SEASONAL HABITAT USE DYNAMICS OF THE MUSKRAT
advertisement

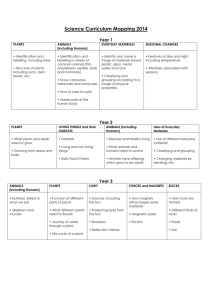
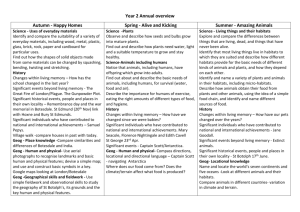
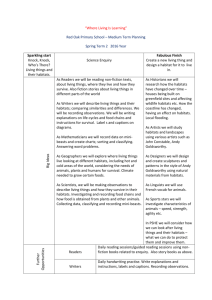
RH: McDonald---Habitats of muskrats in Oklahoma USE OF HABITAT DURING DROUGHT BY THE COMMON MUSKRAT (ONDATRA ZIBETHICUS) IN SOUTHWESTERN OKLAHOMA BRANDON MCDONALD Department of Biology, Midwestern State University, Wichita Falls, TX 76308 Present Address: Department of Biological Sciences, Cameron University, Lawton, OK 73505 Correspondent: bmcdonal@cameron.edu ABSTRACT---Status of the common muskrat (Ondatra zibethicus) in the southern High Plains and western Rolling Plains has remained uncertain due to a scarcity of records. I examined diversity of riparian habitats, frequency of occurrence of habitats, availability of habitats, and use of habitats by muskrats within an irrigation drainage system in southwestern Oklahoma during prolonged drought. Six major habitats were supported in 31 km of drainage channels. These included, in order of descending frequency, emergent cattails, prairie sedge meadow, cattail-forested transition, forest, turf grass, and pioneer mudflat. Of habitats in drainage systems, 60% were available for muskrats as defined by presence of water. Availability differed among habitats; prairie sedge meadow had highest availability and emergent cattails was the lowest in availability. Use of habitats differed significantly among habitats and was driven by availability. RESUMEN---El estatus de las almizcleras (Ondatra zibethicus) en las altiplanicies meridionales y las planicies ondulantes occidentales se ha mantenido incierto debido a la escasez de récordes. Yo examiné la diversidad de hábitats ribereños, la frecuencia de ocurrencia de diferentes tipos de hábitats, la disponibilidad de dichos tipos de hábitats, y el uso por las almizcleras de un sistema de drenaje de irrigación en el suroeste de Oklahoma durante un periodo de sequía prolongado. Treinta y un kilómetros de canales de drenaje sostenían una riqueza de seis tipos principales de hábitats; incluyendo en orden de frecuencia descendiente: anea emergente, juncia de prado, anea en transición a forestación, anea forestada, césped y marisma pionera. El 60% de los hábitats del sistema de drenaje fueron clasificados como disponibles para las almizcleras de acuerdo a la presencia de agua. La disponibilidad difería entre los tipos de hábitats; la juncia de prado tenía la disponibilidad más alta mientras que la anea emergente tenía la más baja. El uso de los hábitats por las almizcleras variaba significativamente entre los tipos de hábitats, y era determinado por su disponibilidad. The common muskrat (Ondatra zibethicus) is the most widespread and abundant semi-aquatic fur-bearer in North America. Its extensive geographical range coincides with an abundance of suitable wetland habitats (Wilner et al., 1980; Parker and Maxwell, 1984; Dalquest and Schultz, 1992; Clark and Kroeker, 1993; Virgil and Messier, 1996; Engeman and Whisson, 2003). Arid regions of North America where, rainfall is annually and seasonally variable, watercourses often are ephemeral, and permanent bodies of water are few and isolated, mark the southwestern boundaries of the geographic range of muskrats. Southwestern populations persist in isolated habitats such as desert water holes, man-made reservoirs, and agricultural irrigation systems (Dixon, 1922; Storer, 1937; Errington, 1951). Isolation of habitats is attributed to a lack of wetland corridors across southwestern margins of the range (Bolen et al., 1989). Verified records of muskrats in the High Plains-Rolling Plains interface are few and isolated in Texas (Bailey, 1905; Jones et al., 1988; Stangl et al., 1989; Dalquest and Schultz, 1992; Schmidly, 2004a) and Oklahoma (Caire et al., 1989; Braun and Relevez, 2005; McDonald, 2006a). During the 20th century, muskrats largely have disappeared from tributaries of the Pecos and Canadian rivers in western and habitats in northern Texas, respectively (Schmidly, 2004b; F. B. Stangl, pers. comm.) McDonald (2006b) suggested that gradients of abundance of muskrats from east to west and northeast to southeast, as previously described by Caire et al. (1989), are associated with large-scale spatial variations in environmental factors (e.g., average annual