C2 4.3 SAA pharmaceutical applications LN + HO
advertisement

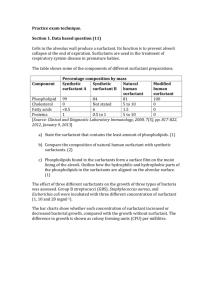
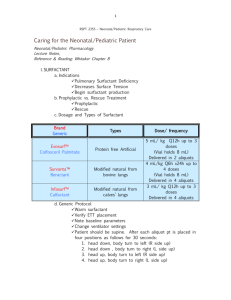
PHARMACEUTICAL APPLICATIONS OF SURFACTANTS A. PHYSIOLOGICAL EFFECTS OF SURFACTANTS 1. Antibacterial properties The cationic surfactants have bactericidal activity against a wide range of gram-positive and some gram-negative organisms. The effect is exerted by the agent becoming adsorbed at the cell surface which causes a change in the permeability of the lipid cell membrane. Essential materials can then leach out of the cell causing cell lysis and death. They may be used on the skin especially in the cleansing of wounds, and aqueous solutions are used for cleaning contaminated utensils. Cetrimide solutions (0.1 - 1%) are used for cleansing the skin, wounds and burns (e.g. Savlon®, Savloclens®) cleansing contaminated vessels, for storage of sterilised surgical instruments and for cleaning polythene tubing and catheters. Solutions of cetrimide are also used in shampoos to remove scales in seborrhoea. Benzalkonium chloride dilute solutions (1 in 1000) may be used for the pre-operative disinfection of skin and mucous membranes. It is also used as a preservative for eyedrops. It is also used as a throat antiseptic (e.g. Oraldettes®). Cetylpyridinium chloride is commonly used in throat lozenges (e.g. Cepacol®, Cepacaine®, Cetoxol® , Medikeel® - this has benzocaine as well). 2. Respiratory distress syndrome in premature neonates In a baby born prematurely, organs systems are not fully developed. We know that surfactants influence surface tension. The fully developed lung has surfactant (lipids and apoproteins) which enables the lung to expand. In the premature neonate, there is a deficiency of endogenous lung surfactant that results in higher alveolar surface tension, causing “stiff” and poorly compliant lungs. Large inflation pressures are required to expand the lungs and lung collapse occurs. A pulmonary surfactant is used called colfosceril palmitate (Exosurf Neonatal®) an intractracheal suspension. 3. Effect of surfactants on the skin Repeated application of surfactants such as detergents causes defatting of the skin and brings about a change in the pH of the skin surface. The skin swells and the surfactant becomes adsorbed onto the skin. If the skin cracks, lower layers of the skin are exposed to bacteria which may result in infection. 4. Dioctyl sodium sulphosuccinate This is used as a faecal softening agent with a laxative. It increases penetration of water and therefore softens the faeces, easing constipation. When used in a water-miscible base, it is used to soften wax in the ear e.g. Waxsol®7. B. SURFACTANTS IN PHARMACEUTICAL FORMULATION 1. Solubilizing agents 1.1 Phenolic disinfectants Phenolic compounds such as cresol, chlorocresol and chloroxylenol are frequently solubilized with soap to form clear solutions which are widely used for disinfection. Chloroxylenol is solubilized with an alkali soap such as potassium ricinoleate (soap made by saponification of ricinoleic acid with potassium hydroxide). The disinfectant properties are potentiated by the presence of a low concentration of surfactant (concentration below the CMC). This is due to a reduction in interfacial tension between the liquid phase and the cell wall of the organism, which facilitates the adsorption and spreading of the phenolic compound over the surface of the organism. At higher concentrations above the CMC the rate of penetration of the antibacterial decreases nearly to zero. This is because the drug is now solubilized in the micelle, resulting in a decrease in the effective concentration. 1.2 Preparations for ophthalmic use The low solubility of steroids in water presents a problem in their formulation for ophthalmic use. The solution must have optical clarity so oily suspensions or solutions cannot be used. Nonionic surfactants are used for solubilization and produce clear solutions which are stable to sterilisation.Most commonly used are the polysorbates (Tweens) and sorbitan esters (Spans). 1.3 Preparations of water-insoluble vitamins The polysorbate nonionic surfactants have been used to prepare aqueous injections of vitamins A, D, E and K. If taken orally, oil-soluble vitamins have an extremely unpleasant taste. They can then be formulated either as o/w emulsions OR as aqueous solutions of solubilised vitamins. The solubilised systems have advantages regarding stability e.g. it has been found that vitamin A is more resistant to oxidation in solubilised systems than when emulsified. 