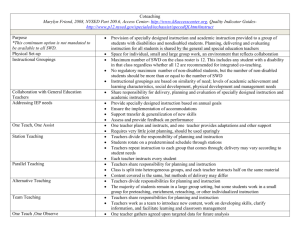
Full report - Proposal Project Management System (PPMS)
advertisement

` Project Director(s): Glen C. Rains, Ashfaq Sial Ahmad Project Title: Evaluation of Sanitation as an IPM tool for SWD Control in Blueberry Problem and Justification: Fruit production in the US has recently been challenged with a new invasive insect pest, spotted wing drosophila (SWD), Drosophila suzukii Matsumura (Diptera: Drosophilidae). The SWD, a native of Eastern and Southeastern Asia (1), is a devastating pest of small and stone fruits. Since it’s first detection in California in 2008 (2, 3), the SWD has spread throughout the United States (4) causing significant losses in crop yield and quality, and risk of even more profound damage. The SWD is highly polyphagous insect (5, 6) and presents a major threat to soft- and thinskinned fruit crops including blueberries, caneberries (blackberries and raspberries), cherries, strawberries, peaches, and grapes worldwide. SWD larvae have also been observed feeding on other wild and cultivated hosts including pears, persimmons, figs, loquat, currents, mulberry, buckthorn, and dogwood (5). Based on descriptions of SWD biology, SWD adults mate within 1-2d of emerging. Females can lay more than 600 eggs in their lifetime, usually 1-3 eggs per fruit. Eggs are inserted just under the skin of fruit, but paired respiratory filaments are left projecting from fruit, which allow the number of eggs to be assessed microscopically. Eggs develop in 1-3d, larvae in 5-7d, and pupae in 3-15d. Therefore, under optimal conditions of about 20 °C, one generation can complete development in about 8 -10 d leading to 15 generations per year (7). Because of this short generation time, SWD populations can increase to potentially devastating levels rather quickly. Estimates of 100-fold population increase every 2 wk are plausible. Actual loss statistics have been more difficult to generate, however, potential losses due to damage caused by SWD in fruit crops in the United States have been estimated at $850-900 million annually (8, 9). Georgia is among the top three blueberry producing states in the US (NASS 2012). Blueberries are the number one fruit crop in Georgia with an annual farm gate value of $250 million (10) and economic impact of $1 billion on the state economy (A. A. Sial, Pers. Comm. with Georgia Blueberry Growers Association). Since its first introduction in Georgia in 2010, SWD infestations have led to 15-20% loss of blueberry crop annually (A. A. Sial, Pers. Comm. with Georgia Blueberry Growers Association). Blueberries produced in the Southeastern states are primarily marketed as fresh fruit in the US as well as export markets. The fresh fruit marketers have zero tolerance for SWD infestation. Detection of a single larva in fruit samples can result in rejection of the entire shipment. It can be difficult to determine if fruit are infested by SWD at harvest because they often appear otherwise sound. Unfortunately, currently available traps and baits are useful for determining fly presence only, but are not reliable predictors of fly density and fruit infestation risk. While this aspect of SWD monitoring is actively being investigated (11, 12), SWD management is currently achieved primarily through preventative insecticide applications (2, 13-15). The number of insecticides available is limited to those with SWD activity and sufficiently short preharvest intervals (≤3 days) to allow their use on frequently picked crops such as blueberries. The most effective insecticides available for use against SWD are primarily broad-spectrum chemicals including organophosphates, pyrethroids, and spinosyns (13-16), the use of which is further ` complicated by annual application restrictions, preharvest intervals, and trade related issues with residue tolerances. In order to meet the zero tolerance policy for SWD mandated by the marketers, growers have to make calendar-day weekly insecticide applications, which are reapplied if feasible in the event of rain, resulting in as many as twice weekly applications as a preventive measure. In general, fruit growers are reluctant to apply broad-spectrum pesticides due to residues and