Prepared by: Nicole Gasbarro, PharmD ACCP
advertisement

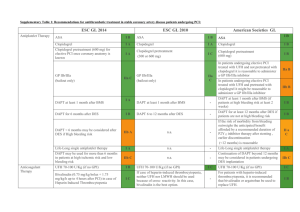
Prepared by: Nicole Gasbarro, PharmD ACCP-Cardiology PRN e-Journal Club Newsletter___________________________________ DAPT Trial: 12 or 30 Months of Dual Antiplatelet Therapy after Drug-Eluting Stents Due to conflicting studies in the literature, the potential benefits and risks of extending dual antiplatelet therapy (DAPT) beyond 12 months remain uncertain. The DAPT trial, a randomized, double-blind, placebo controlled trial of 9961 patients, sought to determine the benefits and risks of continuing DAPT beyond 12 months after placement with drug-eluting stents. Subjects were enrolled within 72 hours of the index stent placement and were then followed on open-label thienopyridine, plus aspirin for 12 months. At 12 months, eligible subjects were then randomized to continue thienopyridine or placebo, each in addition to aspirin, for an additional 18 months. The co-primary efficacy endpoints were definite or probable stent thrombosis and major adverse cardiovascular and cerebrovascular events (MACCE). The primary safety endpoint was incidence of moderate or severe bleeding. From months 12 to 30, the group that continued thienopyridine had a significantly lower cumulative incidence of stent thrombosis (0.4% vs. 1.4%; HR 0.29 [95% CI 0.17-0.48]; p < 0.001) and MACCE (4.3% vs. 5.9%; HR 0.71 [95% CI 0.590.85]; p < 0.001) as compared to the placebo group. Continued thienopyridine was also associated with a significantly lower incidence of myocardial infarction (MI) (2.1% vs. 4.1%; HR 0.47 [95% CI 0.37-0.61]; p < 0.001). The rate of death from any cause was 2.0% in the continued thienopyridine group and 1.5% in the placebo group (HR 1.36 [95% CI 1.00-1.85]; p = 0.05). Lastly, the rate of moderate or severe bleeding was significantly higher in the continued thienopyridine group as compared to the placebo group (2.5% vs. 1.6%; HR 1.0 [95% CI 0.4-1.5]; p = 0.001). The authors concluded that DAPT beyond 12 months, as compared with aspirin alone, significantly reduced the risks of stent thrombosis and MACCE but was associated with an increased risk of bleeding.1 Questions and Answers For what clinical scenario would you extrapolate this data? This data could be used to support the decision to continue DAPT 12 months after drug-eluting stent implantation in a patient with stable coronary disease, one or more risk factors for stent thrombosis, adherent with thienopyridine therapy, and free from ischemic events and moderate or severe bleeding episodes. Approximately 37.7% of patients in the study underwent PCI for stable angina, making stable coronary disease the most common indication. Approximately 50.9% of patients presented with at least one risk factor for stent thrombosis. Lastly, patients included in the study had to be free from ischemic events and moderate or severe bleeding, as well as adherent with thienopyridine therapy.1 An important question to ask ourselves looking at this data, however, is if the benefit of reduced stent thrombosis and ischemic events is worth the risk of bleeding or potentially death in a patient with stable coronary disease. The majority of patients included in this study represented a population with a lower risk of ischemic events. The authors do not provide information on how many patients had multiple risk factors for thrombosis, which could help support the decision to extend DAPT in a patient with stable coronary disease. What are some differences between this study and the others you alluded to? The DAPT Trial is one of the largest DAPT trials completed. Compared to older trials, the study’s definition of stent thrombosis was more clearly defined and endpoints were adequately powered. Prepared by: Nicole Gasbarro, PharmD Compared to the latest