Proposal 1.6 (1 Dec 2011)
advertisement

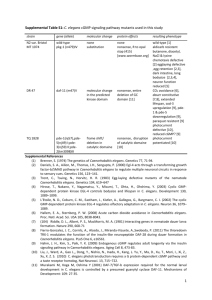
Effect of celastrol on C. elegans with increased beta-amyloid production due to an increase in local cholesterol Project Proposal Lim Eu Gene 3A315 Jarel Teo Shao Hean 3P310 Hwa Chong Institution Mentor: Mrs Har Hui Peng Introduction Background Alzheimer's Disease (AD) is a neurodegenerative disease characterized by the loss of neural tissue. It is also a type of dementia that impairs memory, thought, and behavior. Victims of AD experience problems with language, decision-making ability, judgment, and personality. The cause of AD is not known, but closely tied to the onset of the disease is the accumulation of extracellular amyloid-β plaques and neurofibrillary tangles. These plaques and tangles lead to the degeneration of neurons via apoptosis. Aβ proteins are formed by cleavage of the amyloid precursor protein. The amyloid precursor protein (APP) is regularly processed at the cell surface of neurons and is also processed intracellularly after being trafficked into the cell by lipid rafts. Lipid rafts are specialized microdomains within plasma membranes which are rich in cholesterol and shingolipids. The existence of lipid rafts is still debated, since they cannot be conventionally imaged due to their small size (~20 nm). Protein distribution in lipid rafts is therefore observed biochemically (e.g. detergent-resistant membrane (DRM) isolation). The role of lipid rafts is to regulate signal transduction and trafficking. After APP is brought into a cell by lipid rafts, it is cleaved by β-secretase (Bace1) and γ-secretase (Marquer et al., 2011). This cleavage produces a soluble N-terminal fragment (sAPPβ) and an amino acid amyloid βpeptide (Aβ). Research investigating the effect of an increase in local cholesterol on lipid-raft endocytosis discovered that an increase in cholesterol resulted in rapid endocytosis, as well as the clustering of β-secretase enzymes and APP in lipid rafts. This indicated that increased 2|Page cholesterol levels may not directly cause an increase in the production of Aβ, but be an indirect cause by bringing the β-secretase enzyme and amyloid precursor protein in close proximity. Also, an increase in intracellular APP leads to a greater rate of production of the Aβ peptide, which then accumulates extracellularly to form insoluble, neurotoxic plaques. (Marquer et al., 2011) Celastrol, a chemical derived from the Chinese “Thunder of God Vine” is a potent antioxidant known to have anti-cancer effects. Research has shown that celastrol greatly inhibited beta-amyloid plaque formation by reducing the beta-cleavage of amyloid precursor protein. It is also known for its ability to cross the blood brain barrier and offer neuroprotection in animal models of Parkinson’s disease and Huntington’s disease. Previous work has revealed that celastrol can prevent neurodegeneration and extend the life span of a transgenic mouse model of amyotrophic lateral sclerosis. Celastrol displays potent antiinflammatory activities in vivo and has been shown, for example, to be beneficial against allergy-induced asthma as well as rheumatoid arthritis. (Paris et al., 2010) Since celastrol has recently emerged as a new possibility of a natural neuroprotective agent, our team has decided to study the effects of celastrol so as to identify it as a potential cure for Alzheimer's Disease and even other neurological diseases. 3|Page Rationale In our project, we will be using celastrol extracted from the “Thunder God of Vine” plant. Paris et al., (2010) has discovered that celastrol is a potent Aβ lowering compound in transgenic mice models of AD and is possible that celastrol can also improve cognition and tau pathology in these transgenic mice models. Caenorhabditis elegans (or in short C. elegans) are used as the test system in this study. It shares many similar biological traits and genes with humans, thus the results from this study could be examined for application in humans. In addition, it has a short lifespan that allows results to be obtained within a short duration. Increased local cholesterol will serve as a mutagen in this project. According to Marquer et al., (2011), an increase in cholesterol level will increase the rate of lipid raft endocytosis, which indirectly leads to the increase in production of Aβ, which then accumulates extracellularly to form insoluble, neurotoxic plaques. 