Nuclear Extract Protocol:
advertisement

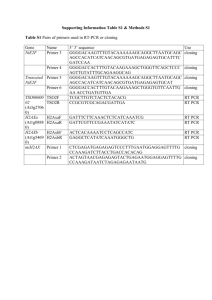
Nuclear Extract Protocol: Buffers for 6 livers: 90 ml Homogenization Buffer Protease Inh. None DTT 90 µl (1 mM) PMSF 450 µl (200 mM) 195 ml Cushion Buffer Protease Inh. None DTT 195 µl PMSF 975 µl 60 ml Nuclear Lysis Buffer Protease Inh. None DTT 60 µl PMSF 300 µl Stock of 200 mM PMSF prepared with isopropanol, final concentration is 0,1 mM On ice: 1. Cut livers into small pieces using scissors 2. Add 6 ml Homogenization Buffer (8ml/liver! Don’t forget to add DTT and PMSF to the buffer!) 3. Dounce with 10 strockes, cool on ice and apply another 10 strockes 4. Filter in 50 ml tube through 2 layer of presoked gaze in HomoBuffer, change gaze if necessary to get rid of cell debris 5. add HomoBuffer to 8 ml/liver (so total 48 ml for 6 livers) 6. Mix homogenate with 2 volumes of Cushion Buffer (96 ml) 7. add 10 ml of Cushion Buffer to the bottom of the centrifuge tube (do not touch the sides!!!) 8. Split the homogenate into the tubes (run it slowly down the side of the tube) 9. balance the tubes (within 0,05 g of each other) 10. centrifuge @ 24000 rpm for 1 h @ 1ºC (SW-32-TI) take 1 ml sample from the supernatant for Western analysis 11. Dry in cold room inverted for 10 min 12. Suspend the pellet in 10 ml Nuclear Lysis Buffer/liver (re-suspend in 1-2 ml first, transfer the solution into the homogenizer, don’t decant!!! wash the bottom of the tube with the rest of the buffer) 13. apply 10-15 strokes with pestle A to homogenize the pellet and lyse the cells Robertson Lab 14. Transfer the nuclei into a small beacon with a stirring bar and bring up the volume to 60 ml (for 6 livers) 15. add drop wise 1/20 volume (3 ml) of saturated (NH4)2SO4 over 25 min, on ice! 16. centrifuge @ 20000 rpm for 30 min @ 1 ºC (SS-34), keep supa 17. add 0,4 g/ml of finely powdered (NH4)2SO4 over 25 min on ice! 18. centrifuge for 1 h @ 24000 rpm @ 1ºC (ultracentrifuge) 19. Suspend the pellet in 3 ml of Dialysis Buffer 20. Dialyse 3 ml of NE against 100 ml of Dialysis Buffer, in the cold room under stirring 21. Change dialysis buffer after 2-4 h, repeat after another 4 h 22. centrifuge @ 1 ºC (Eppendorf) for 10 min 23. collect supernatant 24. Measure concentration (Bredford 2µl, 5 µl) 25. aliquote into 100 µl samples 26. store @ -80 ºC Buffers used: 20 mM 0,2 mM 20% 100 mM 1 L Dialysis Buffer Hepes pH 7,9 20 ml of 1 M EDTA 0,4 ml of 0,5 M Glycerol 200 ml of 100% KCl 7,455 g 100 ml Nuclear Lysis Buffer 10 mM Hepes pH 7,9 1 ml 10% Glycerol 10 ml 100 mM KCl 0,7455 g 3 mM MgCl2 100 µl 0,1 mM EDTA 20 µl 250 ml Homogenization Buffer 0,3 M Sucrose 25,67 g 10 mM Hepes pH 7,9 2,5 ml of 1 M 10 mM KCl 833 µl of 3 M 0,1 mM EDTA 50 µl of 0,5 M 2,2 M 10 mM 10 mM 0,1 mM 300 ml Cushion Buffer Sucrose 225,72 g Hepes pH 7,9 3 ml of 1 M KCl 1 ml of 3 M EDTA 60 µl of 0,5 M Robertson Lab Robertson Lab