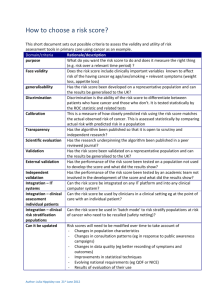
Responsibilities
advertisement

WuXi AppTec is expanding its cell therapy manufacturing facilities in the Philadelphia Navy Yard. The expansion will establish WuXi as a center of excellence for cell & gene therapy, supporting growing customer demand with single-source contract development and cGMP manufacture capabilities for allogeneic and autologous cell-based therapeutics. Expanding departments include process development, manufacturing science and technology, and both clinical and commercial manufacturing. Because of this expansion, we have some exciting opportunities we are currently recruiting for. Here are the areas of expertise we are looking for great talent in: Cell & Gene Therapies Vector Production Virology & Viral Clearance Molecular Biology & DNA Sequencing QC/Analytical Testing Technical Development – Custom Assay Development Commercial Biologics Manufacturing Process Development Quality Assurance Project Management If you or any one you know are interested in learning more about the opportunities @ WuXi AppTec, please visit our website: https://careers-wuxiapptec.icims.com/jobs Also, please feel free to reach out to: Jennifer Royal Recruiter | Talent Acquisition WuXi AppTec, Inc. 4751 League Island Blvd. Philadelphia, Pa 19112 Jennifer.Royal@wuxiapptec.com - Email @xjenroyalx - Twitter https://www.linkedin.com/pub/jennifer-royal/31/535/a22 - LinkedIn OUR PLATFORMS: Cambridge, MA Application Scientist Responsibilities: Provide expert advice on our next-generation sequence (NGS) analysis application, assist customers in troubleshooting and developing custom analysis tools, provide training, and develop documentation and support materials, and develop and maintain positive relationships with customers. Work with our informatics group to give feedback on our systems and defining strategies and content to further improve the analysis and interpretation of NGS data. Basic Qualifications: PhD with experience in NGS data analysis, or MS with additional hands on experience required. At least 3 years’ experience in NGS data analysis required. Scientific writing and bioinformatics experience is a plus. Must be willing to travel up to 25%. Outstanding problem solving and interpersonal skills. Strong written and verbal communication and training skills. Cambridge, MA Bioinformatician, Cancer Genetics Responsibilities: Work with our team of scientists and informatics staff on continued development and implementation of the tumor vs germline analysis based on our informatics pipeline applied to whole exome and whole genome datasets. Analyze our informatics systems for accuracy and content, and to further improve the analysis and interpretation of tumor-specific mutations. Basic Qualifications: Include a PhD and a thorough understanding and experience in working with algorithms and software applications for high-throughput sequence data (NGS) and Bioinformatics. Significant programming experience is required (Unix shell programming and Python/Perl/Java/C++), as well as familiarity with some of the mainstream OUR PLATFORMS: software packages for secondary analysis, such as BWA, GATK, MuTect, VarScan2, etc. Have a good overview of the key public reference databases for clinical variation data. Additional experience in tumor biology and genetics would be an advantage. Be charged with implementing a compelling global brand vision and communicating that vision with passion and energy inside and outside the company. Build a marketing strategy and program that anticipates global industry and competitive developments; creates superior, customer-focused, competitive strategies; and establishes innovative approaches to execute those strategies, in order to: Assist business development with in-license evaluations and strategy to expand the portfolio Support early stage pipeline and in-license products assets to position for future market success Collaborate with and utilize the commercial and communications teams to develop and deliver marketing goals Lead and foster individual and team development by identifying and arranging challenging assignments, formal training, or other experiences that will ready the commercial team for business success and professional growth OUR PLATFORMS: Cambridge, MA Oncologist-Director Responsibilities: Work with our team of scientists and informatics staff on continued development and implementation of the tumor vs germline analysis based on our informatics pipeline applied to whole exome and whole genome datasets. Work with our informatics group to give feedback on our systems and defining strategies and content to further improve the analysis and interpretation of tumorspecific mutations. Train outside users and the testing and validation of new versions of the sequence analysis tools. Basic Qualifications: Include MD, with or without a PhD, with clinical training and experience in oncology as well as past research in tumor genetics and biology. Experience