Post-Lab: Using a SNP to Predict Bitter
advertisement

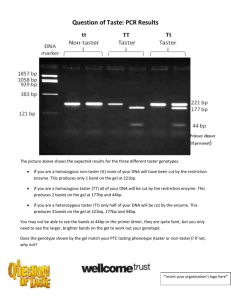
IB Biology 2 / IHS Name: _________________________________________________ Per. __________ Post-Lab: Using a SNP to Predict Bitter-Tasting Ability 1. Write the names of your teammates in the order (from left to right) they loaded their DNA in your gel. Consult the gel map if you don’t remember. __________________________ , _________________________ , __________________________ , _________________________ 2. Determine your PTC phenotype. First, place one strip of control taste paper in the center of your tongue for several seconds. Note the taste. Then, remove the control paper, and place one strip of PTC taste paper in the center of your tongue for several seconds. How do you and your teammates describe the taste of the PTC paper as compared to the control: strongly bitter, weakly bitter, or no taste other than paper? __________________________ , _________________________ , __________________________ , _________________________ 3. Determine your PTC genotype. Observe the photograph of the stained gel containing your PCR digest and those from your teammates. Orient the photograph with the wells at the top. Use the ideal gel photograph to help interpret the band(s) in each lane of your gel. a) Locate the marker DNA lanes on either side of the gel. Because our gels ran longer than the ideal gel, the bands of DNA appear more spread out than they are in the ideal gel. The marker was a plasmid called pBR322 that was digested with a restriction enzyme called BstNI. Locate the bands corresponding to each restriction fragment: 1,857 bp, 1,058 bp, 929 bp, 383 bp and 121 bp. The 121-bp band may be very faint, even not visible. Can you see marker DNA on your gel? __________________ . b) Locate the lanes containing each student’s undigested PCR product (U). There should be one prominent band in this lane with a size of about 221 bp. Compare the migration of the undigested PCR product with that of the 383-bp and 121-bp bands of the marker. For how many students is undigested DNA visible on your gel? ____________ out of _____________ . c) To score the alleles, compare the digested PCR product (D) with the uncut control. It will be one of three genotypes: t t nontaster (homozygous recessive) shows a single band in the same position as the uncut control. TT taster (homozygous dominant) ideally shows two bands of 177 bp and 44 bp. The 177-bp band migrates ahead of the uncut control; the 44-bp band may be faint. If you see the 177-bp band, but not the 44 bp band, you can still score the genotype as TT. Tt taster (heterozygous) ideally shows three bands that represent both alleles – the 221-bp band from one parent, and the 177-bp and 44-bp bands from the other parent. The 221-bp band will be stronger (more intense) than the 177-bp band. (If the 221-bp band is less intense than the 177-bp band, the student has an incomplete restriction digest of genotype TT.) The 44-bp band may be faint. If you see the 221-bp and 177-bp bands, but not the 44-bp band, you can still score the genotype as Tt. Additional faint bands at other positions occur when the primers bind to chromosomal loci other than the PTC gene and give rise to nonspecific amplification products. (We tried to minimize this by placing our PCR tubes on ice until we could run PCR.) 4. Record team results in Table 1, below. Use tally format: Table 1. Correlation of PTC genotype with phenotype for Table ___ . Genotype from gel Phenotype: from taste Phenotype from taste photograph test: strong taster test: weak taster TT Tt tt Can’t tell genotype Phenotype from taste test: nontaster 5. Record class data in Table 2, below. Use tally format: Table 2. Correlation of PTC genotype with phenotype for the class, period ___ IB Biology Genotype from gel Phenotype: from taste Phenotype from taste Phenotype from taste photograph test: strong taster test: weak taster test: nontaster TT Tt tt Can’t tell genotype 6. How well does the TAS2R38 genotype predict PTC-tasting phenotype? Use our class data in your response. 7. What is one kind of evidence that would indicate whether the ancestral state of TAS2R38 is taster? (Hint: think evolutionary tree!) 8. What is one plausible, scientific reason that could explain why a) …a student has no bands in both his U and D lanes? b) …a student has the 221-bp band in her U lane, but no bands in her D lane? c) …a student with a T t genotype doesn’t taste PTC? (This is a hard one… try it!) 9. In the space to the right, sketch a graph of one of the PCR amplification cycles used in this lab. (Temperature should be the y-axis, and time the x-axis. 10. How did the HaeIII enzyme discriminate between the single nucleotide polymorphism in TAS2R38? Be specific. 11. How does having a “G” at position 145 in both his alleles of the TAS2R38 gene render a person unable to taste PTC? Explain in detail. 12. In a forensic crime lab like CODIS (the Combined DNA Index System of the FBI), SNPs are not used to exonerate or incriminate suspects. Instead, they use thirteen STR loci in their DNA analysis. Some of these loci have more than 20 alleles in the population! Explain what Short Tandem Repeats are by consulting Neil 9e (pp 420-421) or by going online at http://www.biology.arizona.edu/human_bio/activities/blackett2/str_description.html . A diagram might be useful. 13. State two different ethical issues raised by human DNA typing experiments like ours. 14. A 2001 study of 344 populations worldwide by Guo and Reed* estimated the frequency of the PTC nontaster allele at 0.48. For our class data, calculate the frequency of the nontaster allele. Show all work and circle your answer. (Hint: our class is certainly not in HWE!)