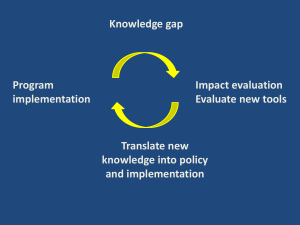
Strategic Case - Department for International Development
advertisement

Market-Shaping for Access to Safe, Effective and Affordable Health Commodities Business Case June2012 Contents Page Contents ................................................................................................................... 2 Acronyms ................................................................................................................. 3 Intervention Summary ............................................................................................. 4 What support will the UK provide? ......................................................................... 4 Why is UK support required?.................................................................................. 4 What are the expected results? .............................................................................. 5 Strategic Case .......................................................................................................... 7 A. Context and need for a DFID intervention .......................................................... 7 B. Impact and Outcome that we expect to achieve .............................................. 17 Appraisal Case ....................................................................................................... 19 A. What are the feasible options that address the need set out in the Strategic case? .................................................................................................................... 19 B. Assessing the strength of the evidence base for each feasible option ............. 22 C. What are the costs and benefits of each feasible option? ................................ 25 D. What measures can be used to assess Value for Money for the intervention? 28 E. Summary Value for Money Statement for the preferred option ........................ 30 Commercial Case ................................................................................................... 31 A. Why is the proposed funding mechanism/form of arrangement the right one for this intervention, with this development partner? .................................................. 31 B. Value for money through procurement ............................................................. 31 Financial Case ........................................................................................................ 33 A. What are the costs, how are they profiled and how will you ensure accurate forecasting? .......................................................................................................... 33 B. How will it be funded: capital/programme/admin? ............................................ 33 C. How will funds be paid out? ............................................................................. 33 D. What is the assessment of financial risk and fraud? ........................................ 34 E. How will expenditure be monitored, reported, and accounted for? .................. 34 Management Case ................................................................................................. 35 A. What are the Management Arrangements for implementing the intervention? 35 B. What are the risks and how these will be managed? ....................................... 37 C. What conditions apply (for financial aid only)? ................................................. 42 D. How will progress and results be monitored, measured and evaluated? ......... 42 Logframe .............................................................................................................. 43 Annex: Cost Benefit Analysis .............................................................................. 44 Endnotes ................................................................................................................ 58 Acronyms ACT API ART ATM cGMP CHAI CBA CYP DCVM D4T EID EMT EPs EFV FDC FDF FIND GAVI GAAP GDPP GFATM GHG GHI GoI HMG HPV IATT IRR LARC LCND LLIN LPV MAR MDR M&E MOH mRDT NMCP NPV PEPFAR PPPY POC PQ RDT RHSC RMNH RNTCP ROI SARPAM SCMS SLD SRA SRP TDF VfM VL ZDV Artemisinin-combination therapies Active pharmaceutical ingredient Anti-retroviral therapy Access to Medicine Current Good Manufacturing Practice Clinton Health Access Initiative Cost Benefit Analysis Couple Years Protection Developing countries vaccines manufacturers Stavudine Early infant diagnosis Executive Management Team Emerging Powers Efavirenz Fixed dose combination Final drug formulation Foundation for Innovative New Diagnostics Global Alliance for Vaccines and Immunisation Generally Accepted Accounting Principles Global Development Partnership Programme Global Fund for AIDS, TB and Malaria Greenhouse Gas Global health initiative Government of India Her Majesty’s Government Human Papillomavirus Inter-Agency Task Team Internal rate of return Long-acting reversible contraceptives Lower Cost New Diagnostics Long-lasting insecticide treated nets Lopinavir Multilateral Aid Review Multi-drug resistant Monitoring and evaluation Ministry of Health Malaria rapid diagnostic test National Malaria Control Programs Net present value (US) President’s Emergency Fund for AIDS Relief Per person per year Point of care Pre-qualified Rapid diagnostic test Reproductive Health Supplies Coalition Reproductive, Maternal and Newborn Health Revised National TB Control Programme Return on Investment South Asia Regional Programme for Access to Medicines Supply Chain Management Systems Second-line drugs Stringent Regulatory Authority Structural Reform Plan Tenofovir Value for Money Viral Load Zidovudine Intervention Summary What support will the UK provide? A range of DFID Departments will together contribute up to a maximum of £35m towards the implementation of an integrated, coordinated portfolio of supply- and demand-side market-shaping activities across a range of priority health commodities, such as antiretrovirals, antimalarials, diagnostics, vaccines, contraceptives and TB treatments. The portfoliowill be implemented by the Clinton Health Access Initiative (CHAI) supported by an accountable grant from DFID. Progress will be monitored by an oversight structure that will assess results and risks and provide strategic guidance. The structure will have at its apex anoversight committee, chaired at CEO level in CHAI and DG level in DFID, that will host a high-level, bi-annual meeting. Based on performance, the committee will discuss overall financing for the portfolio, including whether additional financial support is required. It will also discuss prioritisation of commodities and markets, including new and emerging opportunities. Other partners will be engaged to participate as necessary.Supporting this high-level discussion, will be a range of relevant policy-, region- and country-specific oversight groups that will track results, risks and discuss strategic direction and policies (particularly with regard to knowledge transfer and capacity-building). Why is UKsupport required? The Strategic Case sets out the importance of better access to safe, effective and affordable medicines, vaccines and other health commodities (such as diagnostics and contraceptives) for faster progress towards the health and other MDGs. While access to medicines has always been a constraining factor, there is a particular opportunity now becausenew research and innovative technologies are being harnessed to produce more effective and affordable health commodities, bettersuited to resource-constrained settings, for the prevention, diagnosis and treatment of a wide range of diseases and health conditions. Access to medicines is hindered by a range of market and government failures across the complex value chain that stretches from discovery, testing, approval, manufacture, regulation, procurement, distribution and delivery. These includepoor demand forecasting, weak procurement practices, limited incentives for manufacturers to invest in R&D and to scale up production, a lack of clear regulatory pathways for suppliers and many more. Donors (including the UK) and developing countries spend billions of dollars on health commodities each year. Coordinated market-shaping on both the supply- and demand-side, such as facilitating the entry of new generic suppliers, improving the process chemistry for priority compounds, aggregating demand and using pro-active procurement tactics, can transform the functioning of markets to deliver much lower prices, better quality products and greater security of supply, so allowing for greater availability of quality assured essential medicines for the poor. Such market-shaping interventions have contributed significantly to the 95% decline in prices for first-line antiretrovirals for HIV over the past decade, and to 80% decline for Malaria treatment prices1. The same has been proven for diagnostics: the Foundation for Innovative New Diagnostics (FIND) supported the development of GeneXpertMTB/Rif which is able to rapidly diagnose TB and give indicators of drug resistance and for which FIND has negotiated stepped price reductions for machines and commodities bought for developing countries based on volumes. This is projected to increase access to diagnosis and treatment for the poor and consequently to reduce transmission of the disease. DFID’s approach to market-shaping for health commodities rests on a number of principles: 1. Interventions should be undertaken with the full support and buy-in of developing country 2. 3. 4. 5. authorities, and in coordination with the relevant international stakeholders active in the respective disease or commodity area Supply- and demand- interventions need to be well coordinated and tailored to the specific features of individual health commodity markets, based on robust market assessments. Sustainability needs to be mainstreamed. This means working to build market-shaping capacity whether in developing country Ministries of Health or else in global partnerships and communities that seek to secure better market dynamics outcomes. The approach should focus supplier engagement on key ‘emerging power’ suppliers, such as India, China and South Africa, given their transformative market potential, as shown by experience in Anti-Retroviral Therapy (ART) and other commodities. Any market-shaping intervention should have sufficient scope to cover the priority health commodities in which DFID invests significant resources – both through bilateral and multilateral channels – as well as to complement the investments that DFID is making in new product research and development.. DFID has already invested in access to medicines interventions following the above principles through a grant to CHAI which, in conjunction with partners, has yielded substantial results in access to key commodities. Overall, the grant’s work on first- and second-line ART will secure over US$700m in savings on spend on HIV treatments from 2011-15. An independent review of the grant undertaken by HLSP states that; ‘DFID support has helped to improve the affordability, availability and quality for AIDS and Malaria drugs provided by Indian and other manufacturers, and increased capacity in African countries to access these drugs. This has been done through negotiating pricing, increasing competition through reducing entry barriers for new producers, facilitating reduction in costs through improving production processes, and improving demand forecasting and the flow of market information more generally. By helping to increase the number of people in poor countries who can access these drugs, DFID support has directly contributed to achievement of MDG targets on communicable disease and access to medicines.’ The UK has committed to increased investment in health commodities over the next few years – both bilaterally and through multilateral channels such as the Global Fund for AIDS, TB and Malaria (GFATM), the GAVI Alliance, UNITAID, and UNFPA– thus there are greater opportunities for UK intervention in this area. Intervention now will not only increase the value for money of UK investments in commodities, but will also result in sustainable price reductions for developing countries. What are the expected results? The intervention is expected to generate total global savings of US$1.5 billion by 2015, principally through lower prices for key health commodities. This figure is based on a highly conservative assumption of no global increase in access to key medicines. The total savings delivered rises to US$2.6 billion based on a more dynamic trajectory of global access. A key driver of the savings will be greater engagement of ‘emerging power’ pharmaceutical industries through 10 to 15 new priority health commodities, approved by a Stringent Regulatory Authority (SRA) originating from India, China and South Africa This will be sufficient to allow 2.3 - 2.8 million additional patients to be reached with safe and effective health commodities such as HIV, TB, and malaria treatments, as well as contraceptives. Additionally, savings on new diagnostic products will allow for an additional 550,000 additional patients to be tested for HIV, malaria, and TB. Key outputs include a 45% reduction in prices (from a 2012 baseline) for WHO-preferred HIV first-line and second-line HIV regimens, a 50% decrease in lowest price for quality assured MDR-TB treatment and up to a 30% reduction in the cost per test result for Point of Care CD4 tests to assess eligibility for HIV treatment initiation (over and above price reductions CHAI are expecting to gain through their programme with UNITAID)and two new suppliers of TB molecular tests at 50% of current prices. Additionally, the intervention will work towards a 35% decline in the prices of long-acting reversible contraceptives (LARCs), a 30% price reduction in at least two vaccines supported by GAVI, significant reductions in prices for malaria rapid diagnostic tests (mRDTs) and affordable prices for up to 5 more new malaria treatments (e.g. injectable artesunate and new Artemisinin-Combination Therapies (ACTs) e.g. DHAPQP and ASPY) The Appraisal Case presents a cost-benefit analysis and return on investment analysis of the proposed intervention. It finds that the Internal Rate of Return of the intervention is at least 228% (assuming no increase in global access over the project period) and 305% (based on a dynamic trajectory) through to 2020. Further, and very rarely for a DFID grant, the proposed intervention also has a positive return on investment. In other words, it more than pays for itself through savings on DFID’s spend on health commodities. UK savings on spend on ART through GFATM and UNITAID alone are enough to give the intervention aReturn on Investment (ROI) of at least 5% a year. Were spend through bilateral programmes and savings on vaccines and other commodities to be included the ROI would be much higher. While it is difficult to assess the impact of increased access to affordable quality assured drugs, it is clear that through increased access to medicines, this intervention will directly contribute to achieving MDGs 4, 5 and 6 (Child health, maternal health and HIV, Malaria and other communicable diseases) and to the essential drugs target of MDG 8 (Global partnerships). Further, since the UK’s investment in these commodities both reduces poverty (by enabling people to live productive lives and by reducing out of pocket spend on health), this intervention will indirectly contribute to MDG 1(Poverty reduction). Strategic Case A. Context and need for a DFID intervention The need and the opportunity Despite significant efforts and subsequent advances over the past few years, accelerated progress is needed if the health MDGs are to be reached. There are still substantial gaps in access to services, including: 10m people in developing countries who require anti-retroviral therapy for HIV but who do not receive it;2 215m women globally who want to delay or avoid pregnancy but do not have access to modern contraceptive methods;3 65% of children under 5 years in sub-Saharan Africa do not sleep under an insecticide treated net, and more than 75% of prescribed antimalarial drugs in many high malaria burden countriesare ineffective, rather than the more effective artemisinin combination therapies.4 Addressing these gaps in access is increasingly feasible because innovative technologies and new research are being harnessed to offer the prospect of more effective and affordable commodities. These opportunities are summarised below by disease area: HIV: International global health donors and developing country partnersenabled more than 6 million people to receive treatment by 2010. Recent advances in HIV prevention may result in a declining incidence of HIV infection. A recent study demonstrated that ART is 96% effective in preventing HIV transmission in serodiscordant couples, emphasizing the potential benefits of diagnosing and treating patients as early as possible5. However, modelled scenarios of resource need demonstrate the unsustainability of scaling up ART at current costs (for both drugs and delivery) given the current context limited domestic funds and declining external resources. If efficiencies can be made, however, using improved testing technologies and optimising delivery channels to treat more patients, significant progress could be achieved toward a reduction of both new infections and mortality among infected patients. TB:The advent of easy to use, potentially affordable tests to diagnose drug sensitive and multi-drug resistant (MDR) TB has raised the potential for significant advances in the treatment and hence reduced transmission of TB. Shortened regimens are currently being trialled for both drug sensitive and drug resistant TB; the former may reduce the incidence of MDR TB through improved treatment adherence. MDR TB is a largely unaddressed public health problem, with less than 5% of incident cases being treated in quality assured programmes6. Improvements will only be possible if the costs of testing and treating drug sensitive and MDR TB can be reduced and the products made more accessible to local markets. Malaria:A significantly improved treatment – Artesunate - has been identified for severe malaria, with a 22.5-34.7% relative risk reduction in mortality over alternatives78. Furthermore, new