C2 Colloids
advertisement

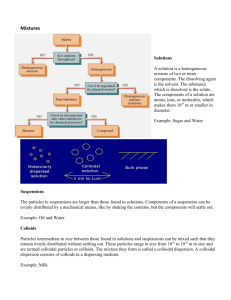
Page 1 DISPERSED SYSTEMS A disperse system may be defined as a system in which one substance (the dispersed phase) is dispersed as particles throughout another (the dispersion medium or continuous phase). We can classify these systems on the basis of the physical shape and size of the dispersed particles. Matter exists as gas, solid or liquid, so many different types of dispersed systems are possible. The dispersed material may range in size from particles of atomic and molecular dimensions to particles where the size is measures in millimetres. Because of this wide range, we can classify disperse systems on the basis of the mean particle diameter of the dispersed material. NB. Size limits are somewhat arbitrary as there is no distinct transition between the 3 systems. COLLOIDS TYPES OF COLLOIDAL SYSTEMS Lyophilic Colloids In these systems, there is affinity between the colloidal particles and the dispersion medium (solvent-loving). Owing to their affinity for the dispersion medium, such materials form colloidal dispersions or sols with relative ease. To obtain these colloids, we merely dissolve the material in the solvent being used e.g. gelatin in water. The affinity of the particles for the solvent renders these systems thermodynamically stable. Solvation occurs in these colloids - the attachment of solvent molecules to molecules of the dispersed phase. In the case of hydrophilic colloids in which water is the dispersion medium, this is termed hydration. Dispersed phase - organic molecules e.g. gelatin, acacia, insulin, albumin, rubber and polystyrene. The first 4 form hydrophilic colloids i.e. the dispersion medium is water. The last 2 are dissolved in nonaqueous, organic solvents - lipophilic colloids. Note that a material that forms a lyophilic colloid in one liquid such as water, may not do so in another liquid such as benzene. So BOTH phases are important. Lyophobic colloids The dispersed phase has little, if any, attraction for the dispersion medium. These are lyophobic (solvent-hating) and their properties differ from those of lyophilic colloids. The main reason is the absence of a solvent sheath around the particles. The dispersed phase of these colloids is generally composed of inorganic particles Page 2 dispersed in water e.g. gold, silver, sulphur. Because there is no affinity for the solvent, spontaneous coalescence occurs which indicates that these systems are unstable and that attractive forces exist between the particles. As these do not form spontaneously, it is necessary to employ special methods to prepare these colloids. Either dispersion or condensation methods are used: Dispersion methods ► Coarse particles are reduced in size by the use of high-intensity ultrasonic generators operating at frequencies in excess of 20 000 cycles per second. ► A second dispersion method involves the production of an electric arc within a liquid. Owing to the intense heat generated by the arc, some of the metal of the electrodes is dispersed as vapour, which condenses to form colloidal particles. Condensation methods ► These involve a high degree of initial supersaturation followed by the formation and growth of nuclei. Supersaturation may be brought about by a change in solvent or a reduction in temperature. ► Chemical reaction such as reduction, oxidation or hydrolysis.. 3. Association colloids We have already dealt with this class. These association or amphiphilic colloids consist of surface-active agents which aggregate into micelles. Below the CMC, they exist as monomers, at and above the CMC, aggregates form (micelles) which may contain 50 or more monomers The size of these micelles lies in the colloidal size range. Formation is spontaneous, provided that the concentration of the SAA in solution exceeds the CMC. SIZE AND SHAPE OF COLLOIDAL PARTICLES Particles lying within the colloidal size range possess a surface area that is enormous compared with the surface area of an equal volume of larger particles. This results in many of the unique properties of colloidal dispersions. The shape adopted by colloidal particles is important, since the more extended the particle, the greater its specific surface and the greater the opportunity for attractive forces to develop between the particles of the dispersed phase and the dispersion medium. OVERHEAD Examples of shapes that may be assumed. Physical Pharmacy Page 3 OPTICAL PROPERTIES OF COLLOIDS When a strong beam of light is passed through a colloidal sol, a visible cone is formed. This results from the scattering of light by the colloidal particles. This is the FaradayTyndall Effect. The ultramicroscope allows us to examine the points of light responsible for the Tyndall cone. An intense light beam is passed through the sol against a dark background at right