precipitation) across Oklahoma. Seasonal variation in rainfall in western portions of Oklahoma contributes to temporally narrow corridors of dispersal and seasonal fluctuations in otherwise suitable habitats. Combinations of natural effects and anthropogenic alterations of the landscape likely have left western populations disjunct (McDonald, 2006b), although viability of populations and connectivity of suitable habitats remain speculative (McDonald, 2006a). Glass (1952) noted that muskrats in Oklahoma are of interest to researchers examining the ecology of a species in marginal environmental conditions. Drought in southwestern Oklahoma that peaked in 2006, afforded an opportunity to investigate ecological characteristics of a peripheral population of muskrats in marginal conditions. I examined an irrigation drainage system and described the diversity of riparian wetland habitats, relative frequencies of habitats, availability of habitats, and use of habitats by muskrats. MATERIALS AND METHODS---Study Area---The Blair irrigation drainages are human-made channels lying parallel and perpendicular to the Altus-Lugert Main Irrigation Canal in and around the town of Blair, Jackson County, Oklahoma (Fig. 1). Nine drainages total ca. 31 km of drainage channels. Channels are steep banked (50-90◦) and reach heights at banks of 6 m. Drainages function in collecting excess surface water from occasional heavy rainfall and overflows of the main irrigation canal; thereby, preventing damage from floods to property, soil, and crops near Blair. The drainages have existed as permanent sources of water since the early 1940s, when the United States Bureau of Reclamation constructed the Altus-Lugert Irrigation District. Drainage channels now support riparian habitats and associated wetland flora and fauna. Classification of Habitat---I randomly selected 75-m sampling transects to represent every 200-m length of each drainage channel. I randomly assigned two sampling points per transect, each intersecting the channel by 90º allowing for a cross-section measurement of 34 environmental variables within the drainage channel and extending into the bank-side herbaceous belt. Most variables estimated were regarded previously as significant to suitability of habitat for muskrats (Wilner et al., 1980; Nadeau et al., 1995). Environmental variables included height of bank, slope of bank, cover of bank-side by grasses, cover of bank-side by forbs, cover of bank-side by woody plants, cover of bankside by litter, cover of bank-side by bare soil, width of bank-side herbaceous belt, cover of bank-side over-story canopy, width of channel, width of water, depth of water at bank, depth of water between bank and center of channel, depth of water at center of channel, greatest depth of water, percentage water capacity of channel, percentage of open water (i.e., no cover by aquatic vegetation), cover of submerged vegetation at bank, cover of submerged vegetation at ¼ of channel, cover of submerged vegetation at center of channel, cover of floating vegetation at bank, cover of floating vegetation at ¼ of channel, cover of floating vegetation at center of channel, cover of emergent sedge-rush-grass vegetation at bank, cover of emergent sedge-rush-grass vegetation at ¼ of channel, cover of sedge-rushgrass vegetation at center of channel, cover of cattails at bank, cover of cattails at ¼ of channel, cover of cattails at center of channel, cover of over-story canopy at center of channel, distance to nearest tree, diameter at breast height (dbh) of nearest tree, distance to nearest neighboring tree, and dbh of nearest neighboring tree. I used a 1-m² PVC frame to estimate values of surface cover. I estimated cover of overstory canopy with the aid of a handheld densiometer (Model-C, R. Lemmon Forest Densiometers, Bartlesville, Oklahoma). I used a 2-m PVC rod marked with 0.1-m increments to measure depth of water, height of bank, and dbh. Principal-components analysis was used to determine which variables significantly contributed to overall variation among transects. Loadings on components >0.30, with >100 samples, were accepted as significant, equivocally at the 99% confidence level (i.e., P < 0.01; following Hair et al., 1987; Tabachnick and Fidell, 1989; McCarigal et al., 2000; Perez-Neto et al., 2003). I used results of