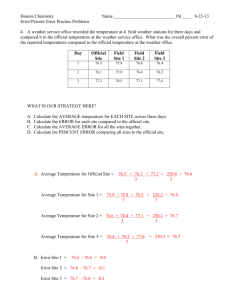
Theory

Overall comments
There are a number of ideas that you touch on but only enough to make the reader curious or confused. One example is the sensitivity of the diode laser to the backscattered light. It is more than OK to say “see Page x or Section y of Ref. Z for further details”, but if you aren’t going to fully explain what is going on, you need to do a good job of sending the reader to appropriate place in the appropriate source for them to investigate further if they so choose.
You guys really only looked at the coarse energy structure of the Rubidium (85/87 a and B) as opposed to the fine structure. So a lot of what you wrote doesn’t accurately represent your investigations.
The abstract provides very little insight into what you will be discussing in the paper.
Your description of the equipment is very scattered and random. Due to the level of detail provided, I would suggest killing off everything in this section aside from your description of the diode laser which you take more time to explain (or you could further expand some of the other descriptions to make them more useful). Since you are not actually describing the experiment as a whole, you will want to make it clear that the purpose of this section is simply to describe some of the major components of the experimental apparatus, and then point the reader to a reference to see how it all fits together.
Should he talk about the interferometer?
Saturated Absorption Spectroscopy
By Brendan Bulthuis
o
Saturated Absorption Spectroscopy
Abstract o
Equipment
The Laser
The Heating Cell
The Photodetectors
Mirrors, Beamsplitters, and Lenses o
Theory
The Two-Level Model
The Multi-Level Model
Rubidium Atomic Structure
Experiment-Specific Theory o References
Abstract
Spectroscopy is the use of light, typically from lasers, and its absorption by an atomic vapour to determine particular aspects of its atomic structure. In the case of saturated absorption
spectroscopy, the light beam is very strong and saturates the sample with the necessary photons, and a weaker beam is used to probe these effects. One such effect is the hyperfine splitting of otherwise degenerate energy levels in an atom.
Equipment
The Laser
The laser used in this experiment is a diode laser. Diode lasers have multiple advantages over the use of other types of lasers, most notably the lower relative cost of purchase and upkeep. Despite their low cost, they still produce high-power beams that are also tunable. For Rubidium, the high power is required to produce observable electron transitions, and the tuning capability allows for refinement of the beam to operate between certain energy levels.
However, there are some disadvantages. The first is that they have a large linewidth, approximately
50 MHz, in comparison to those of atomic transitions. In the case of Rubidium, the linewidth is around 5 MHz (Linewidth is the width of a spectral line produced by absorption or emission of photons by atoms). A smaller linewidth is preferred in order to observe the spectral phenomena with reasonable precision. The second is that these lasers are very sensitive to feedback, ie. output light which is scattered back into the laser.
Figure 1 : Diode Laser Structure with Collimating Lens and Diffraction Grating.
Thankfully, both these issues are resolved by the simple addition of a collimating lens and a reflective diffraction grating (see figure 1). The lens straightens the diverging beam into an almost non-diverging beam. The diffraction grating reflects up to 15% of the beam back into the laser, but the remaining 85% becomes a new output beam with a line-width of up to 1 MHz. As the reflection of light back into the laser is now controllable, it is now less sensitive to feedback as well. As a bonus, the diffraction grating allows for further fine-tuning to observe atomic absorption. [1]
The Heating Cell
The heating cell heats the rubidium until it is in a gaseous state, at about 45ËšC. This is important to the observation of photon absorption, as the gas state interacts more easily with the laser beam than the liquid or solid states. A protrusion of metal is located in the heating cell to condense excess rubidium. Since it is a good conductor, it stays cooler than the cell, ensuring that excess rubidium condenses on the protrusion rather than the windows of the cell. [2]
The Photodetectors
This experiment uses two photodiode detectors. These detectors have a linear response to the voltage of the incoming beam between 0 and -11.0 V. In case of weak signals, the detectors have a knob on the back to control the gain, ranging from 10 MΩ to 333 Ω in ten steps. [2]
Mirrors, Beamsplitters, and Lenses
These three components are quite self-explanatory, and are used profusely in this experiment to manipulate the laser beam. The mirrors reflect the beam in certain directions. The beamsplitters split the laser beam in certain ratios, allowing a certain amount through and reflecting the rest in another direction. Lenses are used mainly to cause the beam to diverge. [2]
Theory
Spectroscopy, in this experiment, is the use of light absorption to determine the energy levels of electron transitions in an atom. SAS saturates the sample with a strong beam of light to excite a maximum number of electrons in order to make more precise measurements of these energy levels.
