introduction
advertisement

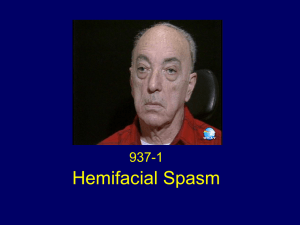
ב"ה Facial Nerve Palsy after Acute Exposure to Dichloromethane R.M. Jacubovich, MD, MOccH, D. Landau MD, Y. Bar Dayan, MD, MHA, M. Zilberberg, MOccH, L. Goldstein MD MHA. Correspondence: Lt. Colonel Dr. Yaron Bar-Dayan, Deputy Surgeon general IAF, MD, MHA. Surgeon general headquarters IAF Adress: 16 Dolev St. Neve Savyon, Or Yehuda, Israel. Phone: 0097236341039 Fax number: 0097237377468 E-mail: bardayan@netvision.net.il ABSTRACT BACKGROUND: Dichloromethane poisoning affects predominantly the central nervous and the cardiovascular systems, and results from both carboxyhemoglobin formation and direct solvent-related narcosis. Exposure is frequently occupational and related to paint-stripping, and several reports have described severe adverse affects as well as fatalities. Conflicting reports regarding peripheral nerve toxicity have been found with no reports of clinical acute toxicity heretofore. CASE REPORT: We present a case report of a patient who developed a facial nerve palsy after acute occupational exposure to Dichloromethane. The patient was part of a paint removing crew who have worked without proper protecting measures and were thus exposed to high levels of Dichoromethane. RESULTS: The patient was involved in Paint stripping with Dichloromethane, and developed acute facial nerve palsy. Other known causes of facial palsy were excluded, and although idiopathic palsy cannot be ruled out, there were no corroborating findings. Carboxyhemoglobin levels taken after a significant delay were normal. CONCLUSION: This is the first article that describes a case of Facial Nerve Palsy related to acute dichloromethane exposure, indicating a possible peripheral neurotoxic effect of this solvent. KEYWORDS: Dichloromethane; Methylene chloride; Facial nerve palsy; Paint stripping; Paint remover; Toxicity; Ventilation; Occupational exposure. INTRODUCTION Dichloromethane ( DCM or Methylene chloride ) is a clear, colorless liquid with a mild sweet odor that can be detected at concentrations of 100-300 parts per million (ppm) (ATSDR ,1993 ; ATSDR, 1998; Mohammad and Stefanos, 1999). Dichloromethane is lipophilic and is an excellent solvent for preparations of paint removers, degreasing agents, aerosol propellants, paint and varnish thinners, fire extinguishers, adhesives and in a variety of other industrial settings (Zenz et al.,1994). Exposures to high concentrations of Dichloromethane are generally occupational (ATSDR, 1998). The principal route of human exposure is inhalation. Skin absorption is usually small because of rapid evaporation. Following absorption, DCM is distributed mainly to the liver, brain and adipose tissue (ATSDR, 1993). The liver is the primary site of metabolism. Metabolic conversion to carbon monoxide occurs and the half-life of carboxyhaemoglobin is almost three times that following an equivalent inhalation of carbon monoxide. This is because hepatic biotransformation to carbon monoxide is dependent on the enzymatic metabolic rate, and the rate at which DCM is released from tissue stores. Consequently, carboxyhaemoglobin production may continue for several hours following cessation of exposure to DCM (Hayes & Laws, 1991; Leiken et al., 1990 ). Hepatic conversion occurs via two pathways (Hayes & Laws,1991): 1. Mixed functions oxidase system of cytochrome P450. A high affinity low capacity pathway, forming carbon monoxide, carbon dioxide and chloride, via a formylchloride intermediate. This pathway is associated with detoxification and is saturable at a few hundred ppm. 2. Cytosolic transformation (glutathione transferase dependent) where formaldehyde and formic acid intermediates are produced. This is a low affinity high capacity system associated with intoxication which shows no indication of saturation up to vapour concentrations of 10,000 ppm. The extent to which each pathway contributes to total hepatic metabolism varies in humans, especially with exposure levels. Thus, toxicity extrapolation between high and low doses is complex. Methylene chloride and its metabolites are chiefly excreted via the lungs with small