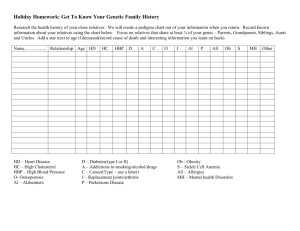
Good practice points
advertisement

Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand Provisional National HBP/Upper GI Tumour Standards Working Group 2013 Citation: National HBP/Upper GI Tumour Standards Working Group. 2013. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional. Wellington: Ministry of Health. Published in December 2013 by the Ministry of Health PO Box 5013, Wellington 6145, New Zealand ISBN 978-0-478-41583-4 (online) HP 5783 This document is available through the Ministry of Health website: www.health.govt.nz or from the regional cancer network websites: www.northerncancernetwork.org.nz www.midland cancernetwork.org.nz www.centralcancernetwork.org.nz www.southerncancernetwork.org.nz Contents Introduction ............................................................................................................... 1 Background ..................................................................................................... 1 Objective ......................................................................................................... 2 How the HBP/upper gastrointestinal cancer service standards were developed ........................................................................................................ 2 Equity and Whānau Ora .................................................................................. 3 Summary of the clinical standards for the management of HBP/upper gastrointestinal cancer services ....................................................................... 4 Summary of standards..................................................................................... 5 1 Prevention and Early Identification................................................................... 9 Rationale ....................................................................................................9-10 Good practice points ...................................................................................9-10 2 Timely Access to Services ............................................................................. 13 Rationale ....................................................................................................... 13 Good practice points ...................................................................................... 14 3 Referral and Communication ......................................................................... 15 Rationale ....................................................................................................... 15 Good practice points ...................................................................................... 15 4 Investigation, Diagnosis and Staging ............................................................. 18 Rationale .................................................................................................. 18-23 Good practice points ................................................................................. 18-23 5 Multidisciplinary Care ..................................................................................... 25 Rationale .................................................................................................. 25-28 Good practice points ................................................................................. 25-28 6 Supportive Care............................................................................................. 29 Rationale ....................................................................................................... 29 Good practice points ...................................................................................... 29 7 Care Coordination ......................................................................................... 31 Rationale ....................................................................................................... 31 Good practice points ...................................................................................... 31 8 Treatment ...................................................................................................... 33 Rationale .................................................................................................. 33-40 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional iii Good practice points ................................................................................ 33-40 9 Follow-up and Surveillance ........................................................................... 42 Rationale .................................................................................................. 42-45 Good practice points ................................................................................ 42-45 10 Clinical Performance Monitoring and Research ............................................. 46 Rationale .................................................................................................. 46-48 Good practice points ................................................................................ 46-48 Appendix 1: National HBP/Upper GI Tumour Standards Working Group Membership .................................................................................................. 49 Appendix 2: Glossary ............................................................................................. 51 Appendix 3: References ......................................................................................... 59 iv Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Introduction Background Cancers of the upper gastrointestinal tract mostly arise in the oesophagus, stomach, liver, biliary system, pancreas and duodenum. (Cancers covered in these standards include hepatobiliary, pancreaticoduodenal and gastro-oesophageal cancers; the group as a whole is referred to as ‘HBP/upper GI’ throughout this document.) These cancers are particularly formidable to treat because of the many different types of cancer involved, complex anatomy, the magnitude of surgical treatment required, the requirement for multi-modal management and a generally poor prognosis relative to most other cancers. The median survival of patients with pancreatic cancer in New Zealand is only 92 days (Phillips et al 2002); as such, pancreatic cancer is the most common cause of HBP/upper GI cancer death. In comparison with the five-year survival rate for patients with all cancers in New Zealand, 60 percent, the five-year survival rate for patients with stomach cancer is 20 percent (Ministry of Health 2006). Māori have 25 percent worse survival than non-Māori after diagnosis (Soeberg et al 2012). Another challenge is that HBP/upper GI cancers are not common. In 2009, there were 253 new cases of primary liver and biliary cancers, 472 new cases of pancreatic cancer, 370 new cases of stomach cancer and 257 new cases of oesophageal cancer (Ministry of Health 2012a). Individual district health boards (DHBs) therefore do not see high volumes of them. When this is coupled with higher treatment-related morbidity and mortality, especially for oesophagectomy, hepatectomy and pancreatoduodenectomy, it is no surprise that there is a relationship between hospital and surgeon volume and patient outcome. The debate about the best model for the delivery of cancer care for rare and complex tumours in New Zealand (Beenen et al 2013) emphasises the need to make best use of our available expertise and facilities and strive to provide the best quality care, and equitable access to it. There is also variation in intervention rates and outcomes between DHBs and, overall, these may be less than they should be. Data suggest that the resection rate for gastro-oesophageal cancers in New Zealand is 15 percent (34–53 patients per year) – this is lower than it should be when compared with the international published literature (Martin 2002). There are incidence inequalities between Māori and non-Māori for these cancers. For example, between 1988 and 1997, Māori had higher standardised incidence rates of pancreatic cancer (7.3 per 100,000) compared with other ethnic groups, and Māori females had the highest reported female rate in the world (7.2 per 100,000) (Phillips et al 2002). Most of the survival disparity between Māori and non-Māori patients is accounted for by higher rates of co-morbidity, more limited health service access, and lower rates of cancer treatment (Hill et al 2012). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 1 Another challenge is the high rates of chronic hepatitis B and C infections in New Zealand, which account for 75 percent of all primary liver cancers (Weir et al 2002). Virtually all hepatocellular carcinoma in Māori is caused by chronic hepatitis B infection; alcoholic liver disease is less prevalent in these patients than in non-Māori (Chamberlain et al 2013). Half of patients living with hepatitis B infection are not aware of it. Similarly high rates of gastric cancer are also seen in Māori. Most are distal cancers, rather than proximal tumours, and are associated with high rates of infection with helicobacter pylori.1 Improving the outcomes of patients with HBP/upper GI cancer requires useful data on the process, treatment and outcomes of the cancer journey. By defining and monitoring standards it will, in time, be possible to make informed decisions about best treatment for these patients in New Zealand, and to find ways to improve cancer care. Objective Tumour standards for all cancers are being developed as a part of the Ministry of Health’s ‘Faster Cancer Treatment’ (FCT) programme’s approach to ensuring timely clinical care for patients with cancer. The standards will promote nationally coordinated and consistent standards of service provision across New Zealand. They aim to ensure efficient and sustainable best-practice management of tumours, with a focus on equity. The standards will be the same for all ethnic groups. However, we expect that in implementing the standards DHBs may need to tailor their efforts to meet the specific needs of populations with comparatively poorer health outcomes, such as Māori and Pacific people. How the HBP/upper gastrointestinal cancer service standards were developed These standards were developed by three working groups – gastro-oesophageal, hepatobiliary and pancreaticoduodenal – representing key specialities and interests across the HBP/upper GI cancer pathway of care. Each group was chaired by a lead clinician (see Appendix 1), and had access to expert advisors in key content areas, including the Northern Cancer Network’s Māori Leadership Group. These standards recognise the need for evidence-based practice. Numerous evidence-based guidelines and standards already exist, so the standards in this document have largely been developed by referring to established national and international guidelines in the HBP/upper GI tumour stream (see Appendix 4). Where no clear evidence was available, expert opinion was obtained through the National HBP/Upper GI Tumour Standards Working Group and its advisors. 1 2 Personal communication D Sarfati and T Blakely 2013. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Tumour-specific national standards were first developed for lung cancer in the Standards of Service Provision for Lung Cancer Patients in New Zealand (National Lung Cancer Working Group 2011); these standards have already made improvements to service delivery and clinical practice. Subsequently provisional standards have been developed for an additional ten tumour types: bowel, breast, gynaecological, lymphoma, melanoma, myeloma, head and neck, sarcoma, thyroid and upper gastrointestinal. The Ministry of Health required all tumour stream work groups to: Maintain a focus on achieving equity and whānau ora when developing service standards, patient pathways and service frameworks by ensuring an alignment with the Reducing Inequalities in Health Framework and its principles (Ministry of Health 2002). Equity and Whānau Ora Health inequities or health disparities are avoidable, unnecessary and unjust differences in the health of groups of people. In New Zealand, ethnic identity is an important dimension of health disparities. Cancer is a significant health concern for Māori, and has a major and disproportionate impact on Māori communities (Ministry of Health 2008). Inequities exist between Māori and non-Māori in exposure to risk and protective factors for cancer, in incidence and outcomes, and in access to cancer services. Barriers to health care are recognised as multidimensional, and include health system and health care factors (eg, institutional values, workforce composition, service configuration and location), as well as patient factors (eg, socioeconomic position, transportation and patient values). Addressing these factors requires a population health approach that takes account of all the influences on health and how they can be tackled to improve health outcomes. A Whānau Ora approach to health care recognises the interdependence of people; health and wellbeing are influenced and affected by the ‘collective’ as well as the individual. It is important to work with people in their social contexts, and not just with their physical symptoms. The outcome of the Whānau Ora approach in health will be improved health outcomes for family/whānau through quality services that are integrated (across social sectors and within health), responsive and patient/family/whānau-centred. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 3 In preparing these standards, the Northern Cancer Network’s Māori Leadership Group provided expert advice to the working groups based on eight principles to identify and address issues of equity in access, process and outcome. These principles were specifically a commitment to mana whenua and Māori communities, Whānau Ora, health equity, self-determination, indigeneity, ngā kaupapa tuku iho, whole-of-system responsibility and evidence-based approaches. These principles are woven throughout the standards, and support the inclusion of supporting rationale, good practice points and monitoring. These standards will address equity for Māori patients with HBP/upper GI cancers in the following ways. The standards suggest offering screening to Māori 10 years earlier than for non-Māori. The standards focus on improving access to diagnosis and treatment for all patients, including Māori and Pacific. Barriers to attendance (eg, mobility, cost, co-morbidities and compliance issues) will be identified at patients’ first specialist assessment (FSA). Ethnicity data will be collected on all access measures and the FCT indicators, and will be used to identify and address disparities. Good practice points include health literacy and cultural competency training for all health professionals involved in patient care. Information developed or provided to patients and their family/whānau will meet Ministry of Health guidelines (Ministry of Health 2012c). Summary of the clinical standards for the management of HBP/upper gastrointestinal cancer services Format of the standards Each cluster of standards has a title that summarises the step of the patient journey or the area on which the standards are focused. This is followed by the standard itself, which explains the level of performance to be achieved. The rationale section explains why the standard is considered to be important. Attached to most of the clusters of standards are good practice points. Good practice points are either supported by the international literature, the opinion of the National Upper GI Tumour Standards Working Group or the consensus of feedback from consultation with New Zealand clinicians involved in providing care to patients with HBP/upper GI cancer. Also attached to each cluster are the requirements for monitoring the individual standards. 