Retrospective Protocol Template - Rutgers Biomedical and Health
advertisement

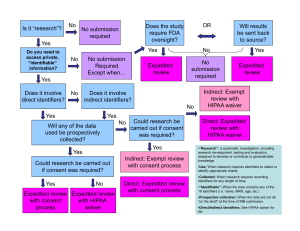
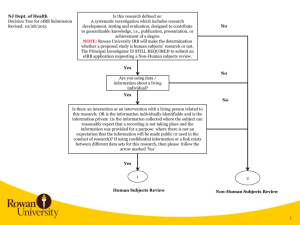
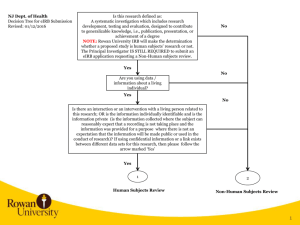
Study Title: Principal Investigator: Study Sites: (should be the same as the eIRB application) 1. Purpose/Specific Aims Clearly state the overall purpose of the study. [Note: IRB reviewers come from a diversity of backgrounds. Therefore, avoid the use of acronyms and highly technical language.] 1.1 Objectives Create objectives—statements outlining specifically what will be achieved by the study— that derive directly from the overall purpose. 1.2 Hypotheses Express scientific hypotheses—statements about expected relationships between variables— that are testable and that include measureable outcomes/endpoints as described in the Research Design and Methods section of the protocol. Hypotheses must correspond directly to the objective(s). 2. Background and Significance Provide a succinct review of the relevant scientific literature to justify the proposed study. Include key references but not a complete literature review. Include relevant preclinical data, such as animal studies or other human studies utilizing similar drugs, devices, procedures, leading up to, and supporting the proposed research, if applicable. Address the importance of the knowledge that may reasonably be expected to result for your discipline (e.g., clinical, diagnostic, etc.) and to society generally (e.g. increased understanding of disease, etc.). 3. Research Design and Methods Describe the design of the study. A retrospective record review means that the data are already in existence when the project is submitted for IRB review (already collected for nonresearch purposes). 3.1. Date range for record review (end date must precede IRB submission date) Dates must be the same on the eIRB application For example: Record review from 1/1/2009 through 12/30/2014 Determine how long it will take to complete the study including data analysis For example: the study will take 2 years to complete. Protocol Version 1.0 - mm/dd/yy Add protocol title PI name 3.3 Sample Size: Estimate the number of records you plan to review, should be the same as eIRB application. 3.4 Record Selection Outline inclusion and exclusion criteria for records. Provide details on how records will be identified and by whom (For example: are you querying a data base or is someone providing a list and if so whom. You may use titles rather than specific names in the protocol to minimize amendments due to personnel changes. (e.g., the Surgical Information Systems Manager for the RWJUH Operating Room Trauma Registry). 3.4.1 Inclusion Criteria Describe characteristics for the selection of records. Provide all relevant demographic (e.g., age, ethnicity), biomedical (e.g., disease status, laboratory values, pregnancy) and behavioral characteristics (e.g., cognitive abilities, mood) relevant for inclusion and exclusion. 3.4.2 Exclusion Criteria Describe what relevant demographic, biomedical or behavioral characteristics exclude records from the research. 4. Study Variables (Variables listed here should include those listed on your data collection tools. 4.1 Independent Variables or Interventions Indicate what variables you will be using to compare groups (e.g., past treatments) or what variables you will use to predict outcomes (e.g., demographics). 4.2 Dependent Variables or Outcome Measures Clearly define the outcomes/variables you will be extracting from the record with justification for the information collected as it relates to your objectives 4.3 Chart Review Process: Data Abstraction: Provide a complete list of all electronic data bases and/or paper records to be reviewed and who will perform the review. Suggest listing study role instead of actual name to minimize amendments due to personnel change. Indicate the source or location of the data bases and records If more than one database is being used, list the variables used to link the databases and who on the study team will manage the data List any data management services you may use (e.g., RedCap) Should be the same as listed on the eIRB application. Note that patient lists and/or protected health information ‘preparatory to research’ is to determine the feasibility of a particular research idea; such information may not be used to conduct research. Page 2 of 4 Add protocol title PI name Note that data containing Protected Health Information cannot be stored on personal or mobile devices such as laptops and memory sticks. Coded Identifier List (link between unique record code generated for the research and identifiers needed for the record review (e.g., DOB, MRN, name, dates of services). List all identifiers you will collect in this file and the unique ID you will assign as associated with the record, Should be the same as listed on the eIRB application Explain when and how this file with the link between personal identifiers and the data will be destroyed. (NOTE) If a waiver of consent is granted, identifiers should be destroyed with no possibility of linking the data with these identifiers as soon as possible after data are transcribed and verified for accuracy. This affords respect and privacy protections to persons whose data was reviewed/used without their consent. In contrast to destroying the link, Rutgers University requires that the data set on which you perform analyses and develop manuscripts must be kept for a minimum of 6 years. Data Collection Form (de-identified variables to be analyzed): Include a data abstraction sheet or excel file with the unique record code you have assigned and all data elements or variables you will collect from each record (this data form should have no identifiers). In the case that you are obtaining information with identifiers (names, dates, etc.) from a database and then removing identifiers please provide both tools/sheets (with and without identifiers) so it is clear that you are removing all identifiers. 4.4 Risks of Harm Generally, the risk for record review studies is likely breach of confidentiality. Address the steps to be taken to minimize this risk including timely destruction of the link between identifiers and health data 4.5 Potential for Benefit The individuals whose records you are reviewing are not likely to receive any benefit from the research. However, society and the investigators may benefit from the knowledge gained. 4.6 Consent Procedures Record review studies require a waiver of consent and hippa authorization waiver and justification for those waivers to include the following: 1) determination of the risk involved 2) the reasons why the waiver will not adversely affect the rights and welfare of the individuals and 3) why the research could not reasonably be carried out without Page 3 of 4 Add protocol title PI name the waiver. This should correspond with the e-irb application section for informed consent. 5. Statistical Analysis Describe the statistical methods to be employed. Clinical relevance of the results as well as statistical significance should be discussed. 6. Reporting Results 6.1 Professional Reporting Describe your plan to share the results of your research with the scientific community. 7. Bibliography Include all references cited in the text. Page 4 of 4