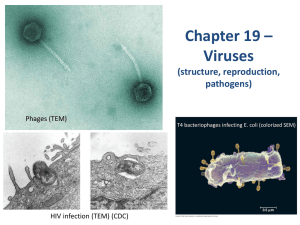
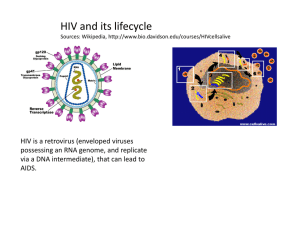
Virological decay after commencing HAART
advertisement

Virological decay after commencing HAART: proposal for analyses of UK CHIC data (Proposal from Rachel Hughes at the University of Bristol based on an idea originally put forward by John Walsh at one of our SC meetings. Circulated to SC 24/04/2013 by CS) Background A significant proportion of individuals commences HAART with a high viral load and may take months to fully suppress their viraemia. It has been shown that viral load decreases rapidly within the first week of starting treatment1,2,3. However, after the first week, and within the first months, the rate of decline is much slower, due to the release of HIV viruses by macrophages and other long-lived cells of the lymph nodes3,4. After 2-3 months some individuals may develop HIV drug resistance such that their viral load increases sharply, possibly reaching pre-treatment levels. For those individuals who have not yet developed HIV drug resistance, their viral loads are expected to continue declining slowly, eventually becoming undetectable. During the slow decline phase clinicians may be tempted to increase monitoring or switch drug therapy, even when this slow decline is predictable and the individual is expected to achieve viral suppression. Aim The aim is to develop a model that can predict time to suppression (undetectable viral load) after commencing treatment based on repeated post-treatment measurements of viral load. Population All patients in UK-CHIC that: 1. were antiretroviral naive at baseline, 2. started HAART including an NNRTI, boosted PI or integrase inhibitor (if numbers permit); 3. have a CD4 measurement and viral load measurement 0-3 months before starting HAART; 4. have two or more post-HAART viral load measurements within the first year. End point HIV viral load below assay limit of quantification; o Complication: Data will contain viral load measurements with differing assay limits of quantification. Use the upper limit of quantification (200 copies per ml); o Complication: Sensitivity analyses will assess time to confirmed suppression (two consecutive measurements below 200 copies per ml). Censor data Switch of drug regime Lost to follow up Data analysis The proposed model must account for: 1. the longitudinal structure of the data 2. non-linear trends of the viral load trajectories 3. censoring due to dropout or switch of drug-regimen Longitudinal structure The repeated measurements of viral load will be modelled in a mixed-effects framework, which accounts for variability between individuals and within individuals. Non-linear trajectories To model the viral load trajectories I will consider the following: 1. Fractional polynomials (within a linear mixed framework); 2. Linear piece-wise model with either: a. fixed number of knots (change-points) and locations for all individuals; or b. number of knots and locations can vary among individuals5; 3. Non-linear mixed effects model. Censoring The mixed-effects model will include all individuals’ viral load measurements up to time of censoring or end-point. Two approaches to account for censoring will be considered: 1. Mixed-effects model of viral load measurements only, with inverse-probability weighting to account for informative censoring; 2. Joint model of viral load and time to dropout or switch of drug regime Without inverse probability weighting, Option 1 above assumes that censoring (or missing data) due to dropout or switching regime is only dependent upon past observed viral load measurements. For example, a clinician switches a drug-regimen based on the slow decline in viral load measurements. If the reasons for censoring are dependent upon other factors (such as drug toxicity or based on CD4 measurements) then the censoring is referred to as “informative”. In the presence of informative censoring Option 1 may lead to biased parameter estimates. Informative censoring can be accounted for using inverse probability weighting (Option 1) or joint modelling of the longitudinal marker and the censoring mechanism6 (Option 2). Outcomes Generate tables of risk of virological failure for key time points (in line with BHIVA monitoring guidelines ); Generate an online risk prediction tool; Consult with experts (such as John Walsh) to further develop tools based on this work that would be useful for clinicians. Utility Reduce patient anxiety about slow VL decline; Reduce unnecessary repeat VL and genotypic testing (and perhaps even treatment switches) among patients with slow decline rates or high baseline viral loads. References 1. Ho, D. D., Neumann, A. U., Perelson, A. S., Chen, W., Leonard, J. M., and Makowitz, M. (1995). Rapid turnover of plasma virus and CD4 lymphocytes in HIV-1 infection, Nature, 373, 123–126. 2. Wei, X., Ghosh, S. K., Taylor, M. E., Johnson, V. A., Emini, E. A., Deutsch, P., Lifson, J. D., Bonhoeffer, S., Nowak, M. A., Hahn, B. H., Saag, M. S., and Shaw, G. M. (1995). Viral dynamics in human immunodeficiency virus type 1 infection, Nature, 373: 117-122. 3. Tan, W. Y. (2000). Stochastic Modeling of AIDS Epidemiology and HIV Pathogenesis, World Scientific, River Edge, New Jersey. 4. Perelson, A. S., Essunger, O., Cao, Y. Z., Vesanen, M., Hurley, A., Saksela, K., Markowitz, M., and Ho, D. D. (1997). Decay characteristics of HIV infected compartments during combination therapy, Nature, 387, 188–191. 5. Huang, Y. (2012) Segmental modelling of viral load changes for HIV longitudinal data with skewness and detection limits. Statistics in Medicine, DOI: 10.1002/sim.5527 6. Philipson, P.M., Ho, W.K. and Henderson, R. (2008) Comparative review of methods for handling drop-out in longitudinal studies. Statistics in Medicine, 27: 6276-6298.