co-formulated ACT products (e.g., DHA-PPQ, AS-PY) and rapid diagnostics have recently been introduced for the treatment of uncomplicated malaria; others are in the pipeline. These tools show potential for a significant reduction in mortality, particularly among children, and lay the foundation to halt the development and spread of drug resistant malaria globally. Vaccines: A number of new and increasingly sophisticated vaccines became available in the last decade—including the pneumococcal conjugate vaccine, the human papillomavirus (HPV) vaccine and the rotavirus vaccine. In coming years, new vaccines (for example, dengue, hookworm, leishmaniasis, malaria and improved TB) may also become available. Accelerating the global rollout for these vaccines offers the potential for huge impact on child mortality rates. Contraceptives:While long lasting contraceptives are considered more clinically effective compared to other contraceptive methods, uptake has been slow due in large part to its high cost. Similar gains as seen in ART markets are anticipated if a competitive sustainable market can be fostered. This requires not only greater volumes of products at lower costs, generated through a competitive generic market, but also products of internationally accepted quality. Opportunities in other reproductive health commodities, such as female condoms, will also be explored. While these examples have the potential to substantially reduce the burden of disease and accelerate progress towards the MDGs, action needs to be taken to ensure that potential is realised. Experience highlights the risk that market and systems bottlenecks will stand in the way of rapid progress. This risk is compounded by the challenging funding situation in global health (with recent developments at the Global Fund for AIDS, TB and Malaria (GFATM) being one of the more widely known and documented) that will threaten the finance available for health commodities. This situation risks an erosion of the advances made in access to treatment over the past several years and limits scope for scale-up. The challenge The challenge of making these new therapies widely accessible at affordable prices is highlycomplex. Effective access to medicines depends on a complex value chain that stretches from discovery, testing, approval, manufacture, regulation, procurement, distribution and delivery before reaching appropriate use. As the figure below shows, there are a number of characteristics that can hinder or prevent the evolution of mature markets. These include: weak domestic purchasing power, and unpredictable and fragmented donor financing poor demand quantification at country level, and poor aggregation of demand at regional and international levels poor product selection (often leading to highly fragmented product portfolios) weak procurement practices, with limited pooling or coordination across purchases and purchasers limited availability and use of market information to inform buying uncertain routes to market for new products or suppliers (e.g. lack of clear regulatory pathways) weak incentives for manufacturers to invest in current Good Manufacturing Practice (cGMP) compliant facilities high barriers to market entrance for some products, compounded by uncertain demand and high market development risks Discover Develop & Test • Low commercial incentives • Limited product development expertise in public sector • Limited R&D/trial capacity in developing countries • Limited ID and Dev Country expertise in private sector Approve & Register Scale-up & Manufacture • Poor demand data • IP challenges • Limited capacity for some products • Regulatory • Unclear pathways • Limited harmonisation • Very limited capacity at country level – approval, quality, pharmacovigilence Procure Distribute Prescribe & Dispense • Weak logistics, infrastructure and information • Poor coverage • Leakage • Mark-ups • Inefficiencies • Low & variable financing • Multiple channels • Poor quantification • Poor use of pricing info • Non-transparent Demand & Use • Affordability/ lack of social financing • Limited access • Social barriers • Poor information • Low health literacy • Poor adherence • EMLs/STGs • Limited HR • Informal sector • Limited regulation • Unethical promotion • Poor practice Source: DFID Access to Medicines framework Increasing coverage and appropriate use of commodities therefore requires a combination of complementary interventions if impact is to be optimised. The value chain will only be as strong as its weakest link. For example, the benefits of investment in new product development will be severely compromised if market barriers limit manufacturing scale-up. Equally, the value of health systems strengthening will be reduced if the supply of safe, affordable health commodities is inadequate. Feasibility of intervention in market-shaping and rationale for UK support Health commodities account for a large proportion of global health spending. For example, around 60% of GFATM expenditure and 65% -75% of Global Alliance for Vaccines and Immunisation (GAVI) expenditure is on commodities. As a large contributor to these and other multilateral and bilateral partnerships, the UK’s investment in health commodities is significant. These contributions to the demand side, through better demand forecasting and assurances of purchase, help to strengthen commodity markets 9 by attracting new manufacturing entrants and production scale-up. For example, over the 2001-2010 period, GAVI (in which DFID accounts for about a third of total financing) increased its supply base from five manufacturers to thirteen, including seven based in emerging markets.10 The emerging powers are well placed to contribute to more effective utilisation of increased global health spending, through their growing pharmaceutical industries. Healthcare companies from emerging powers – particularly India and China – have played an important role in reducing prices and improving access to vaccines, contraceptives, HIV/AIDS and malaria medicines for millions of people worldwide. For example the Indian Serum Institute was involved in developing a new vaccine - the MedAfriVac A meningitis vaccine - designed specifically for Africa’s meningitis affected countries. Emerging powers are increasingly investing in research and development capacity to identify new health technologies. There is consensus that complementary interventions in shaping the structure and dynamics of markets can secure much greater gains. For example, for first-line treatments for HIV, GFATM’s willingness to fund the purchase of generic medicines, the establishment of the WHO PreQualification Programme, together with engagement of largely-Indian generic manufacturers to produce new fixed-dose combination (FDCs) formulations contributed synergistically to the price per person per year for first line combination therapy falling by over 95%11,12 It is unlikely that any one of the interventions would have succeeded alone; GFATM’s willingness to procure was dependent on a cheaper supply of quality assured drugs, while suppliers willingness to invest in manufacturing was dependent on a firmer market The chart below illustrates the evolution of first-line ART prices over the last decade. Global Health Initiatives (GHIs) are developing strategies to move from a relatively passive “market taking” position (ensuring efficient procurement within the current market structure) to an active “market shaping” approach. In May 2011 the 23rd GFATM board meeting approved the recommendations of the Market Dynamics Committee, which identified potential to avert costs upward of $250m over 5 years, support better treatment outcomes and ensure commodity security for high-risk products through strategic engagement in ART markets.1314 GAVI has approved a new Supply and Procurement Strategy to seize opportunities for more active engagement in vaccine markets.15In addition, UNITAID was established in 2006 with the specific remit to use market interventions to improve access to treatment for HIV/AIDS, malaria and tuberculosis in developing countries. UNITAID plays a unique role for these diseases by identifying specific market shortcomings in the area of diagnosis, prevention and treatment. However, what is clear is that successful market intervention cannot be secured by Global Health Initiatives (GHIs) alone, as demonstrated by the ART example above. And indeed, many opportunities for increased value for money gains through more active and strategic engagement have been missed (Grace 2010).16 A number of the drivers of poor market functioning lie outside of the remit or reach of such agencies. For example, while the GFATM made clear that it would fund the purchase of high-quality ART, it was reliant on the WHO Pre-Qualification project to approve drugs. And while WHO can pre-qualify products, it can’t provide direct technical assistance to potential manufacturers to build capacity to meet regulatory requirements (i.e. reduce market entry barriers). Source: MSF (2011) Untangling the Web of Antiretroviral Prices The story of the market entry of generic Tenofovir (TDF) illustrates the way that market-shaping interventions can add significant value. In 2007, most first-line ART comprised zidovudine (ZDV) and stavudine (d4T) based regimens. In 2009 TDF became the preferred first line regimen in the WHO Guidelines on the basis of lower toxicity, a better side effect profile and better efficacy (less resistance). At that time, the Indian company, Matrix Laboratories (now Mylan Laboratories), was the only generic producer to make Tenofovir (TDF), which raised concerns among countries about price and supply security. Physicians in developing countries tended to be comfortable with the status quo. CHAI, with its grant from DFID, worked on the demand side to assure countries of TDF’s therapeutic superiority and that Matrix supply was of good quality. They worked on the supply side to assure Matrix of sufficient demand from countries to expand production capacity and with other suppliers to enter the market. Additionally, since 60-70% of the cost of ART is in the Active Pharmaceutical Ingredient (API), CHAI also undertook work on process chemistry to reduce the cost of production and to facilitate entry of new API and raw material suppliers into the market. The impact of this engagement was to bring down the price for the leading Tenofovir-based regimen by over a third saving several hundred million dollars. The chart below illustrates the evolution of prices and generic suppliers of TDF. Source: MSF (2011) Untangling the Web of Antiretroviral Prices Even where progress has been rapid, such as in TDF market interventions, prices remain high in relation to LIC health budgets and there is a significant challenge in rolling out Fixed Dose Combinations (FDCs). Furthermore, progress with the market evolution for other product categories – such as long-lasting insecticide treated nets (LLINs), MDR TB drugs, quality assured hormonal contraceptives and some vaccines – has been less dramatic than for first-and second-line ART. For these reasons, market-shaping forms a central part of the UK’s approach to access to medicines (ATM). The chart below illustrates how it fits into DFID’s overall portfolio of ATM activities – that seeks to promote innovation for new products, strengthen the enabling environment for the scale-up of manufacturing, procurement and registration and strengthen the capacity of developing country health systems to deliver safe, affordable and effective medicines and other health commodities to their own populations. Discover Develop & Test Approve & Register Scale-up & Manufacture Procure Distribute Prescribe & Dispense Demand & Use Push Investment – e.g. PDPs Innovation Pull Mechanisms e.g. AMC Regulatory Paths and Capacity e.g AMRH TRIPS Flexibilities/Patent Pools/IP – Enabling Environment Global Funds and Market dynamics/engagement - e.g. UNITAID, CHAI, GFATM, GAVI Industry Good Practice - e.g. IGFAM Affordable Medicines Facility Malaria Southern Africa Regional Programme on Access to Medicines Health Systems Medicines Transparency Alliance Bilateral Country Programmes Source: DFID Access to Medicines framework Theory of change The theory of change that guides DFID’s approach to market-shaping is that interventions on the supply- and demand-side can complement other access to medicines interventions and act as a powerful ‘accelerator’ to enhance access to safer, more effective and more affordable health commodities. Thus inputs (investments in the market shaping process) lead to outputs (market shaping interventions), lead to outcomes (global savings through reduced medicines prices and improved availability of essential, quality assured medicines through, for example, engaging emerging market suppliers) lead to impact (improved health outcomes and contributions to achieving the MDGs by, inter alia, at least 2.3 million additional patients receiving effective health commodities) Examples of market-shaping interventions in the theory of change are set out in the diagram. Supplyside activities include: A. Optimising sourcing of API and other raw materials B. Facilitating the entry of new suppliers (of both raw materials and finished product) C. Working to find ways to improve production process chemistry – of both API, finished products, formulations, etc. D. Working to facilitate regulatory approvals (either internationally – US FDA, EMA, WHO PQ or else, crucially, registration within developing countries17) E. Optimising product design (e.g. formulations, tailoring for specific markets, like paediatrics) F. Negotiating prices with suppliers based on anticipated demand On the demand-side, examples of activities with developing countries, normative agencies and Global Health Initiatives include: G. Working to secure the inclusion of commodities in international (e.g. WHO) guidelines H. Producing more robust forecasts of demand (including building developing country capacity in I. this area Using innovative financial approaches (such as volume guarantees) to secure better outcomes J. Using the range of pro-active procurement tactics (at both the level of GHIs and developing countries), such as market consolidation through product selection. These market-shaping activities can be complemented by a range of complementary in-country access interventions - the objective of which is not principally market-shaping - but which contribute indirectly to healthier commodity markets. Examples include: K. L. M. N. Strengthening of supply chains Activities to support new product introduction into health systems Health worker training Supporting shifts in the prescribing behaviour of physicians MARKET-SHAPING INTERVENTIONS COMPLEMENTARY IN-COUNTRY ACCESS INTERVENTIONS DEMAND-SIDE SUPPLY-SIDE SUPPLY AND DEMAND-SIDE: INTERVENTIONS COORDINATED AND PRIORITISED A B SUPPLIERS OF ACTIVE PHARMACEUTICAL INGREDIENTS (API), RAW MATERIALS AND CHEMICALS GLOBAL HEALTH PARTNERSHIPS D GENERIC SUPPLIERS C ORIGINATOR SUPPLIERS E OTHER DONORS F H J DEVELOPING COUNTRIES I REGULATORY AND STANDARD-SETTING G AGENCIES AND FRAMEWORKS K M HEALTH SYSTEMS L 2 N LESSONS LEARNED SHARED AT ALL LEVELS A B C Optimise Sourcing Facilitate New Entrants Improve Process Chemistry D E F Facilitate Regulatory Approvals Optimise Product Design Negotiate Prices G H International Guidelines Inclusions I J Demand forecasting Guaranteed volumes Modify Procurement K S/C management L Product introduction M Health worker training N Prescribing behaviour • Coordinated market-shaping leads to more suppliers, affordable and sustainable prices, supply security, better quality standards, greater country capacity to forecast, procure and adopt. • This results in improved access to safe, affordable and effective commodities and improved health outcomes. This comprehensive, integrated market-shaping approach isunderpinned by a number of principles: Firstly, interventions should be undertaken with full support and buy-in of developing country authorities, and in coordination with the relevant international stakeholder partnerships active in the respective disease or commodity area Second, supply- and demand- interventions need to be well coordinated and tailored to the specific features of individual health commodity markets – based on robust market assessments. Third, sustainability needs to be mainstreamed into all work. This means working to build marketshaping capacity – whether in developing country Ministries of Health or in global partnerships and communities that seek to secure better market dynamics outcomes. Fourth, the approach should focus supplier engagement on key ‘emerging power’ suppliers, such as India, China and South Africa, given their transformative market potential, as shown by experience in ART and other commodities. Fifth, interventions should complement other interventions that DFID supports in market-shaping and access to medicines. This includes investments in multilaterals such as UNITAID, GFATM and GAVI, as well as in-country programmes such as the South Asia Regional Programme for Access to Medicine (SARPAM). Finally, any market-shaping intervention should have sufficient scope to cover the priority health commodities in which DFID invests significant resources – both through bilateral and multilateral channels – as well as offer the potential to seize opportunities for other commodities as they emerge. DFID’s grant to the Clinton Health Access Initiative (CHAI) that began in 2008 has adopted this integrated, market-shaping model. Through work across both sides of the market, this grant, in conjunction with partners, has yielded substantial results in access to key commodities for HIV, TB and Malaria. These results have been documented and validated in an independent review undertaken by HLSP. The evaluation states that: ‘DFID support has helped to improve the affordability, availability and quality for AIDS and Malaria drugs provided by Indian and other manufacturers, and increased capacity in African countries to access these drugs. This has been done through negotiating pricing, increasing competition through reducing entry barriers for new producers, facilitating reduction in costs through improving production processes, and improving demand forecasting and the flow of market information more generally. By helping to increase the number of people in poor countries who can access these drugs, DFID support has directly contributed to achievement of MDG targets on communicable disease and access to medicines.’ Results achieved by the existing CHAI grant include, but are not limited to: CHAI was instrumental in bringing about the reduction of Lopinavir/ritonavir (LPV/r) from $600 per patient per year (pppy) to $400 pppy, a reduction of 33%, and moving the market away from a single supply situation. CHAI’supstream work to reduce the cost of production can be directly linked to the 33% price reduction in leading Tenofovir-based first-line ART regimens. Overall, the grant’s work on first- and second-line ART will secure over US$700m in savings on spend on HIV treatments from 2011-15. CHAI’s work in South Africa was one factor contributing to the expected savings of US $250300 million for South Africa in 2011 alone. CHAI contributed to the increase in the number of pre-qualified artemisinin-combination therapies (ACT) producers going from 2 to 6. The independent assessment also stresses the importance of the integrated, supply- and demandside model: ‘CHAI’s supply side work is often seen as the primary catalyst of cost savings, the denominator in the Value for Money (VfM) equation of economy, efficiency and effectiveness. However, CHAI’sdemand side work is also essential from a VfM perspective, for two reasons: i) making the demand side less opaque is often the means by which to leverage supply side changes, especially for nascent or rapidly scaling up markets, and ii) demand side work is needed to ensure that quality, low cost medicines and health technologies are used appropriately by prescribers and patients; and only through appropriate use will the health impact be seen.’ Demand-side activities have included supporting development of national treatment guidelines, supporting country forecasting and procurement planning, helping to expedite commodity registration, supporting new product introduction and strengthening supply chain management. The independent evaluation notes several positive examples of CHAI having helped to build capacity in this area noting that, ‘throughout its activities, CHAI has emphasised building capacity of local partners. The statement of “they sit with us” was mentioned by many of the government officials in both countries to describe the way CHAI works’. However, it also notes that, ‘there is some indication that CHAI’s result-oriented approach may have compromised capacity-building efforts.’ Based on this last point, the evaluation recommends more even and pro-active oversight of capacity-building activities in any future partnership. Alignment with UK Strategic Priorities The proposed programme maps closely to a range of coalition government development policy objectives. First of all, inadequate and over-priced access to medicines is an important driver of poverty through loss of productivity while increasing household expenditures. Out-of-pocket spending on health, including medicines, often drives or keeps households below the poverty line in lowincome countries. The 2011 MDG Gap Taskforce report18 found that in the public sector, generic medicines are only available in 38% of facilities, and on average cost 250% more than the international reference price. In the private sector, those same medicines are available in 63.3% of facilities, but cost on average about 610% more than the international reference price. High prices often render medicines unaffordable, with common treatment regimens costing a low-paid government worker several days' wages. More specifically, the results will contribute to a number of DFID objectives as set out below: In terms of the focal areas in DFID’s business plan: International commitments: Increasing reliable access to affordable quality health commodities is a necessary component to meeting the health MDGs (SRP 1.2). Access to medicines is an MDG indicator itself (Target 8.E: In cooperation with pharmaceutical companies, provide access to affordable essential medicines in developing countries. Access to medicines is also a necessary condition of progress on the health MDGs (MDGs 4, 5 and 6). Value for money: The focus of this project is to increase value for money in global health spending through increased transparency (SRP 2.0), strengthened accountability for access to health commodity results (SRP 2.4) and reduced prices for key essential commodities Wealth creation: The programme focuses on improving the competitiveness of pharmaceutical markets, and increasing the engagement of the private sector, including through appropriate market entry for quality assured suppliers from emerging powers into developing countries Role of women: Improved access to medicines is essential to improving reproductive, maternal and neonatal health.19Since women are the main caregivers in most societies, increasing the availability of lower cost medicines will save them time (since drugs should become increasingly available at lower level facilities) and money. The programme will specifically support work to improve the function of markets for contraceptive commodities and has the potential to expand focus to other reproductive and maternal health commodities. Additionally, this programme contributes to UK’s Emerging Powers (EPs) agenda as set out in the Global Development Partnership Programme (GDPP) approach and strategy. Through engagement with pharmaceutical companies in China, India and other countries as well as through the positive regional impact of strengthened procurement and regulation in countries like South Africa, there will be positive development impact in DFID priority countries and positive global impact in terms of healthier markets for health commodities. Improving value for money, results focus and transparency of multilateral spend were primary objectives of DFID’s Multilateral Aid Review (MAR). Securing improvements in spend and market shaping for health commodities, in particular through our contributions to GFATM, GAVI, UNITAID, UNFPA and other agencies, were identified as priority areas for attention. . DFID’s Framework for Results for Reproductive, Maternal and Newborn Health (RMNH) states that ‘the UK will invest in continued innovation in products and support stronger procurement and supply chains to prevent stock outs and increase availability of essential commodities (e.g. contraceptives, medicines and supplies for maternity care). Increasing global and national market efficiencies make quality products cheaper to developing countries and increase value for money.’ It further commits that ‘action at the international level will improve value for money for reproductive health commodities. The UK will seek improved market efficiencies for the supply of quality and affordable contraceptives, contributing to an estimated cost savings of £20 million a year.’ The Malaria Framework for Results states that ‘active engagement in markets – by improving demand forecasting, working directly with suppliers to improve production processes and accelerate market entry or by coordinating procurement – could achieve significant improvements in value for money (better prices, supplier performance, product specification and market security.’ Finally, the Operational Plan of DFID’s Human Development Department commits to global support for access to medicines to generate costs savings for key global health commodities. Specific targets set out are global savings of: (i) over £20m p.a. for contraceptive implants and injectables, equating to over 2 million pregnancies avoided and (ii) $140m for first line anti-retroviral regimens and $100m for second line. B. Impact and Outcome that we expect to achieve Impact: The savings generated through the interventions are sufficient to allow 2.3 - 2.8 million additional patients to be reached with safe and effective health commodities such as HIV, TB, and malaria treatments, as well as contraceptives. Additionally, savings on new diagnostic products would allow for an additional 550,000 additional patients to be tested for HIV, malaria, and TB. Outcome: Outcomes are in two key areas; (i) global savings on total spend on medicines (in countries with access to generic prices) and (ii) engagement of Chinese, Indian and South African pharmaceutical industry in producing high-quality health commodities for developing countries. More specifically, the outcomes are: (i) Net Present Value (NPV) of total global savings generated by the interventions planned through to 2015 of US$1.45 billion by 2020 (based on the highly conservative assumption of constant global access to key medicines). The NPV of savings rises to $2.53 billion (based on baseline assumptions around the trajectory of global access). (ii) Between 10 to 15 new products, approved by a Stringent Regulatory Authority (SRA) originating from India, China and South Africa These results will be underpinned by a number of outputs to be delivered by 2015: Optimal drugs for HIV and TB: 45% reduction in prices for WHO-preferred HIV first-line and second-line HIV regimens, including specific focus on prices in priority hyper-endemic countries in Southern Africa Up to 50% decrease in lowest price for quality assured MDR-TB treatment Three additional SRA assured HIV and MDR-TB products supplied from Indian, Chinese and RSA sources Lower-priced, high-quality diagnostics: Up to 30% reductions in the cost per test result received (over and above price reductions through UNITAID interventions) for at least 2-3 Point of Care CD4 products to test for HIV treatment initiation Scale up of usage of Point of Care (POC) testing for HIV in priority hyper-endemic countries in Southern Africa Increase in the number of suppliers of effective TB molecular tests distributing tests through the private sector at affordable prices 3 new SRA-approved POCdiagnostic products from China or India Lower-priced high-quality contraceptives and vaccines: Reduction of more than 35% in the prices of long-acting reversible contraceptives (LARCs) in low and middle income countries A 30% price reduction in at least two vaccines supported by GAVI (including at least one from an Indian or Chinese manufacturer). Increased availability and supply of malaria diagnostics and treatments: Significant reduction in prices for malaria Rapid Diagnostic Tests (RDTs) and up to 5 more new malaria treatments (e.g. injectable artesunate and new ACT products e.g. DHAPQP and ASPY) from Indian and Chinese manufacturers submitted to a Stringent Regulatory Authority (SRA). These results are the outcome of outputs and activities with both producers and countries where the drugs and commodities will be used. On the country side more robust demand forecasts, stronger regulatory pathways, and improved product selection will send signals to producers to facilitate price reductions, registration of products for regulatory approval, and investment in production. This balance of supply and demand side will be monitored through agreed country- and policy-specific project oversight arrangements. These arrangements will include monitoring of other crucial outcomes, such as capacity-building.An appropriate capacity-building indicator will also be developed as part of the ongoing monitoring and evaluation process. Recognising the technical nature of the issues, as well as the challenges of better understanding complex market dynamics and attributing impact to different interventions, an independent entity will be retained to support both monitoring of progress and to provide a rigorous approach to evaluation and lesson-learning. Appraisal Case A. What are the feasible options that address the need set out in the Strategic case? The previous section set out the strategic case for DFID support for market shaping interventions to enhance access to safer, more effective and more affordable health commodities. It also set out a number of principles – such as country ownership, collaboration with global partners and programmes, coordination between supply- and demand-side activities, ‘emerging power’ engagement, tailoring to individual commodity market conditions and mainstreaming of capacitybuilding – that are fundamental to success. This section appraises options for delivering those interventions. It also assesses the costs and benefits of the ‘do-nothing’ counterfactual. The following three options have been identified. They are set out, together with an assessment of their feasibility below: Option 1. The ‘no intervention’ counterfactual: Under this option, no market-shaping intervention would be undertaken to complement UK investment in health commodities through multilateral and bilateral channels. Is this option feasible? This is a feasible option and will be appraised in the cost-benefit analysis section below. Option 2. Deliver individual commodity-specific supply- and demand-side interventions through different organisations with specialist policy or in-country experience. All interventions described in the portfolio would be carried out by one or more organisations with relevant expertise. A mechanism would be put in place to coordinate and communicate across different implementing entities. DFID would manage the different set of activities through individual contracts. Is this option feasible?: Market-shaping is a highly specialist and emerging area. There are insufficient organisations with the specialist skills in the various commodities to make this option implementable (although it is an objective of DFID’s market-shaping engagement to develop the market-shaping landscape in this direction). Further, the capacity requirements and costs associated with putting in placean effective mechanism to meet the principle of supply-demand coordination are highly significant. It would also imply significant additional programme management capacity within DFID to let and manage the different contracts. For these reasons, Option 2 is not considered a feasible option to implement this intervention. Option 3. Deliver supply- and demand-side interventions across the commodity range in an integrated manner through a single market-shaping grant to the Clinton Health Access Initiative (CHAI). Feasibility: CHAI has the experience, technical capacity, presence on the supply- and demandside, track-record of results, commitment and shared objectives necessary to implement the proposed intervention. CHAI’s capacity to implement the individual elements of the proposed portfolio has been reviewed by an independent consultant and by DFID policy leads and incountry health advisers.Competency in key output areas is evidenced as follows: HIV drugs – As demonstrated through their work on the previous DFID grant, CHAI has relied on a network of collaborations and partnerships to progress HIV drug access, including working extensively with the Indian generic industry. They worked in partnership with the Gates Foundation, UNAIDS, WHO, and others in developing a set of drug and drug optimization priorities that have become the basis for global drug optimization research and development and for the UNAIDS Treatment 2.0 Drug optimization agenda. CHAI forged collaborations with major drug procurers and funders including country governments, bilateral funders, implementers and GHIs to coordinate procurement and drug selection opportunities to maximize impact. One example is the creation of a concerted paediatric ART procurement system with the Inter-Agency Task Team (IATT), GFATM, Supply Chain Management System (SCMS), and UNITAID to compensate for possible market failures associated with a transition from the UNITAID program to one funded by Global Fund. On the demand-side, CHAI continues to leverage its strong incountry presence as a convenor of implementing partners to facilitate decisions and transitions to better drug regimens in country. HIV spending efficiency – CHAI has worked with the support of UNAIDS and the Gates Foundation, at the specific request of governments in several countries to analyse national spending for HIV treatment. UNAIDS would like to expand this collaboration to advance the Treatment 2.0 agenda into additional government programmes, upon request. Malaria – In Malaria, CHAI actively collaborates with the WHO, GFATM, African Leaders Malaria Alliance, Medicines for Malaria Venture, and Zagaya (Amyris’ non-profit arm). CHAI is also an active member of the WHO led ACT Taskforce where they work with John Snow International, the President’s Malaria Initiative (US), and UNICEF. CHAI also works with malaria drug suppliers; where their partnerships parallel the partnerships with HIV drug suppliers in nature and strength. TB drugs – CHAI’s work to improve the price and supply security for high quality MDR-TB drugs relies on a network of key partnerships. CHAI has been and will continue to work in close collaboration with the Global Drug Facility and Stop TB department at WHO, the Bill and Melinda Gates Foundation, UNITAID, WHO-SEARO, India’s National TB Treatment Program, and other stakeholders to pursue a range of market shaping efforts which include supporting and growing volumes, expediting regulatory approvals, improving supplier selection, improving forecasting and visibility and supporting accelerated case finding. Simultaneously, CHAI has engaged with key suppliers on the demand side by leveraging existing relationships with ART manufactures who also serve the MDR-TB drugs market, and continues to build newer associations with final drug formulation (FDF) and active pharmaceutical ingredient (API) manufacturers in India and China. Vaccines – CHAI’s vaccine pricing work is built upon foundational partnerships with key stakeholders such as the Gates Foundation, GAVI and UNICEF. CHAI has worked in close collaboration with these organizations across a range of initiatives, which, inter alia, include negotiating lower prices of rotavirus vaccine and accelerated introduction of pneumococcal vaccine in Ethiopia. CHAI has been engaging with major multinational as well as Developing Countries Vaccines Manufacturers (DCVMs), and, would work towards building and strengthening newer association with others. Diagnostics – The diagnostics component of this intervention will be implemented in phases: technology screening, product design and development, test