principal-components analysis and qualitative notes to determine major habitats present within the drainage system. I assigned titles to habitat categories following a plant-functional-group method similar to Harris and Marshall (1962), Day et al. (1988), and Galatowitsch and van der Valk (1996). I used a handheld global positioning system (Garmin eTrex - Legend, Garmin International, Olathe, Kansas) to assign latitude and longitude coordinates of sampling points along each of the drainages. Coordinates in conjunction with United States Geological Survey topographical maps were used to precisely record all pertinent spatial information. Relative Frequency of Habitats---Based on results of principal-components analysis and qualitative data, I subdivided the drainage system into the following habitats: emergent cattails (thick cattails), prairie sedge meadows (thick cover of grasses, sedges, rushes), forest (thick cover by over-story canopy), cattail-forest transition (cattaildominated channel with minimal but significant cover by over-story canopy), pioneer mudflat (high erosion potential and bank-side vegetation sparse and dominated by early succession taxa), and turf-grass (bank-side cover of Bermuda grass Cynodon dactylon). These six habitats parallel the marsh community, drought-induced, model of gradient succession of Harris and Marshall (1962), where sequence of community seres is 1) annual weeds, 2) perennial emergents, 3) composite of annuals, perennials, and woody plants, and 4) woody plants (a list of plants in each habitat is available from the author). I assigned transects to appropriate habitats and calculated total counts for transects in each habitat. Because number of transects in each habitat were unequal, I used relative frequencies. I calculated relative frequencies by dividing number of transects of each habitat by total number of transects in the drainage system. Values of relative frequency were square-root-arcsine transformed as outlined by R. High (http://www.uoregon.edu/~robinh/statistics.html) and compared with chi-squared analysis. All statistical analyses were performed with PAST version 1.39 (Hammer et al., 2001). Availability of Habitats---Availability was defined as transects with a minimum depth of water of 0.10 m. Optimal depths of water for inhabitance by muskrats are 0.151.2 m (Allen and Hoffman, 1984; Schmidly, 2004b); however, based on prior experience, 0.10 m was judged minimally suitable for a combination of dispersal activities and residency. I divided number of available transects within each habitat by total number of transects (available and unavailable) within each habitat to obtain percentage availability to be compared among all habitats. I square-root-arcsine transformed data on availability of habitat and compared relative availability among habitats with chi-squared analysis. Use of Habitats---I examined each transect for evidence of muskrats to diagnostically indicate use of habitats. Verifiable evidence included burrows, feeding stations, and tracks. This census method has been employed routinely and is regarded as non-intrusive, cost-effective, and labor efficient (Errington, 1940; Sprugel, 1951; Brooks and Dodge, 1986; Nadeau et al., 1995). I recorded burrows and evidence of feeding as old or recent. Active burrows were characterized by runways leading to the entrance, as well as proximal recent evidence of feeding (middens). I visually estimated age of feeding evidence as recent or old based on degree of desiccation and color of plant remains. I recorded counts and GPS coordinates of burrows and evidence of feeding for each transect. I plotted recent and old evidence on a hand-drawn map of the Blair drainage system to depict an overall pattern of activity within the drainage system. I pooled counts of burrows and evidence of feeding, both recent and old, for each habitat. I calculated relative frequency of evidence by dividing number of stations with evidence for each habitat by total count of stations with evidence in the drainage system. Values for percentage frequency were square-root-arcsine transformed and compared across habitats with chi-squared analysis. I used linear-regression analyses to examine relationships between frequencies of evidence of muskrats among habitats (response variable), where predictor