(DLAR) LECTURE ORIENTATION HANDOUT
advertisement

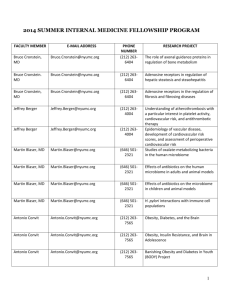
NYU School of Medicine Division of Laboratory Animal Resources (DLAR) LECTURE ORIENTATION HANDOUT 1 Introduction Research institutions must establish and provide resources for proper care, use, and humane treatment of animals used in research, testing and education. This is in compliance with applicable federal, state, and local laws and regulations such as Animal Welfare Regulations or AWRs (CFR 1985), Public Health Service Policy on Humane Care and Use of Laboratory Animals, or PHS Policy (PHS 1996). Also, the Guide for the Care and Use of Laboratory Animals (Guide, 2011) was written by ILAR to strongly affirm the conviction that all who care for or use animals in research, teaching or testing must assume responsibility for their well-being. An Institutional Animal Care and Use Committee (IACUC) is established as an oversight to guide in interpretation and evaluation of animal care and use programs. This handout will provide researchers with information regarding DLAR animal facilities and the care and use of laboratory animals. Topics to be discussed are the following: 1. Introduction to DLAR staff and contact personnel 2. Requirements to access the animal facilities 3. Working in the animal facilities 4. Institutional guidelines and policies 5. Guidelines in the animal facilities 6. Training /Seminars and Refresher Courses DLAR website: http://dlar.med.nyu.edu. Please refer to the DLAR website if you have inquires or email : dlar@nyumc.org. You can read complete documents of all DLAR/IACUC guidelines and policies, and other information. 2 1. INTRODUCTION TO DLAR STAFF Copies of the DLAR directory are posted conspicuously on the walls in each of the DLAR facilities near telephones. Below is the phone list of contact personnel: Animal Husbandry Facility Supervisor: Dental & Parasitology, Varick and Satellites Facility Supervisor: Sloan /MSCAF Facility Supervisor: Skirball Operations Manager Associate Director of Operations Dental Facility Parasitology Facility Office Extension Pager 212-998-9982 3-8128 646-754-2287 646-754-5250 212-998-9982 3-6648 Blackberry/Cell 917-583-0211 646-285-8601 917-748-4895 646-745-6584 347-860-1342 866-1033 866-1498 Veterinary Services Supervisor: Veterinary Technicians Senior Vet Tech: Sloan/MSCAF Senior Vet Tech: Dental & Parasitology , Satellites, Varick 646-501-2238 646-501-2285 212-998-9982 917-608-5615 Senior Vet Tech: Skirball Training Coordinator Clinical Veterinarian Associate Director, Veterinary Services Director, DLAR, Attending Veterinarian 3-5310 646-501-2282 646-501-2283 646-754-5232 646-285-8553 917-284-3451 347-461-7252 646-754-5247 2. REQUIREMENTS TO ACCESS DLAR ANIMAL FACILITIES Users (Research Staff and Principal Investigators or PI) of animal research facilities can only obtain access to the animal rooms if their names are included on an approved “Animal Use Protocol”, after completion of the following: A. attendance of DLAR/IACUC lecture orientation B. Completion of mandatory web-based animal training (Collaborative Institutional Training Initiative CITI, https://citiprogram.org/default.asp), C. Participation on basic hands-on training, workshop on “Biomethodology”, for rodent users. This is mandatory for the new research staff who have no experience or limited experience in live animal work. D. Facility tour (will be conducted after approval of study protocol) 3 Skirball-SCAF, Parasitology-OPH, Varick-VCAF, and Satellites research facility animal rooms are accessed electronically; therefore, users need a Personal Identification Number (PIN code), or an electronically activated IDs to enter the rooms. PIN codes will be assigned to the user after finishing the tour of the animal facility. Dual access to animal facilities is not encouraged, to prevent cross-contamination between facilities. For example, if a user has access in Skirball, that user cannot go to Dental facility and return to Skirball, due to the potential risk of fomite (object capable of carrying pathogens) pathogen transmission between facilities. After the tour, the user must take the Security Access Form (with the signature of Principal Investigator and Training Coordinator/or Facility Supervisor who will conduct the tour) to the Security Office at MSB 182, so the ID card can be activated. ID is used to open the entrance door of the animal facility. For the Dental Facility, the user will be given access after the tour. DLAR will submit the user’s name to NYU Washington Square Security Services to activate the ID. For other facility users, please read DLAR website for specific instructions on your facility tour. PIs and research staff must perform their experiments in accordance to their animal use protocol, as approved by the Institutional Animal Care and Use Committee (IACUC). Laboratory animal users may be asked by IACUC at any time to perform procedures listed in the IACUC animal protocol. Only trained research staff that are listed on the approved protocol are allowed to perform approved experimental procedures and access the animal facility. If the user has no experience in certain technical procedures, they can contact DLAR to sign up for a “Rodent Basic Techniques Workshop -Biomethodology” training session. It is against DLAR policy to let a friend or colleague accompany the user into the facility. Also, newly hired research staff that have not undergone proper orientation and tour are not allowed to go inside the animal facility even if accompanied by an existing user. Cameras must not to be used inside the animal facilities unless you obtain express written approval from either the DLAR Director or DLAR Associate Director. The proper procedure to enter the facility will be taught in detail during the guided tour. Donning of Personal Protective Equipment (PPE) is a must when entering the facility. Leave all lab coats in the lab or hang it at the entrance of the animal facility. Download the tour checklist at http://dlar.med.nyu.edu/training-and-education. 3. WORKING IN THE ANIMAL FACILITY Web Animal Order (WAO) from certified vendors/or non-vendor sources Submit a Web Animal Order (WAO) Form (http://www.med.nyu.edu/dlar/forms/) to DLAR by contacting this number : 646-754-4623, to sign up for access to animal ordering. A DLAR staff member will contact the user with further information on logon name, temporary password, web ordering instructions, and assist the user in ordering the animals. 4 Orders should be placed on Monday before 4:00 p.m. for delivery on Friday of the same week, or Tuesday/Wednesday of the following week, depending on the vendor. See list of certified vendors (http://dlar.med.nyu.edu/wao/vendor-web-sites). Standing orders can be established by submitting a WAO form that includes in the Special Request field, a statement indicating that it is a standing order and specifying the frequency of delivery, e.g. weekly, bi-weekly or monthly, etc. (refer to the detailed instruction of WAO). To import animals from non-vendor sources, submit a WAO form to DLAR office (or contact the personnel) to obtain a logon and password as directed above. For non-vendor sources, the Veterinarian must review the health reports and facility practices of the exporting facility prior to approval. Once approved, animals may be designated for quarantine (average 8 weeks) and/or rederivation following quarantine for a period of time based on pathogen risk analysis. Contact the Veterinary Services Supervisor (tel. 646-501-2238) for detailed activity on the quarantine procedures. Refer to DLAR website for a tutorial in web animal ordering. (http://dlar.med.nyu.edu/animal-orders-and-billing/web-animal-orders-login) Cage Cards and Bar Codes Individual animal cages will have cage cards for the purpose of identification and census. PIs or users order cage cards at the beginning of the experiment and/or upon the arrival of animals. These will be prepared by the DLAR animal facility supervisor, and cage cards will be placed on each animal cage when the animals have arrived. Subsequently, if more cage cards are needed, users can order them by filling out a DLAR Husbandry Service Request Form (available in animal facilities, or at the DLAR website (http://www.med.nyu.edu/dlar/forms ), place the form in a “drop off station” located in the vestibule areas of the animal facilities, or contact directly the Facility Supervisor for the follow-up requests. Specify if the requested cage cards are for non-breeding or breeding cages. Bar coding is a system used to capture and count electronically the number of cages in an animal room. Individual room census is broken down by PI. Bar coding allows for accurate census data to calculate per diem charges. See below for an example of a cage card and note the information necessary to order cage cards; Cage cards should be removed from an empty cage to prevent further per diem charges. These cage cards should be placed in the drop box located in the exit area of the facility. It is very important that the user writes the date of card removal on the back of the card to avoid further per diem charges. These charges could be up to an additional 3 weeks. Do not keep the cage cards. Do not place the old card behind another cage card, because the cage card not returned to DLAR will still be charged for up to 3 weeks. Write on the back of the cage card “RETURN” if you want DLAR to return your cage card to you after removing it from the census. Contact person’s name on the cage cards should be updated, to ease communication. 