evaluation, approval by normative bodies, in-country pilots, in-country registrations, and product uptake. At each of the stages, CHAI will engage with partners who have the necessary skills and experience. The list of partners is evolving; currently, partners include: PATH (project design and evaluation); FIND (product design); WHO and CDC (approval by normative bodies); Ministries of Health (in-country pilots, registrations and product uptake); and the US President’s Emergency Fund for AIDS Relief (PEPFAR), GFATM and USAID(for product uptake). Over the last five years, CHAI has developed a relationship with most of these bodies that they can leverage for this project. Contraceptives – DFID, USAID, and UNFPA support the Reproductive Health Supplies Coalition (RHSC), a global partnership of public, private, and non-governmental organizations to bring together diverse agencies and groups with critical roles in providing contraceptives and other reproductive health supplies. These include multilateral and bilateral organizations, private foundations, governments, civil society, and private-sector representatives. DFID and RHSC have requested that CHAI be involved in developing RHSC’s capacity to use market shaping approaches similar to CHAI’s approach in the HIV space to increase RHSC’s capacity to execute these strategies independently. This work would coordinate closely with the Concept Foundation which has been retained by the RHSC to improve the quality of RH commodities. Option 3 will therefore be appraised in the cost-benefit analysis section below. Option 4. Deliver supply- and demand-side interventions across the commodity range in an integrated manner through a single market-shaping grant to a multilateral, or other, agency. Feasibility: For a range of other health issues, such as health financing, health workers, health metrics, aid effectiveness for health and many more, multilateral agencies – frequently the WHO – or partnerships take a global leadership role and have significant implementation capacity. In the market-shaping space, the need for direct engagement with pharmaceutical companies rules outdirect engagement of normative agencies like the WHO on conflict of interest grounds. UNITAID was established in 2006 to drive access to diagnostics and medicines for HIV, TB and Malaria through specific market-shaping interventions. It is a financing, rather than an implementing, entity so it also delivers projects through implementing partners, such as CHAI, if selected for funding. However, in this case UNITAID could not support a broad-based portfolio such as the one proposed due to the boundaries of its mandate – for example, it does not cover immunisation or reproductive health. While several multilaterals, such as GAVI and GFATM, have taken steps to more pro-actively shape markets in recent years and build their internal capacity, they do not have (and should not have) the mandate, capacity or reach to implement this sort of portfolio of activities. Following analysis and consultation with key partners and stakeholders active in this area, it is assessed that there is no other organisation than CHAI able to execute a comprehensive, integrated portfolio such as that proposed effectively. This option is therefore not considered to be a feasible approach to implementing this intervention. B. Assessing the strength of the evidence base for each feasible option The quality of evidence for each of the three options is rated in the table below: Option Evidence rating There is strong evidence, based on analysis and evaluations, that a 1. The “do passive approach by the global health community to commodity nothing” markets leads to missed opportunities and sub-optimal outcomes in counterfactua terms of prices, supply security and access. Comparative analysis l can be made across different disease and commodity areas for varying market-shaping efforts. There is strong evidence (including from the evaluation undertaken 3. Deliver of the existing DFID grant) that CHAI interventions have been supply- and effective in accelerating access to safe, effective and affordable demand-side health commodities through activities such as supplier engagement, interventions price negotiations and enhanced demand forecasting. through CHAI Climate and Environment Appraisal A climate and environment appraisal of each of the two feasible options was undertaken. This included analysis against the criteria set out below.A summary of the results of this analysis is provided in the following section that identifies expected negative impacts and positive impacts/opportunities. Feasibility Criteria 1 2 3 Description Weighting Integrates environmental and climate change issues as part of market shaping activities. Promotes good environmental management and minimises environmental impacts throughout the value chain of health commodities. Raises awareness of the impacts of climate and environmental change on infectious disease transmission 2 3 3 Note: each CSC is weighted 1 to 5, where 1 is least important and 5 is most important based on the relative importance of each criterion to the success of the intervention. Option 1 – The ‘no intervention’ counterfactual Negative impacts: without market shaping to complement UK investment in health commodities there is a high risk that opportunities will be missed in terms of improving prices, supply and access. This will reduce secondary impacts including improved health and increased wealth and further limit indirect benefits such as a reduction in vulnerability to climate change and better environmental management. Furthermore, opportunities will be missed to help: 1) raise environmental standards and management within the pharmaceutical sector and the whole value chain; and 2) raise awareness of climate and environmental change impacts on health and susceptibility to infectious diseases. Positive impacts/benefits: few tangible benefits of the counterfactual were identified and insufficient information is available to assess the potential benefit of environmentally poor suppliers not entering the market. Option 3 – Deliver supply-side and demand-side interventions across the commodity range in an integrated manner through a single market-shaping grant to CHAI a)Impact of intervention on the climate and environment The intervention is likely to be associated with a number of direct and indirect negative impacts on climate change, environmental degradation and community resilience. These include the following. Increase in greenhouse gas (GHG) emissions. These may result fromoffice based activities and increased travel and transport. To mitigate these CHAI seeks to minimise this through its working practices. GHG emissions already arise from transport of health commodities and storage of vaccines, rapid diagnostic kits and drugs, and may increase as more commodities become available. Insufficient information is available to fully assess the incremental impact of this intervention, but the implementation of environmental policies will help reduce these impacts. Research, development and manufacturing impactson the environment. Facilitating the entry of new suppliers (of raw materials and finished product) from emerging powers (e.g. China, India) as part of supply-side market shaping activities carries the potential risk of increasing environmental impacts. These include air and water pollution; noise;pharmaceutical manufacturing and packaging waste production (including biosafety hazards); and resource use (e.g. energy and water) from pharmaceutical plants. These will be significant in countries where environmental regulations are in their infancy and/or poorly enforced. Similarly, the pharmaceutical industry is associated with low manufacturing efficiency, in part due to the complex chemistries required to make medicines. It is often resource intensive and waste output is high,20 although this is something the project seeks to change. Insufficient information is available to appropriately assess the significance of these impacts in part given the different locations of the new suppliers. However, this impact can be reduced by ensuring, as part of the projects due diligence that suppliers have certified environmental management systems in place that meet national and international standards. The intervention has a number of potentialindirect positive impacts/opportunities resulting from significant improvements in access to safe, effective and affordable medicines, vaccines and other health commodities (e.g. diagnostics and contraceptives). These include: The opportunity to raise the standard of environmental management in the pharmaceutical sector. Not only is there an opportunity for CHAI to raise the profile of environmental and climate change issues through its network of collaborations and partnerships, but also to ensure manufacturers either have, or are working to have, environmental management systems that meet national and international standards. Access to reproductive health can reduce environmental impacts. A community’s ability to mitigate or respond to climate change is significantly increased if women are able to plan their families21, as women play a unique role in the stewardship of natural resources22. In general, access to family planning results in lower fertility rates and hence lower population pressure on natural resources. Impact of climate change and environment degradation on the intervention The success of global responses to malaria, TB, HIV and other infectious diseases can be undermined if other root causes such as climate change, growing poverty and environmental degradation are not considered. Climate variability and change, environmental degradation have the potential to expand the geographical range of vectors and therefore the risk of “vector-borne” diseases.Drought, famine and flooding can lead to outbreaks of disease and migration and force “climate refugees” to move into endemic regions where poor nutrition can further reduce their immunity. While this has negative health consequences, it may enhance the impact of the interventions, since increased numbers of cases will benefit from reduced commodity prices. Climate change can however have negative effects on the intervention by hamperingdistribution of and physical access to medicines and/or make it more difficult to target the “most vulnerable” groups. Summary of Climate and Environment Assessment Option 1: “No intervention” has been rated as no/low positive and negative impacts as a result of missed opportunities for the environment/climate change. Option 3: “Deliver interventions across the commodity range through a single market-shaping grant to CHAI” has the potential for medium/low opportunities for climate change and the environment if climate and environmental measures are mainstreamed throughout the intervention. Medium/low negative impacts are also identified with this option although these are manageable risks that can be easily mitigated with few residual impacts. Final Categorisation of Options Option Climate change and environment negative impacts and risks 1 C 3 B/C Definition of Categories: A High potential risk / opportunity B Medium / manageable potential risk / opportunity C No / Low potential impact / opportunity D Core contribution to a Multilateral Organisation Climate change and environment positive impacts and opportunities C B/C C. What are the costs and benefits of each feasible option? Cost-benefit analysis (CBA) has been applied to compare the value for money of the proposed portfolio of activities against the ‘do nothing’ counterfactual. The CBA was undertaken on each component individually and then aggregated to allow for the whole portfolio to be assessed. Unusually for a proposed DFID grant, a Return on Investment (ROI) approach was also applied to assess the portfolio. This narrower measure allows for an assessment of the impact of the intervention on DFID’s own budget – rather than the wider global, developmental and economic impacts as covered by CBA. This analytical approach is based on the savings that the intervention achieves on DFID’s own spend on health commodities (through its contributions to multilaterals like GFATM, UNITAID, GAVI, UNFPA and others).The full CBA and ROI analysis is set out in a more detailed annex. The methodology and results are set out below. Assessing Benefits: Counterfactual market dynamics have been set up for the different commodities. The graph below shows the counterfactual/baseline trajectory for the focal first- and second-line HIV regimens. (These activities would be supported under Component A.1 which is the largest component and accounts for around a quarter of the grant by inputs.)The counterfactual assumes that a number of factors will reduce prices for HIV regimens in the absence of this intervention. These factors include rising demand and implementation of more pro-active market-shaping activities by key purchasers such as GFATM and large countries. Based on these trends, the rate of decline of prices is expected to be moderate (in the absence of the CHAI activities that have driven declines over the past few years.) leaving prices for the WHOpreferred tenofovir-based first-line regimen 23% lower in 2015 than at the end of 2011. For second-line drugs, the counterfactual is expected to result in a 14% reduction in prices to 2015. In terms of the CBA, these are conservative assumptions. Prices for high-demand ART for which no market-shaping interventions have been undertaken in the past few years (e.g., zidovudine, stavudine) have remained broadly constant. A.1. Maximizing Value for Money and Ensuring Sustainable Supply of HIV Treatment Figure 1: Graph to Show Price Reductions for HIV Regimens *Prices quoted before 2011 are Low Income Averages and CHAI Ceiling Prices respectively **2nd line prices before 2011 use TDF+3TC+LPV/r, rather than TDF+3TC+ATV/r for future prices because ATV/r is a relatively new product The graph above also shows the intervention trajectory. The combination of supply- and demandside activities targeting new product uptake, cost reduction for new/existing ART and long-term market sustainability would accelerate price reductions leading to 47% and 39% lower prices, as compared to the 2012 baseline, for first- and second-line regimens respectively. Prices for firstline drugs would be 31% lower in the intervention case, and second-line prices 29% lower, as compared to the counterfactual. Additional benefits (which are not monetised) include greater national capacity and more effective programme decision-making (in key countries). The graphs assume that prices remain constant post-2015 which is when the proposed intervention ends. The prices are rolled forward to 2020 since the impact of the intervention is captured in to the future and these impacts need to be captured in the CBA. Based on the assumed price dynamics, a financial stream of benefits can be calculated by multiplying savings per patient per year by the total number of patients on treatment. This calculation depends on assumptions about overall patients on treatment (in generically-accessible countries). Two scenarios were established. The first assumes a linear scale-up of patients with access to ART based on historical trends (and within that a switching towards the preferred tenofovir-based therapies). The second (highly) ‘conservative’ scenario assumes patients on ART remaining constant at its 2010 (and within that total a similar switching towards tenofovir-based therapies). The table below sets out the benefit streams. Under the linear scale-up, $310 million of savings are achieved by 2015 and $1,450m by 2020. Under the conservative scenario, $182 million is saved by 2015 and $704 million by 2020. 