in oncology drug development or with tumor sequence data is a plus. At least 5 years’ experience in clinical and research oncology is required. OUR PLATFORMS: Cambridge, MA Director of Marketing Responsibilities: Build a marketing strategy and program that anticipates global industry and competitive developments; creates superior, customer-focused, competitive strategies; and establishes innovative approaches to execute those strategies Have strong marketing experience in a US or global company that commercializes genomics products, diagnostics or therapeutics, with a focus on rare disease and oncology a notable plus. Demonstrated science acumen is essential. Develops marketing intelligence and informing product and business development Owns the collateral end products Manages scientific writing process (contracted, drafted from internal resources) Conference selection, participation design and management Develop and implement customer-focused strategies and tactics to optimize growth, share, and profitability for all our products aimed at using the genome to improve medicine Assist business development with in-license evaluations and strategy to expand the portfolio Support early stage pipeline and in-license products assets to position for future market success Collaborate with and utilize the commercial and communications teams to develop and deliver marketing goals Lead and foster individual and team development by identifying and arranging challenging assignments, formal training, or other experiences that will ready the commercial team for business success and professional growth Basic Qualifications: BS/BA in bioinformatics, engineering or life science, MS life science field (preferred) or MBA 5+ years’ experience marketing in clinical or life science companies. Knowledge of genomics and diagnostics a major plus. OUR PLATFORMS: Senior Staff Scientist II, Bioanalytical Responsibilities: • Designs, plans, and caries out experiments and associated research assignments. • Independently devise procedures or studies to search out causes and remedies for problems. • Performs and/or oversees work to attain objectives. • Operates and/or oversees the operation of equipment to acquire experimental data. • Utilizing the effective time management to complete assigned tasks on time with high accuracy • Advise Junior Scientist on the effective time management to increase efficiency. • Compiles, calculates, plots, and analyzes, and with experience, interprets data; records and maintains data in accordance with accepted standards and practices. • Assumes complete responsibility for assigned projects, such as acting as study director/ principal investigator, and assumes a continuing responsibility for contributing ideas related to this area of investigation. • Prepares reports, oral and written, covering work objectives, methods, and results, which may include recommendation for further experimentation. • May act as a project coordinator. • Thorough understanding of Good Laboratory Practices and EPA, FDA, or other regulations pertaining to the study. • Promotes implementation and compliance with XBL safety policies, practices, and procedures in order to maintain a safe work environment and minimize or eliminate injury. • Maintains and enhances technical expertise, knowledge, in the Bioanalytical area which include up-to-date-technology in method development and sample analysis for parent drugs and its metabolites in various biological matrices. • Prepare scientific presentation in-house and in scientific meetings • Thorough understanding of Protocol Design (Pre-clinical and Clinical Studies) • Guides and counsels employees in order to contribute to the goals of XBL for both the long and short period and to encourage personal growth. • Identifies safety hazards and initiates corrective action. Assists in the safety training for subordinates and/or less experienced staff. • Supervises and participates in training of Staff Scientist, Associate Scientist, Assistant Scientist, and Laboratory Assistant. • Other duties as assigned. OUR PLATFORMS: • Act as an expert in designated area of technology either Software or hardware related to the Bioanalytical techniques. Basic Qualifications: • Bachelor’s or M.S. degree in chemistry, biology, or equivalent • At least 7-10 years of relevant experience in animal research field or equivalent • Any similar combination of education and experience Job Function: Research & Development Primary Location: Plainsboro NJ Validation Specialist Responsibilities: • Responsible for planning, scheduling, execution and leading validation project assignments in coordination with the IT Manager and in accordance with Good Laboratory Practices (GLPs) and FDA 21 CFR Part 11. • Validation assignments include validation, qualification, and implementation of new and existing, processes, equipment, software, and instrumentation systems. • Leads cross-functional project teams in the development and