variables included frequency and availability of habitat . RESULTS---Classification of Habitats---Principal-components analysis yielded eight variables that contributed to significant variation among 120 transects. The first two components (Fig. 2) explained 62% of variation among transects. The first three components collectively explained 77% of variation. Loadings on component 1 were dominated by open water (0.55), cover of cattails at bank (-0.53), cover of cattails at center of channel (-0.50), and distance to nearest tree (-0.31). Loadings on component 2 were dominated by distance to nearest tree (-0.83) and bank-side over-story canopy (0.31). Loadings on component 3 were highest for cover by bank-side grasses (0.63), cover of emergent sedge-rush-grass vegetation (0.48), cover of bare soil (0.36), and bank-side overstory canopy -0.35). Relative Frequency of Habitats---Habitats significantly differed (X ² = 24.07, df = 5, P < 0.01) in relative frequency of occurrence throughout the drainage system (Fig. 3). Emergent cattails (40% of total) was the most abundant habitat, while pioneer mudflat (5%) and turf grass (6%) habitats were the rarest habitats in the drainage system. Availability of Habitats---Within the Blair drainage system (31 km), 60% (18.5 km) was available for habitation by muskrats (Fig. 1). Values for relative availability differed significantly (X ² = 20.5, df = 5, P < 0.01) among the six habitats, prairie sedge meadows (96% availability) were more available than all other habitats (Fig. 3). Use of Habitats---A total of 21 old and 17 recent stations with evidence of muskrats was identified within the drainage system (Fig. 1). Drainages a and b had evidence of feeding, but no recent or abandoned burrows. Evidence of feeding was accompanied by burrows throughout the remainder of drainages in the system (i.e., drainages c- i). Relative frequencies of evidence differed significantly among the six habitats (X ² = 13.2, df = 5, P < 0.03), with prairie sedge meadows accounting for the most use by muskrats (Fig. 3). Results of linear regression indicated a non-influential relationship between evidence of muskrats and frequency of habitat (r² = 0.09, P = 0.55). A significant model was revealed between evidence of muskrats and availability of habitat (r² = 0.87, P < 0.01). DISCUSSION---Habitat heterogeneity in the Blair drainage system is due to a disturbance regime including anthropogenic and natural disturbances. However, drought (a natural disturbance) is the most influential factor on prairie-wetland ecosystems (Shay and Shay, 1986) and central to the theme of my study. Due to drought, only 60% of drainage channels provided habitat for muskrats (Fig. 1). Such drawdowns (Kroll and Meeks, 1985; Shay and Shay, 1986; Thurber et al., 1991) initially are driven by belowaverage rainfall, but subsequently compounded by prevalence of cattail-dominated habitats. Cattails gain a competitive advantage over other wetland plants during drought and rapidly form monotypic stands (Harris and Marshall, 1962; Urban et al., 1993; Shay and Shay, 1986; Gunderson, 2001). Once a monotypic community of cattails is established, rates of transpiration increase, thereby, further reducing available water (Brenzy et al., 1973; Crundwell, 1986; Sanchez-Carrillo, 2004). The high frequency of occurrence of habitats with emergent cattails (40% of total) combined with decreased availability (only 40% available) illustrates fragmentation of habitats within the drainage system. Other habitats with water were separated by extensive stretches of dry habitats with emergent cattails. Muskrats seeking suitable habitat are forced to travel without cover of water, thereby, exposing them to increased risks of predation (Errington, 1940; Sprugel, 1951; Erickson, 1963; Thurber et al., 1991; Clark and Kroeker, 1993). Peripheral populations suffer increased mortality from predation under fluctuating water levels and dependence on marginal habitat for cover (Boyce, 1978; Messier et. al., 1990; Virgil and Messier, 1996, 2000). Stability of water level is the primary limiting factor for populations of muskrats (Sather, 1958; Errington et al., 1963; Clark and Kroeker, 1993; Virgil and Messier, 1996, 2000). Distributional patterns of evidence of muskrats combined with results of linear regression indicate that