2012 2013 2014 2015 2016 2017 2018 2019 2020 Linear scaleup savings $167.M $48.2M $95.5M $150.2M $176.1M $202.0M $227.9M $253.8M $279.7M Conservative Savings $12.7M $31.8M $56.9M $81.1M $88.8M $96.6M $104.3M $112.0M $119.7M The above methodology has been applied to the extent possible across all the components of the intervention. (For some components – where commodities are newer, forecasts are unreliable and pricing information less available – it has not been possible to undertake CBA; it is probable therefore that benefits have been underestimated) Costing Under Option 3, $56M (£35M) will be required to fund all proposed activities over three years. The component by component breakdown is set out in the Financial Case. (Costs are presented in US dollars to allow for consistency with the benefit analysis.) D. What measures can be used to assess Value for Money for the intervention? The individual components of the proposed portfolio have been reviewed extensively – both by an independent consultant as well as by DFID’s own policy and country-level teams.. Net Present Value/Internal Rate of Return Using a 3.5% discount rate (as recommended for global public goods), the NPV of projectable savings from the portfoliois estimated to be $1.44-2.53bn1 for the period 2012-2020.This is likely to be an underestimate since CHAI are currently only able to project the savings for a subset of components. CHAI’s work in low-cost diagnostics, vaccines, mRDTsand malaria treatment are all expected to generate additional substantial savings. The table below presents, by component,the investment required over the life of the grant (2012-2015); the NPV of savings over the same period; and the NPV of savings in the longer term (2012-2020). During the project lifetime (2012-2015), these savings, combined with the costs of the associated interventions (a total of $27.55m) yield an internal rate of return (IRR) of441% on the ‘conservative’ scenario and an IRRof585% for the ‘linear scale-up’, and 443% and 586% respectively through to 2020. When the full costs of the project ($56.05m) are included, the IRR is 220% and 305% respectively by 2015, rising to 228% and 310% by 2020; this calculation excludes additional savings from the remaining components with un-calculated benefits. Summary of NPV Calculation Cost of investment (2012-2015) Project HIV Treatment (A.1) POC CD4 (A.2) HIV Spending (A.3) MDR-TB (B.1) Low-cost new diagnostics (C.1) LARCs (D.1) Savings (2012-2015) (NPV) Linear Conservative scale-up Conservative Linear scale-up $11.11 $7.10 $153.2M $99.7M $267.3M $99.7M $561.4M $426.9M $1,156.8M $426.0M $4.87 $3.23 $73.0M $39.0M $177.0M $39.0M $361.3M $54.9M $849.8M $54.9M $6.55 $1.25 Not Calculated Not Calculated Not Calculated Not Calculated $14.6M $14.6M $68.6M $68.6M Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated 0 0 0 Vaccines (D.2) $2.54 mRDTs (E.1) Malaria Treatments (E.2) Sharing Lessons Learned Global applied overhead IRR2 NPV $5.71 Not Calculated 1 Savings (NPV) Not Calculated $4.58 Not Calculated $2.20 $6.92 2012-2020 2012-2020 0 228% (Conservative) – 310% (Linear scale-up) $1.45 Billion - $2.53 Billion Based on the ‘conservative’ and ‘linear scale-up’ scenarios for ART access respectively 2Excludes savings from 6 areas that have not be estimated Return on Investment As noted above, a Return on Investment (ROI) approach has also been applied to selected components of the proposed intervention. The methodology used was to identify the share of total expected savings on commodities for key multilaterals – based on their expected volume and distribution of future spending. Next, the total share attributable back to the DFID budget could be calculated based on assumptions of UK contributions. The chart below sets out the findings of this analysis. It is estimated that (based on the linear scale-up scenario) the programme will, up to 2020,in NPV terms, save $59.2m from DFID’s spend on first-line ART (through GFATM which accounts for a third of all patients on treatment in generically-accessible countries and will save $19.7 million in spend on second-line ART (through UNITAID which accounts for 60% of adults on second-line treatment). The direct savings to the DFID budget from the ART work alone exceed the total costs of investment of the whole portfolio and mean the intervention has a positive ROI. Based on the savings associated with the ART component alone, the annual ROI of the whole portfolio is 3% over the period 2012-2020. Assessing the ART component alone as a self-standing intervention gives and ROI of 24% a year. Component area First-line Regimens Second-line Regimens Total HIV savings ROI analysis Total Savings (Linear scale-up) $1,304.1M $145.6M $1,449.8M Savings on UK spend $73.4M $24.7M $98.1M Sensitivity Analysis A number of stress test were applied to the analysis above. Given the high returns, the intervention is robust to sensitivity analysis. Applying an 8% discount rate reduces the NPV to a range of $1.11-1.94bn. Even if only 10% of the expected savings are achieved, the intervention still has a positive NPV (of $95-204m) (at a 3.5% discount rate). The breakeven price reduction for the intervention is that the portfolio delivers just 4% of the forecast savings. In concrete terms, this would mean the intervention accounting for a US$2 decrease in the price of the preferred first-line regimen and a US$4 reduction in the price of second-line treatments. It is also important to stress that the CBA analysis is conservative throughout – not just in terms of the assumptions but also in that it excludes significant potential areas for savings, such as vaccines, where the analytics are not sufficiently robust. However, huge opportunities for price reductions exist for new, emerging and possible future vaccines such as Japanese Encephalitis, Typhoid, Hexavalentand Cholera. As an example, the anticipated annual demand in developing countries for Hexavalent vaccines (combining Diphtheria, Tetanus, Pertussis, Hepatitis B, Polio, and Hib) would be over 200 million doses in the 2015-2020 period. Based on very preliminary discussions with suppliers, it is anticipated that market-shaping interventions for this vaccine alone could generate very significant savings over the programme period. Given the UK’s high level of investment in vaccines, this would bring important savings to DFID’s own budget as well. E. Summary Value for Money Statement for the preferred option The value for money arguments for this intervention are compelling at several levels. First of all, the health commodities to which this programme will promote access are highly costeffective. WHO estimates of cost-effectiveness of selected commodities are: - Immunisation: Recent studies indicate that through scaling up vaccine development and delivery over the next 10 years across 72 countries could prevent the deaths of 6.4 million children, ultimately saving more than $151b in treatment costs and producing $231b in economic benefits23. - Family Planning: DFID’s recent framework document on family planning cites family planning as a ‘best buy’ in global health and development, due to its low cost and far reaching benefits24. Estimates indicate that by meeting the global unmet need for family planning,savings of $5.1bn that would otherwise be required for maternal and newborn care can be made25. - ACTs: DFID analysis indicates that the cost per life saved for children under age 5 treated for malaria with ACTs is between $171 and $20926,,making ACT an extremely cost effective regimen for uncomplicated paediatric malaria27 - ART: Current research suggests that the long term economic benefits of providing treatment to patients in need will be largely offset, if not completely exceed, program costs within 10 years of investment. Specifically, they estimate that monetary benefits range from 81% to 287% of program costs over this timeframe 28 Secondly, market-shaping interventions can have dramatic ‘accelerating’ effects to reduce the prices of health commodities and enhance access. The proposed portfolio has a net present value of $1.5bn based on very conservative assumptions and $2.6bn based on an assumption of a continued trend in the scale-up of patients with access to ART. These NPVs are associated with Internal Rates of Return of 330% and 440% respectively. Thirdly, in addition to the wider global and developmental impacts measured in the NPV, this intervention has a more direct VFM impact on DFID’s own budget. Through savings on DFID’s spend on ART alone through GFATM and UNITAID, the intervention will pay for itself – generating a Return on Investment of at least 5% a year. Commercial Case A. Why is the proposed funding mechanism/form of arrangement the right one for this intervention, with this development partner? The Appraisal section has set out the case that Option 3 (Deliver supply- and demand-side interventions across the commodity range in an integrated manner through a single market-shaping grant to CHAI) is the only feasible option for effectively delivering the proposed intervention, and principles, set out in the Strategic Case. The Appraisal Case also established that Option 3 offers highly-compelling value for money. Based on this context, DFID proposes to use an accountable grant to support the interventions. In addition to the above, and in line with the How To Note of March 2010, an accountable grant has been assessed as the appropriate arrangement since: 1. CHAI is a not for profit organisation. 2. The request for support for the activities was initiated by CHAI rather than DFID 3. The proposal and this accompanying Business Case demonstrate that the proposal and the proposed approach achieves value for money and maximum impact. B. Value for money through procurement CHAI’s founding objective is essentially value for money - specifically in global health commodities – as a means to enhance access. This value for money objective is mainstreamed into all levels of its operations. CHAI’s lean management team and small back-office teams both in-country and at the headquarters level, keep salaries and policies in line with its budget constraints. As a result of this approach, CHAI offers a solid value for money proposition and is able to efficiently and effectively handle procurements across a broad spectrum of commodities and countries. At the management level, CHAI has a tight core of senior-level executives supported by a broader network of regional and program managers that oversee the operations of CHAI’s programs around the world. This flat management structure is set up to ensure comprehensive monitoring and guidance to CHAI’s programs, while keeping costs low. All of CHAI’s management have taken significant pay cuts from their previous private sector roles to be a part of CHAI; as such, CHAI’s salary levels are well below industry averages29. A recent survey by InsideNGO found CHAI to be in the 25th percentile for ex-pat salaries for analyst, specialist, technical/senior associate, programme manager, and deputy country director level positions. For coordinators, senior programme managers, and country directors, CHAI ranked at the 50th percentile. Despite these salary levels, CHAI’s reputation, achievements and ambitious goals allow it to attract the highest levels of skilled professionals with significant experience in both private and public sectors; many of whom are considered experts in their fields by the international community. Bringing this level of expertise to bear on AIDS, TB, malaria, and other programme areas without the high costs that are typically associated with it represents a high degree of value for money. CHAI continues to keep its costs down through its policies, which emphasize lower cost alternatives to typical operating expenses. For example, CHAI’s travel policy states that only economy class airfares will be reimbursed and that whenever possible, travel arrangements should be made 14-21 days in advance in order to minimize costs. Similarly for hotels, CHAI’s policy ensures that country teams negotiate reasonable hotel rates in advance. If negotiated rates are not available, staff are expected to perform due diligence to identify moderately priced accommodations. CHAI also keeps costs low for other expenses, relying largely on Skype for long distance calls and employing a large team of highly trained volunteers, most of whom bring several years of experience in the private sector, to support its small staff both in-country and at headquarters. In parallel to ensuring its cost effectiveness through organizational policies, CHAI recognizes the sacrifices that its staff has made to be a part of its mission and is striving to achieve a balance between cost savings and attracting/retaining the talent that it needs to thrive. Financial Case A. What are the costs, how are they profiled and how will you ensure accurate forecasting? Components Maximizing Value for Money and Ensuring Sustainable Supply of HIV Treatment (Grant A.1) Accelerating Introduction and Scale Up of Point-Of-Care HIV Diagnostics (Grant A.2) Maximising Value for Money of HIV Spending for Universal Access to ART (Grant A.3) Improving Pricing and Supply Security for High Quality MDR-TB Drugs (Grant B.1) Accelerating Market Entry of Highly Accurate and Lower Cost New Diagnostic Products (Grant C.1) Increasing Access to Long-Acting Contraceptives (Grant D.1) Improving Vaccine Market Dynamics for Price Negotiations (Grant D.2) Ensuring Rapid and Sustainable Scale up of Supply of Malaria Rapid Diagnostic Tests (Grant E.1) Ensuring Rapid and Sustainable Scale up of Supply of Quality Malaria Treatments (Grant E.2) Sharing lessons learned and new market assessments e.g. for family planning commodities, therapeutic feeding or other products Total Costs (US$ millions) Total Costs (GBPmillions) $11.11M £6.94M $7.10M £4.43M $4.87M £3.04M $3.23M £2.02M $6.55M £4.10M $1.25M £0.78M $2.54M £1.59M $5.71M £3.56M $4.58M £2.86M $2.20M £1.37M Global Applied Overhead $6.92M £4.32M TOTAL $56.05M £34.99M B. How will it be funded: capital/programme/admin? This project will be funded over three years from programme funds from a range of DFID Departments as follows: Human Development Department DFID India (from GDPP) £15 million £11 million Additional resources up to a total maximum of £9m may also be provided towards implementation of the work plan from DFID South Africa (£1.5-2m) and DFID China (£3-5m) (both from GDPP) as well as from DFID Uganda (all subject to further consultation and approval). There are no contingent or actual liabilities. No other donor will co-finance the work plan itself. However, a number of other funders will support the objectives of different components in different ways – either through CHAI or other modalities. Important funders of CHAI include the Gates Foundation, the Children’s Investment Fund Foundation (CIFF), ELMA Philanthropies, UNITAID and the Norwegian, Swedish and Australian bilateral aid agencies. C. How will funds be paid out? Funds will be paid out through a DFID Accountable Grant to CHAI using DFID’s standard template. Payment will be Quarterly. CHAI has requested payment of some or all of the quarterly tranches in advance. DFID procedures allow for this. HDD will work with FCPD, CHAI and others to identify mutually satisfactory arrangements. The financial management of the project will be led by the Health Services Team (HST) in DFID in London, which will manage all claims for reimbursement. HSTwill coordinate contributions from other DFID Departments. Payment timing across financial years will be forecast annually to meet DFID requirements. D. What is the assessment of financial risk and fraud? Low. CHAI has a strong track-record of sound financial management of the 2008-2012 grant from DFID as well as in managing resources from other agencies, such as GFATM and UNITAID. CHAI has a set of internal control mechanisms that mitigate the risk of fraud and other financial risks. Since the vast majority of programme funds will be managed from CHAI’s Boston Headquarters, CHAI will be able to closely monitor grant funds. Additionally, the relatively small portion of funds that will be administered by the Country teams will be subject to review on a monthly basis that will involve the monthly reconciliation of cash reports and bank statements. Internal audits will be conducted periodically to ensure that funds are utilized and accounted appropriately. Moreover, since the bulk of expenditures under the Accountable grant will consist of salaries and benefits, CHAI’s centralized payroll system will provide transparency and accountability with respect to compensation payments made to programme personnel. These payments will be reviewed on a monthly basis by both programme managers and CHAI Headquarters finance staff to ensure that the funds have been appropriately charged to the grant. This fact together with Internal controls, provide a considerable degree of reassurance that the financial risks for this programme will be low and manageable. E. How will expenditure be monitored, reported, and accounted for? Financial records will be maintained in adherence to United States’ Generally Accepted Accounting Principles (US GAAP). Rigorous monitoring and accounting of expenditure will be facilitated by a dedicated project structure in the CHAI financial management system. Internal monthly reviews of all expenditure will be conducted by project and finance personnel to ensure compliance with programme arrangements. Moreover, CHAI maintains a set of codified financial policies and procedures that will govern how programme funds are utilized and accounted. Detailed financial reports (Statements of Expenditure) will be provided to DFID on a quarterly basis in line with the Accountable Grant agreement. The reports will be submitted along with requests for disbursements (quarterly invoices). The reports will be reviewed and certified in attestation of their veracity and completeness by a CHAI officer with certification authority. Any assets procured under this programme will be treated in accordance with DFID procedures, and declared to the DFID programme manager. An asset inventory register will be maintained for equipment whose purchase price meets the DFID capitalization threshold. If assets remain at the end of the programme, a disposal strategy shall be arranged in consultation with the DFID Programme Manager. CHAI will submit annual audited accounts for each of the financial years covered by the Accountable Grant. These will be certified by an independent firm of professional auditors and will show DFID’s grant as a separate item of income. Management Case A. What are the Management Arrangements for implementing the intervention? CHAI will be responsible for implementing the agreed portfolio of activities. The key focal point within CHAI will be the Executive Vice President for Access Programs who will coordinate with CHAI Country Directors and other Executive Vice Presidents as appropriate. Guided by the lessons learned in the implementation and independent evaluation of DFID’s first grant to CHAI, measures will be put in place to ensure strong DFID oversight of operational management of the project. A partnership monitoring structure (as specified as Output 7 in the logframe) will be put in place to monitor results and risks, provide strategic guidance and identify new opportunities. The structure will aim to ensure that all country-level activities (both supply- and demand-side) are aligned with country government priorities, are coordinated with other development partner activities and mainstream capacity-building objectives. It will also ensure that, at the global policy level, activities undertaken under the grant, are embedded within relevant global frameworks and coordinated with the activities of other development partners and relevant stakeholders. The structure will also seek to ensure that, at all levels, opportunities for learning and innovation are maximised. The respective focal points within DFID and CHAI for management of the grant will be the Senior Policy Advisor, Market Dynamics and International Engagement, Human Development Department, DFID and the Director, Director of Institutional Relationships & Reporting, CHAI. The partnership monitoring structure (PMS) will have three levels: - At the apex a high-level oversight committee, chaired at CEO level in CHAI and DG level in DFID. Other partners (such as key financiers of CHAI like the Gates Foundation and the ELMA Foundation, will also be engaged to participate as necessary. This meeting will discuss programme results (against logframe objectives), risks and issues for strategic guidance. DFID country offices from emerging powers will be represented in this oversight committee. - Progress on the individual, commodity-specific components of the portfolio will also be reported on a six-monthly basis against the specific