2 binding independent of each other, CO2, H+ and Cl– primarily function as a consequence of each other on the affinity of hemoglobin for O 2. We shall consider the transport of O2 from the lungs to the tissues first. In the high O2 environment (high pO2) of the lungs there is sufficient O2 to overcome the inhibitory nature of the T state. During the O2 binding-induced alteration from the T form to the R form several amino acid side groups on the surface of hemoglobin subunits will dissociate protons as depicted in the equation below. This proton dissociation plays an important role in the expiration of the CO2 that arrives from the tissues (see below). However, because of the high pO2, the pH of the blood in the lungs (≈7.4 – 7.5) is not sufficiently low enough to exert a negative influence on hemoglobin binding O2. When the oxyhemoglobin reaches the tissues the pO 2 is sufficiently low, as well as the pH (≈7.2), that the T state is favored and the O2 released. 4O2 + Hb <——> nH+ + Hb(O2)4 If we now consider what happens in the tissues, it is possible to see how CO2, H+, and Cl– exert their negative effects on hemoglobin binding O2. Metabolizing cells produce CO2 which diffuses into the blood and enters the circulating red blood cells (RBCs). Within RBCs the CO2 is rapidly converted to carbonic acid through the action of carbonic anhydrase as shown in the equation below: CO2 + H2O ——> H2CO3 ——> H+ + HCO3– The bicarbonate ion produced in this dissociation reaction diffuses out of the RBC and is carried in the blood to the lungs. This effective CO 2 transport process is referred to as isohydric transport. Approximately 80% of the CO 2 produced in metabolizing cells is transported to the lungs in this way. A small percentage of CO2 is transported in the blood as a dissolved gas. In the tissues, the H+ dissociated from carbonic acid is buffered by hemoglobin which exerts a negative influence on O2 binding forcing release to the tissues. As indicated above, within the lungs the high pO2 allows for effective O2 binding by hemoglobin leading to the T to R state transition and the release of protons. The protons combine with the bicarbonate that arrived from the tissues forming carbonic acid which then enters the RBCs. Through a reversal of the carbonic anhydrase reaction, CO2 and H2O are produced. The CO2 diffuses out of the blood, into the lung alveoli and is released on expiration. In addition to isohydric transport, as much as 15% of CO 2 is transported to the lungs bound to N-terminal amino groups of the T form of hemoglobin. This reaction, depicted below, forms what is called carbamino-hemoglobin. As indicated this reaction also produces H+, thereby lowering the pH in tissues where the CO2 concentration is high. The formation of H+ leads to release of the bound O2 to the surrounding tissues. Within the lungs, the high O2 content results in O2 binding to hemoglobin with the concomitant release of H +. The released protons then promote the dissociation of the carbamino to form CO2 which is then released with expiration. CO2 + Hb-NH2 <——> H+ + Hb-NH-COO– As the above discussion demonstrates, the conformation of hemoglobin and its oxygen binding are sensitive to hydrogen ion concentration. These effects of hydrogen ion concentration are responsible for the well known Bohr effect in which increases in hydrogen ion concentration decrease the amount of oxygen bound by hemoglobin at any oxygen concentration (partial pressure). Coupled to the diffusion of bicarbonate out of RBCs in the tissues there must be ion movement into the RBCs to maintain electrical neutrality. This is the role of Cl- and is referred to as the chloride shift. In this way, Cl– plays an important role in bicarbonate production and diffusion and thus also negatively influences O2 binding to hemoglobin. Representation of the transport of CO2 from the tissues to the blood with delivery of O2 to the tissues. The opposite process occurs when O2 is taken up from the alveoli of the lungs and the CO2 is expelled. All of the processes of the transport of CO2 and O2 are not shown such as the formation and ionization of carbonic acid in the plasma. The latter is a major mechanism for the transport of CO2 to the lungs, i.e. in the plasma as HCO3–. The H+ produced in the plasma by the ionization of carbonic acid is buffered by phosphate (HPO42–) and by proteins. Additionally, some 15% of the CO 2 is transported from the tissues to the lungs as hemoglobin carbamate. Role of 2,3-bisphosphoglycerate (2,3-BPG) The compound 2,3-bisphosphoglycerate (2,3-BPG), derived from the glycolytic intermediate 1,3-bisphosphoglycerate, is a potent allosteric effector on the oxygen binding properties of hemoglobin. The pathway to 2,3BPG synthesis is diagrammed in the figure below. The pathway for 2,3-bisphosphoglycerate (2,3-BPG) synthesis within erythrocytes. Synthesis of 2,3-BPG represents a major reaction pathway for the consumption of glucose in erythrocytes. The synthesis of 2,3-BPG in erythrocytes is critical for controlling hemoglobin affinity for oxygen. Note that when glucose is oxidized by this pathway the erythrocyte loses the ability to gain 2 moles of ATP from glycolytic oxidation of 1,3-BPG to 3phosphoglycerate via the phosphoglycerate kinase reaction. In the deoxygenated T conformer, a cavity capable of binding 2,3-BPG forms in the center of the molecule. 