5 Animals with Health Issues Veterinary Services staff are notified when animals are showing clinical abnormalities, such as weakness, rough hair coat, emaciation, dermatitis , hunched-back position, or if an animal is found to be moribund. The Veterinary Technician (Vet Tech), with guidance from the Supervisor or Clinical Veterinarian, will explain the finding on a VETERINARY SERVICES CARD placed on the cage. The Vet Tech will communicate clinical recommendations to the contact person or PI .Certain situations dictate an immediate response from the user. These include: the animal is suffering from pain or distress, post-operative complication, is moribund, etc. Veterinary Services will make every attempt to contact the laboratory before taking further steps to alleviate the condition, which may include euthanasia.. Animals Found Dead Animal facility personnel collect the dead animals in each room daily. They are placed in a plastic bag with proper identification labels, and kept in a refrigerator marked CARCASS near the exit area. Dead animals are stored for seven (7) days in the designated refrigerator so users can retrieve them when necessary. In addition, a note will be placed on the cage of where the dead animal was found, e.g. 1 FD (found dead) and date. The user may also look at the inside of the animal room door. The log of all dead animals is maintained there on a day to day basis (Dead Animal Log Sheet Form). Transfer of Cages If the user needs to transfer animal cages between rooms in an NYUMC facility, between NYUMC facilities, or to an outside facility, the user will submit a Transfer Request Form (http://www.med.nyu.edu/dlar/forms/). This form is submitted to the Facility Supervisor where the animal cages are originally located. The user will place a TRANSFER CARD on the animal cage(s) to be moved. The transfer request will be processed and approved by a DLAR Veterinarian. The contact person or user will be notified if when the transfer has been completed. Transfer is usually completed in 3 days. Request for Pharmaceuticals To request antibiotics, analgesics or controlled drugs and other supplies from DLAR, users should fill out a Drug Purchase Request Form which can be obtained from the animal facility, or download from http://www.med.nyu.edu/dlar/forms/, and drop it to Skirball Vet Tech office. The form should be submitted or faxed to DLAR office (646-754-5268) at least one day ahead of desired pick-up time. The medical drugs can be picked up at Skirball Vet Tech Office from 6 11:00 to 11:30 am on Tuesdays, Wednesdays, and Thursdays. Only those with approved experimental protocols can avail of this service. DLAR staff will further explain proper recording of use and storage of these medical/controlled drugs in their respective laboratory rooms. All controlled drugs MUST be kept in the laboratory in a double-locked safe or in a securely locked box within a substantially constructed locked cabinet in accordance with IACUC policy. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/secure_storage_of_controlled_substanc es.pdf. The Controlled Substances Use-Log Form should be used to record usage. This form must be submitted to DLAR office together with the empty bottle in order to purchase new controlled substances. When picking up the ordered substances, you must bring your ID. Never use expired controlled substances of other drugs on survival procedures. Controlled substances when they expire shall be returned to DLAR vet services office, or used if approved in the study protocol for terminal procedures. Please the policy in detail on the website. http://webdoc.nyumc.org/nyumc_d6/files/iacuc2/Policy_Use_of_Expired_Medical_Materials_Re gulated_Species.pdf Request for Husbandry/Veterinary Services Users who would like veterinary services to perform specific procedures on their animals, must fill out the Husbandry or Veterinary Request Form and specify the requested procedure. Some of these procedures are: euthanasia, blood collection, use of rodent anesthesia machine, immunizations, tissue collection, tail biopsy , identification of animals, special water and feed, ordering cage cards, or management of breeding colonies, etc. These forms are available for download from http://www.med.nyu.edu/dlar/forms/ or in each animal facility. Submit the completed form to the Facility Supervisor