record in the rock earth student notes
advertisement

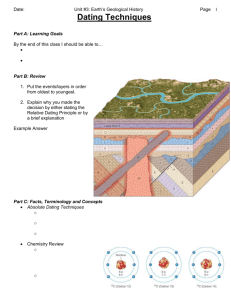
Record in the Rock: What Processes Shape our Earth? Geology: __________________________________________________________________ _________________________________________________________________________ Importance: • Understanding of the ______________ that _____________ our Earth, so we can better _________________ potential __________________ Earth’s Internal Structure: Characterized _____________________________________________________________ _________________________________________________________________________ Crust is the ______________ most layer of Earth, consists of _______ types: • Continental: __________________________________________________________ ___________________________________________________________________ • Oceanic: ____________________________________________________________ ___________________________________________________________________ Age of The Earth & Geologic Time Principle of Uniformitarianism: • The order in which __________________________________________________________ ________________________________________________________________________. • The same processes that are at work ___________ were at work in the ___________ • *The _____________ is the _________ to the __________! Relative Dating: _________________________________________________________________ _______________________________________. We use a number of principles and laws to do this: 1. Law of Original Horizontality – ________________________________________________ ________________________________________________________________________ 2. Law of Superposition - ____________________ ______________________________________ ______________________________________ _____________________________________ • Scientist use law of superposition by using ____________ age (something ____________ to the age of something else) • Problem: ________________ – buried surface that represents a break in the rock record 3. Lateral Continuity- layers of sediment __________ in _________ directions when they form. 4. Law of Cross-cutting Relationships - _____ ___________________________________ ___________________________________ ________________________________________ ________________________________________ 5. Inclusions: _______________________(rock pieces) _________________________________ 6. Faunal Succession: _____________________ _______________________________________ • Index fossil: __________________________________________ __________________________________________ __________________________________________ Correlation: matching ___________ by __________ ___________ in different places • 1st person to use correlation was ______________ _______________ • Smith & others developed the Geologic ____________ – a diagram of the _______________ of rock ____________ in an specific area, in order of _______ Early Estimates of Earth’s Age • Methods • Sedimentation – estimated the total ______________ of the earth’s ______________ rocks (rate of sedimentation) • • • Range from ____ million to _____ billion years Problems • Rate can _________ at different ___________ & ____________ • ______ accurate way to measure thickness • Started with different ___________________ 2. “Salt” Method – estimated the _______ ___________ of the oceans, then _____________ it with the __________ at which salt is being _______________ to the oceans. • Range from ____ million to _____ billion years • • Problems with Salt Method • 1. __________________ at first the oceans contained __________ water • 2. did ______ account for _____ way salt is added to or removed from the oceans • 3. Each scientist _________________ amounts of salt 3. Kelvin Method – assumed Earth was ______ ____________ rock, he measured rate of Earth’s ______________ to _______________. Also, took into account heat coming from within the Earth and from the ________ • • Range from ______ million years to _________ million years Problems • No one knew about ___________________ Absolute Age Dating Absolute Dating: Enables scientists to determine the ______________ _______________ age of rocks and other objects Radioactive Decay : • Radioactive isotopes __________ ________ into other elements by _____________ radiation • An element is defined by the number of ______________ it contains • As the ______________of protons __________________ with each emission, the original radioactive isotope (parent) gradually converted to a _________________ element (daughter) • Ex: Uranium – 238, _____________, changes into Lead -206, ____________________ Radiometric Dating Dating an object using _________________ isotopes As the _____________ of parent atoms ___________________ during radioactive decay the number of __________________ atoms ___________________ Rate of decay ______________ changes, but is based on elements __________ -life Half life: ________ it takes for ____________ of the isotope to break down into another __________________ Uranium 238 use to determine age of_________- ________________ things Carbon used to determine age of once ___________________ things Dendrochronology o Dendrochronology: use _________ ______________ to determine absolute age o Uses tree rings called ________________ rings (early & _________ season) o The _________ of the rings depends on certain _______________ in the environment o Trees from the __________ geographic region tend to have the same ________________ of ring width for a give span of time o By _____________________ the ____________ in these trees scientists have established tree ring ____________________ up to 10,000 years Ice Cores o Ice Cores: they contain a record of past environmental conditions in annual __________________ of _______________ deposition o ________________ ice tends to have ___________ bubbles and ____________ crystals than winter ice o Ice-core chronologies study ______________ cycles and __________________ Varves o Varves: bands of ___________________ light and dark colored _______________ of sand, clay and silt • Occur with _________________ deposition of sediments around ___________ usually • ______________-sized particles in summer (________) • Thinner, ___________-grained particles in winter (_________) • Scientists can date cycles of glacial sedimentation over periods as long as __________ years Calculating Half Lives: The time for ____________ of the ________________________ substances in a given sample to undergo _________________. After one __________________ there is ___________ of original sample left. After two half-lives, there will be ______ of the ________ = ________ the original sample. Example 1: You have 100 g of radioactive C-14. The half-life of C-14 is 5730 years. How many grams are left after one half-life? How many grams are left after two half-lives? Example 2: The half-life of iodine-131 is 8 days. If you start with 36 grams of I-131, how much will be left after 24 days?