indicators set out in the components themselves. Within DFID, the counterparts will be disease-specific policy leads within Policy Division. Wherever possible, reporting and oversight will be integrated within existing forums with an interest in market dynamics. For example, in the area of contraceptives, this will be the Reproductive Health Supplies Coalition (RHSC). Similarly, the work on Efficiency and Effectiveness of spending on HIV will be overseen by the UNAIDS-led group concerned with implementation of the Strategic Investment Framework. Work on vaccines will coordinate with GAVI’s emerging Supply and Procurement Strategy activities. - Finally, at the country-level, on both the supply- and demand-sides, CHAI will report to DFID incountry health advisers on progress, with the level of regularity to be agreed in county and reporting on a bi-annual or annual basis being optimal. Where appropriate this reporting will engage other relevant stakeholders, such as country governments and other development partners. A key focus of discussions will be to ensure appropriate focus on, indicators of, and approaches to, capacity-building. Where possible, this reporting will be aligned with existing donor-government access to medicines forums. For example, in China, the reporting will be integrated into the Government-Development Partner Working Group on Global Health. The table belowwill guide the organisation of oversight of country-level demand-side activities. The table sets out the distribution of activities in the proposed portfolio geographically and by commodity area. The oversight structure will ensure that all these demand-side activities are adequately covered. Wherever in-country activities are on-going, a DFID focal point will be identified. The focal point for activities in Southern African countries will be DFID’s regional health adviser for access to medicines based in Pretoria (in coordination with DFID country health advisers where appropriate). Where DFID does not have a health-specific presence, such as Cambodia or Cameroon, progress will be reported directly to the Senior Policy Adviser in Policy Division, London. Where additional demand-side activities are agreed following discussions (as an example, demand activities for LARCs may emerge), incountry oversight and coordination arrangements will be put in place. A plan for management of oversight will be agreed within the first three months of implementation. Geographic Distribution Grant Component Cambodia Cameroon Ethiopia India Kenya Liberia Malawi Mozambique Nigeria Rwanda South Africa Swaziland Tanzania Uganda Zambia Zimbabw e HIV Treatment Point-Of-Care HIV Diagnostics* Value for Money of HIV Spend MDR-TB Drugs This project will directly benefit other High Burden Countries beyond India, though CHAI do not anticipate further in country demand roles New Diagnostic Products Long-Acting Contraceptives CHAI do not anticipate an in country role for this project Vaccine Market Dynamics Malaria Rapid Diagnostic Tests Quality Malaria Treatments** In addition to the above, CHAI are in the process of identifying 1 to 3 additional countries to engage with Key: Direct DFID Funded Engagement Funding from other donors, leveraging DFID support for Supply projects and Demand work in other countries B. What are the risks and how these will be managed? Steps have been taken to ensure that risks are fully identified and mitigated through a systematic approach to program management and risk identification and resolution. The oversight structure outlined in Section C below will be used to manage risks. Several overarching risks were identified in the independent evaluation of the first grant. The same independent consultant also reviewed the proposed portfolio and identified programme and implementation risks. Some risks that were included in the first grant to CHAI were not borne out during programme implementation. Key among these was the risk of adverse reaction from the UK pharmaceutical industry. The earlier grant to CHAI had identified potential for opposition from originator pharmaceutical companies. However, this did not materialise. In fact, there is now a clearer consensus than in 2008 among originator companies that generic companies have a central role in enhancing access to their products (for example through technology transfer). We have therefore not included this risk here. The remaining risks can be classed into six categories, with associated mitigating actions as follows:- 1. Market leverage (CHAI may not have the same degree of leverage to move the market in the absence of a procurement role as it had with UNITAID): CHAI has certainly achieved significant market leverage through the UNITAID programme for some commodities. However, this is only one tool. Good evidence exists that it can continue to have real leverage as this role phases out, and CHAI are actively refining their approach to maximizing leverage through the addition of new tools or a refinement of existing ones, such as the procurement consortium approach. Probability of risk: Medium. Impact after mitigating actions: Limited. While there remains a risk that new or refined tools relating to procurement may not be quite as effective as the leverage CHAI had with UNITAID, they are nevertheless likely to make a substantial contribution to price reductions. This will be aided by the advocacy of the commission on life saving commodities. Furthermore, demand side activities are only one component of the intervention, supply side activities are likely to result in increased competition and hence reduced prices. 2. Supply-side engagements (refusal of key manufacturers to engage in discussions and negotiations or preference to work with others): CHAI works with all suppliers who are willing to make appropriate commitments to quality and cost. Thus, suppliers are incentivised to engage with CHAI, as they are working with the knowledge that the decision to accept input from CHAI is their option, but also that their competitors will also have access to the same information and assistance that they do. Suppliers are also free to work with other providers of support if they prefer. This underlines the importance of coordination with other players as well as the need for engagement with multiple suppliers. This increases the probability of success, even if one or more suppliers choose not to work with, or fail to succeed in a collaboration with CHAI Probability of risk: Low. Impact after mitigating actions: Very limited. It is clearly preferable that as many suppliers as possible engage with CHAI; however, even if a few choose not to, CHAI’s work with those that do will mean that they can increase manufacturing efficiencies and so reduce prices. This will give them an advantage over their competitors, which is likely to prompt them to ask for CHAI to work with them or to get similar support from elsewhere. 3. Demand-side engagements (inability to obtain commitments from partner governments to support programs and facilitate policy changes where appropriate): To meet objectives and build sustainable programs on the ground, CHAI works in close partnership with governments from the start of any programme development. The proposed policy ensures that there is buy-in at each phase and that all partners are committed to working together towards the stated goals. Probability of risk: Medium, particularly where procurement decisions are not based on costeffectiveness. Impact after mitigating actions: Low to medium. It is difficult to assess, a priori, how many partner governments are influenced by factors other than cost effectiveness and for which products. The engagement of other stakeholders in-country can further mitigate against this risk by increasing pricing transparency of medicines and ensuring that procurement supported by donor agencies is evidence based. 4. New products (new products may not establish superiority (or non-inferiority where prices are lower) to existing regimens): This is a large risk routinely faced by pharmaceutical companies. This strongly supports a portfolio approach to both optimisation of existing products in parallel with the development of new products. Probability of risk: High, Impact after mitigating actions: Low. The mixed portfolio will ensure that the optimisation of existing products contributes to pharmaceutical budget savings 5. Global engagements (potential for overlapping activities and role redundancy): This is an ongoing challenge in the public health arena, and CHAI aims to minimize overlap and redundancy by working through collaborative partnerships. The oversight structure proposed for this programme will ensure that all activities are coordinated and aligned with common frameworks and communities. It should also be noted that there are some activities where some level of duplication or complementary approaches to the same outcome are more productive than a single approach. An example is the benefit to using multiple forecasting methodologies in some cases to validate the results of any one forecast. Probability of risk: Low, Impact after mitigating actions: Very low. 6. Capacity: Establishing a pipeline of new, low-cost diagnostics is a new area for CHAI requiring new expertise and partnerships. Similarly engaging in an expanded range of commodities and in new countries, requires an appropriate skills mix and number of staff in a timely manner. CHAI has already begun to engage and develop relationships with low-cost diagnostic developers and has developed a collaboration with PATH to ensure that complementary skill sets are available for this project without a need always to recruit new staff. Probability of risk: Medium to high, Impact after mitigating actions: Low These above risk types apply differently to different components of the intervention. The following table shows, by component, the types of risks that are most likely to affect each areas of work, the probability and likely impact of each risk, specific mitigating actions and the residual impact after mitigation (1 indicates low probability or impact, 5 is high). As part of the grant oversight structure a systematic risk matrix will be developed and monitored on an on-going basis. No Proposal area A.1 HIV Medicines A.2 POC CD4 Principal Risk Probability Impact Mitigating Actions Residual Type 1-5 1-5 Impact Market 2 4 CHAI has succeeded in facilitating reductions 2 leverage in the price of key first-line products (e.g., TDF, EFV, and complementary FDCs) despite the fact that first-line procurement is not part of the UNITAID program. New tools will be developed and existing ones refined. Market 3 4 CHAI has successfully facilitated price 2 leverage A.3 HIV Spending Demand-side 3 engagements 3 B.1 MDR-TB Drugs Supply-side engagements 2 3 Demand-side engagements 4 3 Global engagements 2 2 C.1 Low-cost Supply-side diagnostics engagements 3 2 New products 5 4 Capacity 4 4 reductions for diagnostics in the absence of a large procurement program. In addition, CHAI is actively seeking new partnerships that would augment its leverage in the market and has just been funded to execute a diagnostics procurement program similar to the drugs programme under UNITAID. CHAI has been asked by governments to support MoHs to drive the change proposed in this project. CHAI will select 3-5 countries where the political leadership is strong, decision makers have prioritized this work, and where its teams have strong relationships needed to achieve success. CHAI will link into coordination mechanisms facilitated by UNAIDS in country to ensure alignment with country operationalisation of the strategic investment framework and treatment 2.0 approach. MDR-TB suppliers have already expressed initial interest in the project and India’s NTP seems open to discussing the possibility of volume consolidation. CHAI will leverage this and its strong relationships with TB suppliers to advance this activities under this project The Government of India (GoI) may not be receptive to CHAI’s efforts to increase the size of PQ’ed orders for MDR-TB drugs. CHAI has worked closely with GOI in the past to advance access goals and is well positioned to effect similar changes in TB. The GoI is the largest and most organized single program that could achieve that goal however, it will be critical that CHAI also quantify and consolidate demand in other large programs to ensure that maximum global benefit is achieved from the project.. CHAI has been supporting GDF and participated in the 2012 Joint Donor Review Mission for India’s TB program. CHAI will continue to build on these collaborations, joint forums, and share lessons learned from its efforts CHAI will leverage its previous supply-side work including price reductions for various diagnostics including CD4, VL, EID, and HIV rapid tests. New, promising suppliers have already been identified. To increase market leverage, CHAI is also applying for complementary funding to support POC adoption (including the proposal for Grant A.2) and commodity procurement. To guide product design CHAI has on-theground access to design requirements and preferences via its countries across Africa, Asia and the Caribbean that can be shared directly to suppliers. Establishing a pipeline of new, low-cost diagnostics is a new area for CHAI requiring new expertise and partnerships; there is a risk 1 1 2 1 1 2 2 Demand-side engagement 4 3 Global collaborations 2 2 D.1 LARCs Supply-side engagement 3 4 D.2 Vaccines Supply-side engagement 3 4 Global engagements 2 2 mRDTs Demand-side engagements 3 4 E.2 Malaria drugs Supply-side engagements 2 3 Demand-side 4 4 E.1 in securing these steps in a timely manner.CHAI has already begun to engage and develop relationships with low-cost diagnostic developers in order to lay a foundation on which to build this project. CHAI has also actively developed a collaboration with PATH, as they feel that PATH and CHAI have very complementary skill sets and focus areas for furthering this project. The collaboration is developing very strongly, and CHAI and PATH are working on a complementary proposal to UNITAID to support related work. CHAI will draw from its relevant experiences and existing relationships with countries and governments from its Pima pilot project CHAI will continue to partner closely with PATH, FIND, and CDC to leverage their expertise in product development and commercialization. Based on initial discussions, Merck and Bayer have expressed interest in establishing an agreement to negotiate reduced pricing if donors are willing to increase funding and RHSC is able to provide improve demand side forecast sand ordering procedures. CHAI will build on its previous work with the Gates Foundation, GAVI, and UNICEF over the past 16 months to engage with suppliers, which have led to price reductions for rotavirus vaccine by two-thirds. CHAI recognizes that that vaccine markets are substantially different from those for drugs. Therefore, CHAI will build on its existing capacity in analyzing markets for health commodities and is already adapting strategies to meet the specific needs and risks of the vaccine market. CHAI will also leverage its strong relationships with multinational vaccines suppliers through its initial work. CHAI will continue to partner closely with GAVI and UNICEF on its market analyses to ensure its work is fully complementary CHAI has targeted its efforts in four focal countries where it has a strong programmatic presence and strong relationships with partners and governments. CHAI will build on its efforts providing technical assistance to countries for AMFm and it success achieved through the RDT scale up pilot in Swaziland CHAI has long standing relationships with key generic suppliers based on its work with ACT suppliers (as well as with suppliers of other products including ART) and has facilitated significant price reductions. CHAI is already engaging with suppliers developing products of interest and will leverage its previous experiences to advance this work. CHAI intends to build on the progress made 2 1 2 3 1 2 2 2 engagements CHAI Global engagements 2 2 Capacity 4 4 thus far in Uganda and Nigeria and select 2-4 additional where it has a strong programmatic presence and deep relationships with governments and partners. CHAI will continue to coordinate closely with GFATM, RBM, WHO and other major stakeholders to ensure that market stability activities are efficiently executed Potential inability to retain and recruit industry respected employees. CHAI has been and will continue to refine and update our value proposition to industry experts to be able to continue to recruit, and to better retain subject matter experts. This remains a subject that CHAI is reviewing; however the success of its recruitment to date and its continuing efforts to retain the organization’s highest performers bodes well in this regard. 1 2 C. What conditions apply (for financial aid only)?Not applicable D. How will progress and results be monitored, measured and evaluated? A project of this scale and reach, with multiple, complementary components requires three different evidence-generating processes to provide information for different programme needs, namely monitoring, evaluation and an assessment of the broader impacts. The logframe provides the overarching umbrella for the first two of these. However, a separate articulation of lessons learnt and means of capturing them will be required for the third. 1) Monitoring An on-going monitoring process, incorporating the logframe indicators, is required to provide the implementing team indications of what is working and where additional effort is required. Each project component has its own monitoring and evaluation framework integrated into design, additional to this, however a number of key commodity prices and procurement volumes in implementing countries will also need to be monitored. These are set out Annex:Cost-Benefit Analysis. The implementing team may also define additional monitoring indicators and processes to provide them with a higher level of management information. 