Geomorph Lec 2 – Chap 3 – Weathering
advertisement

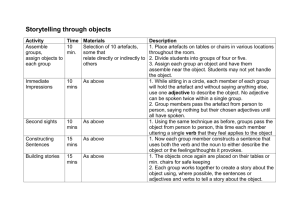
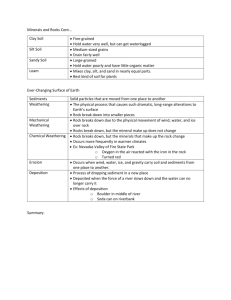
Geomorph Lec 2 – Chap 3 – Weathering Intro – a. Wthrg defined: disintegration & decomposition of rx & mins as a result of physical & chemical processes (does NOT encompass “erosion” and “transportation” b. Changes primarily in situ (in place) c. “Wthrg prepares the way for erosion by weakening the rock & making it susceptible to mass mvmt & removal” i. IMPORTANT concept: materials will continue to break down until they produce end products that are in equilibrium with the environment I. Why is this important? Think about stability field of rx & minerals..and that in nature, the drive toward stability controls a great many processes. Ign, mm rx Pressure Temp Are rx & mins that are precipitated out of a melt at very high P & T going to be stable at low P & T?? What do YOU think? Much like living things, mins have a “comfort zone”, the zone in which they were created…if external environmental conditions start to change, the mins respond by Weathering d. Wthrg zone represents intersection of all Earth’s spheres: bio, hydro, litho, and atmosphere…lots of stress on a mineral that was formed 10 km under Earth’s surface! e. Weathering creates diff “profiles”, depending upon climate: i. Arid climates (no water) produce: 1. angular slopes 2. steep cliffs 3. loose debris at base 4. lots of bedrock exposure 1 ii. Humid climates (much water) produce: 1. smooth, rolling hills 2. gently rounded slopes 3. deep accumulation of weathered material (soil) 4. little exposure of bedrock Dominant wthrg processes change from climate to climate II. Mechanical weathering a. Intro: Wthrg Defined: “breakdown of rx by physical processes, w/ no change in chemical or mineral composition” i. Primary drivers: stresses inside rx, stresses outside the rx ii. Internal stress of rx tend to press outward as overburden stress is released, confining pressure (cp) is reduced (“unloading”). iii. Other stresses include: 1. “Wedging” from ice, salt xls, plant growth 2. Plucking action of colloids 3. Earth-moving by organisms b. Unloading (rock exfoliates) i. High cp results from rock overburden: 1. F=ma 2. Wt=mg 3. Pressure = Force / Unit Area Surface time 1 Lots weight, Surface time 2 Less weight, low cp high cp Rk compressed, high internal forces push out Same high internal forces push against weaker cp, rk expands Fracturing accompanies unloading…occasionally in mining, a face will explosively “fly apart” when unloaded rapidly by a mining operation. 2 Quick aside – Hartmann’s Law – In the strain ellipse caused by differential compression by σ1, σ 2, σ 3, predictable pattern of shear and extension fractures is produced. σ1 (maximum prin stress, always compressional) σ3 (minimum prin stress, sometimes extensional) Shear fractures form at 30o to max principal stress Extension fractures form parallel to max principal stress (You will see this much more in Dr. O’Brien’s Structural Geology course…) BOTTOM LINE: If there are not enough fractures along which a rk can expand when cp drops, more fracturing will occur to accommodate the need for the rk to expand in the shallow subsurface See fig 3-1…fracs become more abundant [spacing decreases] the closer the rk gets to the surface…. And more fracs mean easier penetration by water, salt, plant roots, all of which wedge the cracks open even more. Once the cycle starts, it is self-perpetuating until the rk is reduced mechanically in size to tiny pieces, all of which fall off a cliff face or hill side and fall or slide to the base of a hill. Particularly interesting…exfoliation planes, seen often in granite, and often parallel to the surface…look at the geometry of the local stress field at the near surface: σ3 σ1 Lack of overburden produces vertical σ3, so rock will “unload” in direction of least stress, just like a mineral xl will grow in the direction of least stress, producing foliations during m-m. See Fig 3-2 – classic! 