angles to the place of observation. Although the particles cannot be directly seen, bright spots corresponding to the particles can be observed. The use of the ultramicroscope has declined in recent years since it often does not resolve lyophilic colloids. The electron microscope yields pictures of the actual particles and is now used to observe the size, shape and structure of particles. The light scattering property is widely used for determining the molecular weight of colloids. KINETIC PROPERTIES OF COLLOIDS These properties relate to the motion of particles with respect to the dispersion medium. The motion may be thermally induced (Brownian movement, diffusion, osmosis), gravitationally induced (sedimentation) or applied externally (viscosity). Electrically induced motion is considered in the section on the electric properties of colloids. Thermally : Brownian Motion This results from the bombardment of the colloidal particles by the molecules of the dispersion medium. The velocity of particles increases with a decrease in particle size. If the viscosity of the medium is increased, Brownian motion decreases and finally stops. Thermally : Diffusion Particles diffuse spontaneously from a region of higher concentration to one of lower concentration, until the concentration is uniform. Diffusion is a direct result of Brownian motion. Thermally : Osmotic Pressure The particles in a colloid may consist of aggregates of several molecules, but each aggregate acts as a single unit for colligative purposes. Thus the osmotic pressure exerted by such a colloidal system will be small. Page 4 Gravitationally : Sedimentation The sedimentation of spherical particles may be expressed using Stokes' Law (see suspensions). If particles are subjected only to the force of gravity, then the lower limit of particles obeying Stokes' equation is about 0.5 μm. This is because Brownian movement becomes significant and tends to offset sedimentation and promote mixing. So with increasing particle size, Brownian motion decreases, while the tendency to sediment increases. Applied externally : Viscosity Viscosity is an expression of the resistance to flow of a system under an applied stress. The more viscous a liquid, the greater the applied force required to make it flow at a particular rate. Comparing the viscosity of the lyophobic and lyophilic colloids: The viscosity of colloids is affected by the shapes of particles of the disperse phase. Spheric particles form dispersions of relatively low viscosity, while systems containing linear particles are more viscous. Lyophobic: Viscosity is not much greater than that of the liquid vehicle. This is so even for comparatively high fractions of the dispersed phase. Particles here are symmetric and unsolvated. Lyophilic: In contrast, the apparent viscosity of lyophilic colloids is much greater than the viscosity of the vehicle, even at low concentrations of the dispersed phase. Particles here are highly asymmetric and solvated. ELECTRIC PROPERTIES AND STABILITY OF COLLOIDS These are the properties that depend on, or are affected by, the presence of a charge on the surface of a particle. The presence and magnitude, or absence of a charge on a colloidal particle is an important factor in the stability of colloidal systems. Stabilization is achieved essentially by 2 means: ► dispersed particles can be provided with an electric charge ► each particle is surrounded with a protective solvent sheath that prevents mutual adherence when the particles collide (only significant in lyophilic systems). Page 5 Stabilisation of lyophobic colloids Lyophobic colloids are thermodynamically unstable and the particles are stabilised only by the presence of electric charges on their surface. These charges originate from preferential adsorption of specific ions from solution. The like-charge produces a repulsion that prevents coagulation of the particles. Four researchers, Verwey, Overbeek, Derjaguin and Landau developed a theory called the DLVO theory that describes the stability of lyophobic colloids. To understand it, we must first consider the attractive and repulsive forces that operate between approaching particles, then to examine the combined effect of these opposing forces. OVERHEAD : Net potential energy of interaction graph Repulsive forces The absorbed charges influence the distribution of positive and negative ions in the layers of solution that surround each particle. This distribution is also influenced by thermal motions in the solution, therefore each particle is surrounded by an EDL similar to the distribution we saw previously. When 2 particles approach each other, the atmospheres of counterions or diffuse parts of the EDL overlap. A redistribution of charge occurs in each layer and there is work involved in distorting the diffuse layers. Therefore a net repulsive energy