2) Evaluation The logframe also provides a basis for an evaluation of outputs to inform both the implementers and DFID of whether the project is on track to meet its stated targets. The monitoring information noted above will provide the base data and contextual information for at least an annual evaluation (more frequently if required) . This will be the foundation for policy and programme discussions within the partnership monitoring structure set out in the Management Arrangements section above. 3) Assessment of Impacts An assessment of the broader impacts of the project is also required to capture the synergies from an integrated approach and also to capture lessons from the implementation process. This will be designed during the inception phase of the programme and will aim to capture not only whether the project has achieved its intended impacts, but also what was required to achieve them and whether the impacts were broader than expected and sustainable. Thus it will include questions about the impactthat the project has had on prices for the targeted commodities in countries not directly involved in the implementation; the level of stakeholder engagement at country level and the factors involved in sustaining it; the degree to which implementation has used existing health system and other processes compared with new process that have been set up in countries; and challenges that remain unaddressed Representatives from DFID and CHAI will form a steering committee to contract with independent groups on an on-going basis to review, evaluate and assess the impact of individual programs throughout the whole period of the proposed grant. The role of the steering group will be both formative (e.g. review of progress against objectives contributing to periodic reviews and supporting the partnership monitoring structure) as well as assessing impact, using more objective quasi-experimental measures as much as possible to explore complex dynamics and attribution as well as to learn lessons. This process will engage stakeholders both within DFID and outside. Due to the fact that a large proportion of the proposed program of work is due to have greatest impact towards the end of the grant period, it can be expected that there will be a scale up of progress and results during the life of the grant. The resources for these evaluations will come from the lesson-learning/new product evaluation line of the budget. It should also be noted however that this may limit CHAI’s ability to explore new product opportunities and these competing pressures will have to be managed throughout the grant. Logframe: Quest number of logframe for this intervention: 3506436 Annex: Cost Benefit Analysis Introduction This Annex serves as technical background to the Cost-Benefit Analysis (and Return on Investment analysis) set out in the Appraisal Case of the Business Case. Cost-benefit analysis (CBA) has been applied to compare the value for money of the proposed portfolio of activities against the ‘do nothing’ counterfactual. The CBA was undertaken on each component individually and then aggregated to allow for the whole portfolio to be assessed. However it is worth noting that by evaluating each component separately the CBA does not measure the synergistic benefits of the entire programme of work as a whole. The benefits of this project are far greater than merely the sum of their parts as barriers at both a supply and demand level to medicines and diagnostics are substantially decreased. Unusually for a proposed DFID grant, a Return on Investment (ROI) approach was also applied to assess the portfolio. This narrower measure allows for an assessment of the impact of the intervention on DFID’s own budget – rather than the wider global, developmental and economic impacts as covered by CBA. This analytical approach is based on the savings that the intervention achieves on DFID’s own spend on health commodities (through its contributions to multilaterals like GFATM, UNITAID, GAVI, UNFPA and others). Assessing Benefits A bottom-up, component-by-component, approach has been used to identify benefits. Counterfactual market dynamics have been set up for the different commodities. This section will go through each component in turn to set out the parameters, expectations and dynamics for each health commodity. A.1. Maximizing Value for Money and Ensuring Sustainable Supply of HIV Treatment The graph below shows the counterfactual/baseline trajectory for the focal first- and secondline HIV regimens. (These activities would be supported under Component A.1 which is the largest component and accounts for around a quarter of the grant by inputs.) The counterfactual assumes that a number of factors will reduce prices for HIV regimens in the absence of this intervention. These factors include rising demand and implementation of more pro-active market-shaping activities by key purchasers such as GFATM and large countries. Based on these trends, the rate of decline of prices is expected to moderate (in the absence of additional CHAI activities that have driven declines over the past few years.) leaving prices for the WHO-preferred tenofovir-based first-line regimen 23% lower in 2015 than at the end of 2011. For second-line drugs, namely Atazanavir/ritonavir and tenofovir/lamivudine,the counterfactual is expected to result in a 14% reduction in prices to 2015. In terms of the CBA, these are conservative assumptions. Prices for high-demand ARVs for which no market-shaping interventions have been undertaken in the past few years (e.g., zidovudine, stavudine) have remained broadly constant3. 3 See MSF (2011),‘Untangling the Web of Antiretroviral Prices’ for recent price dynamics of ARTs Figure 2: Graph to Show Price Reductions for HIV Regimens *Prices quoted before 2011 are Low Income Averages and CHAI Ceiling Prices respectively **2nd line prices before 2011 use TDF+3TC+LPV/r, rather than TDF+3TC+ATV/r for future prices because ATV/r is a relatively new product The graph above also shows the intervention trajectory. The combination of supply- and demand-side activities targeting new product uptake, cost reduction for new/existing ART and long-term market sustainability would accelerate price reductions leading to 47% and 39% lower prices, as compared to the 2012 baseline, for first- and second-line regimens respectively. Prices for first-line drugs would be 31% lower in the intervention case, and second-line prices 29% lower, as compared to the counterfactual. Additional benefits (which are not monetised) include greater national capacity and more effective programme decisionmaking (in key countries). The graphs assume that prices remain constant post-2015 which is when the proposed intervention ends. The prices are rolled forward to 2020 since the impact of the intervention is captured in to the future and these impacts need to be captured in the CBA. Based on the assumed price dynamics, a financial stream of benefits can be calculated by multiplying savings per patient per year by the total number of patients on treatment. This calculation depends on assumptions about overall patients on treatment (in genericallyaccessible countries). Two scenarios were established. The first assumes a linear scale-up of patients with access to ART based on historical trends (and within that a switching towards the preferred tenofovir-based therapies). The second (highly) ‘conservative’ scenario assumes patients on ART remaining constant at its 2010 (and within that total a similar switching towards tenofovir-based therapies). The table below sets out the benefit streams. Under the linear scale-up, $310 million of savings are achieved by 2015 and $1,449m by 2020. Under the conservative scenario, $182 million is saved by 2015 and $704 million by 2020. 2012 2013 2014 2015 2016 2017 2018 2019 2020 Linear scaleup savings $16.7M $48.2M $95.5M $150.2M $176.1M $202.0M $227.9M $253.8M $279.7M Conservative Savings $12.7M $31.8M $56.9M $81.1M $88.8M $96.6M $104.3M $112.0M $119.7M A.2. Accelerating Introduction and Scale Up of Point-Of-Care HIV Diagnostics Figure 3: Percentage reduction in global access pricing for POC HIV Diagnostics Counterfactual: A ~3.5% annual price reduction is estimated through to the end of the grant in 2015. This assumption is based on historical pricing trends witnessed with Becton Dickinson conventional CD4 products in the absence of market-shaping work. Intervention: It is estimated that price reductions can be primarily achieved by focusing on reducing the costs of key commodity components. Based on CHAI analytical data it is assumed that these costs amount to $7.65 per test in 2011, with a 10% reduction from the baseline achievable by the end of 2012, 20% in 2013, rising to a 30% reduction in 2014. The additional anticipated benefits of this work would be: • Lower prices for at least 2-3 POC CD4 products (globally) allowing earlier initiation of ART and better outcomes for patients (in 5-7 countries) • Greater efficiency in ART service delivery for national programs (in 5-7 countries) • More sustainable market (for POC suppliers) and greater supply security Figure 4: Graph to Show Anticipated CD4 Tests Required (Conventional V POC) In addition, significant cost savings will be achieved through the adoption of POC CD4 testing as a replacement for traditional forms of CD4 testing and a subsequent reduction in wastage. Estimations across seven proposed focus countries suggest that demand for POC CD4 testing will reach 5.6 million by 2015 and as high as 12.8 million by 2020. CHAI data on POC testing from 3 pilot countries suggests that the wastage rates associated with CD4 testing are consistently around 45%4, as compared to a repeatedly demonstrated zero wastage with CD4 POC testing5. With the consequent reduction in levels of repeat testing, not only will the unmet needfor CD4 tests be substantially reduced, but the cost per test result received will be reduced by 30% to 60%. Further the analysis considers that these savings are only achievable under the intervention proposed, as they are contingent on combining CHAI’s unique strengths working closely with suppliers to accelerate the roll out of products and in partnership with governments to ensure efficient and effective deployment in country. UNITAID is also currently beginning to finance interventions in this area with similar objectives. DFID-supported activities will be complementary and reinforce progress towards targets. The potential savings from this work component are significant, coming as they do from two potential sources (pricing savings and wastage savings). Even if we are to assume that POC CD4 testing has a far higher wastage rate than the 0% found in pilot tests, for example 5% wastage, the cumulative savings are anticipated to total $118 million by 2015, rising to $538 million by 2020. The breakdown is set out in the table below. As a sensitivity analysis we have also calculated a much higher POC CD4 wastage rate of 15% and found that the savings would still be significant totalling $97m by 2015 and $445 by 2010. 2012 2013 2014 2015 2016 2017 2018 2019 2020 Pricing Savings $0.7M $2.9M $6.5M $8.3M $10.3M $12.4M $14.7M $17.0M $19.2 M Wastage Savings (at 5%) $9.8M $20.5M $30.5M $39.1M $48.6M $58.5M $69.2M $80.2M $90.1 M 4 The time from test to result means that results are not always communicated to patients A.3. Maximising Value for Money of HIV Spending for Universal Access to ARVs Figure 5: Graph to show reduction in non-commodity unit costs in 5 focus countries Counterfactual: A very limited level of savings is projected to occur without CHAI intervention. These savings would be mainly driven by laboratory cost reduction in South Africa as result of the predicted revision of guidelines and forms. In addition, some savings (far smaller in scale) should be achieved in personnel costs in Zambia over the next few years Intervention: Although the potential scale of savings in this area is huge across 5 focus countries, the savings anticipated are based on CHAI’s assumptions on what savings per patient per year are achievable in each country (broken out by categories in ARTs, Labs, Personnel, and Other). The savings achievable in this area have been broken down into conservative and high end to give a better idea of the levels of savings that could be achieved here. Both levels represent significant potential levels of savings. The additional anticipated benefits of this work would be: • Decreased unit cost of ART service delivery through earlier patient commencement on treatment, reductions in new HIV infections, greater national capacity to manage HIV spending and programmatic response (in 3-5 countries) • Potential to catalyze similar changes in other countries using newly developed tools/benchmarks 2012 2013 2014 2015 2016 2017 2018 2019 2020 Total Savings (Low) $5.5M $12.8M $14.7M $16.5M $18.4M $20.3M $22.1M $24.0M $25.8M Total Savings (High) $12.9M $30.0M $34.3M $38.6M $42.9M $47.3M $51.6M $55.9M $60.2M B.1. Improving Pricing and Supply Security for High Quality MDR-TB Drugs Figure 6: Graph to show Reduction in Spend for Quality Assured TB treatment Counterfactual: The above graph is relatively conservative because it assumes that India will exit the SRA-approved market in 2015 without further intervention and that the number of patients on these drugs in the rest of the world will not increase from 2010 levels. We are working on the assumption that India would leave the SRA approved product market in favour of the Indian market, consequently the country would not stop buying MDR drugs, but the shift would have global implications due to the volume of demand India represents.Despite this, the long term historical price trend for Cycloserine and Kanamycin, the two drugs included in this analysis, has been increasing significantly.6 In 2010 and 2011 tenders, the price was more or less stable despite volumes increasing nearly two fold with the inclusion of India's order for 11,550 MDR patient courses. Therefore, it could be argued that in a “do nothing” counter factual, the price of Quality Assured TB treatment would not only not improve, but might even increase. As such, we have assumed that pricing as flat at baseline is likely the most conservative and realistic outcome. Intervention: The key benefitof intervention in this field is not price savings, but instead to ensure the long term stability of the market (post 2015) and a healthy API base. If left to natural forces there may be additional entrants and short term price gains; at best these gains would likely be reversed by India's market exit in 2015 and the resulting sharp drop in demand in SRA approved products. In addition, we believe that cost savings are possible through the provision of assistance to formulators currently in the regulatory pipeline to ensure that they become qualified in a shorter time frame, which should result in the realisation of modest price improvements. The anticipated benefits of this work would be: 6Cs 321%, Km 617% between 2001 and 2011; Source: MSF March 2011. • • • Lower prices for 2 high-quality second line drugs (SLDs) (in India) and future reductions for other high burden countries leading to better patient outcomes and decreased spread of resistance More attractive and sustainable market (for SLD suppliers) and greater supply security Potential price reductions for additional SLDs As a result of the above, the estimated financial savings for this work are relatively modest and this reflects the importance of ensuring stability in the market long term following India’s anticipated departure from the market by the end of 2014. Savings (India) Savings (RoW) 2012 2013 2014 2015 2016 2017 2018 2019 2020 $7.0M $10.7M $16.0M - - - - - - $1.6M $2.4M $4.0M $4.0M $4.0M $4.0M $4.0M $4.0M $4.0M C.1. Accelerating Market Entry of Highly Accurate and Lower Cost New Diagnostic Products Counterfactual: No price reductions anticipated Intervention: CHAI anticipates price reductions for Lower Cost New Diagnostics (LCND), with the scale to be determined following initial discussions with suppliers in Q2 2012.These would not include the three tests discussed earlier and would be products from Indian and Chinese manufacturers. The anticipated benefits of this work would be: • Launch of at least 3 new high-quality, low-cost tests (CD4, EID, viral load, TB, or malaria) leading to higher patient retention, decreased spread of resistance (assuming treatment available for diagnosed patients), better outcomes for patients (in Resource Limited Settings) and further price reductions for diagnostic commodities • More attractive, sustainable market for (diagnostic technology suppliers) and greater supply security • Greater attention to servicing customers such as public laboratories and Governments by including repair and replacement services, clear and low pricing on commodities and a focus on point commercialisation activities Given the uncertainties around pricing and priority commodities, no financial benefits are currently attributed to this component. D.1. Increasing Access to Long-Acting Contraceptives Figure 7: Graph to show Percentage Reduction in Purchase Price of LARCs Counterfactual: Withan average price of Jadelle at $22 and Implanon at $20/implant in 2010, we have assumed that the Implanon price has fallen to $18/unit in 2012 based on information from Merck. In this scenario it is further assumed that Implanon pricing will fall to $16.50/unit once 4.5M women are on Implanon in developing countries, which is based on the proposed Merck deal. In our forecast we estimate that this will occur in 2014. Intervention: A price reduction of ~29% on average is estimated between Implanon and Jadelle and/or greater use of equally effective LARCs. The anticipated benefits of this work would be: • Lower prices for Implanon and Jadelle leading to fewer unwanted pregnancies, maternal deaths, and better health outcomes for women of childbearing age. • Maternal and newborn health services savings from fewer abortions, pregnancyrelated deaths and disabilities, and infant deaths • New capacity for demand-side coalition • Further cost savings and outcomes improvements in longer-term Based on these market dynamics, annual global savings of US$9m are achievable by 2015, rising to US$17m in 2020. However, since the analytics do not support attribution to CHAI engagement, these savings are not added to the total benefits associated with this intervention. D.2. Improving Vaccine Market Dynamics for Price Negotiations Counterfactual: No price reductions anticipated Option 3: At least 