1.0 PURPOSE To lay down the procedure for preparation of
advertisement

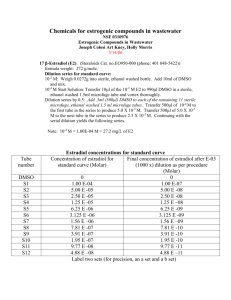
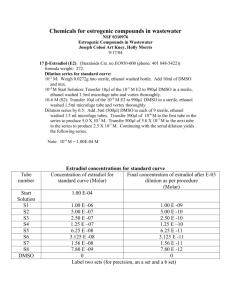
1.0 PURPOSE 1.1 2.0 SCOPE 2.1 3.0 Microbiologist ACCOUNTABILITY 4.1 5.0 This SOP covers the preparation of cell suspension and is applicable in Microbiology Laboratory. RESPONSIBILITY 3.1 4.0 To lay down the procedure for preparation of microbial cell suspension. Head – Quality Control PROCEDURE 5.1 Select the type of organism to which 10-100 cell suspensions need to be prepared. 5.2 Take a loop full of culture from the selected organism and inoculate in 2 ml of Nutrient broth or Soybean Casein Digest broth. 5.3 Mix vigorously in vortex mixer for uniformity. 5.4 Label the inoculated tubes with the date, name of the organism. 5.5 Preparation of serial dilution 5.5.1 Prepare a series of 9 test tubes with each of 9 ml Sterile Saline. 5.5.2 Label the tubes serially by numbering from 1to 8 and mark the remaining one 9 as Negative control (blank) for saline. 5.5.3 Now pipette out one ml of culture from the labeled culture tubes using sterile glass pipette/micro pipette using sterile tips and transfer in the saline water test tube of 9 ml numbered as 1, mark the dilution as 10 –1 and mix well. 5.5.4 Transfer 1 ml of the content from 10 -1 dilution, using sterile glass pipette/micro pipette with sterile tips and pour them in 9 ml of saline tube labeled as 2, again mix the suspension well and mark the dilution as 10 -2. 5.5.5 Like wise transfer 1 ml for all the remaining tubes up to 10 –8 serially. 5.5.6 Transfer the suspensions properly and mix well after each transfer and label the dilutions correctly. 5.5.7 Incubate the negative control (tube 9/blank) at 30 - 35C for 24 to 48 hours to check any chance of contamination. 5.5.8 After the serial dilution is completed, transfer 1 ml from all the dilutions (10 –1 to 10 -8) and pour them in sterile petri plates under laminar airflow chamber. 5.5.9 Then pour the sterilized nutrient agar / soyabean casein digest agar over the petriplates containing bacterial culture samples, Use Sabouroud chromaphinical dextrose agar in case of fungal cultures. 5.5.10 Preserve all the dilution tubes in the refrigerator below 8C. 5.5.11 Allow the plates to get solidified under Laminar air flow and incubate them in inverted position at 35C for 24- 48 hours in case of bacterial cultures and at 20 –25°C for 48-72 hours in case of fungal cultures 5.5.12 After incubation period, observe the plates and count the number of colonies obtained in each plate. 5.5.13 Select the plates, which contains more approximately 10-100 cfu and note down the dilution from which the colonies found. 5.5.14 Enter the details in Format No.: xxxxx. 5.5.15 Discard all the tubes except the dilution tube whare the 10-100 cfu colonies observed 5.5.16 Use the dilution tubes of bacterial and fungal cultures within 7 days from the date of storage by preserving them in the refrigerator. END OF DOCUMENT