3 You can even find places where these sheeting joints will be parallel to surface in concave upward patterns, like in U-shaped valleys and cirques… c. Freeze-thaw cycle of H2O – VERY IMPORTANT – think…. “potholes” mechanism is simple…when water freezes it expands 9%. So observe a filled crack with water: And the crack when the water freezes: Now thaw the ice, fill the crack with water again… Then freeze it again….. Notice how much the original fracture has expanded. See Fig 3-4 showing a shattered boulder This is an exceedingly important phys wthrg mechanism in the mid latitudes where water is available. Less prevalent at equator or at the poles…good exam question here….WHY?? 4 d. Crystal growth (salt, carbonate, etc) – mechanism of wthrg is essen. same as growth of ice xls…expansion forces rk to expand. Note fig 3-5 – fence post shattered by salt xl growth nr Great Salt Lake Interesting story of two Egyptian obelisks w/ hieroglyphics. One moved to NYC in 1879. Now after 100 yrs in humid climate, the NYC obelisk writing is illegible, while that in Egypt still legible after 3000 yrs. But wthrg can occur in arid climates too. See Fig 3-6 of Egyptian pyramid states of wthrg. e. Thermal expansion / contraction – several interesting experiments suggest this mechanism is minor, except in cases of major forest fires, lightning, strikes, etc. f. Wetting and drying – can help disintegrate shale. This is because a number of clay minerals swell with add’n of water, then contract when H2O dries. This can be a significant wthrg force…swelling in montmorillonite can be up to 100% ! Author mentions pedestals in SW US. g. Colloidal plucking – soil colloids pluck of pieces of rk...importance of process “remains to be determined”. h. Organic activity – “difficult to measure directly….” So its importance as a PHYSICAL weathering agent is subject to debate. However, author admits that heaved sidewalks and rocks pried apart by roots “attest to the force of root growth”. Even lichens attach firmly to rock. III. Chemical weathering a. Intro – defined: “decomposition of rx by processes that change the chem composition of original material” In reality, chem & mech wthrg work together to break up rx at the surface Most vivid example – fractures dramatically increase surface area that is exposed to chem processes – see Fig 3-8 Equilibrium is once again mentioned…most rx at surface are in major disequilibrium relative to the processes at depth under which they originated. As a result, chem processes are at work to change the rock mineralogy and chemistry to products that are stable in Earth’s surface environment. 5 b. Role of water – as in phys wthrg, role is hard to overstate….water is of huge importance in wthrg. Roles include a medium of exchange of elements between rx and atmosphere, participation in actual rxns, and washing away of rxn products to make way for next round of chem rxns. Chem wthrg rates are essentially proportional to amt of precip in an area, so chem wthrg is typically most active in wet climates. Water has unique dipolar structure (Mickey Mouse) that creates + and – electrical poles, making it attractive to many minerals, especially those with OH-: + H H O _ H2O can act in the same way a bar magnet does: that is, “opposites attract” Water is a powerful solvent (dissolver) because: 1. Water’ electrical charge can also pull ions off of mineral surfaces, 2. H2O can surround ions, isolate them, keep them in solution Also, the Hydrogen bonds act to keep the H2Os attracted to each other (+ attracted to -), so there is a lot of surface tension c. Solutions – mineral constituents get dissolved into water, then are moved with surface and groundwater flow. Local chemical conditions (Eh and pH) dictate when and where solution occurs, and when deposition occurs “virtually all chem wthrg involves some solution, during which the original material is broken down chemically” The basic mechanism is: “attraction of dipolar water for incompletely bonded ions” (that is, those that have some excess electrical charge that attracts the H2O), and then water enters the xl structure. H2O essentially latches onto (or surrounds) ions, then carries them off in solution 6 2 types of dissolution: Congruent – all ions stay in solution o Examples – o Gypsum o Halite (salt) o Quartz Another example of congruent…perhaps the most important controlling rxn on the planet….dissolution of calcite. Why important? Linked with atmospheric CO2 (as in global warming…) In terms of wthrg significance, Carbonated water (eg, H2CO3, carbonic acid) is usually found in nature, not just pure H2O, and this has more dissolving power than pure H2O Very important rxn is shown on p.25, the carbonate equilibrium rxn….this controls a lot of transfer of CO2 from atmosphere into ground- and ocean water, and vice versa Incongruent dissolution also very important to wthrg, because new minerals (wthrg products) are formed: o Examples o Feldspar breaks down to form clay minerals o Limestone and marble are dissolved in humid regions of the country, creating caves & sinkholes. After dissolution, these mins are often re-precipitated downstream. o Silica can also be dissolved, but