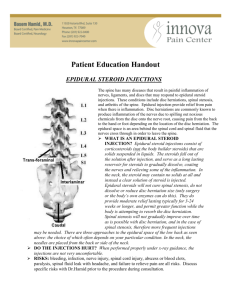
Supplemental PHC Requirements for Medical Necessity for Pain
advertisement

PARTNERSHIP HEALTHPLAN OF CALIFORNIA Medical Necessity Criteria for Pain Management Procedures MCUG3007 - Attachment C MCUP3049 – Attachment B 11/18/14 22521 to 22525 Percutaneous vertebroplasty and percutaneous vertebral augmentation 27096 SI joint injection 62290 to 62291 Discography, Lumbar and Cervical 62360 to 62362 Implantable or replacement device for intrathecal or epidural drug infusion; subcutaneous reservoir 63650 thru 63688 Insertion, revision, or removal of spinal neurostimulator 64479 to 64484 Transforaminal epidural injection Recent well controlled study shows no benefit over placebo for longstanding vertebral fractures/pain. Evidence not as clear for acute fracture; as such, approval will only be considered in patients with 1. Acute vertebral compression fracture (less than 3 months), debilitated and unable to tolerate conservative therapy (minimum of 3 weeks of conservative therapy should be tried first), or 2. Vertebral hemangioma failing conservative therapy, or 3. Malignancy related compression fracture with no neurologic symptoms and pain interfering with activities of daily living. (Same as InterQual criteria, except that longstanding fracture not covered; reference: Treatment of Symptomatic Osteoporotic Spinal Compression Fractures, Journal of the American Academy of Orthopedic Surgeons, March 2011; Spine J. 2012 Nov; 12(11): 998-1005) Remove requirement for trial of NSAIDs and imaging studies. Activity modification for 4 weeks required, unless pain is very severe. Repeat injections only approved if at least 50% decrease in pain lasting at least 8 weeks. Maximum of 3 SI joint injections with steroid covered per 12 months. (Same as previous criteria, except documentation requirements removed) Current medical evidence is that harm outweighs benefit for this procedure. It will only be covered if new and better studies are submitted demonstrating a benefit. Physician review required. (Reference: New York Medicaid redesign team: Basic Benefit review workgroup final recommendations, Nov. 1, 2011) Only medical indication that will be covered is intractable pain caused by malignancy, where a randomized controlled trial has shown short and longterm benefit. Studies in non-cancer pain are observational and of poor quality. (Reference: Pain Physician. 2011 May-Jun; 14(3): 219-248) InterQual criteria followed (covered for complex regional pain syndrome, failed back surgery) except that requirement for psychiatric evaluation is waived except in patients with serious mental illness. Same as InterQual except: remove requirement for documentation of trial of NSAIDS; add requirement for progress note to demonstrate history of radicular PARTNERSHIP HEALTHPLAN OF CALIFORNIA Medical Necessity Criteria for Pain Management Procedures MCUG3007 - Attachment C MCUP3049 – Attachment B 11/18/14 64490 to 64495 Paravertebral facet injections and medial branch blocks 64633 to 64636 Destruction by neurolytic agent, paravertebral facet joint 72285, 72295 Cervical, Thoracic, Lumbar discography symptoms. A minimum of 30 days conservative treatment before eligible for epidural steroid injection. If MRI shows spinal stenosis with no significant impingement, epidural injection is not medically indicated. For repeat injections, a minimum of 50% improvement in pain symptoms on the prior epidural injection is required. Maximum of two levels of transforaminal epidural injections of the spine per 3 months. This may be two levels on the same side or one level bilaterally. The interval between injections must be no more frequently than every 2 months, and the maximum number of injections per site is three per year. Initial injection: Same as InterQual criteria, except documentation of physical therapy for 6 weeks and NSAIDs are not needed. Patient must have a minimum of 12 weeks of symptoms consistent with facet joint pain. The progress note should document a physical examination of the back, including pain elicited with movement. No more than 4 levels will be approved, either 4 levels on one side or two levels bilaterally. Maximum of two MBB per level per year. For repeat injections, a pain log must be submitted showing that the pain relief was consistent with the anesthetic agent used. For example, for a pure bupivacaine block, this is about 2-3 hours; bupivacaine plus minimal dose steroid block could be up 3-4 days for medial branch blocks versus a month to several months for facet joint block. InterQual criteria followed Same as 62290 and 62291 above.