4 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standards of service provision pathway The HBP/upper GI cancer standards are reflected in the following pathway. Summary of standards The standards of service provision for the management of upper gastrointestinal cancer have been divided into 10 clusters: prevention and early identification timely access to services referral and communication investigation, diagnosis and staging multidisciplinary care supportive care care coordination treatment follow-up and surveillance clinical performance monitoring and research. The standards are as follows. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 5 Prevention and early identification Standard 1.1: People are offered evidence-based information on risk factors, prevention and early detection of HBP/upper GI cancer. Standard 1.2: People at significantly increased risk of HBP/upper GI cancer are offered screening for HBP/upper GI cancer. Timely access to services Standard 2.1: Patients referred urgently with a high suspicion of HBP/upper GI cancer receive their first cancer treatment within 62 days. Standard 2.2: Patients referred urgently with a high suspicion of HBP/upper GI cancer receive their FSA within 14 days. Standard 2.3: Patients with a confirmed diagnosis of an HBP/upper GI cancer receive their first treatment within 31 days of the decision to treat. Referral and communication Standard 3.1: Patients with suspected HBP/Upper GI cancer are referred to secondary and tertiary care following an agreed referral pathway. Standard 3.2: Patients and their general practitioners (GPs) are provided with verbal and written information about HBP/upper GI cancer, diagnostic procedures, treatment options (including effectiveness and risks), final treatment plan and support services. Standard 3.3: Communications between health care providers include the patient’s name, date of birth, national health index (NHI) number and contact details, and are ideally electronic. Investigation, diagnosis and staging Standard 4.1: Patients with HBP/upper GI cancer have timely access to experts (including radiologists, endoscopists, pathologists and surgeons) who are appropriately trained and credentialed. Standard 4.2: Patients with HBP/upper GI cancer are diagnosed, investigated and staged in facilities, and with equipment, that meet nationally agreed standards. Standard 4.3: Patients with HBP/upper GI cancer are diagnosed, investigated and staged using nationally standardised protocols. Standard 4.4: The results of the diagnosis, investigations and staging of patients with HBP/upper GI cancer are reported by experts in a standardised and agreed format, encompassing all relevant aspects, and are interpreted in the light of all relevant clinical information. 6 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Multidisciplinary care Standard 5.1: All patients with the following are presented at a multidisciplinary meeting (MDM): a confirmed diagnosis of HBP/upper GI cancer a high suspicion of HBP/upper GI cancer. Standard 5.2: The MDM comprises members of the multidisciplinary team (MDT), including surgeons, radiologists, gastroenterologists, medical and radiation oncologists, cyto/histopathologists and cancer care coordinators. Standard 5.3: Data are available in standardised electronic format for MDM discussion, and include all relevant information about patients and investigations. Standard 5.4: All patients with confirmed HBP/upper GI cancer have their treatment plan discussed at an MDM; recommendations are clearly documented in the patient’s medical records and communicated to the patient and their GP. Supportive care Standard 6.1: Patients with HBP/upper GI cancer and their family/whānau have equitable and coordinated access to appropriate medical, allied health and supportive care services, in accordance with Guidance for Improving Supportive Care for Adults with Cancer in New Zealand (Ministry of Health 2010). Care coordination Standard 7.1: All patients with HBP/upper GI cancer have access to an HBP/upper GI cancer clinical nurse specialist or other health professional who is a member of the MDM to help coordinate all aspects of their care. Standard 7.2: Each regional cancer centre or unit managing patients with HBP/upper GI cancer has designated lead clinicians with specialist knowledge of HBP/upper GI cancers to provide necessary leadership. Treatment Standard 8.1: The treatment of patients with HBP/upper GI cancer is performed by appropriately trained and credentialed experts (surgeons, medical and radiation oncologists, gastroenterologists and interventional radiologists), with a specialty commitment to this field. Access to specialist expertise is available at all times. Standard 8.2: Patients with HBP/upper GI cancer are treated in appropriately equipped facilities offering the required range of services, including interventional radiology and a level one or two intensive care unit. Standard 8.3: Patients with HBP/upper GI cancer are treated according to evidencebased and agreed protocols. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 7 Standard 8.4: Reports of treatment by all specialties of patients with HBP/upper GI cancer (including operation notes) are in a standardised electronic format, and include all of the relevant prognostic criteria. Standard 8.5: Patients with HBP/upper GI cancer have access to a dietitian for assessment and nutritional support before, during and after their treatment. Standard 8.6: Patients with HBP/upper GI cancer are offered early access to palliative care services when there are complex symptom control issues, when a curative treatment cannot be offered or if curative treatment has been declined. Follow-up and surveillance Standard 9.1: When appropriate, patients treated for HBP/upper GI cancer are offered ongoing follow-up at their original referring centre. Standard 9.2: Patient follow-up after HBP/upper GI cancer for the detection of local recurrence and metastases takes place in accordance with international consensus guidelines. Standard 9.3: Follow-up plans include clinical review by appropriate members of the MDT, working in conjunction with the patient, their family/whānau and their GP. Clinical performance monitoring and support Standard 10.1: Data relating to HBP/upper GI cancer beyond the fields required by the Cancer Registry, including treatment data, are reported to existing and planned national repositories using nationally agreed data set fields. Standard 10.2: Data on treatment, clinical outcomes and patient satisfaction are regularly monitored and reported as part of a national audit. Standard 10.3: Patients with HBP/upper GI cancer are offered the opportunity to participate in research projects and clinical trials where these are available. 8 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 1 Prevention and Early Identification Standard 1.1 People are offered evidence-based information on risk factors, prevention and early detection of HBP/upper GI cancer. Rationale There is strong evidence to demonstrate improved peri-operative and long-term outcomes in patients with HBP/upper GI cancer who stop smoking and reduce their alcohol intake. Good practice points 1.1 People are informed that exposure to smoking and alcohol may predispose them to HBP/upper GI cancer. 1.2 Advice on smoking cessation and reducing consumption of alcohol is provided to all those who would benefit from it (Ministry of Health 2011b). 1.3 Primary care clinicians screen for known risk factors for HBP/upper GI cancer, and intervene when they identify people at risk. Monitoring requirements MR1A Monitor smoking cessation initiatives. MR1B Audit results of national public health campaigns for identified risk factors (such as smoking and alcohol consumption). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 9 Standard 1.2 People at significantly increased risk of HBP/upper GI cancer are offered screening for HBP/upper GI cancer. Rationale The early detection of cancer in high-risk individuals is a strategy that will improve patients’ chances of earlier treatment, and their prognosis. Screening should be carried out where it can be undertaken in a cost-effective manner, and should be supported by evidence where possible. Screening should only be undertaken if treatment options are available. Good practice points Gastric cancer 1.4 People at increased risk of gastric cancer are referred to the New Zealand Familial Gastrointestinal Cancer Service according to accepted criteria (Kluijt et al 2012). 1.5 People with a known or suspected E-cadherin mutation are referred to Genetic Health Service New Zealand, the New Zealand Familial Gastrointestinal Cancer Service and a tertiary service managing these patients for consideration of endoscopic screening and prophylactic total gastrectomy. 1.6 Screening gastroscopy is considered for people with Lynch syndrome, familial adenomatous polyposis (FAP), MYH-associated polyposis, Li-Fraumeni syndrome, Peutz-Jeghers syndrome and BRCA2-associated cancer. 1.7 People with known high-risk factors, such as chronic Helicobacter pylori infection (particularly Māori), high alcohol use, heavy smoking, chronic reflux and previous gastric ulcer surgery, are offered early gastroscopy with any new upper GI symptoms. 1.8 Clinicians consider offering screening to Māori 10 years earlier than non-Māori (NZGG 2009). Oesophageal cancer 1.9 Gastroscopy screening for oesophageal cancer is not offered to asymptomatic patients. 1.10 Patients with Barrett’s oesophagus are considered for regular endoscopic screening on a case-by-case basis, in conjunction with international guidelines (AGA 2011; Bennett et al 2012). 10 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 1.11 Patients with known high-risk factors, including high alcohol use, heavy smoking, chronic reflux, achalasia, tylosis and metabolic syndrome, are triaged for rapid access endoscopy if they present with symptoms consistent with upper GI cancer. Hepatocellular carcinoma 1.12 Screening is considered for people at high risk of hepatocellular carcinoma (HCC) in high risk individuals, including those with established liver cirrhosis, regardless of aetiology. 1.13 Ultrasound screening for HCC is performed every six months by appropriately trained personnel in accordance with a standardised protocol covering recall procedures for patients with screening-detected abnormalities (EASL-EORTC 2012; Bruix and Sherman 2011). 1.14 Hepatitis B is a major risk factor for the development of HCC, including in noncirrhotic HBsAg positive patients. The risk of hepatitis B is managed according to the Ministry of Health immunisation schedule and guidelines (Ministry of Health 2013b). 1.15 Patients with the chronic hepatitis B virus receive six-monthly blood tests (from a primary care physician or the Hepatitis Foundation of New Zealand) to monitor liver enzymes and alfa-fetoprotein. Secondary hepatic cancer 1.16 Surveillance for liver metastases in patients with a personal history of colorectal cancer is carried out in accordance with the follow-up recommendations in Management of Early Colorectal Cancer (NZGG 2011). Pancreatic cancer 1.17 People with two or more first-degree relatives who have died of pancreatic cancer and those who have a familial cancer syndrome known to be associated with an increased risk of pancreatic cancer are referred to Genetic Health Service New Zealand and the New Zealand Familial Gastrointestinal Cancer Service (Canto et al 2013). 1.18 Screening of those at high risk commences 10 years earlier than the age at diagnosis of the youngest first-degree relative with pancreatic cancer, and is brought forward a further 10 years if the person is a long-standing smoker. 1.19 Screening of those at high risk is done by magnetic resonance imaging (MRI) scanning, with endoscopic ultrasound with or without fine needle aspiration (FNA) to further investigate any abnormalities detected through MRI (one- to three-yearly). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 11 1.20 A high index of suspicion of pancreatic cancer is maintained for patients with idiopathic acute pancreatitis (over the age of 40), those with new onset diabetes (over the age of 40) and those with chronic pancreatitis (especially when associated with a mass). Pancreatic cystic lesions 1.21 Patients with non-inflammatory pancreatic cystic lesions are investigated in accordance with international guidelines to identify the likelihood of cancer or the risk of developing it (Tanaka et al 2006). Duodenal cancer 1.22 Patients are screened for syndromes that increase the risk of duodenal cancer, including duodenal polyposis and/or some familial cancer syndromes, including FAP, hereditary nonpolyposis colorectal cancer and Peutz-Jeghers. 1.23 Upper gastrointestinal endoscopic screening intervals for duodenal cancer are in accordance with the Spigelman classification (Vasen et al 2008). Monitoring requirements MR1C 12 Monitor extent to which identified population at increased risk are enrolled in surveillance programmes. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 2 Timely Access to Services Standard 2.1 Patients referred urgently with a high suspicion of HBP/upper GI cancer receive their first cancer treatment within 62 days. Standard 2.2 Patients referred urgently with a high suspicion of HBP/upper GI cancer receive their FSA within 14 days. Standard 2.3 Patients with a confirmed diagnosis of HBP/upper GI cancer receive their first treatment within 31 days of the decision to treat. Rationale Timely and equitable access to quality cancer management is important to support good health outcomes for New Zealanders and to reduce inequalities. Key components of successful cancer management include early recognition and reporting of symptoms, expertise in identifying patients requiring prompt referral and rapid access to investigations and treatment. A suspicion of cancer or cancer diagnosis is very stressful for patients and family/whānau. It is important that patients, family/whānau and GPs know how quickly patients can receive treatment. Long waiting times may affect local control and survival for some cancer patients, and can result in delayed symptom management for palliative patients. The standards in this cluster ensure that: patients receive quality clinical care patients are managed through the pathway, and experience well-coordinated service delivery delays are avoided as far as possible. Shorter waits for cancer treatments is a government health target. The FCT indicators (the standards in this cluster) adopt a timed patient pathway approach across surgical and non-surgical cancer treatment, and apply to inpatients, outpatients and day patients. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 13 Good practice points 2.1 When there is a suspicion of HBP/upper GI cancer, the GP or referring specialist refers to, and the referral is received by, the appropriate secondary/tertiary care services within one working day (expert opinion). 2.2 Referrals and reports are distributed electronically (expert opinion). 2.3 Referrers identify barriers to attendance at FSAs (such as mobility, cost, co-morbidities and compliance issues), and address them where possible. 2.4 Strategies are in place to avoid did-not-attends (DNAs), including coordinated clinic times (so that family/whānau can attend together), reminder systems, culturally appropriate information and support services. 2.5 Confirmation of a diagnosis of cancer is tissue diagnosis whenever appropriate. 2.6 If any uncertainty exists regarding a histological diagnosis of cancer, especially in regards to associated precursor lesions and/or marked inflammation, the tissue is reported on by an independent histopathologist. 2.7 Specialists identify barriers to timely assessment and investigation (such as availability of expertise, cost, mobility and language), and address them where possible. 2.8 Specialists primarily responsible for the coordination of assessment, investigation, staging and treatment explicitly and routinely discuss processes with patients and family/whānau. 2.9 Subsequent cancer therapy, such as adjuvant chemotherapy or surgery following neo-adjuvant chemotherapy, is coordinated to avoid clinically unnecessary delay. Monitoring requirements 14 MR2A Track FCT indicators. MR2B Collect and analyse ethnicity data on all access targets and indicators. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 3 Referral and Communication Standard 3.1 Patients with suspected HBP/upper GI cancer are referred to secondary and tertiary care following an agreed referral pathway. Standard 3.2 Patients and their GPs are provided with verbal and written information about HBP/upper GI cancer, diagnostic procedures, treatment options (including effectiveness and risks), final treatment plan and support services. Standard 3.3 Communications between health care providers include the patient’s name, date of birth, NHI number and contact details, and are ideally electronic. Rationale Good communication skills are fundamental to the development of an effective relationship between a patient and health practitioners. Good communication is likely to reduce anxiety, and increase patients’ trust and confidence in cancer care providers. This will increase the chance that they receive the treatment that is most appropriate for them. Good information may improve compliance with treatment, reduce complaints and enhance health outcomes. Communications with other health care professionals There should be rapid and effective two-way information flow between service providers transferring and sharing information on referral, diagnosis, treatment, follow-up and supportive/palliative care. Good practice points Referrals 3.1 All communications between health care providers are electronic where possible (expert opinion). 3.2 The standardised referral letter contains information as to where it should be sent. 3.3 Clinical information provided in the referral letter is agreed by experts in the primary, secondary and tertiary sectors to ensure appropriate prioritisation and decision-making. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 15 3.4 Clinical information is presented in a standardised format. 3.5 A mechanism is in place to ensure that the referrer receives confirmation of the receipt of the referral within one working day of it being sent. 3.6 Where high suspicion of cancer is associated with an inherited pattern or associated with a recognised cancer syndrome, Genetic Health Service New Zealand is involved. Information provided to patients 3.7 Patients specify the way they would like communication to take place early in the care pathway; all patients are entitled to a professional interpreter. Communication may occur through a family member. In some cases, a patient will not want to be informed of the diagnosis or details of their treatment or prognosis (Windsor et al 2008). 3.8 Family/whānau meetings are arranged whenever a patient requests them, and patients determine who attends such meetings. 3.9 The information, treatment and care that patients receive is always culturally appropriate (expert opinion). 3.10 Each new patient with cancer receives information in a language and format appropriate to them; such information meets Ministry of Health guidelines (Ministry of Health 2012c) and covers: general background information about the specific cancer details of treatment options and specific local arrangements, including information about the MDT, support services and whom the patient should contact if necessary details of available support groups and other appropriate organisations details as to when any procedure, test and/or treatment will be undertaken. 3.11 Patients are allowed adequate time to consider treatment options (NHS Wales 2005). 3.12 A professionally trained interpreter is available for all interactions with health care professionals when English is not a patient’s first language. 3.13 All clinicians acknowledge the rights of patients to refuse investigation or treatment. Any discussions on refusal are recorded in a patient’s medical record. Guidance follows the Health and Disability Commissioner Act 1994. 3.14 In the consent process, patients are allowed adequate support, time and information to make informed decisions. Consent discussions are clearly documented. 16 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 3.15 Processes are in place to incorporate information and feedback gained through patient satisfaction surveys. 3.16 All clinicians responsible for communicating with patients and their family/whānau complete health literacy training. Communications between health care providers 3.17 Communications between specialists and referrers occur at least after first assessments, after MDMs and after completion of treatment. 3.18 Communications contain a clear indication of what further role referrers will play in ongoing care. 3.19 Referrers are informed if specialists downgrade an urgent suspected cancer referral to non-urgent (NHS Wales 2005). 3.20 When the referrer is not the GP, all correspondence is copied to the GP. 3.21 Hospital administrative systems have a did-not-attend (DNA) reduction and follow-up policy that entails equity-focused quality assurance. Monitoring requirements MR3A Provide evidence of clear and accessible referral pathways. MR3B Audit actual patient pathways through records of registration of referral, FSAs, dates of diagnosis and first cancer treatments. MR3C Audit correspondence between secondary/tertiary care and GPs. MR3D Provide evidence of culturally appropriate patient and family/whānau satisfaction surveys, and audit complaints processes. MR3E Audit documentation between health care providers. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 17 4 Investigation, Diagnosis and Staging Standard 4.1 Patients with HBP/upper GI cancer have timely access to experts (including radiologists, endoscopists, pathologists and surgeons) who are appropriately trained and credentialed. Rationale It is important that patients with HBP/upper GI cancers have ready access to necessary expertise to allow for efficient, accurate and timely diagnosis, investigation and staging. Incorrect or delayed diagnosis and staging can impair patient outcomes. These cancers are relatively rare, and individual health professionals may have limited experience in diagnosis and staging of them, which makes prompt multidisciplinary review important, in order to ensure optimal treatment decisions are made. Good practice points 4.1 All specialists responsible for the diagnosis, investigation and staging of patients with HBP/upper GI cancers are appropriately qualified, have specialist expertise and are credentialed by the appropriate regulatory body. 4.2 If this expertise is not available, there is an established line of referral to a centre where appropriate expertise is available. Monitoring requirements MR4A 18 Ensure that departments maintain a record of appropriately trained and credentialed staff. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 4.2 Patients with HBP/upper GI cancer are diagnosed, investigated and staged in facilities, and with equipment, that meet nationally agreed standards. Rationale Appropriately resourced facilities are required to accurately diagnose, investigate and stage patients with HBP/upper GI cancers in an acceptable timeframe. Good practice points 4.3 All services involved in the diagnosis, investigation and staging of patients with HBP/upper GI cancer work to high standards of service delivery that encompass management systems, waiting list management, procedural work, examination reporting, provision of clinical advice and quality assurance. Radiology 4.4 Appropriate imaging modalities, such as computed tomography (CT), MRI and positron emission tomography and computed tomography (PET-CT) are provided in accordance with the guidance of local credentialing bodies. Gastroenterology 4.5 Diagnosis of early malignant or highly dysplastic lesions, in particular, is dependent on high-quality endoscopic images, and may require advanced diagnostic endoscopic technologies (including chromoendoscopy, endoscopic ultrasonongraphy and endoscopic mucosal resection); such technologies are available to patients who need them in their regional cancer centre. 4.6 Endoscopic ultrasound with or without FNA is used in preference to CT in the locoregional staging of oesophageal cancer (this is of particular help in identifying more advanced (T4) disease compared to early (T1) disease). Laboratory processes 4.7 Biochemical and immunology testing facilities ensure the availability of agreed tumour markers, so that timely reporting is compliant with nationally agreed investigation pathways. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 19 4.8 Fluorescence in situ hybridisation (FISH) and other advanced methodologies are available in regional cancer centres to facilitate further categorisation of tumours for targeted therapies. 4.9 Lines of referral to a reference laboratory in New Zealand are established, to ensure timely processing of less frequently performed assays. Histopathology 4.10 Histology specimens are processed in an accredited laboratory with adequate fixation and sampling, including specimen photography for complex specimens, to provide high-quality histology slides, including immunohistochemically stained slides. 4.11 Frozen section facilities are available in all centres undertaking surgery for HBP/upper GI cancer. Monitoring requirements MR4B 20 Ensure that departments maintain facilities and equipment against agreed standards. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 4.3 Patients with HBP/upper GI cancer are diagnosed, investigated and staged using nationally standardised protocols. Rationale Variation in practice contributes to worse patient outcomes. Standardisation of protocols for the diagnosis, investigation and staging of HBP/upper GI cancers should improve patient outcomes, ensure equivalent access to appropriate investigation, avoid over-investigation and reduce costs. Good practice points 4.12 Services across the country involved in the diagnosis, investigation and staging of patients with HBP/upper GI cancer unify their protocols, to avoid additional unnecessary studies, make clinically meaningful comparison, permit review of services and outcomes, and facilitate accurate MDM review (NHS Wales 2005). Radiology 4.13 Diagnostic percutaneous liver biopsy may adversely affect prognosis in patients who are potentially eligible for curative surgical treatment; this is only offered following MDM review (expert opinion). 4.14 Contrast-enhanced multiphasic CT and/or MRI is undertaken for the primary diagnostic evaluation of all patients with suspected HBP cancer (NCCN 2011a; 2011b). 4.15 Contrast-enhanced CT of the chest, abdomen and pelvis is undertaken for staging in patients with confirmed HBP cancer (NCCN 2011a; 2011b). 4.16 PET-CT is used for staging of patients with HBP/upper GI cancer on the basis of nationally agreed protocols. Gastroenterology 4.17 Endoscopic retrograde cholangiopancreatography (ERCP) for diagnosis of a dominant biliary stricture is undertaken after an MDM to direct which duct(s) requires drainage and to obtain histological specimens. 4.18 Advanced technical procedures, including cholangioscopy, are available to patients for acquisition of appropriate histology where the clinical suspicion is high but biopsy and cytology specimens are non-diagnostic. 4.19 Serum IgG4 is measured in all patients with a cholangiopathy and/or dominant stricture. In patients where IgG4 disease is suspected, biopsy samples are considered for IgG4 immunostaining. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 21 Laboratory 4.20 Genetic testing is available according to nationally agreed guidelines. Cytology 4.21 Nationally agreed protocols are developed for the collection of cytology samples to provide well-preserved morphology and cell block material for immunohistochemistry. Histopathology 4.22 Specimen handling and processing takes place according to nationally agreed protocols, especially to ensure adequate lymph node yield and evaluation of resection margin status. Surgery 4.23 Staging laparoscopy is indicated prior to resection in most patients with HBP/upper GI cancer; clinicians consider performing this in the referring centre prior to MDM discussion, to avoid the need for patient transfer. Monitoring requirements MR4C 22 Ensure that departments work to written standardised protocols, and supply local copies of documentation to the appropriate MDM (NHS Wales 2005). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 4.4 The results of the diagnosis, investigations and staging of patients with HBP/upper GI cancer are reported by experts in a standardised and agreed format, encompassing all relevant aspects, and are interpreted in the light of all relevant clinical information. Rationale Standardised reporting will decrease error and omissions and help ensure that all relevant information is available for treatment decision-making at MDMs. Waits for imaging investigations may introduce significant delays before clinical diagnosis is confirmed and appropriate treatment can be instituted. This is particularly true for complex investigations (NHS Wales 2005). Good practice points 4.24 Dual reporting of all complex tests takes place, to reduce error. 4.25 Test reports are produced in electronic format within 24 hours of reporting where possible (depending on the test), and are made available to the MDM. 4.26 Reports are supported by relevant clinical information. Radiology 4.27 Imaging review is carried out by a radiologist with a subspecialist interest, to provide the most accurate imaging diagnosis and pre-treatment staging and, through the MDM, facilitate appropriate decisions on further imaging investigations. 4.28 Reports follow the Tumour, node, metastasis staging system (Gormly 2009; Goergen et al 2013), except for HCC, where the Barcelona Clinic Liver Cancer (BCLC) staging system is used (Bruix and Sherman 2011). 4.29 Imaging reports are received by the referrer within two working days of the examination being performed. Gastroenterology 4.30 A structured electronic report is available within two hours of the procedure. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 23 Histopathology 4.31 Reporting of histopathology is standardised, synoptic and structured, and incorporates all relevant criteria as specified in current international consensus guidelines from the American Joint Committee on Cancer (Edge et al 2010). 4.32 Structured reporting protocols are based on agreed protocols published by the Royal College of Pathologists of Australasia. Where these do not currently exist, pathologists consult other appropriate protocols, such as those of the Royal College of Pathologists or the College of American Pathologists. Monitoring requirements MR4D 24 Ensure that MDMs audit diagnostic and investigative results. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 5 Multidisciplinary Care Standard 5.1 All patients with the following are presented at an MDM: a confirmed diagnosis of HBP/upper GI cancer a high suspicion of HBP/upper GI cancer. Rationale The management of HBP/upper GI cancer involves many different specialties, and evidence shows that multidisciplinary care is important in providing best treatment planning and provision along the care continuum. International evidence shows that multidisciplinary care is a key aspect to providing best-practice treatment and care for patients with cancer. Multidisciplinary care involves a team approach to treatment planning and care provision along the complete patient cancer pathway. Cancer MDMs are part of the philosophy of multidisciplinary care. Effective MDMs result in positive outcomes for patients receiving the care, for health professionals involved in providing the care and for health services overall. Benefits include improved treatment planning, improved equity of patient outcomes, more patients being offered the opportunity to enter into relevant clinical trials, improved continuity of care and less service duplication, improved coordination of services, improved communication between care providers and more efficient use of time and resources. Good practice points 5.1 Agreed terms of reference govern MDMs, based on Ministry of Health guidelines (Ministry of Health 2012b); these include written protocols that describe the conduct and content of the meeting, including criteria outlining which patients should be discussed. 5.2 A designated chairperson guides discussion and encourages consensus within the MDM. 5.3 Each case is presented by the referring doctor or designee, unless the patient has already been seen by a specialist. 5.4 An optimal number of cases is discussed at each MDM. 5.5 The MDM meets weekly, and not less frequently than bimonthly. 5.6 The MDM discusses patients prior to their first cancer treatment, to ensure they are staged appropriately and can be offered neo-adjuvant therapy if appropriate. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 25 Monitoring requirements 26 MR5A Ensure that MDMs audit patients registered at MDMs. MR5B Report on cases discussed at MDMs each month (including core data such as referrers, centres, patient data (age, NHI number and date of birth), co-morbidities, diagnoses (provisional or confirmed) and management plans). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 5.2 The MDM comprises members of the MDT, including surgeons, radiologists, gastroenterologists, medical and radiation oncologists, cyto/histopathologists and cancer care coordinators. Rationale A team of specialists with a range of expertise ensures shared responsibility and optimal decision-making. Good practice points 5.7 The cancer care coordinator attending the MDM is the individual identified in Standard 7.1. 5.8 The attendance of other health professionals involved in the care of patients with HBP/upper GI cancer is encouraged. This includes dietitians, palliative care specialists, psychologists, nurses, physiotherapists and whānau support services personnel, as well as representatives from primary health care. 5.9 Māori expertise, including the range of relevant Māori services and providers, is involved in MDMs and networks where appropriate. 5.10 Referring doctors attend MDMs where possible, through videoconferencing where necessary. 5.11 Collaboration between teams supports cover for annual leave, sick leave and public holidays, to avoid cancellation of MDMs (NHS Wales 2005). 5.12 Employers recognise, and resource the amount of time required of certain members of the MDM (eg, histopathologists and radiologists) to prepare for the MDM. Monitoring requirements MR5C Provide minuted evidence of attendance/involvement at MDMs. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 27 Standard 5.3 Data are available in standardised electronic format for MDM discussion, and include all relevant information about patients and investigations. Standard 5.4 All patients with confirmed HBP/upper GI cancer have their treatment plan discussed at a MDM; recommendations are clearly documented in the patient’s medical records and communicated to the patient and their GP. Rationale In order to ensure that the best recommendations are made, all relevant information should be made available in a standardised, preferably electronic format. Timely communication of MDM recommendations will minimise treatment delay. Good practice points 5.13 Patients with suspected or confirmed HBP/upper GI cancer are registered at least 48 hours before the scheduled MDM, to ensure adequate time for radiologists and pathologists to prepare their material. 5.14 Data related to the patient, diagnosis and staging are submitted in a standardised electronic format and distributed to attendees of the MDM. 5.15 Consensus recommendations regarding referrals, further investigations and treatment plans are clearly captured during the meeting and entered into the electronic record, so that they can be collated and analysed locally, regionally and nationally. 5.16 The MDM’s recommendations are distributed to referrers and copied to all members of the MDM and other health professionals involved in patients’ care, within two days of the MDM. 5.17 Consensus recommendations made at the MDM are discussed with patients, so that plans for treatment and care can be made with the involvement of the patient and their family/whānau (expert opinion). 5.18 When a treating clinician does not follow a treatment plan recommended by the MDM, he or she records the reason (expert opinion). Monitoring requirements 28 MR5D Ensure that MDMs audit treatment recommendations and the detail of following communications. MR5E Ensure that MDMs audit their adherence to clinical policies (NHS Wales 2005). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 6 Supportive Care Standard 6.1 Patients with HBP/upper GI cancer and their family/whānau have equitable and coordinated access to appropriate medical, allied health and supportive care services, in accordance with Guidance for Improving Supportive Care for Adults with Cancer in New Zealand (Ministry of Health 2010). Rationale The psychological, social, physical and spiritual needs of cancer patients are many and varied. These needs can to a large extent be met by allied health care teams in hospitals and in the community. Adults with cancer enjoy improved quality of life following needs assessment and provision of supportive care. Non-government organisations, including the Cancer Society, perform an important role in providing supportive care. Good practice points 6.1 All patients and their family/whānau have access to culturally appropriate support services (such as whānau supportive services and Māori patient navigators/cancer coordinators for Māori patients and their family/whānau) and spiritual services (Ministry of Health 2010). 6.2 All patients and their family/whānau have access to support services, including psychology services, appropriate to their needs. Those experiencing significant distress or disturbance are referred to health practitioners with the requisite specialist skills (Ministry of Health 2010). 6.3 Supportive care assessments and interventions are undertaken in suitable facilities and locations that take into consideration patients’ needs for privacy, comfort and mobility. 6.4 Tools (such as Whānau Ora assessments) are developed to meet the supportive care needs of Māori; these are used to inform patient treatment plans and care coordination. 6.5 All staff responsible for engagement with patients and/or their families/whānau undertake cultural competency training. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 29 6.6 Patients and their families/whānau are involved in advance care planning and end-of-life care planning, where appropriate. 6.7 All staff responsible for engagement with patients and/or their families/whānau hold Level 2 Advance Care Planning and end-of-life care planning competencies.2 Monitoring requirements MR6A 2 30 Provide evidence of culturally appropriate patient and family/whānau satisfaction surveys, and audit complaints processes. Through the National Advance Care Planning Cooperative: see www.advancecareplanning.org.nz. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 7 Care Coordination Standard 7.1 All patients with HBP/upper GI cancer have access to an HBP/upper GI clinical nurse specialist or other health professional who is a member of the MDM to help coordinate all aspects of care. Standard 7.2 Each regional cancer centre or unit managing patients with HBP/upper GI cancer has designated lead clinicians with specialist knowledge of HBP/upper GI cancers to provide necessary leadership. Rationale The cancer journey is complex, and it is not uncommon for a patient to be seen by many specialists within and across multiple DHBs and across the public and private sectors. ‘Care coordination’ refers to a system or a role primarily intended to expedite patient access to services and resources, improve communication and the transfer of information between services, address patients’ information needs and improve continuity of care throughout the cancer continuum. Patients and caregivers should be able to access care coordination through a single point of contact through the various stages of the HBP/upper GI cancer journey. Good practice points 7.1 All regional cancer centres have access to a sufficient number of cancer nurse specialists with expert knowledge of HBP/upper GI cancers to work with care coordinators. 7.2 Cancer clinical nurse specialists work collaboratively and in close clinical partnership with lead clinicians (see Standard 7.2). 7.3 Patients are introduced to their cancer care coordinator within seven days of their initial diagnosis, and are provided with their contact details. 7.4 All patients have their supportive care and psychosocial needs assessed utilising validated tools and documented at each stage of their cancer journey, and have access to services appropriate to their needs. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 31 7.5 Systems are in place to develop and implement cultural concordance (where possible) among health care professionals (Jansen et al 2008). 7.6 Patients who travel long distances to regional treatment services have access to information on the Ministry of Health’s National Travel Assistance Scheme (Ministry of Health 2006). Lead clinicians 7.7 Lead clinicians work to a clear job description, have dedicated sessional time allocated for this role and receive management support. 7.8 Lead clinicians coordinate services and ensure that strategies are in place to improve overall coordination of care. 7.9 Lead clinicians are responsible for the allocation of patients to treatment providers, if required. 7.10 Lead clinicians are responsible for the coordination of the care of individual patients through the cancer care coordinator. 7.11 Lead clinicians are responsible for the running of the regional MDM. 7.12 Lead clinicians are responsible for monitoring standards and data collection. Monitoring requirements 32 MR7A Ensure that MDMs provide records of identified care coordinators. MR7B Audit database records and clinical notes on contact points between care coordinators and patients. MR7C Provide evidence of culturally appropriate patient and family/whānau satisfaction surveys, and audit complaints processes. MR7D Ensure that regional cancer provide evidence of job plans, identifying specific roles, allocating sessional time and providing management support. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 8 Treatment Standard 8.1 The treatment of patients with HBP/upper GI cancer is performed by appropriately trained and credentialed experts (surgeons, medical and radiation oncologists, gastroenterologists and interventional radiologists), with a specialty commitment to this field. Access to specialist expertise is available at all times. Rationale Better clinical outcomes for patients will be achieved if complex care is carried out by expert MDTs. Centres need to be large enough to provide the necessary experience to maintain expertise and to justify the number of specialists required to provide cover at all times. Prompt access to expertise is essential, to minimise morbidity, reoperation rates, lengths of hospital stay and mortality. Good practice points Surgery 8.1 Appropriate surgical expertise is available at all times for post-operative care, and appropriate expertise is available to the MDT. 8.2 Surgery is undertaken in accordance with best practice, and in keeping with international guidelines. 8.3 Surgeons undertaking oesophageal surgery are supported by anaesthetists experienced in one-lung ventilation, thoracic epidural and spinal analgesia techniques (NHS Wales 2005). 8.4 Surgeons undertaking hepatobiliary procedures are supported by anaesthetists experienced in low central venous pressure liver surgery and advanced haemostasis support. 8.5 Surgeons undertaking HBP/upper GI cancer surgery work as a team; experienced surgical colleagues are available in the same hospital to manage complications if the operating surgeon is unavailable or on leave. 8.6 Surgeons are supported by appropriately trained theatre staff and surgical assistants (usually training registrars). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 33 Medical oncology 8.7 Systemic therapy (including chemotherapy, biologicals and small molecules) is prescribed, administered and monitored by appropriately trained medical oncologists. 8.8 If systemic therapy is prepared on site, a pharmacist is involved who has adequate training in systemic therapy medications, including dosing calculations according to protocols, formulations and/or preparation. 8.9 Nurses have appropriate training in systemic therapy administration, and the handling and disposal of cytotoxic waste. Radiation oncology 8.10 Radiotherapy is prescribed and supervised by appropriately trained and credentialed radiation oncologists. 8.11 If a radiation oncologist is not considered an HBP/upper GI sub-specialist expert, they consult with a colleague who is. Interventional radiology 8.12 Patients with HBP/upper Gl cancer have access to interventional radiology at all times for life-threatening complications. 8.13 Therapeutic interventional radiology procedures (including percutaneous transhepatic cholangiography, percutaneous transhepatic biliary drainage, radiofrequency ablation and angioembolisation) are available to all patients with HBP/upper GI cancer. Gastroenterology 8.14 Patients with HBP/upper GI cancer have access to interventional endoscopy (including endoscopic stenting, dilation, mucosal resection and radiofrequency ablation) at all times for life-threatening complications. 8.15 Patients have round-the-clock access to procedures for emergency management of complications (including ERCP). 8.16 Gastroenterology services are readily available, in case of the need to intervene for urgent problems including cholangitis, bleeding and obstruction. Monitoring requirements MR8A 34 Ensure that departments maintain a record of appropriately trained and credentialed staff, and supply copies of local documentation to the appropriate MDM (NHS Wales 2005). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 8.2 Patients with HBP/upper GI cancer are treated in appropriately equipped facilities offering the required range of services, including interventional radiology and a level one or two intensive care unit. Rationale Appropriately resourced facilities, including equipment, are required to treat patients with HBP/upper GI cancer to an acceptable standard and in an acceptable timeframe. Level one and two intensive care units should align with the Joint Faculty of Intensive Care Medicine’s definition of an intensive care unit (Joint Faculty of Intensive Care Medicine 1997). Good practice points Surgery 8.17 Resectional surgery of complex HBP/upper GI cancers is supported by a level one or two intensive care unit, a high-dependency unit and round-the-clock access to interventional radiology and interventional endoscopy. 8.18 Operating theatres are equipped with technologies for the treatment of HBP/upper GI cancers, including radiofrequency ablation, argon beam coagulation and high-definition laparoscopy for advanced procedures. Medical oncology 8.19 Systemic therapy is administered in an appropriately accredited facility. 8.20 Medical oncologists have access to timely preparation of cytotoxic agents, nuclear medicine facilities to assess organ function and adequate nursing staff within a dedicated unit. Radiation oncology 8.21 All radiotherapy centres meet the current Radiation Oncology Practice Standards set by the Tripartite Initiative (Tripartite Committee 2011). 8.22 Expert bodies such as the Office of Radiation Safety periodically assess quality assurance for all radiotherapy treatment equipment; such equipment meets the requirements of the New Zealand Radiation Protection Act 1965. 8.23 All radiotherapy centres treat their patients with modern techniques to best meet their cancer, and have access to equipment capable of delivering 4D conformal radiotherapy and intensity modulated radiotherapy. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 35 Interventional radiology 8.24 Diagnostic and therapeutic interventional radiology procedures are readily available in centres treating patients with HBP/upper GI cancer. 8.25 Elective interventional radiology procedures are performed at centres with appropriate interventional radiology and clinical (hepatobiliary surgery and/or gastroenterology/hepatology) expertise following multidisciplinary patient case review. 8.26 Emergency interventional radiology procedures are performed at centres with appropriate interventional radiology and clinical (hepatobiliary surgery and/or gastroenterology/hepatology) expertise following patient clinical review and referral. 8.27 Transfer to a tertiary-level centre is considered if appropriate interventional radiology and/or clinical (hepatobiliary surgery and/or gastroenterology/hepatology) expertise is not available. 8.28 Outcomes are assessed according to an agreed system of quality improvement guidelines and standards of practice. Gastroenterology 8.29 Therapeutic endoscopic facilities and equipment allow the provision of advanced procedures, including endoscopic mucosal resection, radiofrequency ablation, endoscopic stenting and endoscopic ultrasoundguided coeliac plexus neurolysis. Monitoring requirements MR8B 36 Ensure that departments assess facilities and equipment against agreed standards. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 8.3 Patients with HBP/upper GI cancer are treated according to evidence-based and agreed protocols. Rationale The standardisation of treatment protocols is important for many reasons, including optimising patient care; avoiding overtreatment; monitoring the safety, quality and efficacy of treatment; improving outcomes; and comparing outcomes over time and between treatment centres. Good practice points Surgery 8.30 Surgery for HBP/upper GI cancer is based on best current practice and conforms to international consensus guidelines (ANZGOSA 2013a; 2013b). 8.31 Surgeons consult with all patients undergoing surgery and their family/whānau about final disposal of tissue or body parts surgically removed following appropriate pathological examination (some tissue may be retained for future histological reference) (modified NZGG 2009). Medical oncology 8.32 Protocols are in place to cover generic issues pertinent to systemic therapy such as staff grading, training and competencies; prescribing; preparation and dispensing; administration; and disposal of waste and spillage. Radiation oncology 8.33 Formal written treatment protocols specify dose, fractionation, overall treatment time, planning technique and means of verification, plus other appropriate quality assurance measures. Gastroenterology 8.34 ERCP for therapeutic intervention of a dominant biliary stricture is undertaken after the case has been discussed at an MDM and the treatment plan agreed. 8.35 In patients with a resectable tumour who require preoperative biliary drainage, the biliary stent is placed in the ‘non-diseased’ (future liver remnant) lobe of the liver. 8.36 Patients with suspected IgG4 disease are first managed with a trial of corticosteroid therapy. Monitoring requirements MR8C Ensure that MDMs document treatment protocol regarding tumour site and stage, and note any treatment that is outside the MDM’s protocol. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 37 Standard 8.4 Reports of treatment by all specialties of patients with HBP/upper GI cancer (including operation notes) are in a standardised electronic format, and include all of the relevant prognostic criteria. Rationale Standardised reporting is essential for communication between colleagues within MDTs and for meaningful clinical audit and clinical research. Good practice points 8.37 Standardised operation reports are developed. Monitoring requirements MR8D 38 Ensure that MDMs audit diagnostic and investigative reports. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 8.5 Patients with HBP/upper GI cancer have access to a dietitian for assessment and nutritional support before, during and after their treatment. Rationale Patients with HBP/upper GI cancer are prone to nutritional depletion, through the effect of cancer itself and through reduced intake. Such patients are more prone to infection, reduced response to treatment, impaired recovery and worsening prognosis. Nutritional repletion improves quality of life and promotes recovery. Good practice points 8.38 All patients with HBP/upper GI cancer are assessed for nutritional status using a validated nutrition screening tool. 8.39 Patients with significant nutritional depletion are referred for consideration of nutritional support before, during and after treatment. 8.40 A dedicated nutritional support team is available in all centres undertaking treatment of patients with HBP/upper GI cancer, especially if parenteral nutrition is to be provided. 8.41 Nutritional supplementation is offered to all patients, even in the absence of marked weight loss, to improve outcomes, whatever modality of treatment patients has decided (including no treatment). 8.42 Patients have access to a dietitian throughout their recovery period. 8.43 Pancreatic enzyme replacement therapy (as recommended by the Pancreatic Section of the British Society of Gastroenterology) is considered where appropriate, to maintain patients’ weight and increase their quality of life. Monitoring requirements MR8E Ensure that MDMs document nutritional assessments. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 39 Standard 8.6 Patients with HBP/upper GI cancer are offered early access to palliative care services when there are complex symptom control issues, when a curative treatment cannot be offered or if curative treatment has been declined. Rationale A diagnosis of cancer and its subsequent treatment can have a devastating impact on the quality of a person’s life, as well as on the lives of families/whānau and other carers. Patients may face new fears and uncertainties, and may have to undergo unpleasant and debilitating treatments. Patients should expect to be offered optimal symptom control and psychological, cultural, social and spiritual support. They may want to be assured that their families/whānau and carers will receive support during illness and, if they die, following bereavement. Palliative care is the care of people who are dying from active, progressive diseases or other conditions that are not responsive to curative treatment. Palliative care embraces the physical, social, emotional and spiritual elements of wellbeing – tinana, whānau, hinengaro and wairua – and enhances a person’s quality of life while they are dying. Palliative care also supports the bereaved family/whānau (Ministry of Health 2001). The objective of palliative care is to alleviate suffering and provide compassionate care for the patient and their family/whānau. Competence in palliative medicine and sensitivity to people’s beliefs and values are two key prerequisites for a provider of palliative care. Clinicians should form a care plan for palliative patients with a view to ensuring that pain and other potentially distressing symptoms are relieved, dignity is preserved and there is engagement with family/whānau (Ministry of Health 2001). Good practice points 8.44 Screening for palliative care needs, comprehensive assessment and care planning are undertaken at appropriate intervals, to meet the needs and wishes of patients and their family/whānau (NCCN 2012c; Palliative Care Australia 2005). 8.45 Prompt and effective communication takes place between all involved parties (NCCN 2012c; NICE 2004). 8.46 Access to palliative care, decision-making and care planning are based on a respect for the uniqueness of the patient and their family/whānau, independent of their current health status, diagnosis, age, gender, cultural background or geography. Patients’ and their family/whānau’s needs and wishes guide decision-making and care planning (Hospice New Zealand 2012). 40 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 8.47 Patients and carers have access to spiritual care, either from their MDT or from community spiritual care resources (NICE 2004). 8.48 Patients and their family/whānau are offered a range of free, culturally and educationally appropriate high-quality information materials about cancer and palliative care services in a variety of formats (NICE 2004). 8.49 Systems are in place to ensure the views of patients and their family/whānau are taken into account when developing and evaluating cancer and palliative care services. All such services support patients to participate in their own care by offering a range of informal opportunities, such as self-help activities and peer support schemes with community settings (NICE 2004). 8.50 Formal mechanisms are in place to ensure that patients, their carers and family/whānau have access to bereavement care, information and support services (NICE 2004; Hospice New Zealand 2012). 8.51 Interdisciplinary palliative care teams, including New Zealand Medical Councilregistered palliative care physicians and skilled palliative care nurses, are readily available to provide consultative or direct care to patients and their family/whānau (NCCN 2012c). 8.52 All health care professionals take part in educational programmes to develop effective palliative care knowledge, skills and attitudes, covering protocols for the management of cancer pain (World Health Organization), neuropathic pain and the route of referral to palliative care services (NCCN 2012c). 