Word Doc
advertisement

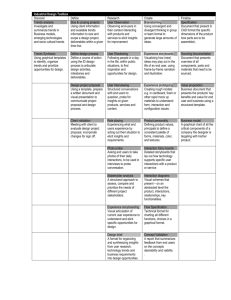
Probe Report Title: Authors: Assigned Assay Grant #: Screening Center Name & PI: Chemistry Center Name & PI: Assay Submitter & Institution: PubChem Summary Bioassay Identifier (AID): Probe Structure & Characteristics: Drawing of Structure CID ** Target Name‡ IC50/EC50 (nM) [SID, AID]† Anti-target Name(s)‡ IC50/EC50 (μM) [SID, AID]† Selecti vity* Secondary Assay(s) Name: IC50/EC50 (nM) [SID, AID]§ ‡Short descriptive name of target or pathway (similar for antitarget, if applicable). †IC50/EC50 value in nM along with the PubChem SID and AID where this value can be found. §For secondary assay provide the following information: Name, IC50/EC50 [SID, AID] *Selectivity = antitarget IC50/target IC50 **Provide the PubChem CID for the probe. Recommendations for the scientific use of this probe: Template G Center Probe Report Guidelines Instructions Probe reports are generated for each structurally unique compound that meets or surpasses the MLPCN requirements for a new probe and the specific characteristics specified in the CPDP for the probe project. The probe report should be submitted at this link (https://mli.nih.gov/mli/upload-probe-reports/) on the MLI website. The first page of each report will adhere to the defined probe template providing a standard format for every report (similar to an executive summary). Following pages should conform to a document one could publish in a journal. Template for the title page, 1st page: It is important to retain the title and order of each section of the 1st page. Title: Provide a suitable title for the probe report. Authors: Provide a list of authors. Authors should be listed according to standard peer-review publication rules for assigning authorship. In the footnote please indicate affiliation and email contact. Probe Structure & Characteristics: Provide a drawing (with stereo-chemistry if known) for the probe. In a table provide the PubChem CID, the target and anti-target names, the IC50/EC50 value in nM. When providing these values, also specify the PubChem SID as well as the PubChem AID where this value is to be found. Recommendations for scientific use of the probe: A brief description how/where/why this probe will be useful. General Guidelines The text of the actual report, i.e., starting on page 2, should be written like a scientific publication and can be structured in any way you deem appropriate. You can include a logo for your center if you choose. An MLPCN center logo may be placed at the top of the first page of the report (second page after the title page). Please ensure that the following pieces of information are present in the report: 1. 2. 3. 4. Scientific Rationale for Project Project Description a. For each assay implemented and screening run please provide i. PubChem Bioassay Name(s), AID(s), Assay-Type (Primary, DR, Counterscreen, Secondary) ii. Assay Rationale & Description (when describing primary screen it would be useful to see standard metrics like, Z’, S:B for the optimized assay.). Table of reagents and source. iii. Center Summary of Results b. Probe Optimization i. Describe SAR & chemistry strategy (including structure and data) that led to the probe. Probe a. Chemical name of probe compound b. Probe chemical structure including stereochemistry if known c. Structural Verification Information of probe SID d. PubChem CID (corresponding to the SID) e. If available from a vendor, please provide details. f. Describe mode of action for biological activity of probe g. Detailed synthetic pathway for making probe h. Center summary of probe properties (solubility, absorbance/fluorescence, reactivity, toxicity, etc.) and i. A tabular presentation summarizing known probe properties Appendices (useful in a tabular form): Template G a. 5. Comparative data on (1) probe, (2) similar compound structures (establishing SAR) and (3) prior probes b. Comparative data showing probe specificity for target Bibliography Tables summarizing information in the text or appendices should contain the following pieces of information: 1) Data tables should contain along with the value the Pubchem SID & AID where the data is to be found. Please indicate in the table whether or not the assay was performed from a powder sample as well as whether or not the powder sample went through structure confirmation method as well as %-purity & method. 2) It would be generally useful to provide a table for the SAR undertaken again with the information in (A) above. If compound is available from a vendor please indicate this. 3) Please provide a table of all AIDs used in the project, while identifying the target, concentration of compound, Bioassay-Type (primary, counterscreen, etc.). 4) If you are proposing a new chemotype as a 2 nd probe then please provide a separate table summarizing the similarities/differences with the previous probe(s). It would also be nice to see what specific characteristic of this probe makes it useful as compared to the previous probes, it might be e.g., something like a different specificity profile, a more extensive SAR, etc. Template G