with much more difficulty than limestone d. oxidation-reduction reactions i. these rxns involve electron transfer 1. oxidation = lose an electron 2. reduction = gain an electron ii. most famous example is O, which is eager to attract electrons in order to fill its shells, and reduce its state from O to O2iii. very common rxn: ferrous iron Fe2+ oxidized to ferric iron Fe3+ while in the presence of O through H2O (the rusting process). This can be a very fast (overnight) reaction, resulting in color changes in “earth tone” range (yellow, brown, red) iv. these rxns also involve many common mins with ferrous (Fe2+) iron: 1. pyroxene 2. amphibole 3. olivine 4. biotite 7 these mins wthr to form various ferric oxide mins: 1. hematite 2. limonite 3. goethite example: olivine in presence of wtr plus oxygen forms hematite oxidation often takes place in presence of wtr because of oxygen in solution however, water can also have oxygen deficient conditions, which leads to reduction Important note, p.26: Removal of Fe from many ferromagnesian silicates (e.g., olivine, pyroxene, amphibole, biotite) greatly WEAKENS them and makes them PRONE TO WEATHERING Note too that reduction occurs in the near surface, but not to the extent that oxidation does, because O2 is abundant in the zone of aeration above the water table. Locally, organic decay in swamps, deep ocean, quiet water, can lead to oxygen deficiency and reducing conditions. These conditions produce, among other things, oil and gas accumulations. e. Ionic exchange This is an extremely important component to chemical wthrg. This is the substitution of one ion for another…requirements are usually similar size, similar charge. Na ----- K Ca <-> Mg <-> Si <-> Al Interesting rule of thumb re: Ionic potential (IP) = Z = charge r radius if IP < 3, ions will remain in solution these include: K+, Na+, Ca++, Fe++, Mg++ Ions with IP > 3 include: Fe+++, Al+++, Si+++ 8 Contrast the ionic potential with cation exchangeability: depending upon the host, cations may be tough to pry out (in feldspars), easy to attract and hold (in clay mins). Sidebar here: clay mins are excellent sites for “fixing” (holding) some types of nasty heavy metals in the xl lattice, keep them in the soil, rather than letting them stay mobile and enter the groundwater supply. f. Hydrolysis – addition of H+ and OH- to mins g. Carbonation – carbonate equilibria equation very important Add’n of CO2 to water in the system will drive to create carbonic acic, which will further dissociate and attack CaCO3: CO2 + H2O H2CO3 H+ + HCO3A companion reaction: H2CO3 + CaCO3 Ca++ + 2HCO3In essence, as you push CO2 into these equations from the left, the rxns get pushed to the right…that is, adding CO2 to groundwater tends to create more H2CO3 (upper eqn), which in turn dissolves CaCO3- (lower eqn) into Ca++ and bicarbonate. If you start to pull CO2 out of the system, rxn will move toward the left, and tend to precipitate CaCO3 (lower eqn). h. Hydrolysis and carbonation – essentially stating that hydrolysis (production of H+) is facilitated by the carbonation rxn This happens a lot in the shallow subsurface around plant roots, which are the sites of a lot of CO2 - the more CO2 available, the more the tendency to produce carbonic acid, which then dissociates to H+ ions and bicarbonate ions (HCO3-) i. Hydration – things are hydrated when water is combined with them…you hydrate yourself when you drink fluids after a workout…. Classic geol case is change from anhydrite (CaSO4) to gypsum (both typically assoc with sedimentary evaporate deposits) Addition of wtr causes expansion and weakening of xl lattice (important phys wthrg effect) 9 Another example of hydration is the swelling of clays mins such as montmorillonite j. Chelation – this involves “grabbing” of metal ions by another compound This is important because metal ions such as Fe and Al, normally immobile during wthrg, can become mobilized, usually by organic compounds IV. Controls on the rate & character of weathering (combination of factors) a. Parent material – what do you start with? i. the resistance of mins to chem & phys wthrg control the resistance to wthrg in general ii. other phys factors also important: 1. joints 2. bedding planes 3. porosity Table 3-2 says it all – the REVERSE of Bowen’s Rxn Series! b. Mineral stability – soluble mins least resistant to chem weathering: 4. Halite 5. Gypsum 6. Calcite 7. Aragonite 8. Dolomite (so these are primarily evaporite and carbonate mins that precipitated out of water to begin with…. Makes sense that they would be easily dissolvable, yes?) chem wthrg products such as the clay minerals & iron oxides illite kaolinite montmorillonite limonite are RESISTANT to wthrg, because they are the end products heavy access mins (rutile, ilmenite, corundum) are inert & resistant silicate min resistance is controlled by bonding (e.g., Si very strongly bonded to O, creating very stable SiO2) and more complex breakdown of sheet and chain silicates involves breaks between other bonds, not the one between Si and O. 10 c. Climate climate very important to weathering…”establishes the temperature and moisture conds at / near Earth surface” NOTE: “THE MOST SIGNIFICANT LONG-TERM CONTROL OF WEATHERING PROCESSES” Number of examples of importance of climate Climate controls freeze-thaw (v. important phys wthrg process) doesn’t happen unless you have moisture and cold temps Climate controls soil types, even to the point where similar soils can develop from diff parent rock types Climate controls the amt & distribution of water, which is extremely important to chem wthrg o Moist tropical regions (near equator) experience very deep wthrg (Fig 3-22) o Arid regions (30o N & 30o S, also high polar latitudes) experience little chem wthrg, because there is no water (Fig 3-22). What little water there is comes down torrentially, and is not effective at infiltrating the soil….so most rainfall in arid regions results in large amt runoff, not infiltration and increased soil moisture Lots of info on Fig 3-22 to ponder In tropics, wthrg is intense & deep, progression develops: Fresh rk > little chem alteration > illite-montmorillonite > kaolinite > alum & iron oxides In deserts, not much wtr, and not much wthrg, except for some carbonates Mid latitudes have mod rainfall and temp, so you see moderate wthrg and soil development Polar regions – little wtr or temp changes, so little wthrg d. Vegetation – also a control on weathering, but very much linked to precipitation / climate an important function of veg is to slow down the rate of precip runoff, and increase infiltration another important function is to contribute – a lot – to soil development in several diff ways: the roots break things up physically as organic material dies, it accumulates on surface, triggering several processes: - releases organic acids (H+) that attack rx - releases organic colloids (particles) that promote ion exchange and chelation 11 - H+ ions nr root tips exchange with rx & mins for nutrient cations for plants: Ca++, K+, Na+, etc. These nutrients act as fertilizers for plants, and this process must be taken into acct when growing crops: must rotate crops or you will quickly draw certain nutrients out of soil if you deplete natural cations, must use fertilizer or plants / crops won’t grow e. Topography altitude slope are the two important topo-related factors that control wthrg at really high altitudes above treeline (i.e., above vegetation), freeze-thaw can be important for phys breakdown of rx at lesser altitudes, vegetation holds wtr & promotes chem wthrg slope controls water table position, and amt of water avail for wthrg typically, steep slopes are not the site of much vegetation…slope must flatten out in order for wtr to accumulate and wthrg to occur to any signif depth to create soils how many steep cliff faces have YOU seen covered with soil??? Also, direction (aspect) of slope faces can be important locally…in N hemi. South-facing slopes are warmer, have less snow that can translate to grdwtr during thaw These can be seen in some famous rule-of-thumb examples: One way to tell “North” in the woods is that moss only grows on the North sides of trees Same (sometimes) for lichen on rx f. Time – also an important control on wthrg Easterbrook suggests a direct relation between time and wthrg (this is true if all other factors are equal, but not true if you start considering presence or absence of wtr, etc). Remember the earlier example of Egyptian hieroglyphics in Egypt (3000 yrs w/ no major wthrg) vs NYC (trashed after 150 yrs in Central Park) (ooh…good exam question!!) 12 g. Weathering Rates summary of wthrg controls: function of 5 controlling factors: parent material climate vegetation topography because of wide variability in the way these factors interact, generalizations about wthrg are difficult but attempts have been made to msr wthrg rates of rx of known ages: exptl lab studies (subject to serious questions, like can you replicate field conditions in the lab?) wthrg dated materials used by man (tombstones, monuments, building stone) study amt of dissolved elements in streams one thing DOES appear to be apparent: “rates of chem wthrg processes decrease with time” however, you MUST consider factors like atmospheric pollution (e.g., acid rain, high levels of CO2 which could promote creation of carbonic acid, H2CO3) V. Effects of wthrg OK, so now it’s time to look at the actual results of processes….how does it change things?