is obtained so a repulsive force will be exerted between the 2 particles. To overcome this repulsion, work or energy is needed to bring the particles closer together. Attractive forces Even when particles aggregate, repulsive forces are exerted, therefore for aggregation to occur, an attractive force must be equal to or greater in its magnitude and range of operation to the repulsive force. Attraction is provided by van der Waal's forces which are additive, so the total attraction between 2 particles will be equal to the sum of all the attractive forces between every atom of one particle and all those of the other. The additive effect increases the range over which this force is exerted. Net potential energy of interaction The net potential energy of interaction is obtained by adding the repulsive and attractive potentials at each distance of separation. As the particles approach each other and a repulsive force is exerted, they either then move apart, or, if there is enough kinetic energy to overcome repulsion, they will move closer together. Page 6 As particles continue to approach, repulsion between surface charges increases the net potential energy of interaction (NPEI) to its maximum value (energy barrier). If the height of this barrier exceeds the kinetic energy of the approaching particles, they will not come any closer, but will move away from each other. However, if their kinetic energy exceeds the energy barrier, the particles continue to approach each other, after which van der Waal's forces become increasingly important compared to electrostatic repulsion. The net potential energy decreases to zero, then becomes negative, pulling particles still closer. The interacting particles will reach the energy depth of the primary minimum and irreversible aggregation i.e. coagulation will occur. It is unlikely that enough kinetic energy can be supplied to the particles to enable them to climb out of the potential energy well, so they are permanently attached to one another. Main determinant of stability: ► The height of the primary energy barrier depends on the density of charge on the surface which is expressed by the surface and zeta potentials, and the resultant magnitude of electrical repulsion. It is also influenced by electrolyte concentration. ► Addition of electrolyte compresses the EDL and reduces the zeta potential. This has the effect of lowering the primary maximum energy barrier and as the concentration of electrolyte increases, the barrier continues to decrease and finally disappears - at this point rapid coalescence occurs. OVERHEAD : Influence of electrolyte concentration on height of energy barrier ► The valence of the ions having a charge opposite to that of the particles appears to determine the effectiveness of the electrolyte in coagulating the colloid. The precipitating power increases rapidly with the valence of the ion. This is known as the Schulze-Hardy rule. e.g. we have a negatively charged colloid and we are going to add both aluminium chloride and barium chloride. Which one would require the larger concentration to result in coagulation of the colloid? Answer: barium chloride since barium is divalent, aluminium is trivalent. ► The energy barrier may also be lowered by adding substances such as ionic surfaceactive agents which are specifically adsorbed within the Stern layer. The Stern and zeta potentials are reduced whereas the double layer is not compressed. Page 7 Stabilisation of lyophilic colloids Lyophilic colloids are stabilised by a combination of EDL interaction and solvation. Both of these stabilizing factors must be sufficiently weakened before attraction predominates and the colloidal particles coagulate Hydrophilic colloids are unaffected by the small amounts of added electrolyte which cause hydrophobic colloids to coagulate; however when the concentration of electrolyte is high, particularly with an electrolyte whose ions become strongly hydrated, the colloidal material loses its water of solvation to these ions and coagulates i.e. a "salting out" effect occurs. Coacervation: when oppositely charged hydrophilic colloids are mixed, the particles may separate from the dispersion to form a layer rich in the colloidal aggregates which is known as a coacervate and is a process of complex coacervation. SENSITIZATION AND PROTECTIVE COLLOIDAL ACTION ► The addition of a small amount of hydrophilic or hydrophobic colloid to a hydrophobic colloid of opposite charge tends to sensitize or even coagulate the particles i.e. it becomes more susceptible to precipitation by electrolytes. This is thought by some to be due to reduction of the zeta potential below the critical value. Others think it is due to a reduction in the thickness of the ionic layer surrounding the particles and a decrease in repulsion between the particles. ► The addition of large