[3]
The basic setup for SAS is shown in figure 2. Two laser beams are sent through an atomic vapour from opposite directions. The probe beam is very weak in relation to the pump beam, and both beams are produced by the same laser, and thus have the same frequency. A photodetector records the intensity of the probe. [4]
Figure 2 : Basic Saturated Absorption Spectroscopy Setup.
The Two-Level Model
Consider a vapour made up of atoms which have only two energy levels, a ground state and an excited state. This is a common model. In the absence of the pump beam, the absorption spectrum recorded by the photodetector would look like that of figure 3. The peak is broadened due to the
Doppler Effect. Basically, if an atom moves parallel to the probe beam and towards (away from) the source, it will perceive the photons as having a higher (lower) frequency, and thus a higher (lower) energy. So, if the photons have a lower (higher) energy than required for excitation, these atoms will absorb those photons, resulting in the broadening of the absorption peak. The tip of the peak corresponds to atoms moving with perpendicular velocity to the probe beam. [3]
Figure 3 : Two-Level atom absorption spectra, without pump beam.
If the pump beam is activated, the absorption spectrum would look like that of figure 4, which is similar to that of figure 3, but with a spike at the middle of the peak. This phenomenon is due to two assumptions, the first being that the pump and probe have the same frequencies at the same times, and the other that they are co-linear but opposite in direction. Because of these reasonable assumptions, an atom moving toward the pump source will absorb pump photons if the frequency is lower than the excitation frequency, and absorb probe photons if the frequency is higher. If the atom moves towards the probe source, the opposite situation occurs. This leaves the Doppler-broadened part of the peak unchanged. If an atom has perpendicular motion, then it will absorb photons from either of the beams, resulting in the characteristic spike. The more pump photons that are absorbed, the larger this peak will be. [3]
Figure 4 : Two-Level atom absorption spectra, with pump beam.
The Multi-Level Model
This model expands on the previous model by allowing multiple excited states. However, this experiment restricts the number of excited states to two, namely an atom with a ground state and an excitation energy split by hyperfine interaction. With both the pump and probe beams, the absorption spectrum in this case would look like that of figure 5, provided the separation between the excited states is less than the Doppler width. The spikes on the right and left represent the two energy levels of the hyperfine splittin g, at resonant frequencies ν
1 and ν
2
. The middle peak at (ν
1
+ν
2
)/2 is called the cross-over resonance. It comes about through atoms moving with velocities such that the pump is in resonance with one transition and the probe is in resonance with the other, which means the atoms will absorb photons from either beam. [3]
Figure 5 : Saturated absorption spectrum for atoms with one ground state and two excited states.
Another possibility is to have two ground states and one excited state. The absorption spectrum looks like that of figure 6. The characteristic dip is due to optical pumping. Basically, the laser excites the atom from the first ground state, g1, to the excited state e. This state has the option of decaying
back to g1, where it can be excited again, or decaying to the second ground state g2, where it stays.
In this case, only the pump beam is strong enough for this process to occur. There are two velocity classes of the atoms that cause this dip: the first occurs when the pump excites g1 to e, which tends to decay to g2. The probe, which excites g2 to e, then sees more absorption. Similarly, if the pump excites g2 to e, which tends to decay to g1, then the probe excites g1 to e and also sees more absorption. [3]
Figure 6 : Saturated absorption spectrum for atoms with one excited state and two ground states.
Rubidium Atomic Structure
The ground state of Rubidium is made up of closed orbital shells (orbitals that are completely filled with electrons), similar to those of the noble gas Krypton, plus one 5s valence electron. As the electrons in the shells do not normally react, the single 5s electron gives Rubidium a spectrum similar to that of Hydrogen. So, the first excited state of Rubidium raises the 5s electron to the 5p.
