MU Manzoor suppl mat
advertisement

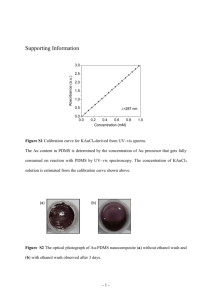
SUPPLEMENTARY MATERIALS Stretchable conducting gold films prepared with composite MWNT/PDMS substrates M U Manzoor1, P Lemoine2, D Dixon2 and J W J Hamilton2, PD Maguire 1 Department of Metallurgy & Materials Engineering, College of Engineering & Emerging Technologies, University of the Punjab, Lahore, Pakistan 2 NIBEC, University of Ulster at Jordanstown, Shore Road, Newtownabbey, Co. Antrim, BT37 0QB, Northern Ireland, UK E-mail:p.lemoine@ulster.ac.uk Amorphous carbon impurities within MWNT thermally decompose below 365oC [7]. TGA measurements performed in this study gave a mass loss below 365C < 2%, however the ash content, or metallic residue was < 0.2 wt.%; probably made of metal oxide and catalyst residue. a b Figure S1. SEM micrographs of the MWNT before (a) and after (b) tip sonication, marker bar = 2μm. The de-aggregation effect of the tip sonication treatment is clear. 1 Figure S2. Raman spectra of the pure PDMS, 4 and 8 % MWNT composites. The D, G and 2D peaks are MWNT peaks. The other vibrations are from PDMS; Si-C symmetric stretching (708 cm-1),CH3 symmetric bending (1262 cm-1), CH3 assymmetric bending (1412 cm-1), CH3 symmetric stretching (2907 cm-1) and CH3 assymmetric stretching (2965 cm-1) For pristine MWNT, the ID/IG and ID/I2D ratio are 0.03 and 0.18, respectively. After sonication, these ratios increase to 0.19 and 1.0, respectively, a sign that the sonication process brings new defects along the tubes. Figure S2 shows the Raman spectra for the different composites, with peak assignments for both the MWNT and PDMS vibrational groups [10]. 2 a b Figure S3. Analysis of the Raman spectra of the CNT/MWNT composites (a) ID/IG and ID/I2D ratio. The dotted lines give the values of these ratios for the pure tip-sonicated MWNT, (b) Normalized intensity ratio rx/r0 to the intensity of the Si-O-Si stretching vibration at 488 cm-1, where the subscript x and 0, respectively, correspond to the composite and pure PDMS. The ID/IG and ID/I2D ratios are generally higher in the composites, with the exception of the 4 % composite. Generally, high ID/IG and ID/I2D ratio in CNT/polymer composites are associated with covalent functionalities neutralizing dangling bonds on the CNT outer shells [8] or with damping of the G vibrations due to polymer intercalation within the bundles [9]. Hence, the trend shown in figure S3a is consistent with the composite prepared with the 4% CNT fraction having poorly dispersed CNT. Changes in the vibrational structure of the PDMS network upon MWNT addition can also be investigated with Raman spectroscopy. A comparison of Raman intensities between samples is difficult as they are affected by scattering, focus conditions and composite homogeneity. However, the ratio of these intensities informs on the relative importance of the vibrational groups and hence can be used to gauge the occurrence of chemical reactions, as demonstrated for ZnO-PDMS composite [11]. In the analysis done in figure S3b, the peak intensity for each PDMS vibration, as determined from figure S2, has been divided by the peak intensity for the Si-O-Si stretching vibration at 488 cm-1. The rationale for choosing this molecular group is based on the well-established hydrosylation cure reaction for PDMS; the vinyl groups in the base and the Si-H bonds in the curing agent react to produce ethylene bridges which crosslinks the PDMS into a 3D network [12]. The number of Si-O-Si groups in the initial mixture (base + cross-linking agent) is the same as in the cross-linked PDMS whereas the numbers of C-H and C-Si bonds change upon cross-linking. Hence, variations in these intensity ratios are good indications of chemical changes resulting from an incomplete curing of the PDMS network. As the curing agent (CA) and base are stoichiometrically matched in the Sylgard 184 formulation, any consumption of the reactants by a reaction with MWNT would affect the final number of crosslinks. These calculated ratios in the composites were then normalized with respect to that of the pure PDMS using the expression; rx r0 = (I⁄I ) 488 PDMS−MWNT (I⁄I ) (3.1) 488 PDMS 3 Figure S3b shows that for 4 % MWNT, there are small variations of intensity ratio (1.15 ± 0.16); all PDMS vibration amplitudes are similar to that observed in the pure PDMS. At all other % MWNT, these intensity ratio exhibit large variations (~ 1.0 ± 0.9), the PDMS molecular groups vibrate with different relative amplitudes with respect to the pure PDMS. In other words, the PDMS in these composites has different bonding environments than pure PDMS, indicative of a reaction between the MWNT and the PDMS cure. 4 Derivate weight loss, au 4% 2% 0% Weight loss, % 100 80 60 40 200 400 600 800 1000 0 200 400 600 800 1000 0 400 600 800 10 % 8% 6% 200 100 Weight loss, % 1000 Derivate weight loss, au 0 80 60 40 0 200 400 600 800 Temperature, °C 1000 0 200 400 600 Temperature, °C 800 1000 0 200 400 600 800 1000 Temperature, °C Figure S4. TGA traces; weight loss and derivative weight losses for the various MWNT/PDMS composites. Figure S4 shows TGA traces for the composite samples, illustrating both the weight loss and weight loss derivative. There are no weight loss steps observed below 200°C, demonstrating that no trapped solvent was present in these samples. However, there is a gradual change in the TGA profile, a second derivative peak appearing upon MWNT addition at higher temperatures. This suggests that the thermal degradations of PDMS are altered by the presence of the MWNT. This shift to a higher temperature peak has previously been associated with a thermal cross-linking reaction [13, 14], implying that the presence of the MWNT reduces the cure yield for PDMS and results in non-crosslinked regions within the PDMS network. It is consistent with the observed longer curing time for the composites and the result of figure S3b. 5 Figure S5. Raman spectra of the pure curing agent (CA) and a CA/MWNT mixture analysed within and outside a MWNT aggregate. The peaks at 488 and 2165 cm-1 respectively correspond to Si-O-Si stretching and Si-H stretching vibrations. The D and G peaks of the MWNT are also shown. Separate experiments were conducted on (base + MWNT) and (CA + MWNT) mixtures to identify more precisely the nature of the reaction between MWNT and PDMS. Using the same approach followed in figure S3, it was found that the Raman intensity ratio of the various PDMS vibrations (i.e. to the Si-O-Si stretching vibration at 488 cm-1) were not significantly different for the pure base and base/MWNT mixture; the MWNT did not react with the base. The situation was different for the CA/MWNT mixture; Raman spectra of pure CA and a CA/MWNT mixture are shown in figure S5. The Si-H stretch vibration of the CA can be seen at 2165 cm-1. The intensity ratio for the 2165 and 488 cm-1 vibrations is 0.88±0.13 for pure CA. For the CA/MWNT mixture it is 0.26±0.10 within MWNT aggregates and 0.87±0.11 outside these aggregates. This clearly shows that CA and MWNT react together. Further confirmation of this came from TGA analysis. Firstly, base and MWNT were mixed together, bath sonicated, the CA was added afterwards. TGA analysis of the cured material showed two peaks of similar heights, as shown for instance for the 8 % sample of figure S4. Secondly, the CA and MWNT were mixed together, bath sonicated, the base was added afterwards. In this case the TGA analysis of the cured material showed a larger second peak, as seen for the 6 % sample of figure S4, indicative of a larger fraction of un-cross-linked component in the material. This is consistent with some of the curing agent reacting first with the MWNT and therefore being unavailable to crosslink the base at a later stage. 6 1. Reference 1. STretchable ELectronics for Large Area applications. Status of the Verhaert infant respiratory monitor, 2010. 6. 2. Lacour, S.P., D. Chan, S. Wagner, T. Li and Z. Suo, Mechanisms of reversible stretchability of thin metal films on elastomeric substrates. Applied Physics Letters, 2006. 88(20): p. 204103. 3. Graz, I.M., D.P.J. Cotton and S.P. Lacour, Extended cyclic uniaxial loading of stretchable gold thin-films on elastomeric substrates. Applied Physics Letters, 2009. 94(7): p. -. 4. Wagner, S., S.P. Lacour, J. Jones, P.I. Hsu, J.C. Sturm, T. Li and Z. Suo, Electronic skin: architecture and components. Physica E: Low-dimensional Systems and Nanostructures, 2004. 25(2): p. 326-334. 5. Tuinea-Bobe, C.L., P. Lemoine, M.U. Manzoor, M. Tweedie, R.A. D'sa, C. Gehin and E. Wallace, Photolithographic structuring of stretchable conductors and sub-kPa pressure sensors. Journal of Micromechanics and Microengineering, 2011. 21(11): p. 115010. 6. Manzoor, M.U., C.L. Tuinea-Bobe, F. McKavanagh, C.P. Byrne, D. Dixon, P.D. Maguire and P. Lemoine, Amorphous carbon interlayers for gold on elastomer stretchable conductors. Journal of Physics D: Applied Physics, 2011. 