availability of habitat (i.e., presence of water) is a more influential limiting factor than structure of plant community in drought conditions. Density of cattails often is emphasized as a vital component of quality of habitat (Allen and Hoffman, 1984), but results regarding how cattail marshes influence peripheral populations of muskrats are absent. Populations in cattail marshes proliferate rapidly and can sustain high mortality (Bailey, 1937), and are thus, ecologically significant source habitats. However, most research has taken place in central regions where environmental conditions are most favorable and large-scale, source-sink, population dynamics and general models of habitat selection apply. Such results might not apply to peripheral populations in marginal conditions, such as those throughout the High Plains-Rolling Plains interface. Muskrats in my study were restricted to isolated habitats, of which 70% were prairie sedge meadows. Muskrats throughout these habitats were established residents as indicated by burrows associated with evidence of feeding. Prairie sedge meadows offered a rich plant community for foraging, although year-round cover provided by dense cattails was lacking. Activities of muskrats in other isolated habitats without significant cover of cattails (e.g., turf grass) support the conclusion that availability drives use of habitats in marginal conditions. Previous authors have suggested that muskrats searching for habitat in average conditions (Errington, 1951; Shanks and Arthur, 1952) and during drought (Kroll and Meeks, 1985) are influenced primarily by continuation of water, leaving composition of plant communities as a secondary and possibly indiscriminate factor in the search. Evidence of feeding by muskrats within drainages a and b were remnants of dispersing individuals based on absence of established burrows, burrowing activity, or temporary shelters indicative of residency or attempted short-term occupancy (Errington, 1939, 1940; Sprugel, 1951). Muskrats in western Oklahoma disperse in autumn (McDonald, 2006a) and those in drainages a and b likely were moving in response to seasonal dispersal cues (Beer and Meyer, 1951), drought (Takos, 1944), or both. Because drainages a and b were 1,400-2,300 m from the nearest water (over land), it is unlikely that resident muskrats from an independent water source would traverse such a distance on regular feeding bouts. However, dispersing muskrats would be attracted to drainages a and b, due to presence of water. Muskrats travel significant distances over land only when driven by dispersal cues (Errington, 1940; Sprugel, 1951; Shanks and Arthur, 1952; Errington et al., 1963; Thurber et al., 1991). Adversities that muskrats face in drought conditions have been illustrated throughout the range of this species (e.g., Bellrose and Low, 1943; Errington, 1939, 1963; Boyce, 1978; Virgil and Messier, 1996). High fecundity, adequate habitats, and dependable dispersal corridors, minimize potential local extirpation for populations in central regions. However, peripheral populations exist in continually marginal environmental conditions (Glass, 1952), which include isolation at the landscape level, lessened viability of local populations through extirpations, temporally narrow corridors for dispersal, fluctuations in otherwise suitable habitats, and increased predation facilitated by exposure during frequent drawdown of water (Sojida and Solberg, 1993; McDonald, 2006b). I thank the Altus-Lugert Irrigation District and landowners in Jackson County for access to properties. Gratitude for critical reviews and assistance with this manuscript is conveyed to F. Stangl, R. Vogtsberger, M. Shipley, M. Rincon-Zachary, M. Husak, and M Santiago. Financial support was provided by the Texas Academy of Science. LITERATURE CITED ALLEN, A. W., AND R. D. HOFFMAN. 1984. Habitat suitability index models: muskrat. United States Fish and Wildlife Service, FWS/OBS-82/10.46:1-27. BAILEY, V. 1905. Biological survey of Texas. North American Fauna 25:1-222. BAILEY, V. 1937. The Maryland muskrat marshes. Journal of Mammalogy 18:350-354. BEER, J. R., AND R. K. MEYER. 