Lab Pure Substance vs. Mixture V2Guide
advertisement

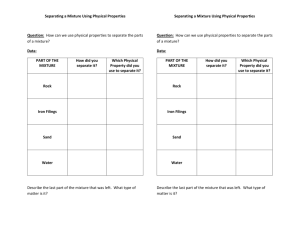
Name:_____________________________________________ Date:_________________ Title: Lab Demo: Pure Substance vs. Mixture Partner/Group _____________________________ Purpose: _____________________________________________________________________________________ Materials: Apparatus: Beakers, test tubes and rack, magnet, bunsen burner, test tube holder Samples of salt, sand, iron filings, powdered sulfur, sugar cubes, water and ethanol (alcohol) Safety: _______________________________________________________________________________________ Procedure: Pictorial Flow Chart _____________________________________________________________________________________________ _____________________________________________________________________________________________ _____________________________________________________________________________________________ _____________________________________________________________________________________________ _____________________________________________________________________________________________ _____________________________________________________________________________________________ 1. Mixture of salt, sand and iron filings, powdered sulfur. 2. Mixture of alcohol and water 3. Device a plan on how to separate the solid mixture and liquid mixture. Activities Activity A Mixture of Solid: a) salt and sand b) Iron Filings and Powdered Sulfur Burning the mixture of iron and sulfur. Activity B Mixture of Liquid: 10 mL samples of water 10 mL aalcohol in separate test tubes. Some Questions to think about. 1. How would you separate the solid mixture of salt and sand? Iron filings and powered sulfur? 1. List the characteristic properties of water and alcohol: Properties appearance odor flammability density melting point boiling point Water Alcohol 2. What would be the density of 50-50 alcohol and water? 3. How would you know if the mixture has more water? Activity C Solubility of Solvent Add sugar cube into the mixture of water and alcohol Describe the solubility of water vs. alcohol as a solvent. Activity D Separation of alcohol and water (Fractional Distillation) Device a plan how would you separate 50-50 alcohol water. Data and Observation: Create a table to organize your observations. Analysis: Use your observation to analyze the result of the separation of mixture. Conclusions: Use the rubric to answer the questions