Research Investigations
advertisement

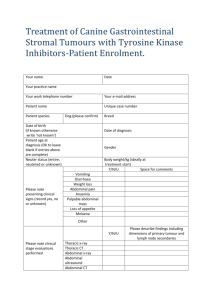
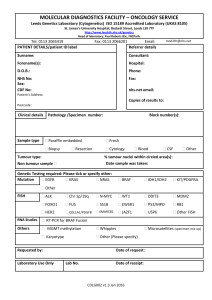
Research Investigations While chemotherapy drugs are highly effective in combating the growth of tumours, resistance to these drugs typically develops during treatment, resulting in treatment failure and progression of disease. Patients then move on to other chemotherapy drugs but these drugs often also become ineffective in halting tumour growth. This phenomenon is called “multidrug resistance”. The research efforts of my research group are focused on better understanding factors that affect the response of tumours to chemotherapy drugs and how tumour cells develop resistance to chemotherapy. Reduction in Tumour RNA Quality and Response to Chemotherapy in Breast Cancer Patients Actively growing tumour cells produce large quantities of a large molecule called ribonucleic acid (RNA). We have observed in a national clinical trial (NCIC-CTGMA.22) that the quality (integrity) of RNA in tumours falls dramatically in about half of breast cancer patients in response to chemotherapy. Another clinical trial in the United Kingdom has recently reproduced these findings. In the MA.22 study, we observed that this drop in quality was highest for the highest doses of chemotherapy drugs. Moreover, low tumour RNA quality mid-treatment predicted complete destruction of the tumour after chemotherapy. In contrast, little change in RNA quality was observed in blood cells from the same patients at any point during treatment. Our findings suggest that a patient’s response to chemotherapy may be effectively measured by monitoring RNA quality early in treatment (after 1-3 cycles). Patients exhibiting reductions in tumour RNA quality would be assured that they are responding to chemotherapy, while patients with high tumour RNA quality after 1-3 cycles could be switched quickly to alternate treatments, such as surgery, radiation therapy, or other drugs. This will be assessed in upcoming clinical trials in collaboration with the Ontario Clinical Oncology Group, Sunnybrook Hospital, McMaster University, the University of Waterloo, and the London Regional Health Sciences Centre. Role of Aldoketoreductases in Resistance to the Anthracycline Class of Chemotherapy Drugs We have recently observed that breast tumour cells that have acquired resistance to a class of chemotherapy drugs called anthracyclines have high levels of protein molecules called aldo keto-reductases (AKRs). The AKRs play a natural role in breast cells, where they stimulate estrogen production. Estrogen, in turn, stimulates rapid growth of breast tumour cells, making them very sensitive to anthracyclines. To counteract this, we observed that anthracyclineresistant cells produce very little of the receptor for estrogen. The cells thus grow more slowly and are not killed effectively by chemotherapy drugs. In addition, the AKRs have the ability to alter the structure of anthracyclines, converting the drug to a form that can no longer reach its target in tumour cells (DNA within the nucleus). In fact, the drug appears to be sequestered in the lysosomes of cells away from the nucleus. Consequently, the altered drug is almost completely ________________________________________________________________ Parissenti – Research Investigations Page 1 of 2 Research Investigations ineffective in killing breast tumour cells. It is also highly toxic to the heart. We have also shown recently that other drugs that inhibit the function of AKRs can prevent this change in anthraycline structure, restoring the ability of this class of drugs to kill tumour cells. In the future we plan to assess whether AKR-inhibitory drugs may improve the ability of anthracyclines to kill tumours in mice, while reducing damage to the heart. Tumour Necrosis Factor Alpha and Response to the Taxane Class of Chemotherapy Drugs Drugs such as paclitaxel and docetaxel (taxanes) are widely used in the treatment of a variety of human cancers, including breast and ovarian cancer. These drugs are known to bind to structures in normal and cancer cells called microtubules that play a role in separation of chromosomes between mother and daughter cells. This binding of the drug to microtubules prevents cell reproduction from taking place, particularly in rapidly proliferating cells such as tumour cells. This is thought to be the primary mechanism of action for taxanes. We have recently discovered that taxanes also promote in tumour cells a dramatic increase in the production and secretion of a death-inducing molecule called tumour necrosis factor alpha (TNFa). This may be a previously unknown mechanism by which taxanes kill tumour cells. Oddly, we have observed that breast tumour cells resistant to taxanes have even higher levels of TNFa. While this initially was puzzling, we also discovered that taxane-resistant cells are resistant to TNFa-induced killing because they have lost the receptor to which TNFa binds (TNFR1) in order to induce cell killing. In contrast, they have another receptor (TNFR2) to which TNFa can bind, which promotes tumour cell survival. Based on these novel findings, we now believe that TNFa plays an important role in tumour response and resistance to taxanes. We hope to be able to block this resistance mechanism in the future, in order to improve the effectiveness of taxane chemotherapy. ________________________________________________________________ Parissenti – Research Investigations Page 2 of 2