2,3-BPG can occupy this cavity stabilizing the T state. Conversely, when 2,3-BPG is not available, or not bound in the central cavity, Hb can be converted to HbO2 more readily. Thus, like increased hydrogen ion concentration, increased 2,3-BPG concentration favors conversion of R form Hb to T form Hb and decreases the amount of oxygen bound by Hb at any oxygen concentration. Hemoglobin molecules differing in subunit composition are known to have different 2,3-BPG binding properties with correspondingly different allosteric responses to 2,3-BPG. For example, HbF (the fetal form of hemoglobin) binds 2,3-BPG much less avidly than HbA (the adult form of hemoglobin) with the result that HbF in fetuses of pregnant women binds oxygen with greater affinity than the mothers HbA, thus giving the fetus preferential access to oxygen carried by the mothers circulatory system. The Hemoglobin Genes The α- and β-globin proteins contained in functional hemoglobin tetramers are derived from gene clusters. The α-globin genes are on chromosome 16 and the β-globin genes are on chromosome 11. Both gene clusters contain not only the major adult genes, α and β, but other expressed sequences that are utilized at different stages of development. The orientation of the genes in both clusters is in the same 5' to 3' direction with the earliest expressed genes at the 5' end of both clusters. In addition to functional genes, both clusters contain non-functional pseudogenes. Hemoglobin synthesis begins in the first few weeks of embryonic development within the yolk sac. The major hemoglobin at this stage of development is a tetramer composed of 2 zeta (ζ) chains encoded within the α cluster and 2 epsilon (ε) chains from the β cluster. By 6-8 weeks of gestation the expression of this version of hemoglobin declines dramatically coinciding with the change in hemoglobin synthesis from the yolk sac to the liver. Expression from the α cluster consists of identical proteins from the α1 and α2 genes. Expression from these genes in the α cluster remains on throughout life. Within the β-globin cluster there is an additional set of genes, the fetal βglobin genes identified as the gamma (γ) genes. The 2 fetal genes called Gγ and Aγ, the derivation of which stems from the single amino acid difference between the 2 fetal genes: glycine in Gγ and alanine in Aγ at position 136. These fetal γ genes are expressed as the embryonic genes are turned off. Shortly before birth there is a smooth switch from fetal γ-globin gene expression to adult β-globin gene expression. The switch from fetal γ- to adult β-globin does not directly coincide with the switch from hepatic synthesis to bone marrow synthesis since at birth it can be shown that both γ and β synthesis is occurring in the marrow. Given the pattern of globin gene activity throughout fetal development and in the adult the composition of the hemoglobin tetramers is of course distinct. Fetal hemoglobin is identified as HbF and includes both α2Gγ2 and α2Aγ2. Fetal hemoglobin has a slightly higher affinity for oxygen than does adult hemoglobin. This allows the fetus to extract oxygen more efficiently from the maternal circulation. In adults the major hemoglobin is identified as HbA (more commonly HbA1) and is a tetramer of 2 α and 2 β chains as indicated earlier. A minor adult hemoglobin, identified as HbA 2, is a tetramer of 2 α chains and 2 δ chains. The δ gene is expressed with a timing similar to the β gene but because the promoter has acquired a number of mutations its' efficiency of transcription is reduced. The overall hemoglobin composition in a normal adult is approximately 97.5% HbA1, 2% HbA2 and 0.5% HbF. Hemoglobinopathies A large number of mutations have been described in the globin genes. These mutations can be divided into two distinct types: those that cause qualitative abnormalities (e.g. sickle cell anemia) and those that cause quantitative abnormalities (the thalassemias). Taken together these disorders are referred to as the hemoglobinopathies. A third group of hemoglobin disorders include those diseases in which there is a persistence of fetal hemoglobin expression. These latter diseases are known collectively as hereditary persistence of fetal hemoglobin (HPFH). Of the mutations leading to qualitative alterations in hemoglobin, the missense mutation in the β-globin gene that causes sickle cell anemia is the most common. The mutation causing sickle cell anemia is a single nucleotide substitution (A to T) in the codon for amino acid 6. The change converts a glutamic acid codon (GAG) to a valine codon (GTG). The form of hemoglobin in persons with sickle cell anemia is referred to as HbS. The underlying problem in sickle cell anemia is that the valine for glutamic acid substitution results in hemoglobin tetramers that aggregate into arrays upon deoxygenation in the tissues. This aggregation leads to deformation of the red blood cell making it relatively inflexible and unable to traverse the capillary beds. Repeated cycles of oxygenation and deoxygenation lead to irreversible sickling. The end result is clogging of the fine capillaries. Because bones are particularly affected by the reduced blood flow, frequent and severe bone pain results. This is the typical symptom during a sickle