associated health and environmental concerns. Additionally, the insecticides are quite expensive and frequent application of insecticides significantly increases the cost of production leading to lower profits for growers. Furthermore, repeated applications with similar broad-spectrum materials could lead to resistance development in SWD rather quickly, compromising the useful life span of these products, and threatening the sustainability of SWD management programs. It is therefore critical to improve currently practiced management programs based solely on prophylactic insecticide applications and develop more sustainable practices to manage SWD in commercial fruit crops by incorporating non-chemical management tactics including cultural control as part of the integrated pest management (IPM) programs. Crop hygiene and sanitation is one of the most commonly used cultural practices to manage fruit fly populations because fallen, damaged, and over-ripe fruit in the orchards serve as a source of attraction for fruit flies and also provides suitable habitat for breeding and larval development. Studies with other fruit fly species have revealed that significantly higher proportion of the fruit on the ground was infested with Fijian fruit fly (Bactocera passiflorae) as compared with that on the tree at a similar stage of maturity (17). In the context of fruit fly IPM, sanitation refers to destruction of all fallen and unwanted fruit from the orchard to ensure that larvae do not survive to pupate in the ground, and later emerge as adult flies. This tactic can help prevent fruit fly egg and larval (maggot) development in infested fruit and deny female adult flies a suitable oviposition site which significantly reduces the fly population in the orchards. A large-scale area-wide destruction of fallen fruit in the orchards resulted in successful control of the Chinese citrus fruit fly (B. minax) in China (17). General fruit fly control recommendations include field sanitation. Sanitation usually entails removal of the fallen fruit by hand which are covered or sealed in plastic and set in the Sun to solarize and kill the eggs and developing larvae. Sanitation is extremely labor intensive and the overall benefit has not been quantified for growers to invest the time and effort. Blueberry producers currently need to optimize time and money spent towards control of SWD. While there are many recommended practices for control, some are labor intensive, expensive and unproven. In addition, as stated earlier, constant use of pesticides also bring about the development of resistance and problems associated with pesticide residue and minimum reentry periods during harvest. As such, the development of sanitation mechanization procedures and determination of effectiveness on controlling SWD would be benefit to growers by developing a procedure that reduces pesticide applications. Packinghouse owners would potentially benefit by reducing the necessary sampling, sorting, freezing and other additional steps due to the reduced infestation of fruit. There will also be a potential benefit to marketers and exporters with a reduced risk of exceeding maximum residue level (MRL) due to the reduction in pesticide use. ` Objectives: The goal of this project was to use a blueberry field sanitizer to eliminate larvae from fallen fruit by rupturing the skin of the fruit on the ground. This was accomplished by mounting a leaf blower on the front of a tractor to move fallen blueberries from between the rows to the row middles (alleys) and developing a roller with modified surface to break open the skins and disrupt the development of the SWD eggs and larvae. To reach this specific goal we completed the following objectives: 1. Developed the field sanitizer for fallen blueberries 2. Evaluated the field sanitation of blueberry crop using sanitizer. 