1951. Seasonal changes in the endocrine organs and behavior patterns of the muskrat. Journal of Mammalogy 32:173-191. BELLROSE, F. C., AND J. B. LOW. 1943. The influence of flood and low water levels on the survival of muskrats. Journal Mammalogy 24:173-188. BOLEN, E. G., L. M. SMITH, AND H. L. SCHRAMM, Jr. 1989. Playa lakes: prairie wetlands of the southern High Plains. BioScience 39:615-622. BOYCE, M. S. 1978. Climatic variability and body size variation in the muskrat (Ondatra zibethicus) of North America. Oecologia (Berlin) 36:1-19. BRAUN, J. K., AND M. A. REVELEZ. 2005. Distributional records and comments on Oklahoma mammals. Texas Journal of Science 57:2-24. BRENZY, O., I. MEHTA, AND R. K. SHARMA. 1973. Studies on evapotranspiration of some aquatic weeds. Weed Science 21:197-204. BROOKS, R. P., AND W. E. DODGE. 1986. Estimation of habitat quality and summer population density for muskrats on watershed basis. Journal of Wildlife Management 50:269-273. CAIRE, W., J. D. TYLER, B. P. GLASS, AND M. A. MARES. 1989. Mammals of Oklahoma. University of Oklahoma Press, Norman. CLARK W. R., AND D. W. KROEKER. 1993. Muskrat population dynamics in experimental marshes at Delta, Manitoba. Canadian Journal Zoology 71:1620-1628. CRUNDWELL, M. E. 1986. A review of hydrophyte evapotransipration. Revue d’Hydrobiologie Tropicale 19:215-232. DALQUEST, W. W., AND G. E. SCHULTZ. 1992. Ice age mammals of northwestern Texas. Midwestern State University Press, Wichita Falls, Texas. DAY, R. T., P. A. KEDDY, J. MCNEIL, AND T. CARELTON. 1988. Fertility and disturbance gradients: a summary model for riverine marsh vegetation. Ecology 69:10441054. DIXON, J. 1922. Rodents and reclamation in the Imperial Valley. Journal of Mammalogy 3:136-146. ENGEMAN, R. M., AND D. A. WHISSON. 2003. A visual method for indexing muskrat populations. International Biodeteriation and Biodegradation 52:101-106. ERICKSON, H. R. 1963. Reproduction, growth, and movement of muskrats inhabiting small water areas in New York State. New York Game and Fish Journal 10:90117. ERRINGTON, P. L. 1939. Reactions of muskrat populations to drought. Ecology 20:168186. ERRINGTON, P. L. 1940. Natural restocking of muskrat-vacant habitats. Journal of Wildlife Management 4:173-185. ERRINGTON, P. L. 1951. Concerning fluctuations in populations of the prolific and widely distributed muskrat. American Naturalist 85:272-292. ERRINGTON, P. L. 1963. Muskrat populations. Iowa State University Press, Ames. ERRINGTON, P. L., R. J. SIGLIN, AND R.C. CLARK. 1963. The decline of a muskrat population. Journal Wildlife Management 27:1-8. GALATOWITSCH, S. M., AND A. G. VAN DER VALK. 1996. The vegetation of restored and natural prairie wetlands. Ecological Applications 6:102-112. GLASS, B. P. 1952. Factors affecting the survival of the plains muskrat, Ondatra zibethicus cinnamomina, in Oklahoma. Proceedings of the Oklahoma Academy of Science 10:484- 491. GUNDERSON, L. H. 2001. Managing surprising ecosystems in southern Florida. Ecological Economics 37:371-378. HAIR, J. F., R. E.ANDERSON, AND R. L. TATHUM. 1987. Multivariate data analysis. Second edition. Macmillan, New York. HAMMER, Ø., D. A. T. HARPER, AND P. D. RYAN. 2001. PAST: palaeontological statistics software package for education and data analysis. Palaeontological Electronica 4:9. HARRIS, S. W., AND W. H. MARSHALL. 1962. Ecology of water-level manipulations on a northern marsh. Ecology 44:331-343. JONES, J. K., R. W. MANNING, C. JONES, AND R. R. HOLLANDER. 1988. Mammals of the northern Texas Panhandle. Occasional Papers, The Museum, Texas Tech University 126:1-54. KROLL, R. W., AND R. L. MEEKS. 1985. Muskrat population recovery following habitat re-establishment near southwestern Lake Erie. Wildlife Society Bulletin 13:484486. MCCARIGAL, K., S. CUSHMAN, AND S. STAFFORD. 2000. Multivariate statistics for wildlife and ecology research. Springer Science-Business Media Inc., New York. MCDONALD, B. K. 2006a. New records of occurrence and notes on the muskrat (Ondatra zibethicus) in western Oklahoma. Proceedings of the Oklahoma Academy of Science 86:93-95. MCDONALD, B .K. 2006b. Distribution, abundance, and habitat affinities of Oklahoma muskrats (Ondatra zibethicus): new insight from trapper reports. Proceedings of the Oklahoma Academy of Science 86:39-45. MESSIER, F., J. A.VIRGIL, AND L. MARINELLI. 1990. Density-dependent habitat selection in muskrats: a test of the ideal free distribution model. Oecologia (Berlin) 84:80385. NADEAU, S., R. DECAIRE, D. LAMBERT, AND M. ST-GEORGES. 