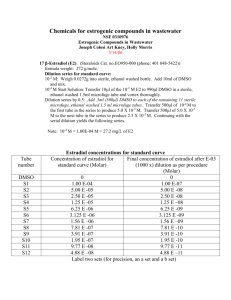
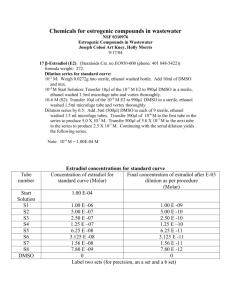
Supplementary materials Besides the two representative
advertisement

Supplementary materials Besides the two representative formulations reported in the main paper, We evaluated the physical properties of twelve additional formulations including A: dimethyl sulfoxide (DMSO)/water (25/75, v/v); B: DMSO/10% dextrose (25/75, v/v); C: DMSO/20% dextrose (25/75, v/v); D: DMSO/blood serum (25/75); E: polyethylene glycol-400 (PEG 400)/water (50/50, v/v); F: PEG 400/water (60/40, v/v); G: PEG 400/water (70/30, v/v); H: ethanol/PEG 400/water (10/50/40, v/v); I: ethanol/PEG 400/water (10/60/30, v/v); J: polyvinylpirrolidone (PVP)-10000; K: PVP-29000; and L: -Cyclodextrins (10 molar ratio of radioiodinated Hyp/Hyp to cyclodextrin) as shown in Figure A-L. Formulations were macroscopically examined by acquisition of digital images in a CN-15 darkroom cabinet under white light and ultraviolet light at 254 nm as excitation wavelength with 4 × magnifications. Microscopic observations over a drop of each formulation were performed. Images were acquired in both bright field and fluorescence illumination modes at 50 × magnifications. Cross-sectional fluorescence intensity profiles (absorbance unit) along red-continuing lines (n=5) over the corresponding drop (µm) were generated. Radioiodinated Hyp/Hyp dissolved either in DMSO/saline (20:80, v/v) (Figure 4 in the main paper); DMSO/water (25/75, v/v); DMSO/10% dextrose (25/75, v/v), DMSO/20% dextrose (25/75; v/v) or -cyclodextrins showed cloudy brownish solutions (A1, B1, C1, L1) without fluorescence (A2, B2, C2, L2). By microscopy, formation of aggregates (A3, B3, C3, L3) and reduced fluorescent intensities (A4, B4, C4, L4) were observed. Cross-sectional fluorescence intensity profiles over a drop of each formulation were distinguished for a dramatically diminished fluorescence signal (A5, B5, C5, L5). In DMSO/PEG400/water (25:60:15, v/v/v) (Figure 4 in the main paper); PEG400/water (60/40, v/v); PEG400/water (70/30,v/v); ethanol/PEG400/water (10/50/40, v/v) and ethanol /PEG400/water (10/60/30, v/v), Radioiodinated Hyp/Hyp formed red solutions (F1, G1, H1, I1) with strong fluorescence under the UV light (F2, G2, H2, I2). Microscopically, these formulations exhibited an intense homogenous fluorescence (F4, G4, H4, I4), as was confirmed by the cross-sectional fluorescence intensity profiles over a corresponding drop (F5, G5, H5, and I5). Among them, the cleanest field without aggregates formation under bright field mode was observed with DMSO/PEG400/water (25:60:15, v/v/v) (Figure 4 in the main paper) in comparison to the other well dissolved radioiodinated Hyp/Hyp formulations (F3, G3, H3, and I3). In contrast, with DMSO/blood serum (25/75, v/v), PEG400/water (50/50, v/v) and in PVP10000 and PVP-29000 systems, radioiodinated-Hyp/Hyp solutions with deprived color intensity (D1, E1, J1, K1) and fluorescent properties (D2, E2, J2, K2) were macroscopically seen. By microscopy, formation of aggregates to some extent was detected (D3, E3, J3, K3) with less pronounced fluorescence signals (D4, E4, J4, K4), as proven by cross-sectional fluorescence intensity profiles analysis (D5, E5, J5, K5).