4|Page Hypothesis If local cholesterol of wild C. elegans is increased, the beta-amyloid plaque accumulated would increase. Movements of the wild C. elegans should be slower or at the same speed of the mutated C. elegans. Under an increase of beta-amyloid plaque due to increase in local cholesterol, if the concentration of celastrol of wild C. elegans is increased, the beta-amyloid plaque accumulated would reduce. Movements of wild C.elegans should be faster as compared to mutated C.elegans. If the concentration of celastrol on mutated C. elegans is increased, movements of the mutated C. elegans should be faster. Objective The objectives of this project are: 1. To study the effects of celastrol on the beta-amyloid plaque concentration, the movement and protein profile of both wild and mutated C. elegans 2. To determine possible sources of neuroprotective agents which can prevent or cure neurological diseases 5|Page Method List of Variables Independent Variables Concentration of celastrol (g/ml) Concentration of local cholesterol (g/ml) Dependent Variables Beta-amyloid plaque accumulation Movement of C. elegans Thrashing of C. elegans Protein profiles of C. elegans Controlled Variables Culture conditions Types of C. elegans used Number of C. elegans per agar plate 6|Page Methods Outline of Method C. Elegans (wild and mutated) + increased cholesterol C. Elegans (wild and mutated) Added celastrol Without celastrol Aβ production SDS-PAGE C. Elegans Movement 7|Page Experimental Procedure I) Experimental Setup A. Preparation of NGM Agar Culture Materials Sodium Chloride (NaCl), Peptone, Agar, 5mg/ml Cholesterol in Ethanol (Not Autoclaved), 1M MgSO4, 1M KPO4 Buffer pH 6.0, Deionised water Apparatus Petri Dish, Conical Flask, 500 ml Glass Bottle, Measuring Cylinder, Electronic Weighing Scale, Beaker Procedure 1. Add 3g NaCl, 17g NGM agar and 2.5M NaOH into a flask. 2. Mix well. 3. Add 970ml of deionised water using a measuring cylinder. 4. Add 1ml 1M CaCl2, 1ml 5mg/ml cholesterol in ethanol, 1ml 1M MgSO4 and 25ml 1M KPO4 buffer. 5. Pour the NGM agar into the plates until the plates are ⅔ full. 6. Leave the plates at room temperature for 2 days. 8|Page B. Preparation of Increased Cholesterol Materials NGM Agar Culture, 5mg/ml Cholesterol in Ethanol (Not Autoclaved) Apparatus Electronic Beam Balance Procedure 1. Weigh additional cholesterol using an electronic beam balance 2. Add the additional cholesterol in the 5mg/ml cholesterol in ethanol solution 3. Use this modified solution to make the NGM Agar Culture C. Preparation of Celastrol Materials Celastrol Crystalline, Dimethyl Sulfoxide (DMSO) solution Apparatus 50ml Falcon Tube, Electronic Beam Balance Procedure 1. Pour 10ml of 100% dimethyl sulfoxide (DMSO) concentration into a Falcon tube. 2. Weigh 0.1g of celastrol crystalline solid and put them in the DMSO solution. 9|Page D. Preparation of Celastrol Materials Resveratrol Crystalline, Dimethyl Sulfoxide (DMSO) solution Apparatus 50ml Falcon Tube, Electronic Beam Balance Procedure 1. Pour 10ml of 100% dimethyl sulfoxide (DMSO) concentration into a Falcon tube. 2. Weigh 0.65g of resveratrol crystalline solid and put them in the DMSO solution. E. Preparation of Bacteria Food Source Materials Nutrient Agar Powder, Deionised Water, E. coli OP50 Starter Culture Apparatus 500ml Glass Bottle, Micro Pipette, Incubator Procedure 1. Using a starter culture of E. coli OP50, isolate a single colony on Petri dish of nutrient agar. 2. Allow the inoculated cultures to grow overnight at 37°C. 10 | P a g e F. Seeding of NGM Agar Plates Materials E. coli OP50 Liquid Culture, NGM Agar Plates Apparatus Micropipette, Glass Rod Procedure 1. Apply 50µl of E. coli OP50 liquid culture onto the one end of the NGM agar plates using a micropipette. G. Chunking of C. elegans Materials C. elegans Culture Plate, Seeded NGM Agar Plates, NaOH, Household Bleach Apparatus Sterilised Scalpel, 50ml Falcon Tube Procedure 1. Pour 20ml 5M NaOH and an equal amount of household Bleach into a 50ml Falcon tube. Mix well. 2. A 5µl drop of the solution should be placed on the other side of the NGM agar plate. 