…what changes to what? Processes combine to produce a VARIETY of Geomorphic Features – - clay formation - granular disintegration - wthrg pits - spheroidal wthrg - chem wthrg & stripping - cavernous wthrg - soils A. Clay Mineral formation These are sheet minerals with lots of cations and anions Sheets can be organized differently 1:1 (Si sheet:Al sheet), 2:1 (Si sheet:Al sheet)), resulting in many diff minerals Fig 3-31 shows how cations and anions are “wedged” in between sheets, causing changes in mineralogy & xl structure 13 These sheets also control how things wthr: 1:1 sheets like kaolinite do NOT have room for excess cations or wtr….so with low cations & anions, they have low CEC (electrically neutral), and do not exert a strong draw to other cations & anions. So these clays do NOT swell. 2:1 sheets include micas, smectite In smectites, Fe2+ and Mg2+ frequently substitute for Al3+ Also, exchange of cations and water is easy and widespread So smectites are known as “swelling clays”…montmorillonite is most well-known smectite In micas, illite & vermiculite are most significant. Illite is v. similar to montmorillonite, so it can alter to that. Chlorite is also a 2:1 sheet, frequently found in soils and in low-grade M-M terrane. Since bonding is strong, no swelling occurs. First-generation clays frequently alter to other clay mins, and mixed-layer clays 2:1:1 are also common Jackson et al (1948) proposed this sequence: Mica > vermiculite > montmorillonite > kaolinite , but this is a bit too simplistic Easterbrook also notes that, with enough time, granites can COMPLETELY weather to clay mins….. B. Granular disintegration – primarily in granites and sandstones – essentially grainby-grain failure and removal from an initially hard rk C. Wthrg pits and etched knobs – common in granitic terranes D. Spheroidal wthrg- produces rounded, boulder-like forms (Fig 3-35) When fspar changes to clay, incr in volume takes place, so xline rk can be broken up (in same way that wtr increases in vol when it changes to ice) Whtrg takes place preferentially along joint planes, which start the rounding process. These look a lot like stream-rounded boulders…! E. Cavernous wthrg (or honeycomb wthrg) Fig 3-36 – mostly a product of granular disintegration F. Deep chem wthrg and stripping – postulated evolutionary sequence that can produce inselbergs (protruding bedrock from a peneplain of wthrd rk) is: 14 Wthrg front pushes downward Pulse pf stripping occurs Process repeats itself Time 1 Time 2 Inselberg Time3 Present surface Bedrock G. Soil development – “perhaps the most important effect of wthrg” (especially if you like to eat….) Soil defined: “a wthrg residue that has become differentiated with depth into horizons” So how do horizons develop? accumulation of organic material at or near surface leaching of parent material to the point that large amts of one or more minerals are removed accumulation of organic material in upper part of profile accumulation of wthrg products with depth what are the horizons? O horizon (decomposing plants, little mineral content) A horizon (dark due to decaying organics, generation of organic acids, and leaching of mins downward – A is known as zone of leaching (occasional E horizon here) B horizon (Zone of accumulation) – material that has accumulated by downward mvmt from O & A horizons, and the parent material below has also been severely weathered – one characteristic is that there is more clay here than above or below There are several variations on the B horizon, including Bt and Bk C horizon (decayed rock horizon) R horizon (unweathered parent rock) H. Soil maturity – some generalizations: the more erosion, the thinner and less mature the soil the less mature the soil, the more it reflects parent rk composition 15 I. Paleosols (ancient soils) Typically preserved by burial They are often associated with uncomformities (makes sense, as unconf is an erosion surface) They are useful in interpretation because they indicate enough time exposed subaerially to have soils develop They can also help us interpret ancient climates J. Soil classification – important skill for soil scientists, especially those who aid farmers END CHAPTER 3 e.g., illite > montmorillonite vermiculite > chlorite smectite > chlorite > kaolinite 16 17