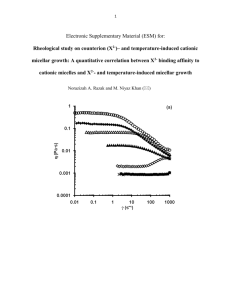
150715225058ManuOralSusChemE2015

SusChemE 2015
International Conference on Sustainable Chemistry & Engineering
October 8-9, 2015, Hotel Lalit, Mumbai
__________________________________________________________________________________________
Aqueous NaOH-Mixed Micellar Catalytic System: Green, Efficient and reusable media for for Cross Aldol Condensation Reaction
Manu Vashishtha
1,2
, Manish Mishra
1
*
, D. O. Shah
1,3
1
Department of Chemical Engineering & Shah-Schulman Center for Surface Science and Nanotechnology,
Faculty of Technology, Dharmsinh Desai University, College Road, Nadiad – 387 001, Gujarat, India
2
Department of Chemical Engineering Indian Institute of Technology, Bombay-400076
3
Department of Chemical Engineering and Department of Anesthesiology, University of Florida, Gainesville,
Florida, USA
E-mail addresses: manu.vash@gmail.com
, manishorgch@gmail.com
, dineshoshah@gmail.com
1. Introduction:
In recent years, there has been increasing interest in mixed surfactant based micellar systems due to the interesting physicochemical properties shown by these mixed compositions [1]. Anionic/cationic surfactant mixtures have been explored for applications like solubilisation, coating, dispersion, and boundary lubrication
[2]. Earlier, we have studied the catalytic effects of aqueous NaOH-cetyltrimethylammonium bromide (CTAB) micellar system on the cross aldol condensation of benzaldehyde and cyclohexanone [3].
In the continuation, present work shows the catalytic performance of mixed micellar system on the base catalyzed cross aldol condensation of benzaldehyde and cyclohexanone to α,ά-bis (benzylidene) cyclohexanone as model reaction. We also investigated the influence of chain length compatibility and the nature of head group of co-surfactant on the micellar stability and its influence on the catalysis (Fig. 1).
Fig.1. Schematic diagram representing the effect of chain length compatibility and nature of head group on molecular packing at the air/water interface .
2. Material and Methods:
All chemicals were from SD fine and used as such. All reactions were performed in a 50 mL reaction tube of reaction station (Radleys Discovery Technologies, UK), 10 mL of QAS aqueous solution (of required concentration) or water (without surfactant) was taken and a mixture of benzaldehyde (10 mmol) and cyclohexanone (5 mmol) was added in the solution under stirring. The NaOH was dissolved in the solution and the reaction mixture was stirred at 60°C for the required period of time. After the completion of reaction, the reaction mixture was neutralized with concentrated HCl and excess of saturated NaCl solution was added to reduce the surfactant concentration below the cmc. The organic phase was extracted with ethyl acetate (10 mL) and was analyzed by gas chromatography (Agilent 5975) having a HP-5 (60 meter, 250 µm diameter) capillary column with a programmed oven temperature from 50 to 280
C, at 1 mL min -1 flow rate of N
2
as carrier gas and
FID detector. The conversion of cyclohexanone was calculated on the basis of its weight percent.
Characterization of micellar stability was carried out by foaming properties, (foamability, foam stability), dynamic and equilibrium surface tension.
SusChemE 2015
International Conference on Sustainable Chemistry & Engineering
October 8-9, 2015, Hotel Lalit, Mumbai
__________________________________________________________________________________
3. Significant Results and Discussion:
In comparison to pure C
16
TAB, C
16
TAB + C
16
OH and C
16
TAB + C
16
PO
4
K were found to be stable and gave higher conversion than other mixed micellar systems. Higher solubilisation of reactants molecules in the stable micellar system helped in the enhancement of conversion. The highest micellar stability was observed for
C
16
TAB + C
16
PO
4
K + which was found to be attributed to both the chain length compatibility and the Coulombic interaction of the surfactant and co-surfactant head groups, resulting in an optimum packing of molecules in micelles. However, the selectivity of the desired product ( 3 )was lesser with C
16
TAB + C
16
OH and C
16
TAB +
C
16
PO
4
K than pure C
16
TAB, which was found to be due to decreased surface charge density on the micellar which resulting to less population of OH - at micellar surface. highest micellar stability was observed for C
16
TAB
+ C
16
PO
4
K + which was found to be attributed to both the chain length compatibility and the Coulombic interaction of the surfactant and co-surfactant head groups, resulting in an optimum packing of molecules in micelles. However, the selectivity of the desired product ( 3 )was lesser with C
16
TAB + C
16
OH and C
16
TAB +
C
16
PO
4
K than pure C
16
TAB, which was found to be due to decreased surface charge density on the micellar which resulting to less population of OH - at micellar surface. Additionally, NaOH amount could be significantly reduced from substrate/ NaOH molar ratio of 3 to 12. However on reducing the catalyst amount the rate of reaction was slowed down but the final conversion and selectivity were almost comparable .
Table 1 Conversion and selectivity towards di (3) and mono (4) condensation products in NaOH base catalysed cross aldol condensation in different pure and mixed micellar solutions a
Selectivity (wt.%)
Entry Surfactant
Conversion
(wt.%)
Product (3) Product (4)
5
6
7
8
9
1
2
3
4
10
11
12 C
C
C
16
TAB + C
5
OH
C
16
TAB + C
6
OH
C
16
TAB + C
8
OH
C
16
TAB + C
10
OH
C
16
TAB + C
12
OH
C
16
TAB + C
14
OH
C
16
TAB + C
16
OH
C
C
16
16
16
16
C
16
TAB
TAB + C
TAB + C
TAB + C
TAB + C
16
16
18
22
OH
OH
PO
4
K
COONa
80
73
68
68
69
75
79
82
80
76
85
67
75
41
34
37
44
58
60
70
60
53
66
17
25
59
66
63
56
42
40
30
40
47
34
83
Conclusion
Catalytic efficiency of mixed micellar system can be tailored by appropriate selection of co-surfactant for base catalyzed cross aldol condensation reaction. The mixed micellar system, C
16
TAB+C
16
PO
4
K and C
16
TAB +
C
16
OH gave enhanced stability than pure CTAB and other mixed micellar system. Stability of micelles helped in the enhancement of conversion by solubilising large number of reactants molecules but decreased in the surface charge density at micellar surface resulted to reduce selectivity of desired product.
References
1.
Cui, X., Jiang, Y., Yang, Ch., Lu, X., Chen, H., Mao, Sh., Liu, M., Yuan, H., Luo, P., and Du, Y., J. Phys.
Chem. B , 2010, vol. 114, no. 23, p. 7808.
2. Y. I. Rabinovich, S. Pandey, D. O. Shah,
, and B. M. Moudgil,
Langmuir 22 (
2006) 6858.
3. M. Vashishtha, M. Mishra, D. O. Shah, Journal of Molecular Catalysis A: Chemical (2015) 143–154