3. Using a sterilised scalpel, cut out a 3mm x 3mm x 3mm cube from the C. elegans culture plate. 11 | P a g e 4. Place the cube face down onto the drop of bleach solution. 5. Incubate the plate for 4 days at 27°C. II) Data Collection A. Plaque Formation (Herndon et al., 2002) Materials 5-fluorescein Isothiocynate, Dimethylformamide, M9 Buffer, Seeded Growth Medium Plate, Apparatus Fluorescence Microscope Procedure 1. Mix 20mg/ml of 5-fluorescein isothiocyanate (FITC) in dimethylformamide. 2. Mix 50µl of the resulting stock dye solution with 200µl M9 Buffer 3. Apply the resulting solution to the seeded growth medium plate 2-12 hours before moving to step 4. 4. Observe the seeded growth medium plate using the fluorescence microscope mounted with FITC filter. B. Worm Movement Materials C. elegans Petri Dish Apparatus 12 | P a g e Paper, USB Microscope, Computer Screen, Transparency Sheet Procedure 1. Place a Petri dish containing C. elegans on a piece of paper 2. Observe the C. elegans with a USB microscope on a computer screen 3. Place a piece of transparency sheet over the computer screen 4. Trace the path of the worm for 30 seconds 5. Obtain the actual distance and the speed of travel of the worm C. Thrashing Assay (Nazir, Sammi, Singh, & Tripathi, 2010) Materials C. elegans Petri Dish, M9 Buffer, Deionised Water Apparatus USB Microscope, Glass Slide Procedure 1. Wash off the C. elegans from their parent plates using the M9 buffer 2-3 times. 2. Place a drop of M9 buffer on a clean glass slide. 3. Place a single worm into the buffer. 4. Allow the sample to stabilise for 30 seconds. 5. Count the number of body bends for 30 seconds using the USB microscope. 13 | P a g e D. Protein Profile (Neo, Tan, Morris, & Ferguson, 2011) Materials Crushed C. elegans Suspension, Polyacrylamide Gel, Coomassie Blue Stain, Apparatus SDS-PAGE Kit, Eppendorf Tubes Procedure 1. Mix 10µl of C. elegans Suspension and 5µl of loading dye into an Eppendorf tube. 2. Place the Eppendorf tubes into a hot water bath at approximately 50°C. 3. Load the Polyacrylamide Gel into the electrophoresis kit. 4. Load 10µl of protein ladder into the first well. 5. Load the subsequent wells with the C. elegans suspension mixed with loading dye. 6. Set the electrophoresis kit to 25mA. 7. Allow the gel to run until the protein column runs to the end of the gel. 8. Stain the gel with Coomassie Blue. Safety Precautions During experimentation, latex gloves will be worn when handling the C. elegans samples. All cultures will be decontaminated by autoclaving before disposal 14 | P a g e Materials General Materials: Beakers Centrifuge tube Measuring Cylinder Petri Dishes Electronic Beam Balance NGM agar for Worm Culture Sodium Chloride (NaCl) Peptone Agar 5mg/ml cholesterol in ethanol (Not autoclaved) 1M MgSO4 1M KPO4 buffer pH 6.0 Deionised water Preparation of C. elegans N2 C. elegans (wild type) FX 776 C. elegans (mutant type) 15 | P a g e Scalpel Glass bead sterliser 5M NaOH Household bleach 16 | P a g e Timeline 17 | P a g e Bibliography Herndon, L. A., Schmeissner, P. J., Dudaronek, J. M., Brown, P. A., Listner, K. M., Yuko, S., et al. (2002). Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature Journal, 419, 808-814. Marquer, C., Devauges, V., Cossec, J., Liot, G., Lécart, S., Saudou, F., et al. (2011). Local cholesterol increase triggers amyloid precursor protein-Bace1 clustering in lipid rafts and rapid endocytosis. FASEB Journal, 25(4), 1295-1305. Nazir, A., Sammi, S. R., Singh, P., & Tripathi, R. K. (2010). Trans-Cellular Introduction of HIV-1 Protein Nef Induces Pathogenic Response in Caenorhabditis elegans. PLoS ONE, 5(12). Neo, A., Tan, D., Morris, C., & Ferguson, A. (2011). Experiment. Retrieved Nov 11, 2011, from Investigating the Effects of St. John's Wort on Drosophila and Cancer: http://projectsday.hci.edu.sg/2011/15-FinalsWeb/Cat-01/1-084/exp.html Paris, D., Ganey, N., Laporte, V., Patel, N., Beaulieu-Abdelahad, D., Bachmeier, C., et al. (2010). Reduction of beta-amyloid pathology by celastrol in a transgenic mouse model of Alzheimer's disease. Journal of Neuroinflammation, 7(17), 1-15. 18 | P a g e