amounts appearing in the urine and bile. Low doses of 14C-labelled methylene chloride were excreted mainly as 14C-carbon monoxide (with 14C-carbon dioxide) whereas high concentrations were excreted in the expirate largely unchanged as 14C-methylene chloride (IPCS 1997). Toxic effects of DCM have been observed following its inhalation, due to direct central nervous system depression (Zarrabietia et al., 2001, Leiken et al., 1990) or from the effect of carbon monoxide on the central nervous and cardiovascular systems (Rioux et al.,1989; Pankow, 1996; Savolainen, 1989; Stewart and Hake, 1976). The most serious manifestations of DCM toxicity are unconsciousness and death, and a number of fatalities have been reported in the literature (Rioux and myers,1989;Savolainen, 1989; Leiken et al.,1990;, Stewart and Hake, 1976; Manno et al., 1989; Manno et al., 1992; Goulle et al., 1999; Novak and Hain, 1990; Winek et al., 1981). Most of these cases were associated with furniture paint stripping. The objective of this article is to describe a case of facial nerve palsy in a soldier who was acutely exposed to Dichloromethane in a paint stripping operation. CASE REPORT Eleven Israeli Air Force soldiers used a liquid paint stripping formulation named TURCO 5873, containing DCM to remove old paint stains from the floor of a small building. Four soldiers worked in the entrance to this building, 4 soldiers in the hall ( 2 meter width and 10 meter length), and 3 soldiers worked in a closed small room, 25 square meters in size, with one door that opened to the hall, and one window 10 cm*10 cm in size. There was no ventilation device in the room. Shortly after they began working they experienced headache, dizziness and throat irritation. They left the room every few minutes as they felt insufficient ventilation and excess solvent vapor in the room-air. They wore no appropriate protective gloves or masks. After a total of about three hours the work was stopped due to excess vapors in the room, and exacerbation of symptoms. Two hours later, one of the soldiers who worked in the small room, reported to the base clinic. This patient was 21 years old, non-obese of average height and weight, who was known to have an atrophic left kidney without additional co-morbities or substance abuse (including tobacco and alcohol). He reported of headache, dizziness and nausea. His vital signs were normal as well as the rest of his physical examination including a thorough neurological exam. The attending physician ordered a change of clothes, and he then received IV hydration and a mild analgesic. Upon waking up the next morning the patient noticed weakness and asymmetry of the left side of his face an immediately went to the base clinic. He was re-interviewed and reported no preceding viral infection, tinnitus, hearing diminution, pain behind the ear, hyperacussis or taste loss. Additionally, there were no reports of lyme disease in the areas in which he lives and works nor did he report any significant travel history. There were no other manifestations indicating lyme disease (typical rash, arthralgia etc.). Physical examination revealed features compatible with left peripheral facial nerve palsy with drooping face and mouth. The palpebral fissure appeared widened and the forehead smooth. Upper and lower parts of the face were affected. There was also a decrease in sensation over the left side of the face. A possible mild weakness of the muscles of mastication on the left side was also noted. The rest of the exam was normal including tympanic membranes and a thorough neurological examination. The patient was referred to the emergency department of a nearby hospital. The diagnosis of peripheral facial nerve palsy was confirmed. Laboratory tests revealed normal kidney and liver function tests and oxygen saturation of 98%. Blood carboxyhemoglobin levels were 0.3-0.4% on serial tests. He was admitted for observation and received a short course of 1mg/kg Prendisolone. After his D/C he had a rapid improvement and an almost complete recovery after about three weeks from the event, although he continued to complain of left temporal headaches for