2. Wetting agents Liquid drops in contact with a solid surface can assume different shapes. We can express this degree of wetting of the solid by the liquid in terms of the contact angle θ. As θ decreases, have greater contact between the solid and the liquid. Physical Pharmacy 1. θ = 0; have complete wetting (cos θ = 1). The droplet spreads out over the solid and completely wets it. 2. θ > 90° and approaches 180°; the contact area between the solid and the liquid decreases and the liquid is said to be non-wetting 3. θ lies between 0° and 90°; liquid is said to be non-spreading and to have partial wetting. SLIDE - DROP OF LIQUID ON A SOLID, FLAT SURFACE. Consider a drop of liquid on a solid, flat surface, have 3 forces working: γSL - tension between solid and liquid γGL - tension between gas and liquid γGS - tension between gas and solid At equilibrium: γGS = γSL + γGL cos θ Area of contact: ↓ ↑ ↑ As wetting occurs, the drop spreads out and the gas/solid surface decreases. This is offset by the increased solid/liquid and gas/liquid area of contact. If a surfactant is added to the system:γGS remains constant γGL is reduced γSL is also reduced; so cos θ must increase and θ must decrease. Wetting agents should move rapidly from the bulk of the solution to the surface as that is where the wetting effect is exerted. Short chain surfactants are best e.g. sodium benzene sulphate as they have a high CMC. We want as much surfactant adsorbed at the surface as possible before micelles start forming. 3. Detergents Detergency is a complex process whereby surfactants are used for the removal of foreign matter from solid surfaces, be it removal of dirt from clothes or cleansing of body surfaces. The surfactant must have good wetting characteristics (short chain SAA) so that the detergent can come into intimate contact with the surface to be cleaned. If the dirt is oily, it may be solubilized in which case the requirements would be for a long chain hydrocarbon and a low CMC. 4. Deflocculating agent The dispersion of hydrophobic powders in aqueous vehicles is difficult as powders of this sort tend either to float on the water surface or to form large floccules (aggregate into loose masses). This causes an apparent increase in the viscosity and it may be very difficult, even after prolonged shaking to attain homogeneity. This may affect the spreading of a lotion, the pourability of a suspension or the ingestion of the correct dose of suspension. Use of a surfactant may overcome these problems as the surfactant is adsorbed at the solid-liquid (particle) interface and this increases the affinity of the particles for the surrounding medium, and decrease the inter-particulate affinity. This should reduce flocculation. 5. Flocculating agent In suspensions a controlled amount of flocculation is often desirable to obtain the required rheological properties and optimum stability. This flocculation may be achieved by the use of a suitable surfactant which alters the affinity of the particle for the medium. 6. Emulsifying agent Synthetic and naturally-occurring surfactants are widely used as emulsifying agents. 7. Additives to ointment and suppository bases Surfactants are included into fatty bases used for ointments and suppositories for a variety of reasons. ► The can modify the rate of release of drugs from the base. The surfactant promotes better contact with body fluids and forms an emulsion at the interface between the base and body environment which may increase exchange of medicament between the two phases. To retard the release rate, surfactants may be added to increase the hydrophilic character of the base. This effects the partitioning of the drug between the base and the body environment. ► Addition of surfactant improves the capacity of the base to take up aqueous liquids. With the addition of a water-soluble surfactant such as sodium lauryl sulphate,greasy ointments are more easily removed from the skin. ► The addition of surfactant promotes better spreading of the base. Cetyl alcohol, an oilsoluble surfactant is used. The polar group aligns at the surface of the oily base and promotes easier contact with the skin.