3. Analyzed and disseminated field test results to stakeholders 4. Assessed stakeholders view of sanitation before and after disseminating results. Results: 1. Develop field sanitizer for fallen blueberries The field sanitizer consisted of two components: blower and roller. The prototype blower was selected and designed to move fallen fruit from between the bushes in the row to the row middle where they will be rolled over to kill or disrupt the development of the eggs and larvae inside the berries. Each component was developed as follows. a. Blower A gas-powered backpack blower (Echo PB-770T) was purchased and modified for mounting to the front of a Ford 2610 tractor (Figure 1). The blower selected could move 756 cfm at an average of 196 mph at full speed. The force of the air from the blower and the angle and shape of the tube had a significant effect on the effectiveness in moving blueberries from between the rows. An adjustable arm was designed to mount on the front of the tractor and hold the end of the blower nozzle. It can be raised/lowered and extended/retracted to improve movement of blueberries between the bushes. Figure 1 Blower mounted to front of Tractor ` The trigger mechanism was changed so that blower speed could be adjusted from the tractor seat. The end of the blower was modified to increase the vertical air pressure to cover more of the variable terrain (Figure 2). A GoPro Silver video camera was also mounted as shown to record the blower action in the field. Figure 2. Modified end of blower tube and GoPro Camera. b. Roller Design A 36” poly roller packer with 18” dia. was purchased from Agri-supply Co. in Tifton, GA. The roller was filled with water and pulled behind the tractor. To improve the blueberry skin disruption, a covering was attached as an outer skin of the smooth roller. Several different attachments were examined as potential “skins”, each purchased from American Floormats (Figure 3a). The final covering was the “Nautilus Pool Mat” and is shown attached to the roller in Figure 3b. The weight of the roller was adjusted by adding water to the hollow roller. Weight was tested in the range of 200 to 400 lbs. The attachment to the tractor was modified so that the roller rotated with the terrain of the row middles. Figure 3 a) Selected materials for roller covering. b) Selected covering on roller. ` The weight of the roller is important to rupture of the fruit skin. However, the ground condition may also be important. If the ground is soft, the roller may push the blueberries into the ground without damaging the fruit. Over all soil types and weights, the heavier the roller the better the ability to disrupt the skin covering of blueberries placed on a test area in Tifton. Data was collected and plotted, however, a computer virus erased the data. We also tested the roller with and without covering. Without a covering the roller is smooth. The material chosen in figure 3b was the best for creating creases and pressure on the berries to allow the type of action needed to disrupt SWD development. Figure 4 is a picture of the entire system. Figure 4 Rear and front view of sanitizer/blower on tractor 2. Evaluate Field Sanitation of Blueberry Crop using Sanitizer. Field testing of the sanitizer was conducted at the University of Georgia Blueberry Research Farm in Alma, GA. The field tests were conducted to evaluate sanitation as a useful method to help control SWD and to evaluate the sanitizer as a useful tool in sanitation. a. Sanitation Treatments/Preparation SWD traps were built, sanitizer tested in tractor, and plots prepared for testing the sanitizer during the Summer 2014 blueberry harvest. The field treatments consisted of the matrix of tests shown in Table 1 for rabbiteye blueberry varieties. Table 1: Treatments and number of reps per treatment in Rabbiteye plots Sanitation Only Sanitation + Spray ½ Sanitation + ½ Spray Spray Only No Treatment Treatment # 1 2 3 4 5 Reps 3 3 3 3 3 ` Figure 5 Field layout for treatments, black lines are blueberry rows. 5 treatments x 3 reps. The spray treatments were with Mustang max applied at 40 GPA and 4 oz/acre of active ingredient. Each test treatment had 3 replicates, each rep covering 4 rows of blueberries, 75 feet long. Data was collected from sanitation of the inner 2 rows (alley 2, fig. 5) in each repetition. The sanitizer was used to blow berries from alley 1 and 3 into alley 2. The blower was then turned off as the sanitizer traveled down alley 2 to have the roller sanitize the berries in the row middle of the alley. Sanitation