several weeks. There is currently no facial asymmetry and only a slight weakness of the affected side. The other ten soldiers involved in the incident were summoned after the patient was diagnosed with facial palsy. Nine soldiers reported symptoms such as headaches, dizziness and nausea for several hours after the event, but were all symptom free by the next morning. They had no abnormal findings on physical examination and a laboratory evaluation that included carboxyhemoglobin levels, liver and kidney function tests was normal. DISCUSSION In the case presented, a worker engaged in paint stripping with a solvent containing DCM, in an enclosed space without adequate body protection, developed facial nerve palsy. Carboxyhemoglobin levels were found in the normal range 27 hours after the exposure. The delay was due to the late development of the facial Nerve palsy and referral to the hospital. The half-life of carboxyhemoglobin is about 13 hours in DCM exposure compared with 4 hours in direct carbon monoxide intoxication (Ratney et al.,1974). The low levels found in this case may be due to the delay in the carboxyhemoglobin sampling. Additionally, carbon monoxide production varies in different exposure scenarios due to the complex metabolic breakdown. Moreover, even after accidental DCM fatality, carboxyhemoglobin blood levels were low despite lethal levels of dichloromethane in the blood (Zarrabetia et al.,2001). Rioux and Myers (1989) reported two workers who were found unconscious in a semienclosed area with a high level of DCM fumes, with initial presenting Carboxyhemoglobin levels of only 5 and 7%. A compilation of data from case reports in the literature, show that symptoms like nausea, light-headedness, dizziness, headaches, correlate with air DCM levels of 1000-5000 ppm, thus, the high probability of a significant exposure in our case. This level is far greater than the ambient exposure standard of 50 ppm recommended by the American Conference of Governmental Industrial Hygienists as the Threshold limit value (TLV). The primary target organ of DCM is the central nervous system (ATSDR,1993; Savolainen, 1989; Hall and Rumack,1990). Both the direct neurologic effects of DCM and carbon monoxide toxicity appear to contribute to the adverse effects of DCM exposure. During acute and intense exposures to DCM, which usually occur in poorly ventilated areas, the direct solvent-narcotic effect may play a greater initial role in central nervous system depression (Savolainen, 1989; Leiken et al.,1990; Hall and Rumack, 1990; Bakinson and Jones, 1985; DiVincenzo and Kaplan, 1981). Clinical peripheral nervous system toxicity of DCM was not described heretofore, although one report has linked the exposure to another chlorinated solvent (trichloroethylene) with facial anesthesia and pupillary response indicating a peripheral cranial nerve injury (Feldam RG and Mayer RF, 1968). Regarding the possible effect of DCM on the peripheral nervous system, there are conflicting reports. Workers chronically exposed to DCM reported excess neurological symptoms compared with a non-exposed referent group, including numbness and tingling in the hands and feet. No evidence, however, of slowed motor nerve conduction velocity in either the ulnar or median nerves was found (Cherry et al., 1981). The effects of organic solvents on the myelin sheath of peripheral nerve tissue, was studied under specified experimental conditions. This study demonstrated that DCM produced complete disorganization of the myelin structure within a few hours after exposure, indicating possible peripheral nerve toxicity (Rumsby and Finean, 1966). Experimental neurotoxicologic evaluation of exposure to high DCM concentrations in rats, revealed no evidence of peripheral nerve pathology (Mattsson et al., 1990) whereas another study revealed a decrease in sciatic motor conduction velocity after intraperitoneal administration of DCM in rats (Winneke, 1981; Pankow et al., 1979). Facial nerve palsy following acute DCM exposure has not been described in the literature. Although known causes of facial nerve palsy (Cocker and Vrabec 2003) such as lyme disease, diabetes