1995. Nonlinear modeling of muskrat use of habitat. Journal of Wildlife Management 59:110-117. PARKER, G. R., AND J. W. MAXWELL. 1984. An evaluation of spring and autumn trapping seasons for muskrats in eastern Canada. Canadian Field-Naturalist 98:293-304. PEREZ-NETO, P. R., D. A JACKSON, AND K. M. SOMERS. 2003. Giving meaningful interpretation to ordination axes: assessing loading significance in principal component analysis. Ecology 84:2347-2363. SANCHEZ-CARRILLO, S., D. G. ANGELER, M. ALVEREZ-COBELAS, AND J. GARATUZAPAYA. 2004. Evapotranspiration in semi-arid wetlands: relationships between inundation and the macrophyte-cover: open-water ratio. Advances in Water Resources 27:643-655. SATHER, H. J. 1958. Biology of the Great Plains muskrat in Nebraska. Wildlife Monographs 2:1-35. SCHMIDLY, D. J. 2004a. Texas natural history: a century of change. Texas Tech University Press, Lubbock. SCHMIDLY, D. J. 2004b. The mammals of Texas. University of Texas Press, Austin. SHANKS, C. E., AND G. C. ARTHUR. 1952. Muskrat movements and population dynamics in Missouri farm ponds and streams. Journal of Wildlife Management 16:138-148. SHAY, J. M., AND C. T. SHAY. 1986. Prairie marshes in western Canada, with specific reference to the ecology of five emergent macrophytes. Canadian Journal of Botany 64:443-454. SOJIDA, R. S., AND K. L. SOLBERG. 1993. Management and control of cattails. Waterfowl Management Handbook, United States Fish and Wildlife Service Leaflet 13.4.13:18. SPRUGEL, G. 1951. Spring dispersal and settling activities of central Iowa muskrats. Iowa State College Journal of Science 26:71-84. STANGL, F. B., S. KASPER, AND T. SCHAFER. 1989. Noteworthy range extensions and marginal distribution records for five species of Texas mammals. Texas Journal of Science 41:436-437. STORER, T. I. 1937. The muskrat as native and alien. Journal of Mammalogy 18:443-460. TABACHNICK, B. G., AND L. S. FIDELL. 1989. Using multivariate statistics. Second edition. Harper and Row, New York. TAKOS, M. J. 1944. Summer movements of banded muskrats. Journal of Wildlife Management 8:307-311. THURBER, J. M., R. O. PETERSON, AND T. D. DRUMMER. 1991. The effect of regulated lake levels on muskrats, Ondatra zibethicus, in Voyageurs National Park, Minnesota. Canadian Field-Naturalist 105:34-40. URBAN, N. H., S. M. DAVIS, AND N. G. AUMAN. 1993. Fluctuations in sawgrass and cattail densities in the Everglades Water Conservation Area 2A under varying nutrient, hydrological, and fire regimes. Aquatic Botany 46:203-223. VIRGIL, J. A., AND F. MESSIER. 1996. Population structure, distribution, and demography of muskrats during the ice-free period under contrasting water fluctuations. Ecoscience 3:54-62. VIRGIL, J. A., AND F. MESSIER. 2000. Assessment of source-sink theory for predicting demographic rates among habitats that exhibit temporal changes in quality. Canadian Journal of Zoology 78:1483-1493. WILNER, G. R., A. FELDHAMER, E. E. ZUCKER, AND J. A. CHAPMAN. 1980. Ondatra zibethicus. Mammalian Species 141:1-8. Submitted 14 September 2007. Accepted ????????????. Associate Editor was Philip D. Sudman. FIG. 1---Drainage system near Blair, Jackson County, Oklahoma: Altus-Lugert Main Irrigation Canal (black and white line oriented north-south), drainages (solid lines ai), available habitats in drainage (wide corrugated line), recent evidence of muskrats (solid circles), and old evidence of muskrats (open circles). FIG. 2---Scatterplot of principal components 1 and 2 calculated from a study of habitats used by common muskrats (Ondatra zibethicus) near Blair, Jackson County, Oklahoma. Vectors (dashed lines) represent significant loadings of variables on component axes: A, cover of cattails at bank; B, cover of cattails at center of channel; C, cover by over-story at bank-side; D, cover of overstory in center of channel; E, open water; F, distance to nearest tree. Triangles indicate transects in relation to respective component axes and vectors of variables. FIG. 3---Histograms displaying a) frequency of occurrence, b) availability, and c) use of habitats by common muskrats (Ondatra zibethicus) near Blair, Jackson County, Oklahoma: EC, emergent cattails; F, forested; CF, cattail-forested transition; PSM, prairie sedge meadow; PM, pioneer mudflat; TG, turf grass. Figure 1 Figure 2 Figure 3