Document 7366588
advertisement

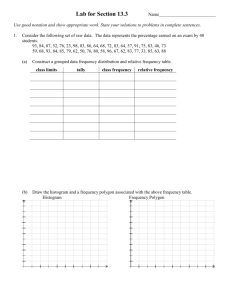
Pesticide Poisoning in Avian Communities Laura O’Byrne 2 Abstract Pesticide application has become an integral part of agricultural practices as anthropogenic population increase has accelerated the demand for food crops. Pesticides increase crop yields by chemically destroying pests such as weeds, insects and fungi. This increase in pesticide use has increased the concentration and amount of pesticides or they’re by -products that remains in soils and water bodies. Residence time of pesticides in environment and extent of the contaminants are reliant on many dynamic variables, biotic and abiotic. Therefore it is difficult to predict residence time, or what concentrations of pesticides will remain in the environment after application. Birds are omnivores whose diet includes seeds, insects and in some cases small rodents or other birds. Subsequently, avian communities often accidentally ingest environmental pesticides or their residues and this causes lethal or sub lethal effects. Introduction Soil, food chain relationships of pesticides have been extensively studied and have become increasingly important since Rachel Carson’s Silent Spring was published in the 1962. This book was instrumental in raising awareness of pesticide (DDT) poisoning in non-target organisms, specifically birds. A pesticide is intentionally applied to the environment and is designed to kill or harm living organisms (Cox, 2006). The top ten most common pesticides are shown in Table 1, with insecticides being the most prevalent in use today. Pesticide application (Figure 1) is succeeded by absorption by crop, adsorption to soil particles and the possibility of leaching or surface run off. Microorganisms, ultraviolet light or vaporization degrades pesticides. Multiple factors determine the 3 fate of a pesticide after application. The specific pesticide applied, the dose and concentration applied will be significant in determining how much of the compound is taken up by the intended pest and how much remains in the environment. Factors influencing infiltration and residence time include, soil type and condition of soil including soil texture, moisture and pH, soil organic matter, hydrologic conditions and how strongly a particular pesticide is bound by soil components. Pesticides disable pests by neurotoxicity, genotoxicity or impaired hormone function. (Cox, 2006). Neurotoxicity would commonly result in the death of an organism. Secondary poisonings are frequent in the food chain, where one organism ingests another who has consumed a pesticide. This process where toxins are magnified in the environment through food webs is called biomagnification. Toxins which were are lower levels when first taken into an organism become increasingly concentrated in its tissues which are then ingested by the next animal on the food chain and so on. Genotoxicity is a process that is not directly toxic to an organism but impairs it’s reproductive functioning through damage to the DNA or RNA. The likelihood that wildlife will be subjected to pesticides in their environment is great due to the widespread use of pesticides in agriculture. Pesticide compounds have a potential to remain in their environment for long periods of time and to travel far distances due aeolian, hydrologic and biological dispersion. A birds diet is varied and commonly they consume a combination of seeds, fruit and insects. These crops are very likely contaminated with pesticides. This makes the avian community vulnerable to unintentional poisoning by pesticides ingestion. Discussion Pesticide toxic effects: 4 Many insecticides are AChE inhibitors such as Malathion and Parathion (George, 1995). AChE inhibition, acetylcholoinesterase is a key enzyme in the nervous system down the neurotransmitter acetylcholine to choline and acetic acid at the synoptic cleft resulting in the next nerve impulse transmitting across the synaptic gap. Inhibition of this enzyme results in paralysis, convulsions and possible death. In George’s 1995 study of grasshoppers, he found that dead birds had been secondarily poisoned by ingesting grasshoppers and the dead birds were found to have and large amount of AChE in their bodies. In addition, this study found that AChE levels of 50% or greater resulted in sub-lethal of lethal effects on avian communities and Blus’ 1997 field studies concurred. According to Mullie and Keith 1993, pesticide application and bird mortality linked but it is unclear whether interaction is direct or indirect, via ingestion or reduction of food supply. Lee et al found evidence of airborne pesticide contamination levels of AChE in animals indicating that point sources of poisoning are not easily traced. Chemistry of Pesticides The EPA (Environmental Protection Agency) sets maximum contamination levels based on individual pesticides, but mixtures are most common in the environment. Many studies, including