treatments were conducted on a weekly basis for all plots, except the ½ sanitation + ½ spray treatment. These treatments were sanitized once every 2 weeks and sprayed in the alternating weeks. b. Data Collection Data for each rep of each treatment were collected as follows. Each treatment was evaluated by three methods. First, traps were placed in the center of one rep of each treatment. Traps consisted of 32 oz. cups with 10-12 holes (3/16” of an inch) 1” below the top of the cup containing ½” of trap mix. These traps were collected weekly and replaced with fresh bait. Trap contents were identified with 15x hand microscope in the lab. Also, for each treatment, 500 fallen berries were collected weekly and put in growth chambers for 2 weeks. Number of emerging flies were counted and used to compare treatments. Lastly, blueberries were counted in 10 foot segment between bushes in the rows where they were inaccessible to the tractor tire or roller. These counts were taken before and after running the blower/roller combination at 3 mph through the field as shown in Figure 6. The berries were counted on the left and right side of the alley. This resulted in an assessment of how effectively the blower moved berries from between the bushes. Berries were not collected in those treatments (4 and 5) that were not sanitized. c. Data Presentation Sanitation data was presented to the Annual Georgia Blueberry Growers meeting in Alma, GA and the SWD OREI Planning Meeting in Atlanta. A pre- and post- presentation survey was ` handed out and growers asked to fill out two short questionnaires before and after the meeting presentation on our sanitation study (Appendix A). 3. Analyze field test results a. Trap Data Due to the amount of spraying at the UGA Blue Berry Research Farm, there were very few SWD found in the traps even in the untreated plots (Treatment 4). Only during harvest were there any collected and then only in small numbers as shown in Table 2. Table 2 – Adult SWD found in traps. Samples taken once per week within rep 2 of each treatment. Date Plot re Rep-Treatment Male Female 6/5/2014 2--1 0 0 2--2 2 0 2--3 1 0 2--4 0 0 2--5 1 0 6/12/2014 2--1 0 0 2--2 0 0 2--3 1 0 2--4 1 1 2--5 1 0 6/19/2014 2--1 1 0 2--2 0 0 2--3 0 1 2--4 0 1 2--5 0 1 6/26/2014 2--1 0 0 2--2 0 0 2--3 0 1 2--4 1 3 2--5 1 2 7/3/2014 2--1 2 7 2--2 3 4 2--3 0 5 2--4 2 1 2--5 3 2 Fallen blueberries collected and incubated for 2 weeks did not show any emergent flies for all 5 weeks of data tested. This somewhat correlated with the lack of SWD found in traps. Subsequently, we were not able to assess if the sanitation mechanism made a difference in SWD ` populations. There were none there to begin with. However, we were able to assess the ability of the blower to move blueberries from between the blueberry bushes so that they could be accessed by the roller. The tables below show the week by week movement of the berries. The columns show the number of berries between the trees before and after running the blower. Therefore, the fewer the berries after, the more berries were moved by the blower. b. Moving Berries with Blower Berries were counted in each treatment before and after running the blower for each week of the test. We ran the blower every other week on the ½ sanitation + ½ sprayer treatment. Week 1 did not have any berries on the ground for treatment 5, so collection began there on week 3. Results are report by rep-treatment (such as 2-1 is rep 2 treatment 1) as shown in Figure 4. Week 1 ` Week 2 Week 3 ` Week 4 Week 5 ` Five Week Total The average percent of berries moved from between the blueberry bushes to where the roller and/or tire could roll over them was 47% for the left and right side of the bushes. The challenges moving forward are: 1. The area around the bushes must be kept clean and void of weeds. Any weeds in the row with the blueberry bushes prevent them from moving to the alley. 2. Some blueberries also get blown into the base of the blueberry bushes, where they get stuck. The direction and number of tubes blowing the blueberries should be modified to blow blueberries away from the bushes when they get into the base of the bushes. 3. To sanitize blueberries closer to the base of the blueberry bushes, the roller should be moved to the outside of the tractor wheel, providing a width of the roller plus the tractor wheel for rolling over blueberries. The figure below is a top view of lego model showing the roller outside the tractor wheel, where it would be next to the base of blueberry bushes, closer to blueberries for sanitation. ` Presentation to Growers Sanitation data was presented to the Annual Georgia Blueberry Growers meeting and the organic blueberry growers meeting. A pre- and post- presentation survey was handed out and growers asked to fill out two short questionnaires, one before and one after the meeting (Appendix A). The first question tracked the respondent’s level of how important they thought sanitation was before and after the presentation. It also asked if they knew what it was before and after the meeting. There were 129 pre-survey respondents, but only 93 post-survey respondents. Whether this had an overall effect on each of the questions is unknown. While there appeared to be only 3 respondents after the meeting that did not know what sanitation was, compared to 39 before, the rest of the results are mixed. The grower responses before and after the sanitation presentation ` are relatively unchanged from 1 through 6, which would be somewhat a lack of importance, the results before and after are relatively unchanged. There was a marked increase in response of importance at the 7-8 level, but then there were actually fewer people post-presentation who believed it was of a higher level of importance (9-10). A follow-up question from the pre-survey revealed that 78% of growers responding had never used sanitation methods. Those that had used sanitation methods reported equipment cleaning and weed control as their sanitation methods. The second post-test question asked if the growers knowledge of sanitations was increased, 1, being very little and 10 being significantly. These results were very encouraging as shown in the bar chart. Over 60% rated their increased knowledge of sanitation as 8 or greater. Fifty-five percent of growers that responded were more likely to try adding sanitation to their SWD program and about that same percentage would pay at least $500.00 to implement the sanitation system presented and developed for this project. However, 19% were not willing to spend any money on sanitation. ` Literature Cited: 1. Lee, J. C., D. J. Bruck, H. Curry, D. Edwards, D. R. Haviland, R. A. Van Steenwyk, B. M. Yorgey. 2011. The susceptibility of small fruits and cherries to the spotted wing drosophila, Drosophila suzukii. Pest Manag. Sci. 67, 1358e1367. 2. Walsh, D. B., M. P. Bolda, R. E. Goodhue, A. J. Dreves, J. Lee, D. J. Bruck, V. M. Walton, S. D. O’Neal, F. G. Zalom. 2011. Drosophila suzukii (Diptera: Drosophilidae): invasive pest of ripening soft fruit expanding its geographic range and damage potential. J. Int. Pest Manage. 2, 1e7. http://dx.doi.org/10.1603/IPM10010. 3. Hauser, M. 2011. A historic account of the invasion of Drosophila suzukii (Matsumura)(Diptera: Drosophilidae) in the continental United States, with remarks on their identification. Pest Manag. Sci. 67, 1352e1357. 4. Burrack, H. J., J. P. Smith, D. G. Pfeiffer, G. Koeher and J. LaForest. 2012. Using volunteer based networks to track Drosophila suzukii an invasive pest of fruit crops. J. Integrated Pest Management 3(4): B1B5. 5. Cini, A., C. Ioriatti, Anfora, G. 2012. A review of the invasion of Drosophila suzukii in Europe and a draft research agenda for integrated pest management. Bull. Insectol. 65, 149e160. 6. Burrack, H. J., G. E. Fernandez, T. Spivey, D. A. Kraus. 2013. Variation in selection and utilization of host crops in the field and laboratory by Drosophila suzukii Matsumara (Diptera: Drosophilidae), an invasive frugivore. Pest Manag. Sci. http://dx.doi.org/10.1002/ps.03489. 7. Kanzawa, T. 1939. Studies on Drosophila suzukii Mats (in Japanese).Yamanashi Agricultural Experimental Station, Kofu, Japan. 8. Bolda, M. P., R. E. Goodhue, F. G. Zalom. 2010. Spotted wing drosophila: potential economic impact of a newly established pest. Agric. Resour. Econ. Update Univ. Calif. Giannini Found. Agric. Econ. 13, 5e8. 