amounts of the hydrophile stabilizes the system as the hydrophile is adsorbed on the hydrophobic particles which then acquire hydrophilic properties. This phenomenon is known as protection and the added hydrophilic sol is known as a protective colloid. ► The protective property is expressed most frequently in terms of the gold number. The gold number is the minimum weight in milligrams of the protective colloid (dry weight of the dispersed phase) required to prevent a colour change from red to violet in 10 ml of a gold sol on the addition of 1 ml of a 10% solution of sodium chloride. Examples of some gold numbers of protective colloids: gelatin (0.005 - 0.01), albumin (0.1), acacia (0.1-0.2), sodium oleate (1-5) and tragacanth (2). Page 8 PHARMACEUTICAL APPLICATION OF COLLOIDS Even though it may not be apparent from the name of a specific drug product, in many instances one or more of the ingredients of a drug product may be considered a colloidal dispersion. A number of pharmaceutical compounds will become colloidal dispersions when added to water e.g. certain inorganic salts, all proteins, surface active agents and gums. Mucilages These are viscous, aqueous systems containing gums, either dissolved or suspended e.g. acacia mucilage and tragacanth mucilage. Gels These are colloidal systems, as will be seen in the section on gels. Collodions Collodions are liquid preparations containing pyroxylin in a mixture of ether and alcohol. Collodions leave a flexible, protective layer over the site of application after being painted on the skin and allowed to dry. They may provide a means of maintaining contact of a drug with the skin for a prolonged period and are also used to seal minor cuts and wounds. The most commonly used preparation is one for removing warts and corns and contains salicylic acid and lactic acid in collodion (keratolytic agent). Ionic versus colloidal ► Silver salts are used externally as germicides/antimicrobials. Ionic silver salts cause a lot of irritation. Colloidal silver chloride, and silver iodide have the same efficacy (plus more) but do not cause irritation. ► Colloidal iron is less astringent than crystalloidal iron. ► Coarsely powdered sulphur is poorly absorbed when administered orally we say that it has poor bioavailability. The same dose in a colloidal solution may be absorbed so completely that it causes a toxic reaction i.e. bioavailability is much higher. Natural and synthetic polymers Polymers are macromolecules formed by polymerization or condensation of smaller, noncolloidal molecules. Natural polymers - proteins are important natural colloids and are found in the body as components of muscle, bone and skin e.g. collagen, albumin. Synthetic polymers – are widely used in a variety of drug delivery systems. (see under Formulation) Blood plasma substitutes (see pamphlet on Rheomacrodex®) “Effective colloids retain water in the vascular space and thereby improve haemodynamics” These are colloidal dispersions with a particle size such that they are retained in the blood vessels for an adequate time e.g. natural polymers such as 10% dextran 40 and hydroxyethyl starch are macromolecules also used as plasma substitutes. Dialysis Because of their size, colloidal particles can be separated from molecular particles with relative ease using the separation technique of dialysis. ► A semipermeable membrane of collodion or cellophane is used, the pore size of which will prevent the passage of colloidal particles but will permit small molecules and ions such as urea,, glucose and sodium chloride to pass through. ► Dialysis occurs in vivo. Ions and small molecules pass readily from the blood, through a natural, semipermeable membrane, to the tissue fluids; the colloidal components of the blood remain within the capillary system. ► The principle of dialysis is utilized in the artificial kidney, which removes small, molecular impurities from the body by passage through a semipermeable membrane. (Polymers in )…Formulation ► The protective ability of hydrophilic colloids is used to prevent the coagulation of hydrophobic particles in the presence of electrolytes. ► Hydrophilic colloids are used in suspensions as suspending agents to retard sedimentation as they increase the viscosity of the medium. ► Polymers are extensively used in drug delivery systems to either protect the drug or modify its release characteristics e.g. tablet coatings such as the cellulose derivatives which are natural polymers from vegetable sources e.g. hydroxypropyl methylcellulose, methylcellulose applied to solid dosage forms to protect drugs from a variety of factors e.g. atmospheric moisture or degradation under the acid conditions of the stomach. e.g. drugs contained in polymer matrices to control release rates e.g. Eudragit® consisting of the polyacrylates which are synthetic but biodegradable