30% price reduction from 2 suppliers for GAVI-eligible countries in 2015 The anticipated benefits of this work would be: • • • • Accelerated entry of new and affordable vaccines leading to reductions in vaccinepreventable illness and death among children and adults More attractive market for suppliers of new vaccines Potential for increased financial support for GAVI and additional strategic investments for additional marketplace improvements Greater capacity (market-shaping knowledge and skills) for key actors in the field (e.g., GAVI) to enable leveraged impact and sustainability Based on these market dynamics, very high annual global savings are achievable. However, since the analytics do not support attribution to CHAI engagement, these savings are not added to the total benefits associated with this intervention. E.1 Ensuring Rapid and Sustainable Scale up of Supply of Malaria Rapid Diagnostic Tests (mRDT) Counterfactual: No price reductions anticipated Intervention: CHAI anticipates an over 50% reduction in irrational use of ACTs in our four focal countries, based on experience from an effective deployment of RDTs in Senegal7, as well as price reductions and greater market share for India/ Chinese suppliers in 2013. Alongside supply-side interventions, CHAI will be working closely with National Malaria Control Programs (NMCPs), WHO and in country partners to address barriers to appropriate usage of and adherence to malaria RDTs. Some examples of in-country work include: 1. Ensure clear national policies are developed, especially in regards to confirmatory diagnosis and management of RDT negative patients 2. Develop and disseminate job aids with the national treatment algorithms 3. Identify additional barriers to uptake of and adherence to RDTs through qualitative and quantitative surveys 4. Test solutions to non-adherence through Operational Research studies 5. Incorporate messages about malaria diagnosis into planned IEC/BCC campaigns The anticipated benefits of this work would be: • • • • Better health outcomes for patients with febrile illnesses in public health sector (in 4 focal countries) Improved planning and targeting of resources for national programs leading to stronger management capacity of NMCPs Lower pricing for high-quality mRDTs (globally) Contributions to global public health knowledge (e.g., operational research publications, best practices Based on these market dynamics, very high annual global savings are achievable. However, since the analytics do not support attribution to CHAI engagement, these savings are not added to the total benefits associated with this intervention. E.2. Ensuring Rapid and Sustainable Scale up of Supply of Quality Malaria Treatments Counterfactual: No price reductions anticipated 7 Major Reduction in Anti-Malarial Drug Consumption inSenegal after Nation-Wide Introduction of Malaria Rapid Diagnostic Tests; PLoS ONE, April 2011 Intervention: It is anticipated that more than 17M patients can be treated with more effective ACTs globally in 2015, with between 375 thousand to 885 thousand patients on artesunate in 4 focal countries in 2015, resulting in ~35 thousand lives saved. It is also intended to achieve SRA/WHO dossier submission from 6-7 supply sources for ACTs, new malaria treatments and artesunate by 2015.The anticipated benefits of this work would be: • • • Better health outcomes for patients with malaria and prevention of drug resistance in countries which are sensitive and containment in countries where resistance currently exists (in 4-6 countries), fewer inpatient admissions and drug administration needs for severe malaria More attractive, sustainable market for suppliers of ACT, artesunate, and other new treatments) and greater supply security Potential for expanded business opportunity for Indian/Chinese generic suppliers, supporting developing country economies Based on these market dynamics, significant market impact, access gains and global savings are achievable. However, since the analytics do not support attribution to CHAI engagement, these savings are not added to the total benefits associated with this intervention. Costing The cost of the intervention is $63.1M (£39.4M) over three years. Details are set out in the table below. Components Summary of Costs* by Option Total Costs Total Costs under under intervention counterfactual Maximizing Value for Money and $0 $12.68M Ensuring Sustainable Supply of HIV Treatment (Grant A.1) Accelerating Introduction and Scale Up of $0 $8.16M Point-Of-Care HIV Diagnostics (Grant A.2) Maximising Value for Money of HIV $0 $5.60M Spending for Universal Access to ARVs (Grant A.3) Improving Pricing and Supply Security for $0 $3.72M High Quality MDR-TB Drugs (Grant B.1) Accelerating Market Entry of Highly $0 $7.35M Accurate and Lower Cost New Diagnostic Products (Grant C.1) Increasing Access to Long-Acting $0 $1.43M Contraceptives (Grant D.1) Improving Vaccine Market Dynamics for $0 $2.92M Price Negotiations (Grant D.2) Ensuring Rapid and Sustainable Scale up $0 $6.40M of Supply of Malaria Rapid Diagnostic Tests (Grant E.1) Ensuring Rapid and Sustainable Scale up $0 $5.26M of Supply of Quality Malaria Treatments (Grant E.2) Sharing Lessons and Market $0 $2.53M Assessments TOTAL $0 $56.05M *Assumes overhead of 15% on all project direct costs and 5% on subcontracts The individual components of the proposed portfolio have been reviewed – both by an independent consultant as well as by DFID’s own policy and country-level teams. Net Present Value/Internal Rate of Return Using a 3.5% discount rate (as recommended for global public goods), the NPV of projectable savings from the portfoliois estimated to be $1.45-2.53bn8 for the period 20122020.This is likely to be an underestimate since CHAI are currently only able to project the savings for a subset of components. CHAI’s work in low-cost diagnostics, vaccines, mRDTsand malaria treatment are all expected to generate additional substantial savings. The table below presents, by component,the investment required over the life of the grant (20122015); the NPV of savings over the same period; and the NPV of savings in the longer term (2012-2020). During the project lifetime (2012-2015), these savings, combined with the costs of the associated interventions (a total of $27.55m) yield an internal rate of return (IRR) of441% on the ‘conservative’ scenario and an IRRof585% for the ‘linear scale-up’, and 443% and 586% respectively through to 2020. When the full costs of the project ($56.05m) are included, the IRR is 220% and 305% respectivelyby 2015, rising to 228% and 310% by 2020; this calculation excludes additional savings from the remaining components with un-calculated benefits. Project HIV Treatment (A.1) POC CD4 (A.2) HIV Spending (A.3) MDR-TB (B.1) Low-cost new diagnostics (C.1) LARCs (D.1) Summary of NPV Calculation Cost of investment Savings (2012-2015) (2012-2015) (NPV) Linear Conservative scale-up Conservative Linear scale-up $11.11 $7.10 $153.2M $99.7M $267.3M $99.7M $561.4M $426.9M $1,156.8M $426.0M $4.87 $3.23 $73.0M $39.0M $177.0M $39.0M $361.3M $54.9M $849.8M $54.9M $6.55 $1.25 Not Calculated Not Calculated Not Calculated Not Calculated $14.6M $14.6M $68.6M $68.6M Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated Not Calculated 0 0 0 Vaccines (D.2) $2.54 mRDTs (E.1) Malaria Treatments (E.2) Sharing Lessons Learned Global applied overhead IRR9 NPV $5.71 Not Calculated 8Based Savings (NPV) Not Calculated $4.58 Not Calculated $2.20 $6.92 2012-2020 2012-2020 0 228% (Conservative) – 310% (Linear scale-up) $1.45 Billion - $2.53 Billion on the ‘conservative’ and ‘linear scale-up’ scenarios for ART access respectively savings from 6 areas that have not be estimated 9Excludes Return on Investment As noted above, a Return on Investment (ROI) approach has also been applied to selected components of the proposed intervention. The methodology used was to identify the share of total expected savings on commodities for key multilaterals – based on their expected volume and distribution of future spending. Next, the total share attributable back to the DFID budget could be calculated based on assumptions of UK contributions. The chart below sets out the findings of this analysis. It is estimated that (based on the linear scale-up scenario) the programme will, up to 2020,in discounted savings terms, save $59.2m from DFID’s spend on first-line ART (through GFATM which accounts for a third of all patients on treatment in generically-accessible countries and will save $19.7 million in spend on second-line ART (through which accounts for 60% of adults on second-line treatment). The direct savings to the DFID budget from the ART work alone exceed the total costs of investment of the whole portfolio and mean the intervention has a positive ROI. Based on the savings associated with the ART component alone, the annual ROI of the whole portfolio is 3% over the period 2012-2020. Assessing the ART component alone as a self-standing intervention gives and ROI of 24% a year. Component area First-line Regimens Second-line Regimens Total HIV savings ROI analysis Total Nominal Savings (Linear scale-up) $1,304.1M $145.6M $1,449.8M Nominal Savings on UK spend $73.4M $24.7M $98.1M Sensitivity Analysis A number of stress test were applied to the analysis above. Given the high returns, the intervention is robust to sensitivity analysis. Applying an 8% discount rate reduces the NPV to a range of $1.11-1.94bn. Even if only 10% of the expected savings are achieved, the intervention still has a positive NPV (of $95-204m) (at a 3.5% discount rate). The breakeven price reduction for the intervention is that the portfolio delivers just 3.7%of the forecast savings. In concrete terms, this would mean the intervention accounting for a US$2 decrease in the price of the preferred first-line regimen and a US$4 reduction in the price of second-line treatments. It is also important to stress that the CBA analysis is conservative throughout – not just in terms of the assumptions but also in that it excludes significant potential areas for savings, such as vaccines, where the analytics are not sufficiently robust. However, huge opportunities for price reductions exist for new, emerging and future vaccines such as Japanese Encephalitis, Typhoid, Hexavalent, Malaria and Cholera. As an example, the anticipated annual demand in developing countries for Hexavalent vaccines (combining Diphtheria, Tetanus, Pertussis, Hepatitis B, Polio, and Hib) would be over 200 million doses in the 20152020 period. The potential demand for a new malaria vaccine could be 90 million does over the same time frame. Based on very preliminary discussions with suppliers, it is anticipated that market-shaping interventions for these two vaccines alone could generate almost $195M in savings over the programme period. Given the UK’s high level of investment in vaccines, this would generate very significant savings to DFID’s own budget as well. Monitoring requirements To track progress a number of prices and other information will be required. These include: - HIV treatment (Output Indicator 1.1, 1.2): o Price at baseline and prices monitored every 6 months for ART in genericaccessible countries o Price differential between existing 1L/2L regimen prices ($169 pppy for 1L and $409 pppy for 2L in 2011) and new negotiated price o Expected number of patients requiring drugs from WHO estimates o Actual sales of drugs to intervention countries - MDR-TB drugs (Output Indicator 1.3): o Price at baseline and prices monitored every 6 months for MDR-TB regimens in MDR TB high burden countries o Price differential between existing quality-assured MDR-TB price ($0.64/Cs unit, $2.60/Km unit, $1.45/PAS unit in 2011) and new negotiated price o Expected number of patients requiring drugs from WHO estimates of need and of diagnosed patients o Actual sales of drugs to intervention countries - POC CD4 tests (Output Indicator 2.1): o Price at baseline and prices monitored every 6 months for intervention POC CD4 tests delivered in focal countries o Price differential between existing POC CD4 tests ($11.81 per Pima test in 2011) and new negotiated price. o Wastage rates for conventional and new POC CD4 tests in intervention countries, to generate wastage accounts based on prices o Trend in volumes of intervention and conventional tests procured in intervention countries - HIV spending (Output Indicator 3.1): o Unit cost per person per year (pppy) of HIV service delivery (e.g., ARVs, laboratory costs, personnel, other) at baseline and monitored every 6 months for intervention POC CD4 tests delivered in focal countries o Numbers of patients treated with ARVs in focal countries - LARCs (Output Indicator 4.1): o Price at baseline (average of Jadelle at $22/implant and Implanon at $20/implant in 2011) and prices monitored every 6 months for intervention LARCs delivered in focal countries o Expected number of patients requiring LARCs from WHO estimates o Actual sales of drugs to intervention countries - Vaccines (Output Indicator 5.1, 5.2): o Price at baseline and prices monitored every 6 months for vaccines given in GAVI countries o Wastage rates for conventional and new POC CD4 tests in intervention countries, to generate wastage accounts based on prices o Expected number of patients requiring vaccines from WHO estimates o Actual sales of vaccines to intervention countries - mRDT (Output Indicator 6.1, 6.2): o o o o - Price at baseline and prices monitored every 6 months for mRDT Wastage rates for RDTs and of ACTs (based on appropriate use of drugs) at baseline and in 2015 (methods to be determined) Expected number of patients requiring mRDT and ACT from WHO estimates Actual sales of RDTs to intervention countries Malaria ACTs and injectable artesunate: o Number of price agreements for ACT signed with suppliers o rice at baseline and prices at 2015 for ACT in focal countries o Number of injectable artesunate doses delivered o Estimated number of lives saved based on number of patients given injectable artesunate and mortality rates Endnotes 1Global Fund (2010) Press Release: Agreements Reduce Prices of Malaria Medicines by up to 80%. http://www.theglobalfund.org/en/mediacenter/pressreleases/Agreements_reduce_prices_of_malaria_medicine s_by_up_to_80__/ 2 DFID (2011) “Towards Zero Infections: The UK’s position paper on HIV in the developing world” 3 DFID (2010). “Choices for Women: planned pregnancies, safe births and healthy newborns. The UK’s Framework for Results for improving reproductive, maternal and newborn health in the developing world.” 4 DFID (2010).”Breaking the Cycle: saving lives and protecting the future.The UK’s framework for results for malaria in the developing world.” 5 Cohen MS, et al. Prevention of HIV-1 infection with early antiretroviral therapy. NEJM. 2011;365(6):493-505. 6Nathanson E et al. (2010) MDR tuberculosis–critical steps for prevention and control. N Engl J Med 363: 1050– 1058 7SEAQUAMAT Group. Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet.2005; 366:717-25. 8AQUAMAT.Artesunate versus quinine in the treatment of severe falciparum malaria: a randomized trial. Lancet.2010; 376(9753): 1647-1657. 9 Grace, C (2010) “A Value for Money Perspective Applied to GHI Market Shaping Activities”. DFID HDRC. 10 GAVI (fortcoming) “Shaping Vaccine Markets” 11 MedecinsSansFrontieres (2011). “Untangling the Web of Antiretroviral Price Reductions. 11th Edition” 12 Grace, C (2010) 13 Results for Development (2011) “Market Dynamics Study: Phase II Report for the Global Fund Market Dynamics Committee (MDC)” 14 Results for Development (2011) “Market Dynamics Study: Phase II Report for the Global Fund Market Dynamics Committee (MDC)” 15 GAVI Supply Task Team (unpublished) “Vaccine Supply and Procurement Strategy - Inception Report” 16 ibid 17Narsai, K (2010). “Impact of Regulatory Requirements on Medicines Access in African Countries”. Pharmaceutical Industry Association of South Africa (2010)reports that, while experiences vary by region, over 60% of companies have experienced registration timeframes for some products excess of 2 years, with more than a quarter of companies experiencing processes taking 3 years and more. A majority of respondents also noted that they had actively taken decisions not to register products in some African countries because prospective returns did not merit the costs of complying with national registration and current good manufacturing practice (cGMP) inspection requirements. 18http://www.undp.org/content/undp/en/home/librarypage/mdg/the_mdg_gap_taskforcereport2011/ 19 See WHO (2011). Priority Medicines for Mothers and Children 2011: http://www.who.int/medicines/publications/emp_mar2011.1/en/index.html 20 Hagen, J (2009) “Take Your Medicine,” www.worldpharmaceuticals.net 21 Roberts, A and Imanguli N (2010) “Sexual and Reproductive Health and Rights of Women and Youth in the Context of Climate Change” http://advocatesforyouth.org/climatechange?tmpl=component&print=1&page 22UNDP (2010) “Gender, Climate Change and Community-Based Adaptation” 23 Ozawa S. During the ‘decade of vaccines,’ the lives of 6.4 children valued at $231 billion could be saved. Health Affairs. 2011;30(6):1010-20. http://www.healthaffairs.org/press/2011_06_10.php 24DFID. Choices for women: planned pregnancies, safe births, and healthy newborns. December 2010. http://www.dfid.gov.uk/Documents/prd/RMNH-framework-for-results.pdf 25 Singh S et al (a). Adding it Up: the costs and benefits of investing in family planning and maternal and newborn health. Guttmacher Institute and UNFPA; 2009. 26DFID.Breaking the cycle: saving lives and protecting the future.http://www.dfid.gov.uk/Documents/publications1/prd/malaria-framework-for-results.pdf 27Davis W, Clarke P, Siba P, et al. (2011) Cost-effectiveness of artemisinin combination therapy for uncomplicated malaria in children: data from Papua New Guinea. Bulletin of the World Health Organization 2011;89:211-220. doi: 10.2471/BLT.10.084103 28Resch S, Korenromp E, Stover J, Blakley M, Krubiner C, et al. (2011) Economic Returns to Investment in AIDS Treatment in Low and Middle Income Countries.PLoS ONE 6(10): e25310. doi:10.1371/journal.pone.0025310 29 Senior level salaries are set out, alongside other financial information, in the IRS 990 form available at: http://www.clintonhealthaccess.org/files/CHAI_2010_Form_990.pdf