8.53 All health care professionals recognise dying patients in a timely manner, and discuss advance care planning and end-of-life goals of care with patients and their family/whānau, using end-of-life care pathways. 8.54 Palliative care services are consulted at any stage through a patient’s cancer journey if symptoms are complex and/or prove difficult to treat. Monitoring requirements MR8F Audit records of proposed plans of care, onward referrals and follow-up responsibilities recorded at MDT reviews and in patients’ notes. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 41 9 Follow-up and Surveillance Standard 9.1 When appropriate, patients treated for HBP/upper GI cancer are offered ongoing follow-up at their original referring centre. Rationale Follow-up promotes recovery and improved quality of life. It is also useful to detect disorders of function, to assess nutritional status, to provide psychosocial support and to audit treatment outcomes (SIGN 2006). The ongoing support of patients with cancer after definitive treatment should ideally take place close to home and family/whānau support, and involve the referring specialist or GP. Good practice points 9.1 Follow-up occurs as close to home and family/whānau as possible. 9.2 Where appropriate, and to reduce travel costs and inconvenience to the patient, shared follow-up arrangements are considered, in which patients have alternating visits with treating specialists and the referring specialist/GP. Monitoring requirements MR9A 42 Audit follow-up practices and the timeliness and appropriateness of investigations. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 9.2 Patient follow-up after HBP/upper GI cancer for the detection of local recurrence and metastases takes place in accordance with international consensus guidelines. Rationale Regular review makes it possible to diagnose early local recurrence and distant metastases. A decision as to whether this will advantage a particular patient needs to be made when developing the follow-up plan. Good practice points Palliative patients 9.3 When all possible curative treatment has been administered and no more curative treatment will be offered, follow-up is based on the identification of symptoms that can be palliated, rather than repeating tumour markers or cross-sectional imaging. 9.4 For patients who have had palliative treatment, follow-up may be provided by the GP, with the support of community and/or specialist palliative care services. Patients are able to contact one MDT-designated hospital specialist for advice on referral to appropriate hospital services in the event of complications such as complex pain, cholestasis/cholangitis, duodenal obstruction or bleeding. Gastric and oesophageal cancer 9.5 After endoscopic treatment of early gastric and oesophageal cancer, a regular surveillance programme is established according to international consensus guidelines. Secondary liver cancer 9.6 For patients who have undergone treatment for colorectal cancer, the detection of liver metastases takes place in accordance with Management of Early Colorectal Cancer (NZGG 2011). 9.7 Patients with resected colorectal liver metastases are followed up for further recurrence, since treatment with curative intent is often possible. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 43 Hepatocellular carcinoma 9.8 Contrast-enhanced multiphasic liver CT and/or MRI is undertaken for surveillance of all patients following hepatic resection or locoregional (thermal ablation) or trans-arterial (chemo/radio embolisation) therapy for HCC, in accordance with international consensus guidelines (NCCN 2011a; Bruix and Sherman 2011). Pancreatic cancer 9.9 Patients who have completed treatment with curative intent and in whom asymptomatic recurrence is identified to be followed up more intensively with regular review by a cancer specialist if they would benefit from therapy in terms of survival. Frequency of Ca19-9 and CT scanning is tailored to the patient, based on international best practice. Duodenal cancer 9.10 Patients who have had a pancreatoduodenectomy for duodenal cancer do not receive follow-up for duodenal polyposis. 9.11 Depending on the clinical situation (particularly the presence of FAP), followup is considered for patients who have had a pancreatoduodenectomy for duodenal cancer because of the possibility of colonic or gastric malignancies. MR9B 44 Audit organisational compliance with surveillance protocol. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 9.3 Follow-up plans include clinical review by appropriate members of the MDT, working in conjunction with the patient, their family/whānau and their GP. Rationale The ongoing involvement of the health professionals who were involved in the diagnosis and referral of a patient with HBP/upper GI cancer is usually important, and is seen as ‘closing the loop’. Follow-up should be carried out as close as possible to the family home. Good practice points 9.12 Treating specialists, in consultation with referrers and GPs (if a GP was not the referrer) makes a clear decision about the most appropriate person to take primary responsibility for follow-up. The patient and their family/whānau are involved in this decision. 9.13 Patients’ GPs receive written copies of follow-up plans within one day of patients receiving the same information. 9.14 Follow-up plans include the frequency of visits; the tests required, and with which designated service; and a nominated point of contact if there is clinical concern. 9.15 Patients receive information about available culturally appropriate support. 9.16 Referral to a psychologist is considered, when appropriate, to promote more effective coping strategies and improve patients’ self-esteem and mood. 9.17 Local palliative care services receive early notification of patients who have undergone palliative treatment. 9.18 Patients and their family/whānau receive information on lifestyle factors, including smoking cessation, exercise, nutrition and psychological support. 9.19 Patients receive equitable access to integrated rehabilitation services (including education, housing, welfare and Whānau Ora services) in a coordinated manner. Monitoring requirements MR9C Audit provision of written follow-up information following agreed surveillance protocols. MR9D Provide evidence of appropriate written information provided to patients and families/whānau, and record names of nominated contacts. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 45 10 Clinical Performance Monitoring and Research Standard 10.1 Data relating to HBP/upper GI cancer beyond the fields required by the Cancer Registry, including treatment data, are reported to existing and planned national repositories using nationally agreed data set fields. Rationale There is currently no national cancer database other than the New Zealand Cancer Registry, which is a population-based register of all primary malignant tumours diagnosed in New Zealand. This is a significant impediment to advancing cancer care in this country. Cancer data-related projects are being undertaken or planned by the Ministry of Health, Cancer Control New Zealand and regional cancer networks. Good practice points 10.1 Where data are collected, they are compiled in accordance with the National Cancer Core Data Definition standards (IT Health Board 2011). 10.2 Dedicated database managers are employed in regional cancer centres to oversee satisfactory data entry. Data veracity is confirmed clinically. 10.3 Patients are informed that their information is being recorded in an HBP/upper GI cancer database to help the MDT propose a treatment plan and to monitor and evaluate access to services. 10.4 Clinicians working in HBP/upper GI cancer treatment and care have access to the database to help inform disease management. Monitoring requirements MR10A 46 Provide evidence of reporting of required patient data sets to national data repositories (as available) at the agreed frequency. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Standard 10.2 Data on treatment, clinical outcomes and patient satisfaction are regularly monitored and reported as part of a national audit. Rationale Accurately recorded and good quality data are essential for monitoring outcomes and comparing clinical services. Audits identify inequalities in service along the patient pathway, including inequities in timely access to services and those based on ethnicity. One of the key findings of Access to Cancer Services for Māori (Ministry of Health 2005) was the limited nature of information on access to cancer services for Māori, including discussion of interventions. Several areas arise from the project as priorities for further research. Good practice points 10.5 Data are reported according to an agreed system of quality improvement guidelines and standards of practice. 10.6 All parts of the patient pathway are investigated and audited, including referral, investigation, management of treatment and patient outcomes, with particular emphasis on interventions to enhance the patient journey for Māori patients and their family/whānau. 10.7 The results of audits are presented at a national multidisciplinary audit meeting, and are available to clinicians within referring units, the cancer networks and the public. 10.8 Surgical outcomes are monitored against international guidelines. Monitoring requirements MR10B Ensure that MDMs provide evidence of participation in regular audits. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 47 Standard 10.3 Patients with HBP/upper GI cancer are offered the opportunity to participate in research projects and clinical trials where these are available. Rationale Advances in evidence-based practice are largely dependent on clinical trials. Although clinical trials are primarily focused on the improvement of care for future patients, there is good evidence that patients treated within a trial setting fare better than those treated outside of a trial setting. This is thought to be due in large measure to the benefits of treatment that is carried out according to documented protocols (NHS Wales 2005). Good practice points 10.9 Information about available trials (regionally, nationally and internationally), including eligibility criteria, is readily available to providers of cancer care, preferably through a website. 10.10 Funds are allocated to support the costs of research personnel, such as research nurses, to facilitate enrolment of patients into clinical trials, data collection and adherence to compliance requirements. 10.11 A coordinated collaborative approach is taken to clinical trials, to ensure that studies are conducted efficiently and results are available in a timely manner. 10.12 Regional cancer centres maintain a listing of clinical trials to which patients are being recruited, and make this available for collation on a national basis. 10.13 Tissue is appropriately collected and stored, and good-quality clinical data is appropriately collected, in order to enhance current and future research opportunities. Monitoring requirements MR10C 48 Ensure that MDMs provide documentation of all open trials/research projects, and record the number of patients entered per trial per year. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Appendix 1: National HBP/Upper GI Tumour Standards Working Group Membership Chair Prof John Windsor, Consultant Surgeon, Auckland DHB/University of Auckland Clinical leads Mr Grant Coulter (gastro-oesophageal lead), Consultant Surgeon, Canterbury DHB Mr Simon Bann (gastro-oesophageal deputy lead), Consultant Upper Gastrointestinal and Laparoscopic Surgeon, Capital & Coast DHB Prof John McCall (hepatobiliary lead), Hepatobiliary and General Surgeon, Southern DHB/Dunedin School of Medicine Associate Prof Jonathan Koea (hepatobiliary deputy lead), Hepatobiliary and General Surgeon, Waitemata DHB Prof John Windsor (pancreaticoduodenal lead), as above Mr Kusal Wickremesekera (pancreaticoduodenal deputy lead), Consultant Hepatobiliary/Upper Gastrointestinal/Laparoscopic Surgeon, Capital & Coast DHB Members Gastro-oesophageal Sue Corkill, Cancer Information Nurse, Cancer Society (Christchurch) Dr Jim Edwards, Consultant Medical Oncologist, Canterbury DHB Dr Gabriel Lau, Consultant Radiologist, Southland DHB Dr Paul Restall, Histopathologist, Auckland DHB Mr Michael Rodgers, Clinical Director of General Surgery, Waitemata DHB Dr David Rowbotham, Clinical Director of Gastroenterology and Hepatology, Auckland DHB Hepatobiliary Mr Adam Bartlett, Consultant Surgeon, Auckland DHB Dr Katriona Clarke, Consultant Medical Oncologist, Capital & Coast DHB Dr Daniel Cookson, Consultant (Interventional) Radiologist, Counties Manukau DHB Sue Lodge, Palliative Care Team Leader, Mary Potter Hospice (Kapiti) Dr David Orr, Consultant Hepatologist and Gastroenterologist, Auckland DHB Dr Stephen Williams, Consultant Radiation Oncologist, Canterbury DHB Dr Mee Ling Yeong, Histopathologist/Clinical Director, Diagnostic MedLabs Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 49 Pancreaticoduodenal Mr Saxon Connor, Consultant Surgeon, Canterbury DHB Dr Susan Hamilton, Consultant Palliative Medicine, Mary Potter Hospice (Wellington) Dr Dean Harris, Consultant Medical Oncologist, Canterbury DHB Dr Chris McKee, Consultant Radiologist, Waitemata DHB Dr Andrew Miller, Consultant Anatomical Pathologist, Canterbury Health Laboratories Petro Nel, Clinical Nurse Specialist, Medical Oncology, MidCentral DHB Dr Hermann van der Vyver, Consultant Radiation Oncologist, Waikato DHB Dr Frank Weilert, Director of Endoscopy, Waikato DHB Ex officio members Sarah Bell, Project Manager, Northern Cancer Network Deirdre Maxwell, Manager, Northern Cancer Network Dr Richard Sullivan, Regional Clinical Director, Northern Cancer Network Dr David Theobald, National Clinical Lead, Gastrointestinal Endoscopy, National Endoscopy Quality Improvement Programme, Ministry of Health 50 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Appendix 2: Glossary Adjuvant therapy Additional treatment to increase the effectiveness of the main treatment (often surgery), such as chemotherapy, systemic therapy or radiotherapy Advance care planning A process of discussion and shared planning for future health care Aetiology The origins or causes of disease Allied health professional One of the following groups of health care workers: physiotherapists, occupational therapists, dietitians, paramedics, prosthetists/orthotists, psychologists, social workers and speech and language therapists Angioembolisation A non-surgical, minimally invasive procedure performed by an interventional radiologist Asymptomatic Without obvious signs or symptoms of disease. In early stages, cancer may develop and grow without producing