pedal reflex - People.vcu.edu
advertisement

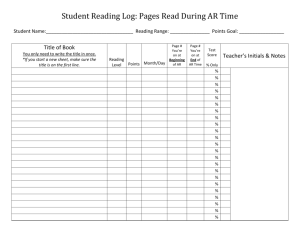
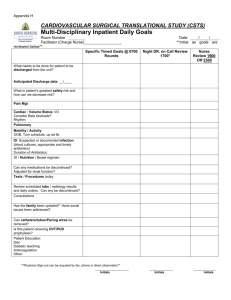
3/21/11 Rabbit Surgeries See procedure notes attached beneath these records. Todd Kitten, x6-7010, cell: 301-4948, IACUC # AM10030 Presurgical examination normal? Yes No Date______________ Initials ______ Notes:_____________________________________________________________________________ Rabbit Mass (kg) 563 Drug Amounts aceprom. (10 mg/ml) ket # ket + xyl + bupren. bup # Card# (ml) Time/Initials 0.6 pedal reflex test 15 min 30 min (87 + 8.7 + 0.013 mg/ml) glycopyrrolate (0.2 mg/ml) 45 min Surgery start Surgery finish Surgical Notes: Parameter Anesthesia Respiration Cardiovascular Cath. placement Blood loss Hemostasis Other Normal/Good Abnormal Comments Initials: Immediate Post-operative observations (behaviors observed, time, initials) Chewing Lifting head Eating paper Repositioned body Sitting upright Other: Return to cage: Time________PM Initials_______ Buprenorphine injection (0.3mg/ml): Vial #______ Vol. (ml)_______ Time______PM Initials______ Follow-up postoperative daily observations: (1= excellent; 5 = poor. Any problems or other observations are recorded below in "Other comments." *NC = cages not cleaned before surgery; JC = cages just cleaned) Item Date_________ Time AM Date_________ Time PM Date_________ Time AM Date_________ Time PM Responsiveness (1-5) Posture/Activity (1-5) Respiration normal? (Y/N) Eating/drinking (Y/N) Urine (1-5; NC; JC)* Feces (1-5; NC; JC)* Buprevol. (ml) norphine Vial # Initials Other comments_____________________________________________________________ _________________________________________________________________________ _________________________________________________________________________ Inoculation and necropsy notes on back. Rabbit _____ Inoculation Notes Date____________ 0.8 ml acepromazine SC Time_______ Initials ______ Strain Inoculated (iv) ______________ Time_______ Initials ______ Incision ____OK _____Clips removed? Other ___________________________________ Initials ______ Necropsy Notes Date____________ 1.2 ml acepromazine SC Time_______ Initials ______ Euthasol (iv, ear vein) _____ ml Time_______ Initials ______ Catheter location: __________________________________ Other necropsy notes: Description of Procedures Performed Date____________ Signed_________________________ For catheterization, rabbits were anesthetized as follows. All rabbits were given 0.6 ml acepromazine SC (10 mg/ml; on the back). At least 10 minutes later, 0.575 ml/kg xylazine + ketamine + buprenorphine solution was injected SC at an adjacent site. (Premix is 1 ml of 100 mg/ml xylazine and 0.5 ml of buprenorphine (0.3 mg/ml) added to a 10 ml bottle of 100 mg/ml ketamine. Final solution is 87 mg/ml ketamine, 8.7 mg/ml xylazine and 0.013 mg/ml buprenorphine.) This was followed by immediate SC injection of 0.1 mg (0.5 ml) per kg glycopyrrolate. See previous pages for exact times and amounts of anesthetic injected. Ten minutes later, the rabbits had their neck, sternum, and ears shaved. Ophthalmic ointment was applied to each eye. This was followed by swabbing of the neck with betadyne and injections of 0.5 ml (2.5 mg) bupivicaine SC at the site where the incision was to be made. Rabbits were then carried to the sterile field and covered with a sterile drape. An incision was made in the neck above the sternum. The right carotid artery was identified and gently dissected away from surrounding tissue. The artery was tied off with suture at the head end, and clamped with a vascular clamp at the base of the neck. An 18G needle was bent and introduced into the artery. This served as a guide for introduction of a catheter into the artery. The needle and clamp were then removed, and the catheter threaded through the artery until resistance was met, indicating contact with the aortic valve or insertion into the left ventricle . The catheter was crimped, tied off with suture, and cut. Sutures were also tied around the artery just caudal to the point of catheter insertion to secure the catheter and prevent bleeding. The short remaining head end of the catheter was tucked under the subcutaneous tissue of the neck, and the incision closed with skin clips. The rabbit was returned to the anesthesia room with a warmed deltathermal pad for observation until recovered. All animals were returned to their cages as soon as they were upright and recovered. At the end of the day, animals received ~0.03 mg/kg buprenorphine SC. Buprenorphine (~0.03 mg/kg) was also supplied every 12 hrs for the next two days. Animals were then monitored every 12 hrs until euthanasia, and provided buprenorphine if a rabbit showed signs of pain. See previous pages for notes on surgery and recovery. Two days after catheterization, an inoculum of one or more bacterial strains was injected into the marginal ear vein, as noted on the previous pages. The inoculum consisted of approximately 107 colony forming units of bacteria, unless otherwise noted. Rabbits were given 0.8 ml (8 mg) acepromazine subcutaneously and placed in a restrainer prior to inoculation. Animals were euthanized the next day. Rabbits were given 12 mg acepromazine subcutaneously and placed in a restrainer. Two ml euthasol (equivalent to ~780 mg of sodium pentobarbital) was then injected into an ear vein. If death did not occur immediately, additional euthasol was injected. Thoracotomy immediately followed euthanasia. All animals were necropsied to determine catheter placement and presence of vegetations on heart valves. Organisms were recovered by homogenizing these tissues in PBS, then spreading the homogenates on agar plates and incubating to allow for bacterial growth. Initials TK NZ XG CM Name Todd Kitten Nicai Zollar Xiuchun Ge Cindy Munro