Rubidium also has two natural isotopes, 85 Rb and 87 Rb, which can be found with 72% and 28% abundance respectively. The former has a nuclear spin quantum number of I=5/2, the latter has
I=3/2. [4]
The notation 2S+1 L’
J is used to describe the different energy levels, where S is the spin quantum number, L’ denotes the angular momentum quantum number (the S,P,D,… orbitals corresponding to the orbital angular momentum quantum number L=0,1,2,…) and J=L+S is the total angular momentum. Thus, for the ground state, we have S=1/2, because of the single electron, and L=0, as the electron is in the S orbital. So, J=1/2, and the ground state is 2 S
1/2
. For the excited state, we have S=1/2 and L=1. But this actually gives two possibilities for J, J=1/2 or J=3/2. So we have two degenerate excited states, 2 P
1/2
and 2 P
3/2
. However, the phenomenon of spin-orbit coupling splits these two levels. [4]
The interaction between the nuclear spin and the electron results in the magnetic hyperfine splitting, with an energy splitting given by
where F=I+J is the total angular momentum quantum number, which includes the nuclear spin, and
C is the hyperfine structure constant. Figure 7 shows the S and P energy levels for 85Rb and 87Rb, with the hyperfine splitting, and a typical unsaturated absorption spectrum.
Figure 7 : Energy level diagrams for 85Rb and 87Rb, and a typical unsaturated absorption spectrum.
Experiment-Specific Theory
The goal of this experiment is to observe the phenomena of SAS, and then measure the
P
3/2
hyperfine splitting of 87 Rb. The experimental setup shown in figure 8 will be used to accomplish these goals.
Following the laser beam in figure 8, the first 10:90 beamsplitter takes 10% of the beam and sends it to a Michelson interferometer. This will be explored later. The remaining 90% is used to observe the saturated absorption. This beam passes through another 10:90 beamsplitter. The 10% portion then becomes the probe, and is directed through the heating cell by the use of two mirrors. The 90% portion is directed to the heating cell by the use of a 50:50 beamsplitter. This becomes the pump beam. The 50:50 beamsplitter is used to allow the co-linear probe beam through so that we can record absorption with the photodetector. [4]
Figure 8 : Experimental Set-up.
The output from this detector can be observed on an oscilloscope, but this by itself is not enough to determine the energies involved. This problem is solved by the addition of the interferometer, which causes two beams to interfere with each other, producing fringes that change with time. By isolating one fringe and sending it into the second photodetector, one can record the frequency of the changing fringe intensity. This intensity is governed by the equation [4]
Using this intensity, one can determine how much the wavelength needs to change to get from one peak to the next, by the use of the following equation
However, this concerns changes in wavelength, not frequency. One can switch to frequency by recognizing that which implies that
So,
Finally, one can overlay the outputs from both photodetectors, then, by the use of a ruler, phrase the energy difference between the P
3/2 energy levels in terms of Δf. [4]
References
[1] Teachspin. (2009). Laser Physics.
http://cistwiki.ufv.ca/pub/Phys382/DLSUserManual/DLS910-
Laserphysics_rev2.0.pdf
[2] Teachspin. (2009). Diode Laser User Manual: Apparatus
Section. http://cistwiki.ufv.ca/pub/Phys382/DLSUserManual/DLS950-Apparatus_Manual_Rev2.0.pdf
[3] Teachspin. (2009). Spectroscopy. http://cistwiki.ufv.ca/pub/Phys382/DLSUserManual/DLS920-
Spectroscopy_Background_rev2.0.pdf
[4] Caltech. (n.d.). Saturated Absorption
Spectroscopy. http://cistwiki.ufv.ca/pub/Phys382/ResearchLines/DLS003-caltech-
Saturated_Absorption_Spectroscopy.pdf
-- BrendanBulthuis - 2012-03-07
Wiki_fig_1.jpg:
Wiki_fig_2.jpg:
Wiki_fig_3.jpg:
Wiki_fig_4.jpg:
Wiki_fig_5.jpg:
Wiki_fig_6.jpg:
Wiki_fig_7.jpg:
Wiki_fig_8.jpg: