APPENDIX Alternative and future approaches to renal denervation
advertisement

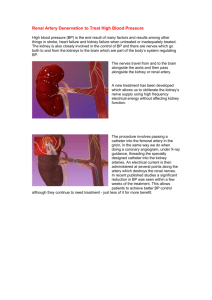
APPENDIX Alternative and future approaches to renal denervation Currently available clinical trial data has primarily been obtained with first- and second generation Symplicity radiofrequency catheter systems and sparked substantial interest in this area, resulting in the initiation of a large number of clinical trials and observational studies in (resistant) hypertension and other potential indications. A total of 45 clinical trials investigating various approaches to achieve RDN are currently listed on clinicaltrials.gov and summarized in Online Table 1. Furthermore, these initial promising results have led to the rapid development of several alternative treatment modalities to achieve renal artery denervation: Radiofrequency ablation Recently, a report on the use of a saline-irrigated radiofrequency ablation catheter (Celcius Thermocool, Biosense Webster, Diamond Bar, California) for RDN was published. The ablation catheter was placed within the renal artery to allow energy delivery in a circumferential, longitudinally staggered manner in 10 patients with resistant hypertension. Energy titration was performed to achieve a 10% to 20% drop in impedance. After 6-month, the systolic/diastolic blood pressure decreased by −21/−11 mm Hg. There was no evidence of renal artery stenosis or aneurysm at repeat angiography, and the authors reported a significant decrease in metanephrine (−12±4; p=0.003), normetanephrine (−18±4; p=0.0008), and aldosterone levels (−60±33ng/l, p=0.02) at 3 months (JACC: Cardiovascular Interventions2012; 5, 758-765). The EnligHTNTM catheter system (St. Jude Medical) has a basket-like design with four separate electrodes allowing simultaneous or sequential activation. In the first-in-human multi-centre, non-randomized study (EnligHTN I) in 46 patients with drug-resistant hypertension the use of this system was safe and resulted in a significant office blood pressure reduction by -28/10, -27/10 and -26/10mmHg at 1, 3 and 6 months, respectively . (Eur Heart J 2013: 34:2132-40) The OneshotTM catheter system (Maya Medical, Saratoga, California, now Covidien) delivers a single RF treatment per artery utilizing a balloon mounted helical electrode configuration. The Vessix V2 RDN system (Vessix Vascular) is a percutaneous noncompliant balloon catheter system with RF electrodes and thermistors mounted on the exterior of the balloon. Once inserted into the renal artery, a 30-second inflation/treatment per renal artery delivers simultaneous RF therapy with independent temperature control to all electrode pairs. It has received European CE Mark approval and is currently being trialled in the international multicentre REDUCE-HTN study. Conference abstracts report BP reduction similar in magnitude to those reported in the Symplicity studies. Ultrasound ablation Other systems use catheters emitting ultrasound energy to achieve RDN. The TIVUSTM catheter (CardioSonic, Tel Aviv, Israel) is a high-intensity, nonfocused ultrasonic catheter system. By applying ultrasonic energy, the TIVUS technology enables localized thermal modulation of the renal vessel wall tissue containing the renal nerves, presumably without thermal damage to the endothelium and media. Swine studies have shown that kidney tissue NE concentrations was reduced by 50% or more at 30- and 90day follow-up without local tissue damage. The PARADISETM catheter system (ReCor Medical, Inc, Ronkonkoma, NY), is an ultrasound catheter placed inside a low-pressure balloon, which, when inflated, allows symmetrical distribution of ultrasound energy into the surrounding artery. Preliminary clinical data for PARADISE were previously reported at EuroPCR 2012 with an average systolic blood pressure reduction of 31mm Hg in 7 patients at 60-days follow-up. The use of external ultrasound is currently investigated by Kona Medical, Campbell CA, The system is designed to deliver ultrasound energy from an external source to the renal nerves. The basis of the non-invasive technique is low-intensity focused ultrasound (LIFU). Peri-vascular pharmacologic ablation Catheter systems for local delivery of neurotoxic drugs are also being investigated. The Mercator BullfrogTM catheter, (Mercator MedSystems, Inc, San Leandro, CA) is composed of a