44(24): p. 245301. 7. Li, J. and Y. Zhang, A simple purification for single-walled carbon nanotubes. Physica E: Lowdimensional Systems and Nanostructures, 2005. 28(3): p. 309-312. 8. Celina-Maria DAMIAN, A.M.P., Horia IOVU, Ethylenediamine Functionalization Effect On The Thermo-Mechanical Properties Of Epoxy Nanocomposites Reinforced With Multiwall Carbon Nanotubes. UPB Scientific Bulletin, Series B: Chemistry and Materials Science, 2010. 72(3): p. 12. 9. McClory, C., T. McNally, M. Baxendale, P. Pötschke, W. Blau and M. Ruether, Electrical and rheological percolation of PMMA/MWCNT nanocomposites as a function of CNT geometry and functionality. European Polymer Journal, 2010. 46(5): p. 854-868. 10. Agarwal, R., P. Tandon and V.D. Gupta, Phonon dispersion in poly(dimethylsilane). Journal of Organometallic Chemistry, 2006. 691(13): p. 2902-2908. 11. Bistričić, L., V. Borjanović, L. Mikac and V. Dananić, Vibrational spectroscopic study of poly(dimethylsiloxane)-ZnO nanocomposites. Vibrational Spectroscopy, 2013. 68(0): p. 1-10. 12. Wong, E.J., Modeling and control of rapid cure in polydimethylsiloxane (PDMS) for microfluidic device applications, in Mechanical Engineering. 2010, Massachusetts Institute of Technology: Massachusetts Institute of Technology. p. 151. 13. Camino, G., S.M. Lomakin and M. Lazzari, Polydimethylsiloxane thermal degradation Part 1. Kinetic aspects. Polymer, 2001. 42(6): p. 2395-2402. 14. Camino, G., S.M. Lomakin and M. Lageard, Thermal polydimethylsiloxane degradation. Part 2. The degradation mechanisms. Polymer, 2002. 43(7): p. 2011-2015. 15. Chassé, W., M. Lang, J.-U. Sommer and K. Saalwächter, Cross-Link Density Estimation of PDMS Networks with Precise Consideration of Networks Defects. Macromolecules, 2011. 45(2): p. 899912. 16. Zhang, Y., C.J. Sheehan, J. Zhai, G. Zou, H. Luo, J. Xiong, Y.T. Zhu and Q.X. Jia, PolymerEmbedded Carbon Nanotube Ribbons for Stretchable Conductors. Advanced Materials, 2010. 22(28): p. 3027-3031. 17. Ci, L., J. Suhr, V. Pushparaj, X. Zhang and P.M. Ajayan, Continuous Carbon Nanotube Reinforced Composites. Nano Letters, 2008. 8(9): p. 2762-2766. 18. Liu, C.-X. and J.-W. Choi, Patterning conductive PDMS nanocomposite in an elastomer using microcontact printing. Journal of Micromechanics and Microengineering, 2009. 19(8): p. 085019. 7 19. Chao-Xuan, L. and C. Jin-Woo, Strain-Dependent Resistance of PDMS and Carbon Nanotubes Composite Microstructures. Nanotechnology, IEEE Transactions on, 2010. 9(5): p. 590-595. 20. Huang, Y.Y. and E.M. Terentjev, Tailoring the Electrical Properties of Carbon Nanotube– Polymer Composites. Advanced Functional Materials, 2010. 20(23): p. 4062-4068. 21. Cha, B., J. Yang and W. Hwang, Selective attachment of multi-walled carbon nanotubes on poly(dimethyl siloxane)substrates. Macromolecular Research, 2006. 14(6): p. 579-583. 22. Zhang, N., J. Xie, M. Guers and V.K. Varadan, Chemical bonding of multiwalled carbon nanotubes to polydimethylsiloxanes and modification of the photoinitiator system for microstereolithography processing. Smart Materials and Structures, 2004. 13(1): p. N1. 23. Xu, J., K.M. Razeeb and S. Roy, Thermal properties of single walled carbon nanotube-silicone nanocomposites. Journal of Polymer Science Part B: Polymer Physics, 2008. 46(17): p. 18451852. 24. Corporation, D.C. Sylgard(R) 184 Silicone Elastomer Kit Base. 2013 [cited 2014; MSDS]. 25. Corporation, D.C. Sylgard(R) 184 Silicone Elastomer Kit Curing Agent. 2014 16-01-2014 [cited 2014; MSDS]. 26. Dooher, T., Multi-walled carbon nanotube/high temperature polymer composites, in Faculty of Computing and Engineering. 2011, University of Ulster: UK. 27. Coleman, J.N., U. Khan, W.J. Blau and Y.K. Gun’ko, Small but strong: A review of the mechanical properties of carbon nanotube–polymer composites. Carbon, 2006. 44(9): p. 16241652. 28. Esteves, A.C.C., J. Brokken-Zijp, J. Lavèn, H.P. Huinink, N.J.W. Reuvers, M.P. Van and G. de With, Garnet particles effect on the cross-linking of PDMS and the network structures formed. Polymer, 2010. 51(1): p. 136-145. 29. Goyal, A., A. Kumar, P.K. Patra, S. Mahendra, S. Tabatabaei, P.J.J. Alvarez, G. John and P.M. Ajayan, In situ Synthesis of Metal Nanoparticle Embedded Free Standing Multifunctional PDMS Films. Macromolecular Rapid Communications, 2009. 30(13): p. 1116-1122. 8