Does the amount of calcium chloride affect the temperature of water?
advertisement

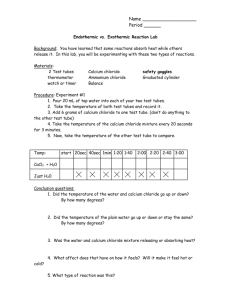
Name_______________________ Question: Does the amount of calcium chloride affect the temperature of water? Title : ________________________________________________________________________________ Hypothesis : ___________________________________________________________________________________ Materials: 150 ml beaker Thermometer (C°) Scoop or plastic spoon Safety goggles Calcium chloride Clock Water Graduated cylinder Procedure 1. Place 75 ml of water in a beaker. 2. Record the initial temperature (°C) of the water. __________ 3. For the first trial, add zero scoops of calcium chloride. Stir for 2 minutes then read the new temperature. In the data table, record the difference between the initial temperature and the temperature after two minutes. 4. Add one scoop of calcium chloride and stir for 2 minutes. Read the new temperature and record the difference between the last reading and the new reading. 5. Add another scoop of calcium chloride and stir for 2 minutes. Read the new temperature and record the difference between the last reading and the new reading. 6. Add another scoop (total of 3) of calcium chloride and stir for 2 minutes. Read the new temperature and record the difference between the last reading and the new reading. 7. Discard your chemical slurry into the discard bucket. 8. Repeat the procedure for trials 2 and 3. 9. Record the average change in temperature in the data table and on the promethean data table. 10. Record the class average change in temperature in your data table. 11. Graph your result and the class result. Data Table Amount of chemical (scoops) Change in Temperature (°C) Trials 1 2 3 Average change in Temperature (°C) 0 1 2 3 Class Data Using your recorded measurements and the class average as the accepted value, determine the percent deviation for all three trials. Amount of Chemical Scoops 0 1 2 3 CLASS average change in temperature (C°) Percent Deviation GRAPH Data Analysis 1. Based on the graph created, describe how the amount of calcium chloride affected the temperature of the water. 2. Imagine you were the supervisor of the Leesburg Department of Public Works and you had to make a decision on whether to use calcium chloride or a stronger liquid chemical on the roads. The temperature of the roadway was -10°C. Would you use the solid calcium chloride or a stronger liquid chemical? Explain your answer using evidence and data. (Hint: think about the change of temperature that was noted in the lab and assume that the amount of calcium chloride used in the lab would cover one square foot of roadway basically the size of a piece of tile on the floor of this room). Conclusion Write a RECALL conclusion (level 2) on a separate piece of paper and attach it to this lab activity.