implementation of the validation plan and protocols. • Prioritizes, manages and executes multiple projects utilizing Project Management methodology. This may include assignment/oversight/review of validation projects to other personnel/teams. • Review and approve all validation documentation (plans, protocols, summary reports, etc), including vendor documentation. • Acts as the primary Validation contact source for vendors, the FDA, and sponsors. Provides support for regulatory submissions and regulatory agency inspections as required. • Responsible for maintaining and updating XBL’s Validation Master Plan (VMP). • Maintains a thorough knowledge of the organization, policies and Standard Operating Procedures (SOPs), adheres to all organizational standards defined in SOPs and the XBL Employee Handbook, and coordinates with IT team activities. OUR PLATFORMS: • Support development of best demonstrated validation practices within the IT department, based on current industry practices and guidelines. Champions continuous improvement of XBL’s practices, documentation, forms, SOPs, and systems within the IT department (including validation) and throughout XBL. • Serve on XBL’s SOP Committee, Review/Harmonize SOPs, and author assigned SOPs to ensure compliance with Company policies and federal regulations. • Provides Business Support/Training to Users for Thermo Scientific’s Watson LIMS System and other Validated Systems as assigned. Other duties as assigned. Basic Qualifications: Bachelor’s degree applicable to a pharmaceutical R&D environment. • Minimum of 5 years’ experience in a regulated, GxP environment validating and qualifying laboratory equipment, computer systems, and processes. • Extensive, in-depth knowledge and understanding of Good Laboratory Practices (GLP) and quality requirements in an FDA-regulated environment along with knowledge of 21 CFR Part 11. • Exceptional written and verbal communication skills with superior organizational skills. • Excellent problem solving skills and the ability to quickly and effectively analyze critical issues and develop assessments for risk and impact. • Ability to utilize Project Management methodologies to manage validation projects. • Ability to work in a high pressure, fast-paced, environment. • Ability to work with various scientists and management across various departmental groups to achieve company’s goals and objectives. • Ability to work independently and manage time with respect to project priorities. • Have thorough understanding of the Bioanalytical Process. • Ability to operate instrumentation within the limits required for validation development and execution, with the appropriate training provided by XBL staff or the vendor if required. • Advanced knowledge and experience of complete MS Office system. • Firm understanding of all associated technology platforms as needed for validation projects. OUR PLATFORMS: • Ability to effectively manage other Laboratory Informatics Systems as assigned Job Function: Research & Development Primary Location: Plainsboro NJ Associate Scientist II, DMPK Responsibilities: • Learn and enhance technical knowledge of aseptic procedures, cell cultures and in vitro assays. • Perform routine laboratory techniques under supervision. • Receive specific instructions on assigned task and expected results. • Maintain research notebooks in a well-organized manner. All necessary documentation is recorded. • Perform routine calculations, tabulate and record results. • Prepare standard solutions used in the laboratory. • Prepare accurate data summary with supervision. • Maintain and calibrate laboratory equipment. • Comply with all safety rules, practice good housekeeping, and maintain safe workplace without supervision. • Assume responsibility for technical accuracy of work. • Understanding of Good Laboratory Practices and EPA, FDA, or other regulation pertaining to the study. • Promote implementation and compliance with XBL safety policies, practices, and procedures in order to maintain a safe work environment and minimize or eliminate injury. • Other duties as assigned. Basic Qualifications: • Bachelor’s or Master’s degree in chemistry, biology, or a related discipline • 1-4 years of relevant experience, or equivalent • Any similar combination of education and experience Ability to follow instructions to perform required procedures. OUR PLATFORMS: • Ability to work on multiple tasks with efficient approach to complete the work on time with high accuracy. • Knowledge of commonly used concepts, practices, and procedures within biological or chemical R&D laboratory setting • Well organized, detail oriented, and adaptable to changes • Effective oral and written communication skills • Effective interpersonal skills • Computer skills: knowledge of internet research, MS Word, Excel, Outlook, and PowerPoint • Good time management skill in order to perform of multiple projects within deadlines • Ability to effectively prioritize workload and manage changes in direction • Ability to work in a team environment • Familiarity with and adherence to regulatory guidelines consistent with XBL requirements Job Function: Research & Development Primary Location: Plainsboro NJ Postdoctoral Research Associate, Met ID Responsibilities: • Identification of drug metabolites using liquid chromatography-tandem mass spectrometry, NMR, and other analytical methodologies. • Elucidation of metabolic pathways involves the identification and quantification of metabolites in biological matrices (e.g., blood, plasma, excreta) using HPLC with radioactivity detection and/or LC-MS/MS. • Conduct in vivo and in vitro metabolism studies. • Handle radio-labeled materials (e.g., 3H, 14C). • Implements new techniques and instrumentation to improve technical operations. • Prepares scientific reports that document the analytical results and interpretation for the identification of drug metabolites. OUR PLATFORMS: • Authors scientific papers, which are published in peer reviewed journals, and presented in scientific meetings. • Collaborate with other scientists in a team to support metabolism studies. • Knowledge of in-vitro and in-vivodrug metabolism processes and related bioanalytical techniques. • Conduct LC-MS/MS analysis and interpretation. Basic Qualifications: • Ph.D. degree in a chemical, pharmaceutical, biological discipline or equivalent • 1-2 years of relevant research experience, or equivalent, is desirable • Any similar combination of education and experience • Experience in structural characterization and identification of drug metabolites using LC/MS/MS. • Experience and knowledge of in vivo and in vitro metabolism. • Experience with radioisotope handling (e.g., 3H, 14C) biochemical techniques is a plus. • Experience and knowledge in design of NMR experiments, operation, and data interpretation as applied to identification of unknowns is a plus. o Basic knowledge of commonly used scientific concepts, practices, and procedures within a biological or chemical R&D laboratory setting o Highly motivated with excellent communication and organizational skills o Well organized, detail oriented, and adaptable to changes o Effective oral and written communication skills o Effective interpersonal skills o Computer skills: knowledge of internet research, MS Word, Excel, Outlook, and PowerPoint are desirable o Ability to learn quickly and effectively in a dynamic laboratory environment o Ability to follow instructions and research plans with limited supervision o Ability to work in a team environment o Ability to understand and adhere to regulatory guidelines consistent with XBL requirements Job Function: Research & Development OUR PLATFORMS: Primary Location: Plainsboro NJ Associate Quality Assurance Specialist Responsibilities: • Maintains a current awareness of EPA, FDA, and OECD Good Laboratory Practice (GLP) regulations and any other related Regulations which may apply to XBL activities. • Evaluates current laboratory practices. Recommends changes or modifications to laboratory procedures in order to facilitate compliance with GLP standards. • Conducts audits (protocol/plan, raw data, and final report audits for accuracy and completeness, verifying that the final reports accurately reflect the raw data generated) and in-lab critical phase inspections of any GLP-regulated study performed at the laboratory for conformance to the final protocol/plan, SOPs, and relevant GLP regulations. • Conducts inspections of laboratories/facilities Conducts audits of employee training files. • Prepares written records of all inspections and audits. Submits findings to the study director,and/or principal investigator (or laboratory supervisor, when appropriate) and management, indicating deviations from or conformance with GLPs, protocol/plan, methods and SOPs. • Reports periodically to QA management regarding quality issues and status of inspection/audit activities. • Assists in reviewing, writing, and maintaining up-to-date SOPs and controlled documents for the QA unit. • Updates the master schedule as required by GLPs. • Other duties as assigned by QA management Basic Qualifications: • Knowledge of EPA, FDA, and OECD Good Laboratory Practice (GLP) regulations and commonly used concepts, practices, and procedures within GLP QA field • Must work well with numbers • Detail-oriented and highly organized • Effective oral and written communication skills OUR PLATFORMS: • • Excellent interpersonal skills Computer skills: working knowledge of Internet, MS Word, Excel, Outlook, SharePoint and PowerPoint • Ability to multitask and work under pressure of multiple projects and deadlines • Ability to effectively prioritize workload and manage changes in direction • Must be able to record and keep essential records for a regulated environment • Familiarity with and adherence to regulatory guidelines consistent with XBL requirements Job Function: Research & Development Primary Location: Plainsboro NJ OUR PLATFORMS: