outline30983
advertisement

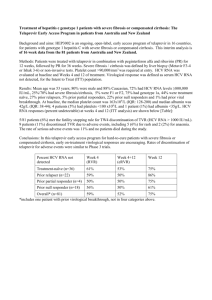
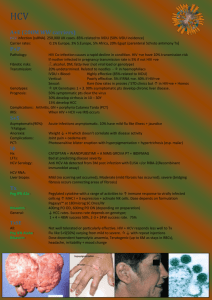
New Advances in and Retinal Management of Hepatitis C (HCV) Treatment Dr. Jim Williamson VAMC Memphis 1030 Jefferson Ave. Memphis, TN 38104 901-523-8990 x5366 Jim.williamson44@gmail.com Course Description: Hepatitis C is a major public health concern, and its treatment may cause ocular sequelae. This course will educate attendees about the management of these patients. Course Learning Objectives: To familiarize attendees with Hepatitis C To discuss the specifics of the disease To review current and new treatment strategies To show attendees how to recognize and manage ophthalmic manifestations of systemic treatment To provide case examples with clinical lab data Outline: I. II. Introduction to Hepatitis C a. Major public health concern b. Leading: i. Cause of chronic liver disease ii. Cause of death from liver disease iii. Indication for liver transplant c. 180 million infected worldwide d. Contact with blood i. Transfusions ii. IV drug use iii. Unsterilized body piercing, tattooing, barbering iv. Co-infection with HIV common Signs and Symptoms a. Acute HCV i. 90% asymptomatic ii. If symptomatic: 1. Occurs 2 weeks to 6 months post-exposure 2. Elevated alanine aminotransferase a. Up to 7 times normal III. IV. 3. Loss of appetite 4. Fatigue 5. Nausea 6. Vomiting 7. Abdominal pain 8. Jaundice iii. Fifteen to 25% will clear the virus without treatment iv. Rest will develop chronic or lifelong infection b. Chronic HCV is hepatitis that lasts >6 months i. Additional signs 1. Lichen planus 2. Mucocutaneous vasculitis 3. Glomerulonephritis 4. Porphyria cutanea tarda 5. Non-Hodgkin B-cell lymphoma 6. 1% develop symptomatic cryoglobulinemia a. Asymptomatic cryoglobulinemia more common Types a. Six HCV genotypes i. More than 50 subtypes b. Genotype 1 most common in US i. Most difficult to treat ii. Subtypes 1a and 1b c. Genotypes 2 and 3 next most common i. Respond more favorably to treatment d. Less common genotypes 4-6 i. Getting more frequent due to cultural diversity e. Knowing genotype helps predict i. Response ii. Treatment duration Tests a. Elevated alanine aminotransferase i. Can be normal in chronic HCV if disease quiescent b. Serologic assays i. Detection of specific antibody to HCV (anti-HCV) 1. Two enzyme immunoassays (EIAs) FDA approved a. Abbott b. ORTHO c. 99% specificity 2. Recombinant immunoblot assay (RIBA HCV) a. Confirm EIA testing b. Not used anymore due to nucleic acid testing ii. Molecular assays 1. HCV RNA Detection (FDA approved) V. a. Used to diagnose and monitor i. Amplicor ii. Cobas iii. Versant b. Sreening only i. Ampliscreen ii. Procleix (HIV-1/HCV) 2. HCV RNA Quantification (4 FDA approved tests) a. Viral load i. Copies/ml ii. IU/ml (preferred units per WHO iii. Used for monitoring iii. Interpretation of HCV assays c. HCV genotype d. Confirmation liver biopsy i. Active inflammation ii. Evolving fibrosis iii. Table of scoring systems for histological stage Current and New Treatments a. Current i. Pegylated Interferon alpha and Ribavirin (up to 50% cure in genotype 1) 1. Pegylated Interferon a. Two types i. Alfa-2b (Pegasys) 1. 12-kd linear polyethylene glycol (PEG) covalently link to the standard interferon alpha-2b molecule 2. 1.5 ug/kg/week based on weight ii. Alfa-2a (PEG-Intron) 1. 40-kd branched PEG 2. Fixed dose of 180 ug/week 2. Ribavirin a. Nucleoside analogue b. Antiviral c. Dosage i. With Pegasys (PO) 1. 800 mg if <65 kg 2. 1000 mg for 65 to 85 kg 3. 1200 mg for 85 to 105 kg 4. 1400 mg for >105 kg but <125 kg ii. With PEG-Intron (SubQ) 1. 1000 mg for <75 kg 2. 