DAPT trials, the DAPT trial differs in length of time of intervention, as well as type of drug-eluting stent, end points, and overall findings as listed below. ITALIC3 (2014) Patients 1850 Comparison 6 vs. 24 month Study drugs Clopidogrel, prasugrel, (ticagrelor ≤ 1%), aspirin Stent-type 2nd > 1st generation 2nd > 1st generation 2nd generation Study end -ST, MACCE -Composite death, -Composite death, points -Bleeding MI, ST, stroke, MI, stroke, urgent thrombolysis in MI, revascularization, major bleeding major bleeding Overall finding ↓ risk ST, MACCE No difference in No difference in for extended ↑ risk bleeding primary endpoint primary endpoint DAPT including bleeding including bleeding ST = stent thrombosis, MACCE = major adverse cardiovascular and cerebrovascular events DAPT (2014) 9961 12 vs. 30 months Clopidogrel, prasugrel, aspirin ISAR-SAFE2 (2015) 4000 6 vs. 12 month Clopidogrel, aspirin ARCTIC-Interruption4 (2014) 1259 12 vs. 18-30 months Clopidogrel, prasugrel, aspirin 2nd > 1st generation -Composite death, MI, ST, stroke, urgent revascularization -Bleeding No difference in primary endpoint ↑ risk bleeding In terms of the primary safety outcome, were there any specific differences in major or minor bleeding? The primary safety end point was the incidence of moderate or severe bleeding as defined according to the GUSTO criteria. There was no significant difference between the randomized treatments with respect to severe or life threatening bleeding, only moderate bleeding, which was defined as bleeding that required blood transfusion but did not result in hemodynamic compromise. Mild bleeding outcomes were not listed in the study or study appendix.1 Is there a role for extended DAPT in secondary prevention? Possibly. The DAPT trial detected an elevated risk of stent thrombosis and myocardial infarction in the 3 month window following discontinuation of thienopyridine treatment, suggesting a potential role for study of lifelong treatment. The CHARISMA trial assigned patients with a high risk of having a cardiovascular event to receive DAPT or aspirin alone for 28 months and showed that overall, DAPT was not significantly more effective than aspirin alone in reducing the rate of MI, stroke, or death from cardiovascular causes.5 The PEGASUS-TIMI 54 trial, which is ongoing, is testing longer-term use of ticagrelor plus aspirin after acute MI. Its results may offer new information on the benefit of prolonged DAPT as secondary prevention of ischemic events.6 References 1. Mauri L, Kereiakes D, Yeh R, et al. Twelve or 30 months of dual antiplatelet therapy after drug-eluting stents. N Eng J Med. 2014; 371 (23): 2155-66 2. Schulz-Schupke S, Byrne R, Ten Berg J, et al. ISAR-SARE: a randomized, double-blind, placebo-controlled trial of 6 versus 12 months of clopidogrel therapy after drug-eluting stenting. Eur Heart J. 2015 Jan 23. pii:ehu523. [Epub ahead of print] 3. Gilard M, Barragan P, Noryani A, et al. Six-month versus 24-month dual antiplatelet therapy after implantation of drug eluting stents in patients non-resistant to aspirin: ITALIC, a randomized multicenter trial. J Am Coll Cardiol 2014 Nov 16. doi: 10.1016/j.jacc.2014.11.008 4. Collet JP, Silvain J, Barthélémy O, et al. Dual-antiplatelet treatment beyond 1 year after drug-eluting stent implantation (ARCTICInterruption): a randomized trial. Lancet. 2014 Nov 1; 384(9954):1577-1585 5. Bhatt D, Fox K, Hacke W, et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N Engl J Med 2006 Apr 20; 354:1706-1717 6. Bonaca M, Bhatt D, Braunwald E, et al. Design and rationale for the prevention of cardiovascular events in patients with prior heart attack using ticagrelor compared to placebo on a background of aspirin-thrombolysis in myocardial infarction 54 (PEGASUS-TIMI 54) trial. Am Heart J. 2014 Apr;167(4):437-444.e5