Reylea, 2008 and Stromberg, 1977 agree that multiple pesticide contamination in the environment is standard and that negative synergistic interactions between pesticides are not well understood. Current laboratory testing is only done on singular pesticides and lab testing is not comparable to the biological factors influencing a natural environment. Although “laboratory results showed no toxic effects of DDD (a metabolite of DDT), 1000 pairs of breeding Grebes were completely decimated in one year” following DDT application (Blus, 1997). Further complicating the pesticide toxic effects are the solvents or ‘inert’ ingredients that are added to the commercial pesticide formulations. These ingredients, which include solvents like ethanol or petroleum, are not required to be disclosed because 5 of their function in the product. Pesticide testing is restricted to active ingredients and for acute toxicity only. This testing does not include inert ingredients or sublethal effects. There is a 30% greater dermal exposure rate with inert ingredients then with active pesticide ingredients alone (Cox, 2006). In the Cox 2006 studies, he found that these inert ingredients could be both biologically and chemically active when added to the pesticide compound. It was determined by Reylea and Cox that these inert ingredients may have toxic effect independent of the pesticide itself and that some solvents increase environmental mobility and persistence of the compound. Metabolites of the pesticide compound themselves are toxic (Schwartz, 1967) and pesticides have a ‘life’ and interact with their surrounding environment. Physiochemical Factors Once a pesticide compound is released into the environment, many biotic and abiotic factors determine its fate. Some of these factors include climate, moisture, soil type, texture and pH and microclimate. Several studies have shown that an increase in moisture will increase both pesticide resilience and toxicity. Mullie and Keith found that shallow depressions also increase concentration and toxicity of pesticide compounds due to leaching and sediment run off from precipitation. Reylea found that multiple pesticides usually result in an increase in the soil pH while Douthwaite and his team determined that although heat increases dissipation of pesticide residues, it can take up to “12 years “ for pesticide residue breakdown in temperate soils. Chemistry and topography dictate pesticide persistence and these vary spatially and temporally. Blumhurst studied pesticide degradation and found that biological activity of microorganisms, which encourages pesticide degradation, is greater near the soil surface. If pesticide leaches, the biological activity decreases which increases its persistence. In studies by Braverman et al, found that dry soil beds “inactivate soil applied herbicides”. The same study found that although high temperatures of +25 degrees Celsius favor herbicide degradation, they also decrease 6 herbicide efficacy. This demonstrates how complicated pesticide application, degradation and persistence are to predict in a natural environment. Biological Components Pesticides are hydrologically pervasive and Reylea found that 60-95% of surface waters contain multiple pesticides. The trend of multiple pesticides increasing the negative effects to biotic communities is well documented in scientific studies. These studies agree that laboratory experiments are one-dimensional and cannot substitute for a natural habitat. Laboratory experiments test one organism and not a whole biotic community. Multiple studies agree that “persistence residence time is not predictable” (Stone, 1984) in a natural environment which is multifactor and not static like the laboratory (Table 2). A 1995 study by Douthwaite showed that DDT was still present in fat tissues of animals although DDT was banned over forty years earlier. In Stones’ study of crickets, they found DDT and it’s metabolites present in 22/25 sampled crickets. This study indicates that pesticide residence time is not predictable and that ecological pathways should be considered in pesticide application. Avian Consequences Birds are opportunistic feeders whose diet varies according to availability, and their life cycle requirements. Adult birds who are granivores often change their diet to an entirely insectivorous diet when mating season begins and continue this diet through their breeding season and well into their chicks’ development (Sibley, 2001). The process of biomagnification is well understood in the scientific community and this process has a crucial effect on avian communities. An avian diet often includes 7 insects that accumulate toxins directly from their soil environment. Korschgen found that pesticide residues or degradation products in soils “directly poison macro invertebrates”, these include, earthworms, beetles, toads snakes and mice. These organisms are common prey for many bird species and by ingesting this food source; the birds accumulate toxins at a much higher rate then in the original organism. Korschgens’ studies included some examples of biomagnification; Earthworms and toads in their study had toxin levels of 5.65ppm and 8.30ppm respectively. The snakes that ingested these organisms further accumulated the toxins in their tissues at a much higher rate of up to 10.3-14.4ppm. The fundamental factor influencing effects of pesticides on avian communities is the timing of the application. Insecticides applied during breeding season have a “negative effect of reproduction, diet and chick mortality (George, 1995). Avian parents insectivorous diet is essential in order for chicks to achieve maximum growth and health (Alsop, 2001). Therefore, a decrease in avian prey during breeding season can result in many dire consequences for parents and chicks (Table 3). Reproductive failure is a well-documented phenomenon in pesticide treated avian communities. This can range from eggshell thinning, which makes the shells more vulnerable to breaking and therefore decreases the viable eggs hatched. Decrease in avian parents diet may result in a lower birth weight of chicks and higher chick mortality due to starvation or malnutrition (George, 1995). Stromberg also found a direct relationship between pheasant egg laying, which declined with an increase in seed treated by pesticides. Mullie and Keith documented a decrease in “fledgling weight, decreased singing and mating rituals that result in a decrease in breeding and future populations of birds". Adult weights of birds also decreased in pesticide treated areas and parents have been known to abandon chicks as a result of sub-lethal toxicity or decrease in food supply. Lethal and sub-lethal effects of pesticide poisoning are at times more subtle and difficult to ascertain. Although rarely documented, farmers have been known to intentionally use insecticides to kill birds in an attempt to preemptively save their crops (Schwartz, 8 1967). This has been confirmed through necropsies of masses of corpses in agricultural areas and subsequent confessions of farmers. The number of intentionally poisoned birds may be vastly underestimated due to the mobility of birds and sub-lethal effects. In human studies dermal and inhalation exposure is the most common form of pesticide absorption. Adverse health effects have been documented in agricultural workers and persons living in close proximity to agricultural fields. Human exposure yields similar health effects to animals exposed that include reproductive disruption, increase in blood pressure and heart rate (Ward, 2000). Wennergren and Stark found evidence of second-generation effects and increased infant mortality in birds from pesticides in environment. Indirect effects of pesticide poisoning include a decrease or alteration in native vegetation due to herbicide application. The result of this is a decrease in potential or preferred habitat of birds that alters the biological community in unpredictable ways. Herbicides have the potential to decrease food supply, reduce possible prey that can culminate in extirpation or even extinction of species in an area (Blus, 1997). Studies by Blus showed that DDT and its metabolites, DDD and DDE have been shown to biomagnify in the food chain. DDT metabolites, which do not have directly toxic effects, can accumulate in body tissues as AChE inhibitors. Geluso determined that pesticides could accumulate in fatty tissues in most organisms. These fat stores of toxin are released during times of stress. Avian lives often include two migrations per year. Migrations, breeding, molt, illness and cold temperatures would all be classified as stress to birds. During these times, the toxins stored in the fatty tissues become released and accumulate in the brain that causes a delayed lethal effect. In studies done by Cox, it was found that birds, specifically gulls and terns that have 9 come into contact with pesticides have initiated homosexual pairings, which results in a zero reproductive success rate. Birds’ exposure to pesticides is considerable given their life cycle and feeding strategies. Exposure can be lethal to sub-lethal but both may be difficult to identify due to the mobility of birds, predation of bird corpses and the secretive nature of certain avian species (Mullie and Keith). Toxicity through biomagnifications, AChe inhibition, reproductive success or intentional poisoning are constant risks for avian communities. Conclusion Bait application has proven to be a less toxic option to pesticide control by reducing non-target organism poisonings due to reduction in dermal or inhalation exposure and reduction in amount chemicals applied (George, 1995). The ripple effect of decreased prey or ingestion of contaminated prey results in wide ranging effects on the food web. A particular species may be eradicated or extirpated which will affect its predators and it’s biotic habitat. Western Meadowlarks