1200 mg for >75 kg 3. Duration VI. a. 48 weeks genotype 1 and 4 b. 24 weeks genotype 2 and 3 c. Genotypes 5 and 6 4. Ribavirin monotherapy is of no value b. New Treatment Standard i. Telaprevir (Incivek) and Boceprevir (Victrelis) 1. Directly targets the HCV protease a. Protein needed for reproduction 2. Addition of this to pegylated interferon + ribavirin is now standard therapy for genotype 1 3. Has increased SVR from 50 to up to 75% 4. Studies a. Telaprevir i. ADVANCE and ILLUMINATE 1. First-time treatment ii. REALIZE 1. For non-responders b. Boceprevir i. HCV RESPOND-2 1. HCV Serine Protease Inhibitor Boceprevir and PegIntron/Rebetol 2 2. Previously treated patients ii. SPRINT-2 1. Serine Protease Inhibitor Therapy 2 2. Untreated patients c. Liver transplant i. HCV can recur in the graft ii. Recurrence is usually indolent 1. Long-term survival rates are relatively high Side effects (10 to 14% d/c therapy due to adverse events) a. Systemic i. Influenza-like (most common about 50%) 1. Fatigue, headache, fever, rigors ii. Psychiatric (about 25%) 1. Depression, irritability, insomnia iii. Abnormal labs 1. Neutropenia (20%) a. Absolute normal count (ANC) = 1500 mm3 b. If ANC 750, dosed decreased 50% c. If ANC <500, discontinued d. Ironically, serious infections uncommon 2. Anemia (33%) a. Growth factors not recommended i. Don’t improve SVR rates VII. ii. Have their own ADRs 1. Cardiovascular and thromboembolic events, pure red cell aplasia, cancer progression, death b. Best way to manage cytopenias is dose reduction c. Eltrombopag i. Orally active thrombopoietin-receptor agonist ii. Stimulates thrombopoiesis iv. Pegylated interferon may induce autoimmune disorders 1. Types a. Autoimmune thyroiditis b. Other autoimmune disorders 2. Chronic HCV may present with features that simulate idiopathic autoimmune hepatitis, including (+)ANA a. May be hard to distinguish between “true” autoimmune disorder and non-hepatitis C-related autoimmune hepatitis with superimposed chronic HCV infection v. Ribavirin 1. Most common SE is hemolytic anemia 2. Ribavirin cleared by kidneys so careful with CKD b. Ophthalmic i. Retinopathy 1. Cotton-wools spots > hemes 2. Mimic DR or HTN retinopathy 3. Macular edema ii. Optic neuropathy 1. Anterior ischemic has been reported iii. Ocular toxicity 1. Decreased visual acuity Treatment Outcome/objectives 1. Rapid virological respone (RVR) a. Undetectable HCV RNA at week 4 of treatment b. Predicts a high likelihood of achieving an SVR 2. Early virological response (EVR) a. >2 log reduction or complete absence of serum HCV RNA 3. Sustained virological response (SVR) a. Absence of HCV RNA 24 weeks after last treatment 4. End-of-treatment response (ETR) a. Undetectable virus after 24 or 48 weeks of treatment 5. Reappearance of HCV RNA a. Virological breakthrough i. While still on therapy b. Virological relapse VIII. IX. i. After treatment is discontinued and an ETR was documented 6. Treatment ineffective a. Null responder i. Failure to suppress serum HCV RNA by at least 2 logs after 24 weeks b. Partial nonresponder i. Decreased by <2 logs IU/mL but never become undetectable Ophthalmic Management a. Baseline exam i. Initial retinopathy from DM or HTN does NOT preclude treatment ii. If retinopathy present at baseline, photos b. Two to 3 months after treatment started i. No retinopathy 1. RTC 2 to 3 months until a. See retinopathy b. Treatment completed ii. Retinopathy 1. Does NOT necessarily halt treatment 2. Depends on severity 3. Photos 4. RTC 6 to 8 weeks a. Compare to photos 5. Coordinate with managing doctor 6. Reversible c. Some risk factors for developing retinopathy i. Vascular disease 1. HTN 2. DM ii. Anemia Case examples a. Multiple case examples i. Baseline to retinopathy to resolution ii. Blood work results