or the Vet Tech. Follow-up your request by constantly communicating with DLAR staff. Sentinel Cages Sentinel cages are maintained by DLAR to track the health status of each animal room. They are located in all animal rooms and are labeled with pink-colored cage cards. The sentinels MUST not be moved from their rack location or transferred by unauthorized personnel. Go to www.med.nyu.edu/dlar/FAQs for more information. Users can track the health status of the room by going to the DLAR website. Special Procedures The following conditions/procedures should be part of the approved IACUC protocol: food and/or water deprivation requirements prior to surgery, special food and/or water requirements of animals, use of chemicals or biohazard materials, and other procedures. Notify the DLAR Facility Supervisor if these activities are to be performed. To request special cards, fill out the Husbandry Service Request Form and submit it to the Facility Supervisor. Examples of these requests are: special food or water for the animals; 7 treatment card; treating animal with chemical or biological hazardous agents; post- operative care or recent anesthesia. If a user is maintaining a breeding colony, a special card must be placed on the cages , e.g. “DO NOT DISTURB” card. Husbandry staff will not handle these cages so the user is responsible for changing the cage, bedding, water and feed in an appropriate time frame or as outlined in the corresponding IACUC protocol. Cleaning/Changing of Cages All rodent cages are changed once a week. The schedule of cage changing and room cleaning is found on the inside wall of animal room. Users should familiarize themselves with the cleaning schedule as room access is limited during that time. Light/Dark Cycle The standard light/dark cycle in every animal room is based on a 12-hour light/ 12-hour dark cycle. The lights go on at 6:30am and go off at 6:30 pm. Maintaining Rodent Cage Populations The IACUC sets standards for maintaining rodent cage populations based on the Guide for the Care and Use of Laboratory Animals (8th ed).The standards are the following : 2 adult rats, <500 grams each in weight per cage, or 5 adult (25-gm each) mice per cage, or 1 pair of breeding mice with one litter. Unless approved by IACUC, harem breeding is not allowed. Veterinary staff will place a SEPARATE BY CARD in front of the cage card when the quantity of animals exceeds the policy standards. DLAR Veterinary Staff will automatically separate the mice without notifying the users if animals are not separated by the specified date. A noncompliance fee will be charged to the PI account. Please read in detail the policy for maintaining mouse cage population ,in this website below. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/IACUC_policy_maintaining_mouse_cage_popula tions.pdf Performing Euthanasia using CO2 For rodent species, CO2 is an acceptable form of euthanasia when used following DLAR guidelines. According to AVMA Panel on Euthanasia, compressed CO2 gas in cylinders is the only recommended source of carbon dioxide because the inflow to the chamber can be regulated precisely. Animals should first be placed into the chamber, followed by the addition of CO2 at a low flow rate (20% of the chamber volume per minute) to complete the process. Rapid gas flows should be avoided since excessive noise and decreased in temperature ("winds") can develop and induce excitement/distress in the animals. Gas flow should be maintained for at least 1 minute after apparent clinical death (approximately 5 minutes total). A timer should be used to ensure adequate length of exposure. 8 It is important to confirm that an animal is dead after removing it from the chamber. This may be accomplished by performing cervical dislocation or decaptitation following CO2 narcosis or by creation of a bilateral pneumothorax. Decapitation is recommended for neonatal rodents under 10 days of age. Please read the whole guideline, below is the website. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/guidelines_for_performing_euthanasia_via_C02. pdf http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/guidelines_for_the_euthanasia_of_rodent_fetuse s_and_neonates.pdf Guidelines on Murine Ulcertaive Dermatitis (MUD) Mice, especially on a B6 background, are prone to the development of a severe debilitating idiopathic dermatitis. This condition is an ulcerated skin lesion on the dorsum, neck or head. It is progressive, poorly treatable and causes itchiness, pain and suffering. Investigators are obliged to detect, manage and when necessary, euthanize animals suffering from this condition. As soon as MUD lesions are detected, treatment must be initiated. If on detection MUD lesions are greater than 10 mm diameter, then the animal must be promptly used or euthanized. Topical treatment using agents approved by the veterinary staff may slow or reverse the onset of MUD, and can be initiated if the lesion is less than 10 mm diameter. If treatment is contraindicated due to experimental considerations, then untreated lesions may be allowed to progress to no larger than 10 mm diameter at which point the animal must be humanely euthanized. Any single inflammatory lesions of the head or face exceeding 3 mm diameter which involve sensory areas (eyes, vibrissae (whiskers), ears, oral cavity) are grounds for immediate euthanasia. Any facial or head lesion totaling in area 10 mm squared is grounds for immediate euthanasia. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/murine_ulcerative_dermatitis.pdf Minimizing Pain and Distress Pain and distress cannot always be adequately evaluated in animals and users must therefore assume that animals experience pain in a manner similar to humans. Decisions regarding their welfare in experiments must be based on this assumption unless there is evidence to the contrary. If animals develop signs of severe pain or distress, they should have the pain or distress alleviated promptly or be euthanized humanely and without delay if the pain cannot be alleviated. Alleviation of such pain or distress takes precedence over continuing or finishing the experiment. If in doubt, users must always seek a professional veterinary opinion before continuing an experiment. When writing the protocol, the user can refer to the website for more information on this topic. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/classifying_pain_and_distress.pdf Humane Endpoints The user should develop humane study end-points when preparing the study proposal. Death as an end-point is generally unacceptable and should be fully justified if required. If pain or 9 distress is unavoidable, then a scientific justification must be provided and approved by the IACUC, and the earliest possible endpoint compatible with answering the scientific question must be employed. Humane endpoints are preferable to death or morbidity, since they minimize pain and distress. Animals must be humanely euthanized if unanticipated pain or distress cannot be treated. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/endpoints_in_animal_study_proposals.pdf Rodent Survival Surgery The importance of utilizing aseptic technique is emphasized. This includes preparation of the patient; preparation of the surgeon; sterilization of instruments, supplies and implanted materials; and the use of operative techniques to reduce the likelihood of infection. These measures are designed to reduce post-surgical complications (e.g., infections and wound dehiscence), improve animal survival rates, and hasten return to the basal physiological functions that were present in the animal prior to surgery. In addition, surgical procedures should be carried out under appropriate local or general anesthesia. There should be adequate monitoring of the depth of anesthesia and effects such as hypothermia, and cardiovascular and respiratory depression. The choice and administration of anesthetic, analgesic and tranquilizing agents should be suitable for the species and appropriate for the purpose of the experiment. The use of such agents should conform to current medical, veterinary or laboratory animal practice. Below is the website where you can read specific guideline to conduct successful aseptic survival surgery in rodents. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/guidelines_for_performing_rodent_surgery.pdf Genotyping of Mice (Tail biopsy) Tail biopsy procedure for DNA analysis, or genotyping must be described in an approved animal protocol. Ideally, mice should be 10-21 days old. At this age, the tail tissue is soft (vertebra are not yet calcified) and the yield of DNA is highest. In addition, prompt analysis of tail tissue allows the desired mice to be identified prior to weaning which can facilitate more efficient use of cage space. Because pain sensory development may be complete, and to further minimize any transient pain or distress, investigators are strongly encouraged to apply local anesthesia to the tail for mice 10-21 days of age. Local anesthesia may be achieved by immersion of the tail in ice cold ethanol for 10 seconds. Alternatively, the tail can be disinfected with 70% ethanol and allowed to dry, followed by an application of ethyl chloride spray, lidocaine cream or other suitable anesthetic as recommended