cell "crisis". Long term the recurrent clogging of the capillary beds leads to damage to the internal organs, in particular the kidneys, heart and lungs. The continual destruction of the sickled red blood cells leads to chronic anemia and episodes of hyperbilirubinemia. An additional relatively common mutation at codon 6 is the conversion to a lysine codon (AAG) which results in the generation of HbC. Electrophoresis of hemoglobin proteins from individuals suspected of having sickle cell anemia (or several other types of hemoglobin disorders) is an effective diagnostic tool because the variant hemoglobins have different charges. An example of this technique is shown in the Figure below. Pattern of hemoglobin electrophoresis from several different individuals. Lanes 1 and 5 are hemoglobin standards. Lane 2 is a normal adult. Lane 3 is a normal neonate. Lane 4 is a homozygous HbS individual. Lanes 6 and 8 are heterozygous sickle individuals. Lane 7 is a SC disease individual. Another effective tool to identify the genotype of individuals suspected of having sickle cell disease as well as for prenatal diagnosis is to either carry out RFLP mapping or to use PCR. An example of the use of these tools can be seen in the Molecular Tools of Medicine page. In addition to the missense mutations that lead to HbS and HbC, a number of frameshift mutations leading to qualitative abnormalities in hemoglobin have been identified. A 2-nucleotide insertion between codons 144 and 145 in the β-globin gene results in the generation of hemoglobin Cranston. The insertion, which is near the C-terminus of the β-globin protein, results in the normal stop codon being out of frame and synthesis proceeding into the 3'untranslated region to a fortuitous stop codon. The result is a β-globin protein of 157 amino acids. In the hemoglobin Constant Spring variant, a mutation in the α-globin gene converts the stop codon (UAA) to a glutamine codon (CAA) so that the protein ends up being 31 amino acids longer than normal. The resultant αglobin protein in hemoglobin Constant Spring is not only qualitatively altered but because it is unstable it is a quantitative abnormality as well. Because the globin gene loci contain clusters of similar genes there is the potential for unequal cross-over between the sister chromatids during meiosis. The generation of hemoglobin Gun Hill and Lepore hemoglobins are both the result of unequal cross over events. Hemoglobin Gun Hill is the result of a deletion of 15 nucleotides caused by unequal cross over between codons 91– 94 of one β-globin gene and codons 96–98 of the other. Generation of Lepore hemoglobins results from unequal cross over between the δ-globin and βglobin genes. The resultant hybrid δβ gene is called Lepore and the βδ hybrid gene is called anti-Lepore. As indicated earlier, the promoter of the δ-globin gene is inefficient so the consequences of this unequal cross over event are both qualitative and quantitative. The thalassemias are the result of abnormalities in hemoglobin synthesis and affect both clusters. Deficiencies in β-globin synthesis result in the βthalassemias and deficiencies in α-globin synthesis result in the αthalassemias. The term thalassemia is derived from the Greek thalassa meaning "sea" and was applied to these disorders because of the high frequency of their occurrence in individuals living around the Mediterranean Sea. In normal individuals an equal amount of both α- and β-globin proteins are made allowing them to combine stoichiometrically to form the correct hemoglobin tetramers. In the α-thalassemias normal amounts of β-globin are made. The β-globin proteins are capable of forming homotetramers (β 4) and these tetramers are called hemoglobin H, (HbH). An excess of HbH in red blood cells leads to the formation if inclusion bodies commonly seen in patients with α-thalassemia. In addition, the HbH tetramers have a markedly reduced oxygen carrying capacity. In β-thalassemia, where the β-globins are deficient, the α-globins are in excess and will form α-globin homotetramers. The α-globin homotetramers are extremely insoluble which leads to premature red cell destruction in the bone marrow and spleen. With the α-thalassemias the level of α-globin production can range from none to very nearly normal levels. This is due in part to the fact that there are 2 identical α-globin genes on chromosome 16. Thus, the α-thalassemias involve inactivation of 1 to all 4 α-globin genes. If 3 of the 4 α-globin genes are functional, individuals are completely asymptomatic. This situation is identified as the "silent carrier" state or sometimes as α-thalassemia 2. Genotypically this situation is designated αα/α– (where the dash indicates a non-functional gene) or α–/αα. If 2 of the 4 genes are inactivated individuals are designated as α-thalassemia trait or as α-thalassemia 1. Genotypically this situation is designated αα/– –. In individuals of African descent with α-thalassemia 1, the disorder usually results from the inactivation of 1 α-globin gene on each chromosome and is designated α–/α–. This means that these individuals are homozygous for the α-thalassemia 2 chromosome. The phenotype of αthalassemia 1 is relatively benign. The mean red cell volume (designated MCV in clinical tests) is reduced in α-thalassemia 1 but individuals are