mellitus, otits media or externa, neoplasia and ramsy- hunt syndrome are highly unlikely it this case because of lack of appropriate anamnestic and physical findings and indolent clinical course, we cannot rule out the possibility of idiopathic facial palsy. Nevertheless the temporal correlation with an acute DCM exposure, and the lack of collaboratory evidence to support the diagnosis of idiopathic facial palsy, raises the possible causal role of DCM toxicity. CONCLUSSION This is the first article, as far as we know, that describes a possible link between acute dichloromethane exposure and facial nerve palsy. REFERENCES ATSDR. Methylene chloride toxicity. Agency for Toxic Substances and Disease Registry. Am Fam Physician 47:1159-1166 (1993) ATSDR. Toxicological Profile for Methylene Chloride. Atlanta, GA:Agency for Toxic Substances and Disease Registry, 1998. Bakinson MA, Jones RD. Gassing due to Methylene chloride, xylene, toluene, and styrene reported to Her Majesty Factory Inspectorate. Br J Ind Med 42:184-190 (1985). Cherry N, Venables H, Waldron HA, Wells GG. Some observations on workers exposed to methylene chloride. BJIM 38:351-355 (1981). Cocker NJ and Vrabec JD, Acute paralysis of the facial nerve. Head and Neck Surgery-Otolaryngology, 3rd Edition, Bailey BJ, Lippincott Williams and wilkins, Philadelphia, 2003. DiVincenzo GD, Kaplan CJ. Uptake, metabolism and elimination of methylene chloride vapors by humans. Toxicol Appl Pharmacol 59:130-140 (1981). Feldman RG, Mayer RF. Studies of trichlorethylene intoxication in man. Neurology. 1968 Mar;18(3):309. Goulle JP, Lacroix C, Vaz E, Rouvier P, Proust B. Fatal case of dichloromethane poisoning. J Anal Toxicol 23:280-283 (1999). Hall AH, Rumack BH. Methylene chloride exposure in furniture-stripping shops: ventilation and respiratory practices. J Occup Med 32:33-37 (1990). IPRS, Methylene chloride toxicity. International Programme on Chemical Safety, 1997. Leikin JB, Kaufman D, Lipscomb JW, Burda AM, Hryhorczuk DO. Methylene chloride: report of five cases and two deaths. Am J Emerg Med 8:534-537 (1990). Manno M, Chirillo R, Dannioti G, Cochea V, Albrizio F. Carboxyhemoglobin and fatal methylene chloride poisoning (letter). Lancet 2(8657):274 (1989). Manno M, Rugge M, Cocheo V. Double fatal inhalation of dichloromethane. Hum Exp Toxicol 11:540-545 (1992). Mattsson RR, Albee RR, Eisenbrandt. Neurotoxicologic Evaluation of Rats after 13 weeks of Inhalation Exposure to Dichloromethane or Carbon Monoxide. Pharmacology Biochemistry and behavior 36:671-681 (1990). Mohammad M, Stefanos NK. Methylene chloride poisoning in a cabinet worker. Environ Health Perspectives, Volume107, 9:769-772 (1999). Novak JJ, Hain JR. Furniture stripping vapor inhalation fatalities: Two case studies. Appl Occup Environ Hyg 23:380-383 (1990). Pankow D, Gutewort R, Glatzel W, Tietze K. Effect of dichloromethane on the sciatic motor conduction velocity of rats. Experientia 35:373-374 (1979). Pankow D. Carbon monoxide formation due to metabolism of xenobiotics. In: Carbon Monoxide (Penney DG, ed). New York: CRC Press, 1996; 25-43. Ratney RS, Wegman DH, Elkins HB. In vivo conversion of methylene chloride to carbon monoxide. Arch Environ Health 28:223-226 (1974). Rioux PJ, Myers RAM. Hyperbaric oxygen for methylene chloride poisoning : report on two cases. Ann Emerg Med 18:691-695 (1989). Rumsby MG, Finean JB. The action of organic solvents on the myelin sheath of peripheral nerve tissue-Chlorinated hydrocarbons. Journal of Neurochemistry 19:1513-1515 (1966). Savolainen H. Carboxyhemoglobin and fatal methylene chloride poisoning. Lancet 2(8665):748-749 (1989). Stewart RD, Hake CL. Paint removal hazard. JAMA 235:398-401 (1976). Winek CL, Collom WD, Esposito F. Accidental methylene chloride fatality. Forenscic Sci Int 18:165-168 (1981). Winneke G. The Neurotoxicity of Dichloromethane. Neurobehavioral Toxicology and Teratology 3:391-395 (1981). Zarrabeitia MT, Ortega C, Altuzarra E, Martinez MA, Mazarrasa O, Calvet R. Accidental dichloromethane fatality: a case report. J Forensic Sci 46(3):726-7. Zenz C, Dickerson OB, Horvath EP. Occupational Medicine 3rd edition, page 721 (1994).