by the attending veterinarian. Local or general anesthesia is required for mice that are 21-day of age or older. http://webdoc.nyumc.org/nyumc/files/animalresources/attachments/Genotyping%20of%20Rodents.pdf Occupational Health and Safety Components outlined in “Occupational Health and Safety in the Care and Use of Research Animals”, and the NYUMC occupational health and safety program include: hazard identification and risk assessment; personnel training; personal hygiene; facilities, procedures, and monitoring; personal protection; and medical evaluation and preventive medicine. The goal 10 of the occupational health and safety program is to identify the hazards, assess the risk(s) associated with those hazards, and eliminate or manage those assessed risks. See the detailed guideline on the website below. Web-based training modules applicable to the type of research being conducted by the research staff include modules on the laws and regulations that govern animal research and occupational health and safety. In this particular situation, the goal of the investigators is to inform the research staff about occupational hazards (e.g., the use of toxic chemicals in animal studies), and be aware of the necessary precautions to be taken to protect against these hazards, notifying them of any possible exposure to hazardous biological, chemical, or physical agents in the workplace. Research staff must inform the investigators of any animal bites, scratches, or injuries received or illnesses that may be related to working with animals, or any work situation that might be hazardous. Use personal protective equipment and clothing, proper animal restraining devices, and other applicable safety devices that are available. http://webdoc.nyumc.org/nyumc/files/iacuc/attachments/guidelines_for_occupational_health_and_safety.p df Disposal of Sharps. Needles, blades, capillary tubes and other sharp objects are properly disposed of in a red, puncture proof container provided in each animal room. When using a syringe with needle, do not recap the needle prior to disposal. Please find all relevant policies and guidelines on the website below: (http://iacuc.med.nyu.edu/policies-guidelines) Personal Protective Equipment (PPE) PPE consistent with DLAR standard procedures will be posted at the entrance to each animal facility and/or each animal room. Training on the use of PPE will be covered during the tour of the facility. To reduce potential tripping, crushing, scraping, cutting and puncture injuries to the feet, research staff must wear foot-hugging, low-heeled shoes constructed of leather or rugged synthetic material, so that the entire foot from toe to heel is covered at least to the lower ankle. The outer garment must be disposable, have long sleeves, and be able to protect the front and side surfaces of the uniform or street clothes. Minimum PPE requirement is as follows: disposable gown, head cover, mask, shoe cover and gloves. The hair cover is worn to minimize loose hanging hair that might be a hazard, and to limit exposure of hair to allergens via animal dander. Gloves of several different types are available to accommodate persons who are allergic or hypersensitive to glove materials. http://webdoc.nyumc.org/nyumc/files/animalresources/attachments/PPE.pdf 11 4. ANIMAL FACILITY GUIDELINES - Adhere to ALL institutional guidelines and policies. - Always be knowledgeable of the protocols with which you are working. - Work only on experimental procedures that are written in your IACUC approved protocol. - Do not escort friends, or new lab members who have not yet undergone DLAR training, orientation and tour, into DLAR facilities. - Do not use cameras/videos inside the facilities unless you have express written permission from the DLAR Director or Associate Director. - Never bring food or drink into the animal facility. - Follow established traffic patterns when working in multiple rooms in the same facility. - Work with animals only inside the hood (biosafety cabinet). - Notify the Veterinary staff immediately with any animal health concerns. - Do not keep animals out of animal facility (only in Berg animal facility) for more than 12 hours unless it is approved in your IACUC protocol. - Take off your lab coat before entering the animal facility (see tour checklist). - Always wear proper PPE when entering the animal facility (see tour checklist). - Communicate with the Facility Supervisor, and/or animal care technicians concerning your special needs. TRAINING/SEMINARS AND REFRESHER COURSES: Please DLAR website http://dlar.med.nyu.edu/seminars-workshops-schedule Last updated (October 15, 2013) 12