declined in Luke’s study due to a decrease in food supply (grasshoppers), an increase in toxicity of surrounding environment, and decreased productivity or reproduction due to sub-lethal effects (George, 1995). IPM goals should be considered, before pesticide applications. In the case of grasshoppers, birds reduce grasshopper populations and maintain them at endemic levels. Adverse effect of pesticides on grasshopper predators may be counterproductive to long-term pest management goals. If the predator is extirpated, the prey may develop a pesticide resilience that would increase the 10 pressure to use more pesticides that would potentially exacerbate the initial problem. Life histories are important factors to consider when determining sensitivity to birds and treated areas. Different organisms have varying sensitivity to various pesticides. Factors such as habitat preference, diet, feeding strategies and systematic relationships vary between species. Although lethal and sub-lethal effects are at times difficult to ascertain, there is enough evidence through laboratory and field studies to show that pesticide toxicity will continue to increase with an increase in pesticide use. Using a ‘cocktail ‘ of pesticides will result in more detrimental effects on all organisms. Biotic and abiotic conditions in the environment are not static and the precautionary principle should be considered before bombarding the environment with more pesticides. Biological controls could be a consideration in order to increase crop yields while decreasing a growing reliance on pesticides. Ecological toxicity is complicated and both short term and long term studies should be employed to gain a stronger understanding of this complex process of pesticide poisoning in avian communities and non target organisms. Ecological toxicity is complicated and need long-term studies to begin to understand pieces of this complex process. 11 Table 1 12 Table 2 13 Table 3 14 Figure 1 15 Bibliography Alsop, Fred, Smithsonian Birds of North America, 2001. Blumburst, Michael, Experimental Parameters Used to Study Pesticide Degradation in Soil, 1996. Weed Technology, Vol. 10, No.1, p 169-173. Blus, L., and Henry, C., Field Studies on Pesticides and Birds: Unexpected and Unique Relations, 1997. Ecological Applications, Vol. 7, No.4, p-1125-1132. Braverman, M., Dusky, J., Locascio, S., Hornsby, A., Sorption and Degradation of Thiobencarb in Three Florida Soils, 1990. Weed Science, Vol. 38, No. 6, p 583-588. Carson, Rachel, Silent Spring, 1962 Cox, C. and Surgan, M., Unidentified Inert Ingredients in Pesticides: Implications for Human and Environmental Health, 2006. Environmental Health Perspectives, Vol. 114, No. 12, p 1803-1806. Douthwaite, R.J., Occurrence and Consequences of DDT Residues in Woodland Birds Following Tsetse Fly Spraying Operations in NW Zimbabwe, 1995. Journal of Applied Ecology, Vo. 32, No. 4, p 727-738. Geluso, K., Altenbach, J., and Wilson, D., Bat Mortality: Pesticide Poisoning and Migratory Stress, 1976. Science, New Series, Vol. 194, No. 4261, p 184-186. George, L., McEwen, L., Petersen, B., Effects of Grasshopper Control Programs on Rangeland Breeding Bird Populations, 1995. Journal of Range Management, Vol. 48, No. 4, p 336-342. 16 Korschgen, Leroy, Soil-Food-Chain-Pesticide Wildlife Relationships in AldrinTreated Fields, 1970. The Journal of Wildlife Management, Vol. 34, No. 1, p 186-199. Lee, S., McLaughlin, R., Harnly, M., Gunier, R., and Kreutzer, R., Community Exposures to Airborne Agricultural Pesticides in California: Ranking of Inhalation Risks, 2002. Environmental Health Perspectives, Vol. 110, No. 12, p 1175-1184. Mullie, W., and Keith, J., The Effects of Aerially Applied Fenithrothion and Chlorpyrifos on Birds in the Savannah of Northern Senegal, 1993. Journal of Applied Ecology, Vol. 30, No. 3, p 536-550. Relyea, Rick, A Cocktail of Contaminants: How Mixtures of Pesticides at Low Concentrations Affect Aquatic Communities, 2008. Oecologia, Vol 159, p 363-376. Schwartz, Henry, Microbial Degradation of Pesticides in Aqueous Solutions, 1967. Water Pollution Control Federation, Vol. 39, No. 10, Part 1, p 1701-1714. Sibley, D.A., The Sibley Guide to Bird Life and Bevohavior, 2001. Stone, W., Overmann, S., and Okeniewski, J., Intentional Poisoning of Birds with Parathion, 1984. The Condor, Vol. 86, No. 3, p 333-336. Stromberg, K., Seed Treatment Pesticide Effects on Pheasant Reproduction at Sub lethal Doses, 1977. The Journal of Wildlife Management, Vol. 41, No.4, p 632-642. Ward, M., Nuckols, J., Weigel, S., Maxwell, S., Cantor, K., and Miller, R., Identifying Populations Potentially Exposed to Agricultural Pesticides Using Remote Sensing and a Geographic Information System, 2000. Environmental Health Perspectives, Vol, 108, No. 1, p 5-12. 17 Wennergren, U., and Stark, J., Modelling Long-Term Effects of Pesticides on Populations: Beyond Just Counting Dead Animals, 2000. Ecological Applications, Vol. 10, No. 1, p 295-302.