catheter tipped with a balloon-sheathed microneedle. After advancement of the catheter into the renal artery, the balloon is inflated with saline, securing the system for injection and sliding the microneedle through the vessel wall. Guanethidine is delivered through the vessel wall into the adventitia. Given locally, guanethidine is known to induce an autonomic denervation directly and through an immune-mediated pathway. Mercator’s preclinical experiments have shown that guanethidine, injected at appropriate concentrations into the adventitial space around renal arteries, selectively ablates the nerves in the adventitia around the renal artery after a single, 20-minute procedure. Online Figure 1. Discrepant effects of antihypertensive treatment and renal sympathetic denervation on office and ambulatory blood pressure levels. Dark gray squares represent the effects of renal sympathetic denervation on office and ambulatory BP levels in subjects with true resistant hypertension receiving renal denervation in five interventional studies. Light circles represent the effects of antihypertensive treatment in office and ambulatory BP levels in different pharmacological studies. Regression line (dotted line) and 95% confidence intervals (dashed lines) for pharmacological studies are presented. (with permission from Parati et al. Circulation 2013: 128:315-7) Online Table 1. Clinical trials on renal denervation [http://clinicaltrials.gov] Company Study name Device Inclusion c (energy) Ardian Symplicity™ *RDN in Patients With Refractory RF office SBP≥ 160 m Catheter Hypertension dose of appropria drugs Ardian Symplicity™ Catheter Symplicity Catheter (Medtronic ®) *RDN in Patients With Refractory RF Hypertension *Renal Denervation in Refractory office SBP≥ 160 least 3 anti-HT RF Hypertension office SBP≥ 160 m dose of at least 3 drugs Symplicity Catheter (Medtronic ®) *RDN in Patients With Uncontrolled RF Hypertension (Symplicity HTN-2) office SBP ≥1 (≥150mmHg in T least 3 anti-HT Ardian Symplicity™ Catheter *RDN in End Stage RENAL Disease RF Patients With Refractory least 3 anti-HTN Hypertension Ardian Symplicity™ *Native Kidney Denervation in Catheter Patients With End Stage Renal dialysis>6 RF (Medtronic ®) RDN in Hypertension (DENER- ESRD on dial uncontrolled HT w Disease Symplicity Catheter office SBP≥ 160 anti-HTN RF HTN) office SBP>14 DBP>90mmHg SBP≥135 and/o mmHg) on at leas drugs including Symplicity Catheter (Medtronic ®) Renal Sympathetic Denervation in Mild Refractory Hypertension RF RF on at least 3 drugs including (daytime SBP 140 DBP 90-94m Symplicity Catheter (Medtronic ®) RDN in Patients With Resistant RF RH (Office SBP≥1 daytime≥135 mmH Hypertension and Obstructive Sleep Apnea Symplicity Catheter (Medtronic ®) Symplicity Catheter (Medtronic ®) RDN in Diabetic Nephropathy RF (DERENEDIAB) RDN in Refractory Hypertension Persistent Prote T2DM RF (PRAGUE-15) office SBP>14 daytime SBP>130 at least 3 an Symplicity Catheter (Medtronic ®) Symplicity Catheter (Medtronic) Renal Artery Denervation in Chronic RF Heart Failure Study (REACH) Renal Sympathetic Denervation and LVEF<4 RF Potential Effects on Glucose diuretic or cert Risk-Factors (Re-Shape) (Medtronic ®) Symplicity Catheter (Medtronic ®) Renal Nerve Ablation in Chronic office BP>140/90 least 4 anti-HTN Metabolism and Cardiovascular Symplicity Catheter HF (NYHA intoleran RF CKD (stag RF office SBP ≥1 Kidney Disease Patients The Effect of RDN on Biological Variables (≥150mmHg in T least 3 anti-HT including a di intolerance t Symplicity Catheter RDN in Patients With Chronic Heart (Medtronic ®) Failure & Renal Impairment Clinical RF HF (NYHA II LVEF<4 Trial (SymplicityHF) Symplicity Catheter RDN in Patients With Uncontrolled (Medtronic ®) Hypertension (SYMPLICITY HTN- RF least 3 anti-HT 3) Symplicity Catheter office SBP≥160m including a d Global SYMPLICITY Registry RF HT; DM; HF; C Study of Catheter Based RDN RF RH on at least 3 (Medtronic ®) Symplicity Catheter (Medtronic ®) Therapy in Hypertension (DEPART) drugs including a an attempt to t spironolac Symplicity Catheter (Medtronic ®) The Effects of RDN on Insulin RF Treatment resista daytime ≥145mmH Sensitivity 3 anti-HTN drugs diureti Symplicity Catheter (Medtronic ®) Denervation of the REnal Artery in Metabolic Syndrome (DREAMS) RF FPG ≥5.6 mmol SBP>130 mmHg use of anti-diabe HTN dru Symplicity