9. Goodhue, R. E., M. Bolda, D. Farnsworth, J. C. Williams, F. G. Zalom. 2011. Spotted wing drosophila infestation of California strawberries and raspberries: economic analysis of potential revenue losses and control costs. Pest Manag. Sci. 67, 1396e1402. 10. Wolfe, K. and T. Shepherd. 2011 Georgia Farm Gate Value Report. The University of Georgia, Center for Agribusiness and Economic Development, College of Agricultural and Environmental Sciences. Athens, GA: 2012. 11. Lee, J. C., L. D. Barrantes, E. H. Beers, H. J. Burrack, A. J. Dreves, L. J. Gut, K. A. Hamby, D. R. Haviland, R. Isaacs, A. R. Nielson, T. Richardson, C. R. Rodriguez-Saona, P. W. Shearer, C. A. Stanley, D. B. Walsh, V. M. Walton, F. G. Zalom, D. J. Bruck. 2013. Improving trap design for monitoring Drosophila suzukii (Diptera: Drosophilidae). Environ. Entomol. (in press). 12. Landolt P. J., T. Adams, H. Rogg. 2011. Trapping spotted wing drosophila, Drosophila suzukii (Matsumura) (Diptera: Drosophilidae), with combinations of vinegar and wine, and acetic acid and ethanol. J. Appl. Entomol. doi:10.1111/j.1439-0418.2011.01646.x. 13. Bruck, D. J., M. Bolda, L. Tanigoshi, J. Klick, J. Kleiber, J. DeFrancesco, B. Gerdeman, H. Spitler. 2011. Laboratory and field comparisons of insecticides to reduce infestation of Drosophila suzukii in berry crops. Pest Manag. Sci. 67, 1375e1385. 14. Beers, E. H., R. A. Van Steenwyk, P. W. Shearer, W. W. Coates, J. A. Grant. 2011. Developing Drosophila suzukii management programs for sweet cherry in the western United States. Pest Manag. Sci. 67, 1386e1395. 15. Haviland, D. and E. H. Beers. 2012. Chemical control programs for Drosophila suzukii that ` comply with international limitations on pesticide residues for exported sweet cherries. Journal of Integrated Pest Management Vol. 3, Issue 2. http://esa.publisher.ingentaconnect.com/content/esa/jipm/2012/00000003/ 16. Timmeren, S. V. and R. Isaacs. 2013. Control of spotted wing drosophila, Drosophila suzukii, by specific insecticides and by conventional and organic crop protection programs. Crop Protection 54, 126-133. 17. Allwood, A. J., A. Leblanc, E. T. Vueti, E. Bull. 2001. Fruit fly control methods for the Pacific Island countries and territories. Pest Advisory Leaflet, Secretariat of the Pacific Community. Plant Protection Service, 40. ` APPENDIX A – Survey, Pre and Post Pre-Meeting Blueberry Workshop Survey – scale of 1-10. 1 equal not important and 10 equals very important. For each answer circle your best response or fill in the blank 1. How important would you rate sanitation as a control measure for SWD in Blueberries? If you do not know what sanitation is, circle 0. 0 1 2 3 4 5 6 7 8 9 10 2. Have you used sanitation to control SWD or other pests in the past? Yes No If yes, what pest and what sanitation method did you use? Pest__________ Sanitation Method ____________________________________ _____________________________________________________________________ 3. How important is SWD control to you for blueberry production? 1 2 3 4 5 6 7 8 9 10 4. How many acres of blueberries do you produce? (fill-in blank) Highbush _____ Rabbiteye ____ High Density ____ 5. What strategies do you employ to control SWD? Circle all that apply Aerial Spraying Ground Spraying Cultivar Selection Scouting Others _________________________________________________ Traps ` Post-Meeting Blueberry Workshop Survey – scale of 1-10. 1 equal not important or not likely and 10 equals very important or very likely. For each answer circle your best response 1. How important would you rate sanitation as a control measure for SWD in Blueberries? If you do not know what sanitation is, circle 0. 0 1 2 3 4 5 6 7 8 9 10 2. Did the presentation increase you understanding of sanitation for SWD control? 1 2 3 4 5 6 7 8 9 10 3. How likely are you to implement sanitation procedures as described in this presentation? 1 2 3 4 5 6 7 8 9 10 4. How much would you be willing to spend as a one-time expense to utilize sanitation as described at the meeting to help control SWD? $0 $100-$200 $200-$500 $500-$1000 $1000-$2500.00 5. Do you believe, based on the presentation, that adding sanitation to your SWD control strategy would reduce pesticide applications? Yes No