symptoms Barcelona Clinic Liver Cancer staging system A staging system used particularly when describing the extent of liver cancer Best practice A method or approach that is accepted by consensus to be the most effective way of doing something, in the circumstances; may or may not be based on evidence Biliary Of the bile duct Biopsy Removal of a sample of tissue or cells from the body to assist in the diagnosis of a disease Cancer journey The individual and personal experience of a person with cancer throughout the course of their illness Cancer Networks Cancer Networks were formed in response to national policy to drive change and improve cancer services for the population in specific areas. There are four regional networks: Northern, Midland, Central and Southern Cancer service pathway The cumulative cancer-specific services that a person with cancer uses during the course of their experience with cancer Carcinoma Cancer of the lining tissue that covers all the body organs. Most cancers are carcinomas Care coordination Entails the organising and planning of cancer care, who patients and family/whānau see, when they see them and how this can be made as easy as possible. It may also include identifying who patients and family/whānau need to help them on the cancer pathway Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 51 52 Chemotherapy The use of drugs that kill cancer cells, or prevent or slow their growth (also see systemic therapy) Cholangiopathy Any disease of the bile ducts Cholangioscopy The examination of bile ducts using an endoscope Chromoendoscopy The examination of gastrointestinal tracts using a fibre-optic endoscope and dyes Clinical trial An experiment for a new treatment Co-morbidities Two or more diseases existing at the same time in the body Computed tomography (CT) A medical imaging technique using X-rays to create crosssectional slices through the body part being examined Confirmed diagnosis (used in FCT indicators) The preferred basis of a confirmed cancer diagnosis is pathological, noting that for a small number of patients cancer diagnosis will be based on diagnostic imaging findings Curative Aiming to cure a disease Decision to treat (used in FCT indicators) A decision to begin a patient’s treatment plan or other management plan, following discussion between the patient and treating clinician. DHB District Health Board End of life care The provision of supportive and palliative care in response to the assessed needs of the patient and family/whānau during the end-of-life phase Endoscope A tubular device with a light at the end that transmits images to aid diagnosis or therapy. Specialised endoscopes may be threaded down the oesophagus to the stomach or beyond, or inserted through an incision in the abdomen. They may also be used to take samples of tissues (see biopsy) or to carry out therapeutic functions, such as inserting stents Endoscopic mucosal resection A technique used to remove cancerous or other abnormal lesions found in the digestive tract Endoscopic retrograde cholangiopancreatography (ERCP) A method by which the bile and pancreatic ducts may be accessed using an endoscope Endoscopic ultrasound Imaging using high-frequency sound waves, carried out inside the body using an endoscope Endoscopist A person trained in the use of the endoscope Endoscopy The examination of the upper gastro-intestinal tract Excision The removal of tissue by surgery Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Familial adenomatous polyposis (FAP) An inherited disorder characterised by cancer of the large intestine (colon) and rectum. People with the classic type of FAP may begin to develop multiple non-cancerous (benign) growths (polyps) in the colon as early as their teenage years Faster Cancer Treatment (FCT) A Ministry of Health programme that will improve services by standardising care pathways and timeliness of services for cancer patients throughout New Zealand Faster Cancer Treatment indicators Measures of cancer care collected through DHB reporting of timeframes within which patients with a high suspicion of cancer access services. The indicators are internationally established and provide goals for DHBs to achieve over time Fine needle aspiration (FNA) cytology The use of a fine needle to biopsy a tumour or lymph node to obtain cells for cytological confirmation of diagnosis First specialist assessment (FSA) Face-to-face contact (including telemedicine) between a patient and a registered medical practitioner or nurse practitioner for the purposes of first assessment for their condition for that specialty First treatment (used in FCT indicators) The treatment or other management that attempts to begin the patient’s treatment, including palliative care Fluorescence in situ hybridisation (FISH) A cytogenetic technique used to detect and localise the presence or absence of specific DNA sequences on chromosomes Gastroenterology The branch of medicine that deals with the digestive system Gastroenterologist A doctor who specialises in diseases of the digestive system Gastroscopy A diagnostic test that enables the inside of the upper digestive tract to be viewed GP General practitioner Haemostasis A process to stop bleeding Hepatocellular carcinoma (HCC) The most common type of primary liver cancer. Most cases of HCC are secondary to either a viral hepatitis infection (hepatitis B or C) or cirrhosis (alcoholism is the most common cause of hepatic cirrhosis). Also called malignant hepatoma Health equality/equity Absence of unnecessary, avoidable and unjust differences in health (Ministry of Health 2002) Health inequality/inequity Differences in health that are unnecessary, avoidable or unjust (Ministry of Health 2002) Hepatectomy Surgical resection of the liver High suspicion of cancer (used in FCT indicators) Where a patient presents with clinical features typical of cancer, or has less typical signs and symptoms but the clinician suspects that there is a high probability of cancer Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 53 54 Histological Relating to the study of cells and tissue on the microscopic level Histopathologist A doctor who specialises in examining tissue samples microscopically in order to make a diagnosis and ensure tumour excision is complete Histopathology The microscopic study of diseased tissue Holistic Looking at the whole system rather than just concentrating on individual components Hospice Hospice is not only a building; it is a philosophy of care. The goal of hospice care is to help people with life-limiting and life-threatening conditions make the most of their lives by providing high-quality palliative and supportive care Immunohistochemistry A technique that uses antibodies to show up specific proteins in tissues seen down a microscope Immunology A branch of biomedical science that covers the study of all aspects of the immune system in all organisms Immunostaining A term in biochemistry that applies to any use of an antibody-based method to detect a specific protein in a sample Immunotherapy Stimulation of a patient’s immune system to attack the malignant tumour cells that are responsible for disease. This can be either through immunisation of the patient, in which case the patient's own immune system is trained to recognise tumour cells as targets to be destroyed, or through the administration of therapeutic antibodies as drugs, in which case the patient’s immune system is recruited to destroy tumour cells by the therapeutic antibodies Intensity modulated radiotherapy An attempt to optimise dose distribution during external beam radiotherapy delivery. Each radiation field is divided into small segments with varying radiation intensity, which allows for target shape, location and the geometry of overlaying tissues. Intensity modulated radio therapy fields are typically designed using computer-driven (or -aided) optimisation. This is often referred to as ‘inverse treatment planning’ Interventional radiologist A doctor who specialises in imaging and the use of imaging techniques to guide the placement of therapeutic devices like stents inside the body Laparoscopy Visualisation of the interior of the abdomen using a special type of endoscope, inserted through a small incision in the abdominal wall Lesion An area of abnormal tissue Local recurrence Local persistence of a primary tumour due to incomplete excision Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Magnetic resonance imaging (MRI) A non-invasive method of imaging, which allows the form and metabolism of tissues and organs to be visualised (also known as nuclear magnetic resonance) Malignant Cancerous. Malignant tumours can invade and destroy nearby tissue and spread to other parts of the body Medical oncologist A doctor who treats cancer patients through the use of chemotherapy and, for some tumours, immunotherapy Medical oncology The specialist treatment of cancer patients through the use of chemotherapy and, for some tumours, immunotherapy Metastases Cancerous tumours in any part of the body that have spread from the original (primary) origin. Also known as ‘secondaries’ Morbidity The state of being diseased Morphology A branch of bioscience dealing with the study of the form and structure of organisms and their specific structural features Mortality Either (a) the condition of being subject to death or (b) the death rate, which reflects the number of deaths per unit of population in any specific region, age group, disease or other classification, usually expressed as deaths per 1000, 10,000 or 100,000 Multidisciplinary meeting (MDM) A deliberate, regular, face-to-face meeting (which may be through videoconference) to facilitate prospective multidisciplinary discussion of options for patients’ treatment and care by a range of health professionals who are experts in different specialties. ‘Prospective’ treatment and care planning makes recommendations in real time, with an initial focus on the patient’s primary treatment. Multidisciplinary meetings facilitate a holistic approach to the treatment and care of patients Multidisciplinary team (MDT) A group of specialists in a given disease area. The MDT meets regularly to plan aspects of patient treatment. Individual patient cases might be discussed at an MDM, to best plan approach to treatments National Health Index (NHI) number A unique identifier for New Zealand health care users Oesophagectomy Removal of part of the oesophagus Oncology The study of the biological, physical and chemical features of cancers, and of the causes and treatment of cancers Palliative Anything that serves to alleviate symptoms due to an underlying cancer but is not expected to cure it Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 55 56 Palliative care Active, holistic care of patients with advanced, progressive illness that may no longer be curable. The aim is to achieve the best quality of life for patients and their families/whānau. Many aspects of palliative care are also applicable in earlier stages of the cancer journey in association with other treatments Pancreaticoduodenectomy Surgical removal of most of the pancreas, the encircling loop of duodenum, the common bile duct and the pylorus (also known as the Whipple procedure) Pathologist A doctor who examines cells and identifies them. The pathologist can tell where a cell comes from in the body and whether it is normal or a cancer cell. If it is a cancer cell, the pathologist can often tell what type of body cell the cancer developed from. In a hospital practically all the diagnostic tests performed with material removed from the body are evaluated or performed by a pathologist Pathology A branch of medicine concerned with disease; especially its structure and its functional effects on the body Patient pathway The individual and personal experience of a person with cancer throughout the course of their illness; the patient journey Positron emission tomography (PET) A highly specialised imaging technique using a radioactive tracer to produce a computerised image of body tissues to find any abnormalities. PET scans are sometimes used to help diagnose cancer and investigate a tumour’s response to treatment Positron emission tomography and computed tomography (PET-CT) An advanced imaging technique combining an injected material (18 Fluorine) which is taken up by cancer cells and a CT scan Primary care Primary-level health services provided by a range of health workers, including GPs and nurses Prognosis A prediction of the likely outcome or course of a disease; the chance of recovery or recurrence Quality assurance All the planned and systematic activities implemented within the quality system, and demonstrated as needed Radiation oncologist A person who is registered as a medical practitioner by the relevant medical board, is a fellow of the Royal Australian and New Zealand College of Radiologists or equivalent and is licensed to prescribe radiation therapy Radiologist A doctor who specialises in creating and interpreting pictures of areas inside the body using X-rays and other specialised imaging techniques. An interventional radiologist specialises in the use of imaging techniques for treatment; for example catheter insertion for abscess drainage Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Radiology The use of radiation (such as X-rays, ultrasound and magnetic resonance) to create images of the body for diagnosis Radiofrequency ablation A procedure that uses a probe called an electrode to apply an electrical current (radiofrequency) to a tumour Radiotherapy (radiation treatment) The use of ionising radiation, usually X-rays or gamma rays, to kill cancer cells and treat tumours Recurrence The return, reappearance or metastasis of cancer (of the same histology) after a disease-free period Referred urgently (used in FCT indicators) Describes urgent referral of a patient to a specialist because he or she presents with clinical features indicating high suspicion of cancer Resection Removal of tissue from the body by surgery Stage The extent of a cancer; especially whether the disease has spread from the original site to other parts of the body Staging Usually refers to the Tumour, node, metastasis system for grading tumours by the American Joint Committee on Cancer Stent A tubular device made of metal or polythene designed to hold open a tube or opening in the body, such as the oesophagus (oesophageal stent) or bile duct (biliary stent) Supportive care Supportive care helps a patient and their family/whānau to cope with their condition and treatment – from prediagnosis through the process of diagnosis and treatment to cure, continuing illness or death, and into bereavement. It helps the patient to maximise the benefits of treatment and to live as well as possible with the effects of their disease Synoptic report A standardised proforma for reporting of cancer Systemic therapy Treatment using substances that travel through the bloodstream, reaching and affecting cells all over the body Tertiary Third level. Relating to medical treatment provided at a specialist institution Tumour, node, metastasis A staging system that describes the extent of cancer, developed by the American Joint Committee on Cancer Ultrasound A non-invasive technique using ultrasound waves (highfrequency vibrations beyond the range of audible sound) to form an image Whānau Māori term for a person’s immediate family or extended family group. In the modern context, sometimes used to include people without kinship ties Whānau Ora An inclusive interagency approach to providing health and social services to build the capacity of New Zealand families. It empowers family/whānau as a whole, rather than focusing separately on individual family members Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 57 X-ray 58 A photographic or digital image of the internal organs or bones produced by the use of ionising radiation Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Appendix 3: References Development of the HBP/upper GI cancer standards was informed by key national and regional policy documents. Those documents that most directly influenced the development of the standards are listed below. Allum WH, Blazeby JM, Griffin SM, et al. 2011. Guidelines for the management of oesophageal and gastric cancer. Gut 60: 1449–72. Bruix J, Sherman M. 2011. Management of hepatocellular carcinoma: An update. Alexandria: American Association for the Study of Liver Diseases. Eckel F, Brunner T, Jelic S, et al. 2011. Biliary Cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology Suppl 5: v65–9. European Association for the Study of the Liver, European Organisation for Research and Treatment of Cancer. 