Catheter (Medtronic ®) Single-arm Study of Symplicity™ RF RDN System in Patients With office SBP≥160m least 3 anti-HT Uncontrolled HyperTensioN in India including a d (HTN-India) Symplicity Catheter (Medtronic ®) Denervation of the REnal Artery in RF Metabolic Syndrome (DREAMS) FPG ≥5.6 mmol/L use of anti-diabet ABPM with SBP without the use o drugs Symplicity Catheter Renal Sympathectomy in Treatment RF daytime SBP>145 (Medtronic ®) Resistant Essential Hypertension, a least 3 anti-HTN Sham Controlled Randomized Trial diureti (ReSET) MDT-2211 System (Medtronic Vascular) RDN by MDT-2211 System in RF Patients With Uncontrolled least 3 anti-HT Hypertension (HTN-J) Standard steerable Marinr RF ablation Catheter (5F RDN in Patients With Advanced office SBP ≥160m including a d RF Heart Failure HF (NYHA I LVEF<3 or 7F) Vessix V2 RDN System Treatment of Resistant Hypertension Using a Radiofrequency RF office SBP≥ 160 least 3 anti-HTN Percutaneous Transluminal therap Angioplasty Catheter (REDUCEHTN) Celcius Thermacool Catheter or Chilli II Cooled Ablation Catheter Impact of Renal SympAthetic RF RH (BP>140/90 DEnerVation on Chronic least 3 anti-HTN HypErtension (SAVE) a diuretic or treatm HTN Biosense Webster Celcius Thermacool catheter Renal Sympathetic Denervation for RF the Management of Chronic least 3 anti-HTN Hypertension (RELIEF) THERMOCOOL® Catheter RDN in Patients With Uncontrolled daytime BP>140 a diuret RF office SBP≥ 160 m DBP≥90 mmHg o Hypertension in Chinese appropriate antiTHERMOCOOL® Renal Sympathetic Modification in catheter Patients With Chronic Renal Failure THERMOCOOL® Renal Sympathetic Modification in catheter THERMOCOOL® catheter RF definite kidney RF RF Renal Sympathetic Modification in catheter Patients With Metabolic Syndrome HF (NYHA II, LVEF<40% o Patients With Heart Failure THERMOCOOL® EH (office SBP≥ and/or DBP Patients With Essential Hypertension Renal Sympathetic Modification in at least three mon RF Metabolic Syndro of T2D Circumferential PVI+RDN Combined Treatment of Resistant (catheter not provided) Hypertension and Atrial Fibrillation St. Jude Medical system Safety and Efficacy Study of Renal RF RH on at least 3 a persistent or paro RF Artery Ablation in Resistant office SBP ≥1 (≥150mmHg in T Hypertension Patients (EnligHTN 1) least 3 anti-HT including a d Maya Medical OneShot Rapid Renal Sympathetic Ablation System Denervation for Resistant RF least 3 anti-HT Hypertension (RAPID) not provided ReCor Medical PARADISE Efficacy and Safety of including a d RF Ultrasound office SBP≥16 Radiofrequency RDN in Drug or DBP ≥100 on a Resistant Hypertension HTN drugs includ Renal denervatIon by ultraSound IVUSE Transcatheter Emission (REALISE) Kona Medical Focused office SBP ≥160m A Safety Evaluation of Renal RH as defined i ESH-ESC gu US Denervation Using Focused office SBP≥ 160 least 3 anti-HT Therapeutic Ultrasound on Patients With Refractory Hypertension not provided Adjunctive Renal Sympathetic Denervation to Modify Hypertension RF office SBP≥ 160 m DBP≥100 mmHg as Upstream Therapy in the anti-HTN drugs a Treatment of Atrial Fibrillation (H- or paroxysm FIB) not provided Sympathetic Activity and Renal RH (office BP>14 Denervation (ReD) on at least 3 a includi a diuret not provided RDN for Management of Drug- --- Resistant Hypertension (INSPiRED) EH under maxim (daytime SBP≥13 mm Hg) on at leas drugs including not provided (drug Effect of RDN on NO-mediated intervention: NG- Sodium Excretion and Plasma Levels monomethyl-L-arginine (L- --- EH (daytime B mmHg on at least of Vasoactive Hormones (RENO) including a d NMMA) not provided DENERVATION of the renAl --- sympathetIc nerveS in hearT Failure HF with evidence dysfunction, LV With nOrmal Lv Ejection Fraction (DIASTOLE) not provided Renal Artery Denervation in Chronic Heart Failure (REACH-Pilot) STUDY COMPLETED RF Congestive HF(N RF indicates radiofrequency; RDN, renal denervation; EH; essential hypertension; RH, resistant hypertension; OSA, obstructive sleep apnoea, T2DM, type 2 diabetes mellitus; HF, heart failure; CKD, chronic kidney disease; CRF, chronic renal failure; AF, atrial fibrillation; anti-HTN, anti-hypertensive; LVEF, left ventricular ejection function; FPG, fasting plasma glucose; PVI, pulmonary vein isolation; IVUSE, intravascular ultrasound emission; *Indicates ongoing study, not recruiting participants Online Figure 1.