generally asymptomatic. The clinical situation becomes more severe if only 1 of the 4 α-globin genes is functional. Because of the dramatic reduction in αglobin chain production in this latter situation, a high level of β 4 tetramer is present. clinically this is referred to as hemoglobin H disease. Afflicted individuals have moderate to marked anemia and their MCV is quite low, but the disease is not fatal. The most severe situation results when no α-globin chains are made (genotypically designated – –/– –). This leads to prenatal lethality or early neonatal death. The predominant fetal hemoglobin in afflicted individuals is a tetramer of γ-chains and is referred to as hemoglobin Bart's. This hemoglobin has essentially no oxygen carrying capacity resulting in oxygen starvation in the fetal tissues. Heart failure results as the heart tries to pump the unoxygenated blood to oxygen starved tissues leading to marked edema. This latter situation is called hydrops fetalis. A large number of mutations have been identified leading to decreased or absent production of β-globin chains resulting in the β-thalassemias. In the most severe situation mutations in both the maternal and paternal β-globin genes leads to loss of normal amounts of β-globin protein. A complete lack of HbA is denoted as β0-thalassemia. If one or the other mutations allows production of a small amount of functional β-globin then the disorder is denoted as β+-thalassemia. Both β0- and β+-thalassemias are referred to as thalassemia major, also called Cooley's anemia after Dr. Thomas Cooley who first described the disorder. Afflicted individuals suffer from severe anemia beginning in the first year of life leading to the need for blood transfusions. As a consequence of the anemia the bone marrow dramatically increases its' effort at blood production. The cortex of the bone becomes thinned leading to pathologic fracturing and distortion of the bones in the face and skull. In addition, there is marked hepatosplenomegaly as the liver and spleen act as additional sites of blood production. Without intervention these individuals will die within the decade of life. As indicated, β-thalassemia major patient require blood transfusions, however, in the long term these transfusions lead to the accumulation of iron in the organs, particularly the heart, liver and pancreas. Organ failure ensues with death in the teens to early twenties. Iron chelation therapies appear to improve the outlook for β-thalassemia major patients but this requires continuous infusion of the chelating agent. Individuals heterozygous for β-thalassemia have what is termed thalassemia minor. Afflicted individuals harbor one normal β-globin gene and one that harbors a mutation leading to production of reduced or no β-globin. Individuals that do not make any functional β-globin protein from 1 gene are termed βo heterozygotes. If β-globin production is reduced at one locus the individuals are termed β+ heterozygotes. Thalassemia minor individuals are generally asymptomatic. The term thalassemia intermedia is used to designate individuals with significant anemia and who are symptomatic but unlike thalassemia major do not require transfusions. This syndrome results in individuals where both βglobin genes express reduced amounts of protein or where one gene makes none and the other makes a mildly reduced amount. A person who is a compound heterozygote with α-thalassemia and β+-thalassemia will also manifest as thalassemia intermedia. The primary cause of α-thalassemias is deletion, whereas, for βthalassemias the mutations are more subtle. In β-thalassemias, point mutations in the promoter, mutations in the translational initiation codon, a point mutation in the polyadenylation signal and an array of mutations leading to splicing abnormalities have been characterized. An interesting and common (up to 30% of persons from Southeast Asia) hemoglobinopathy that has both quantitative and qualitative characteristics is caused by the synthesis of hemoglobin E. Hemoglobin E arises due to a point mutation in codon 26 that changes glutamic acid (GAG) to lysine (AAG). Individulas with this mutation make only around 60% of the normal amount of β-globin protein. The reason for this is that the mutation creates a cryptic splice site such that 40% of the hemoglobin E mRNA is shorter by 16 nucleotides and does not give rise to detectable β-globin protein. There are some individuals in whom the developmental timing of globin production is altered as a consequence of mutation. Persons with hereditary persistence of fetal hemoglobin, HPFH continue to make HbF as adults. Because the syndrome is benign most individuals do not even know they carry a hemoglobin abnormality. Many HPFH individuals harbor large deletions of the δ- and β-coding region of the cluster. There is no deletion of the fetal globin genes and by an as yet uncharacterized mechanism expression of these genes persists in adulthood. As discussed above functional hemoglobin is a heterotetramer. Mutations in either the α-globin or the β-globin genes lead to quantitative and qualitative abnormalities in hemoglobin. Therefore, it should not be surprising that complex compound heterozygosities can result in offspring of individuals harboring different mutations. Source: medical biochemistry web page