2012. EASL–EORTC Clinical Practice Guidelines: Management of hepatocellular carcinoma. Journal of Hepatology 56(4): 908–43. IT Health Board. 2011. National Cancer Core Data Definitions, Interim Standard, HISO 10038.3. Wellington: Ministry of Health. Ministry of Health. 2001. The New Zealand Palliative Care Strategy. Wellington: Ministry of Health. Ministry of Health. 2002. Reducing Inequalities in Health. Wellington: Ministry of Health. Ministry of Health. 2005. Access to Cancer Services for Māori. Wellington: Ministry of Health. Ministry of Health. 2010. Guidance for Improving Supportive Care for Adults with Cancer in New Zealand. Wellington: Ministry of Health. Ministry of Health. 2011a. Targeting Shorter Waits for Cancer Treatment. Wellington: Ministry of Health. Ministry of Health. 2012b. Guidance for Implementing High-Quality Multidisciplinary Meetings: Achieving best practice cancer care. Wellington: Ministry of Health. Ministry of Health. 2013a. Faster Cancer Treatment programme. URL: www.health.govt.nz/our-work/diseases-and-conditions/cancer-programme/faster-cancertreatment-project (accessed 28 June 2013). National Lung Cancer Working Group. 2011. Standards of Service Provision for Lung Cancer Patients in New Zealand. Wellington: Ministry of Health. NCCN. 2011a. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Hepatobiliary Cancers: Version 2 2012. Fort Washington: National Comprehensive Cancer Network (member access only). NCCN. 2011b. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Pancreatic Adenocarcinoma: Version 2 2012. Fort Washington: National Comprehensive Cancer Network (member access only). NCCN. 2012a. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Esophageal and Esophagogastric Junction Cancers. Fort Washington: National Comprehensive Cancer Network (member access only). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 59 NCCN. 2012b. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Gastric Cancer. Fort Washington: National Comprehensive Cancer Network (member access only). NHS Wales. 2005. National Standards for Upper Gastro-intestinal Cancer Services. Cardiff: NHS Wales. Pancreatic Section of the British Society of Gastroenterology, Pancreatic Society of Great Britain and Ireland, Association of Upper Gastrointestinal Surgeons of Great Britain and Ireland, Royal College of Pathologists, Special Interest Group for Gastro-Intestinal Radiology. 2005. Guidelines for the management of patients with pancreatic cancer periampullary and ampullary carcinomas. Gut 54: 1–16. SIGN. 2006. Management of Oesophageal and Gastric Cancer: A national clinical guideline. Edinburgh: Scottish Intercollegiate Guidelines Network. Statewide Cancer Clinical Network. 2010. South Australian Upper Gastrointestinal Cancer Care Pathway. Adelaide: Department of Health, Government of South Australia. Seufferlein T, Bachet JB, Van Cutsem E, et al. 2012. Pancreatic adenocarcinoma: ESMOESDO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology Suppl 7: vii33–40. Verslype C, Rosmorduc O, Rougier P, et al. 2012. Hepatocellular carcinoma: ESMO-ESDO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology Suppl 7: vii41–8. WoSCAN. 2007. The Management of Pancreatico-Biliary and Duodenal Tumours. Regional protocol for the west of Scotland cancer network. Glasgow: West of Scotland Cancer Network Upper GI Cancer Managed Clinical Network Advisory Board. Introduction Beenen E, Jao W, Coulter G, et al. 2013. The high volume debate in a low volume country: centralisation of oesophageal resection in New Zealand. New Zealand Medical Journal 126(1374): 34–44. Chamberlain J, Sarfati D, Cunningham R, et al. 2013. Incidence and management of hepatocellular carcinoma among Māori and non-Māori New Zealanders. ANZ Journal of Public Health (in press). Hill S, Sarfati D, Robson B, et al. 2012. Indigenous inequalities in cancer: what role for health care? ANZ Journal of Surgery 83(1–2): 36–41. Martin IG. 2002. Gastro-oesophageal malignancy in New Zealand: 1995–97. New Zealand Medical Journal 115(1148): 64–7. Ministry of Health. 2002. Reducing Inequalities in Health. Wellington: Ministry of Health. Ministry of Health. 2006. The National Travel Assistance Scheme: Your guide for claiming travel assistance: brochure. URL: www.health.govt.nz/publication/national-travel-assistancescheme-your-guide-claiming-travel-assistance-brochure (accessed 14 August 2013). Ministry of Health. 2008. Health Equity Assessment Tool: A User’s Guide. Wellington: Ministry of Health. Ministry of Health. 2012a. Cancer: New Registrations and Deaths 2009. Wellington: Ministry of Health. National Lung Cancer Working Group. 2011. Standards of Service Provision for Lung Cancer Patients in New Zealand. Wellington: Ministry of Health. 60 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Phillips AJR, Lawes CM, Cooper GJ, et al. 2002. Ethnic disparity of pancreatic cancer in New Zealand. Journal of Gastrointestinal Cancer 31(1–3): 137–45. Soeberg M, Blakely T, Sarfati D, et al. 2012. Cancer Trends: Trends in cancer survival by ethnic and socioeconomic group, New Zealand 1991–2004. Wellington: University of Otago and Ministry of Health. Weir RP, Brunton CR, Blakely TA. 2002. Chronic liver disease mortality attributable to hepatitis B and C in New Zealand. Journal of Gastroenterology and Hepatology 17(5): 582–8. Prevention and early identification American Gastroenterological Association. 2011. American Gastroenterological Association Medical Position Statement on the Management of Barrett’s Esophagus. Gastroenterology 140(3): 1084–91. Bennett C, Vakil N, Bergman J, et al. 2012. Consensus statements for management of Barrett’s dysplasia and early-stage oesophageal adenocarcinoma, based on a Delphi process. Gastroenterology 143(2): 336–46. Bruix J, Sherman M. 2011. Management of hepatocellular carcinoma: An update. Alexandria: American Association for the Study of Liver Diseases. Canto MI, Harinck F, Hruban RH, et al. 2013. International cancer of the pancreas screening consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut 62: 339–47. European Association for the Study of the Liver, European Organisation for Research and Treatment of Cancer. 2012. EASL–EORTC Clinical Practice Guidelines: Management of hepatocellular carcinoma. Journal of Hepatology 56(4): 908–43. Kluijt I, Sijmons RH, Hoogerbrugge N, et al. 2012. Familial gastric cancer: guidelines for diagnosis, treatment and periodic surveillance. Familial Cancer 11: 363–9. Ministry of Health. 2011b. Targeting Smokers: Better Help for Smokers to Quit. Wellington: Ministry of Health. Ministry of Health. 2013b. Hepatitis B. URL: www.health.govt.nz/your-health/conditions-andtreatments/diseases-and-illnesses/hepatitis-b?qt-moh_topic_sheet_tabs=3#qtmoh_topic_sheet_tabs (accessed 26 August 2013). NZGG. 2009. Suspected Cancer in Primary Care: Guidelines for investigation, referral and reducing ethnic disparities. Wellington: New Zealand Guidelines Group. NZGG. 2011. Management of Early Colorectal Cancer – Evidence-based Best Practice Guidelines. Wellington: New Zealand Guidelines Group. Tanaka M, Chari S, Adsay V, et al. 2006. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology 6: 17–32. Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 61 Referral and communication Ministry of Health. 2012c. Rauemi Atawhai: A guide to developing health education resources in New Zealand. Wellington: Ministry of Health. NHS Wales. 2005. National Standards for Upper Gastro-intestinal Cancer Services. Cardiff: NHS Wales. Windsor JA, Rossaak JI, Chaung D, et al. 2008. Telling the truth to Asian patients in the hospital setting. New Zealand Medical Journal 121(1286): 92–9. Investigation, diagnosis and staging Bruix J, Sherman M. 2011. Management of Hepatocellular Carcinoma: An update. Alexandria: American Association for the Study of Liver Diseases. Edge SB, Byrd DR, Compton CC, et al (eds). 2010. AJCC Cancer Staging Manual (7th edition). New York: Springer. Goergen SK, Pool FJ, Turner TJ, et al. 2013. Evidence-based guideline for the written radiology report: Methods, recommendations and implementation challenges. Journal of Medical Imaging and Radiation Oncology 57(1): 1–7. Gormly KLM. 2009. Standardised tumour, node and metastasis reporting of oncology CT scans. Journal of Medical Imaging and Radiation Oncology 53(4): 345–52. NCCN. 2011a. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Hepatobiliary Cancers: Version 2 2012. Fort Washington: National Comprehensive Cancer Network (member access only). NCCN. 2011b. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Pancreatic Adenocarcinoma: Version 2 2012. Fort Washington: National Comprehensive Cancer Network (member access only). NHS Wales. 2005. National Standards for Upper Gastro-intestinal Cancer Services. Cardiff: NHS Wales. Multidisciplinary care Ministry of Health. 2012b. Guidance for Implementing High-Quality Multidisciplinary Meetings: Achieving best practice cancer care. Wellington: Ministry of Health. NHS Wales. 2005. National Standards for Upper Gastro-intestinal Cancer Services. Cardiff: NHS Wales. Supportive care Ministry of Health. 2010. Guidance for Improving Supportive Care for Adults with Cancer in New Zealand. Wellington: Ministry of Health. 62 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional Care coordination Jansen P, Bacal K, Crengle S. 2008. He Ritenga Whakaaro: Māori experiences of health services. Auckland: Mauri Ora Associates. Ministry of Health. 2006. The National Travel Assistance Scheme: Your guide for claiming travel assistance: brochure. URL: www.health.govt.nz/publication/national-travel-assistancescheme-your-guide-claiming-travel-assistance-brochure (accessed 14 August 2013). Treatment ANZGOSA. 2013a. Guidelines For Hospitals and/or Health Services to Assist in Credentialing of Surgeons Performing Gastric Resection in Australia and New Zealand (draft). Belair: Australian and New Zealand Gastric and Oesophageal Surgery Association. ANZGOSA. 2013b. Guidelines for Hospitals and/or Health Services to Assist in Credentialing of Surgeons Performing Oesophageal Resection in Australia and New Zealand (draft). Belair: Australian and New Zealand Gastric and Oesophageal Surgery Association. Hospice New Zealand. 2012. Standards for Palliative Care: Quality review programme and guide. Wellington: Hospice New Zealand. Joint Faculty of Intensive Care Medicine. 1997. Minimum Standards for Intensive Care Units. Review IC-1. Melbourne: Faculty of Intensive Care, Australia and New Zealand College of Anaesthetists. Liverpool Care Pathway. 2009. New Zealand Integrated Care Pathway for the Dying Patient. http://lcpnz.org.nz/pages/home (accessed 16 August 2013). Ministry of Health. 2001. The New Zealand Palliative Care Strategy. Wellington: Ministry of Health. NCCN. 2012c. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Palliative Care. Fort Washington: National Comprehensive Cancer Network (member access only). NHS Wales. 2005. National Standards for Upper Gastro-intestinal Cancer Services. Cardiff: NHS Wales. NICE. 2004. Guidance on Cancer Services: Improving Supportive and Palliative Care for Adults with Cancer. London: National Institute for Clinical Excellence. NZGG. 2009. Suspected Cancer in Primary Care: Guidelines for investigation, referral and reducing ethnic disparities. Wellington: New Zealand Guidelines Group. Palliative Care Australia. 2005. Standards for Providing Quality Palliative Care for all Australians. Canberra: Palliative Care Australia. World Health Organization. WHO’s Pain Ladder for Adults. URL: www.who.int/cancer/palliative/painladder/en (accessed 26 August 2013). Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional 63 Follow-up and surveillance Bruix J, Sherman M. 2011. Management of hepatocellular carcinoma: An update. Alexandria: American Association for the Study of Liver Diseases. NCCN. 2011a. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) – Hepatobiliary Cancers: Version 2 2012. Fort Washington: National Comprehensive Cancer Network (member access only). NZGG. 2011. Management of Early Colorectal Cancer. Wellington: New Zealand Guidelines Group. SIGN. 2006. Management of Oesophageal and Gastric Cancer: A national clinical guideline. Edinburgh: Scottish Intercollegiate Guidelines Network. Clinical performance monitoring and research IT Health Board. 2011. National Cancer Core Data Definitions, Interim Standard, HISO 10038.3. Wellington: Ministry of Health. Ministry of Health. 2005. Access to Cancer Services for Māori. Wellington: Ministry of Health. NHS Wales. 2005. National Standards for Upper Gastro-intestinal Cancer Services. Cardiff: NHS Wales. Appendices Ministry of Health. 2002. Reducing Inequalities in Health. Wellington: Ministry of Health. 64 Standards of Service Provision for Upper Gastrointestinal Cancer Patients in New Zealand - Provisional